Abstract
Persistent sciatic artery (PSA) is a rare embryonic vascular remnant, with PSA aneurysms represent an uncommon yet clinically critical complication due to inherent risks of rupture and neurovascular compression. Endovascular therapy has emerged as a preferred minimally invasive management option, though its success hinges on precise anatomical characterization via multimodal imaging. We report a case of an early 60s female with a 6.5 cm Pillet-Gauffre type IIa PSA aneurysm complicated by ipsilateral femoral artery hypoplasia, who presented with a 2-month history of right hip pain and lower limb numbness without ischemic changes. A systematic multimodal imaging workflow (Doppler ultrasound, computed tomographic angiography, digital subtraction angiography) confirmed the PSA aneurysm diagnosis and clarified its anatomical features. Endovascular treatment was performed, involving covered stent-graft deployment for aneurysm exclusion and targeted coil embolization to address an intraoperative minor type III endoleak. Over 1-year clinical and imaging follow-up demonstrated sustained stent patency, near-complete resolution of the aneurysm sac, and complete resolution of the patient's neurological symptoms. This case highlights the value of multimodal imaging for the accurate diagnosis of large PSA aneurysms and validates the safety and efficacy of endovascular intervention for this rare, high-risk condition. Notably, our findings fill a critical gap in the current literature regarding the midterm outcomes of endovascular management for large PSA aneurysms (>5 cm), providing actionable insights for clinicians encountering similar cases.
Keywords
Introduction
Persistent sciatic artery (PSA) refers to a condition in which the sciatic artery fails to degenerate during the embryonic period, remaining as the primary blood supply to the lower limbs. 1 Beyond its well-documented association with aneurysmal formation (15–44% of cases), 2 PSA holds broader clinical relevance: it can induce lower limb ischemia (via stenosis, thrombosis, or distal embolization), chronic sciatica from neurovascular compression, diagnostic dilemmas (mimicking soft tissue tumors or nerve lesions), and complications in routine vascular interventions if unrecognized. 3 PSA aneurysms themselves are of high risk due to their weight-bearing gluteal-popliteal location, which predisposes to compression, friction, and rupture. 2 The Pillet-Gauffre classification 4 serves as a key guide for treatment strategies, with endovascular therapy having emerged as the preferred minimally invasive approach—one that delivers favorable 1-year midterm outcomes, including 100% clinical success and 81.5% primary patency. 5 But data on endovascular management of large PSA aneurysms (>5 cm) remain scarce, highlighting a need for additional clinical evidence.
We report a case of a Pillet-Gauffre type IIa PSA aneurysm, which presented with a large maximum diameter of 6.5 cm. We provide detailed follow-up data for more than 1 year to validate the midterm efficacy and safety of the intervention. Furthermore, we performed a detailed interpretation of multimodal imaging findings, including Doppler ultrasound, computed tomographic angiography (CTA), and digital subtraction angiography (DSA), which are critical for the accurate diagnosis and treatment planning of this rare anomaly.
Case report
The patient was admitted to The First Affiliated Hospital of Guangdong Pharmaceutical University in June 2023. An early 60s female patient presented with a 2-month history of right hip pain and right lower limb numbness. Her medical history included hypertension, for which she took antihypertensive medications irregularly. Initially attributing her symptoms to lumbar spinal disease, the patient sought acupuncture and massage therapy. During physical examination, the therapist identified a mass in the right buttock. Further clinical assessment revealed a palpable, pulsatile mass measuring approximately 6 cm × 5 cm near the right ischial tuberosity. Bilateral femoral pulses were palpable, with intact distal perfusion to both lower limbs—no ischemia-related changes.
Doppler ultrasound demonstrated an abnormal echogenicity within the right buttock, raising suspicion for a peripheral aneurysm (approximately 5.4 cm × 4.5 cm; Figure 1), with pseudoaneurysm as a differential consideration. CTA confirmed a large PSA originating from the right internal iliac artery, with a fusiform aneurysm and a maximal transverse diameter of approximately 6.5 cm (Figure 2). The right superficial femoral artery (SFA) was notably hypoplastic compared to the left (diameter 3.2 vs. 7.5 mm). Proximal and distal diameters of the PSA adjacent to the aneurysm were 8.5 and 9.5 mm, respectively.
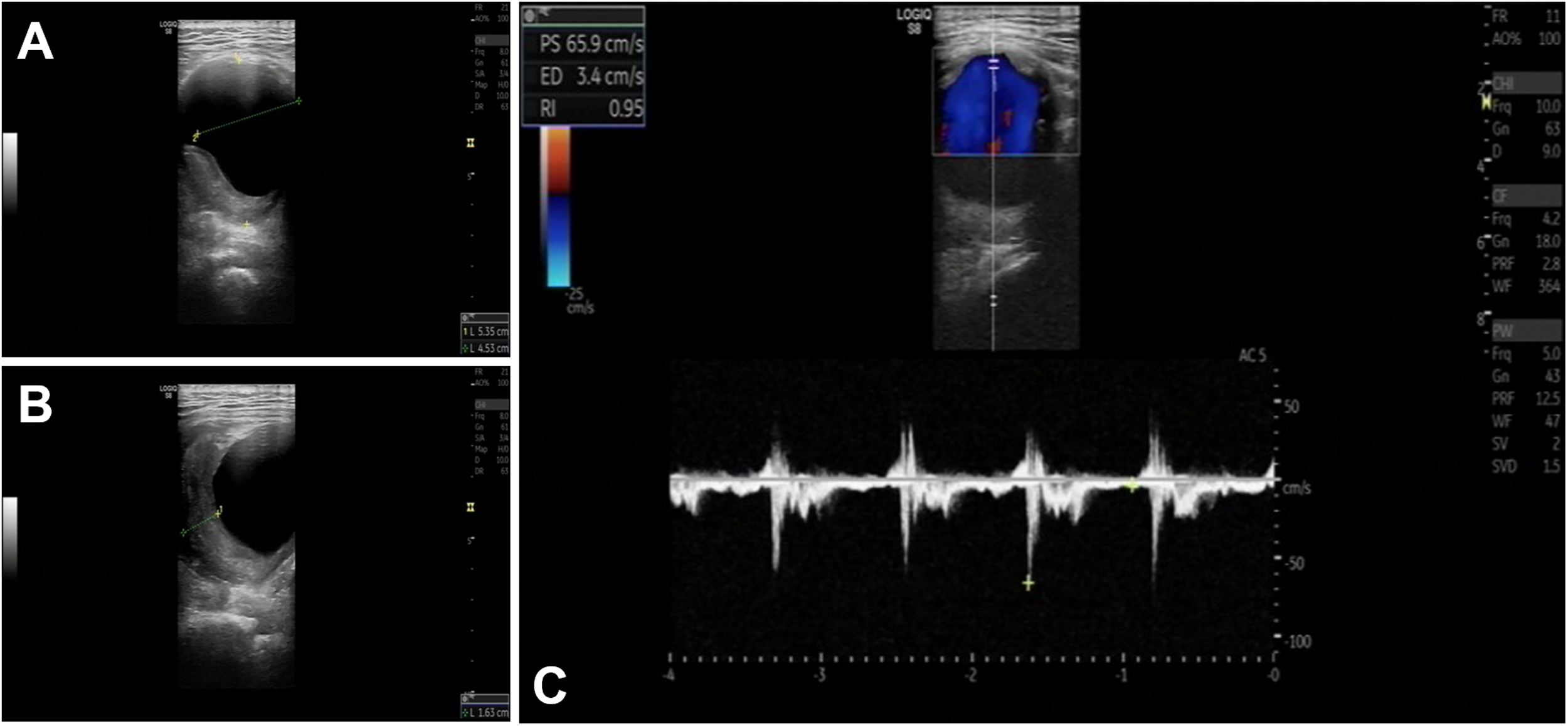
(a) Right PSA aneurysm; (b) mural thrombus in the PSA aneurysm; and (c) PS, ED, and RI of the PSA aneurysm. PSA: persistent sciatic artery; PS: peak systolic velocity; ED: end-diastolic velocity; RI: resist index.

Preoperative CTA: (a) right PSA aneurysm, the maximal transverse diameter is about 65 mm; (b) slender change of superficial femoral artery on the affected side; and (c) reconstruction of the right PSA. CTA: computed tomographic angiography; PSA: persistent sciatic artery.
Subsequent DSA confirmed that the right PSA was the primary arterial supply to the right lower extremity, classified as Pillet-Gauffre type IIa. A 2.7 Fr microcatheter (Progreat, Terumo, Tokyo, Japan) was advanced into the aneurysm sac via the right femoral artery approach. Two overlapping Viabahn stent grafts (W. L. Gore, Flagstaff, AZ) were deployed across the right ischial aneurysm via the left femoral artery approach. For Viabahn stent delivery, the left femoral artery was accessed percutaneously using an 8 Fr vascular sheath (Terumo, Tokyo, Japan), and a 0.035-in Amplatz Super Stiff guidewire (Boston Scientific, Natick, MA) was positioned across the aneurysm to establish stable support. The distal stent (10 mm × 100 mm) and proximal stent (10 mm × 50 mm) were overlapped by approximately 20 mm. Postdeployment angiography demonstrated an endoleak, suspected to be a type III endoleak likely caused by insufficient overlap of stent grafts of the same diameter. Using a perigraft approach, embolization coils (Interlock-18, Boston Scientific; 20 mm × 40 cm and 22 mm × 60 cm) were loosely deployed into the aneurysm sac via a microcatheter, resulting in a reduction of the endoleak (Figure 3).

Intraoperative angiography and deployment of the covered stent.
Postoperatively, the patient's right hip pain and lower limb numbness improved significantly. She was discharged on aspirin monotherapy (100 mg once daily) with a recommended long-term course of treatment to maintain antiplatelet effects and reduce the risk of stent thrombosis. Follow-up CTA at 1-, 6-, and 12-month postintervention demonstrated a well-apposed stent graft: the small type III endoleak resolved completely by the 6-month follow-up, and the aneurysm sac showed nearly complete regression (Figure 4). Notably, the patient's right lower limb numbness had fully resolved at 6 months, with restoration of normal ambulation and unrestricted daily living activities. Distal lower limb perfusion remained unimpaired throughout the follow-up period. The last clinical and imaging follow-up was completed in December 2024.
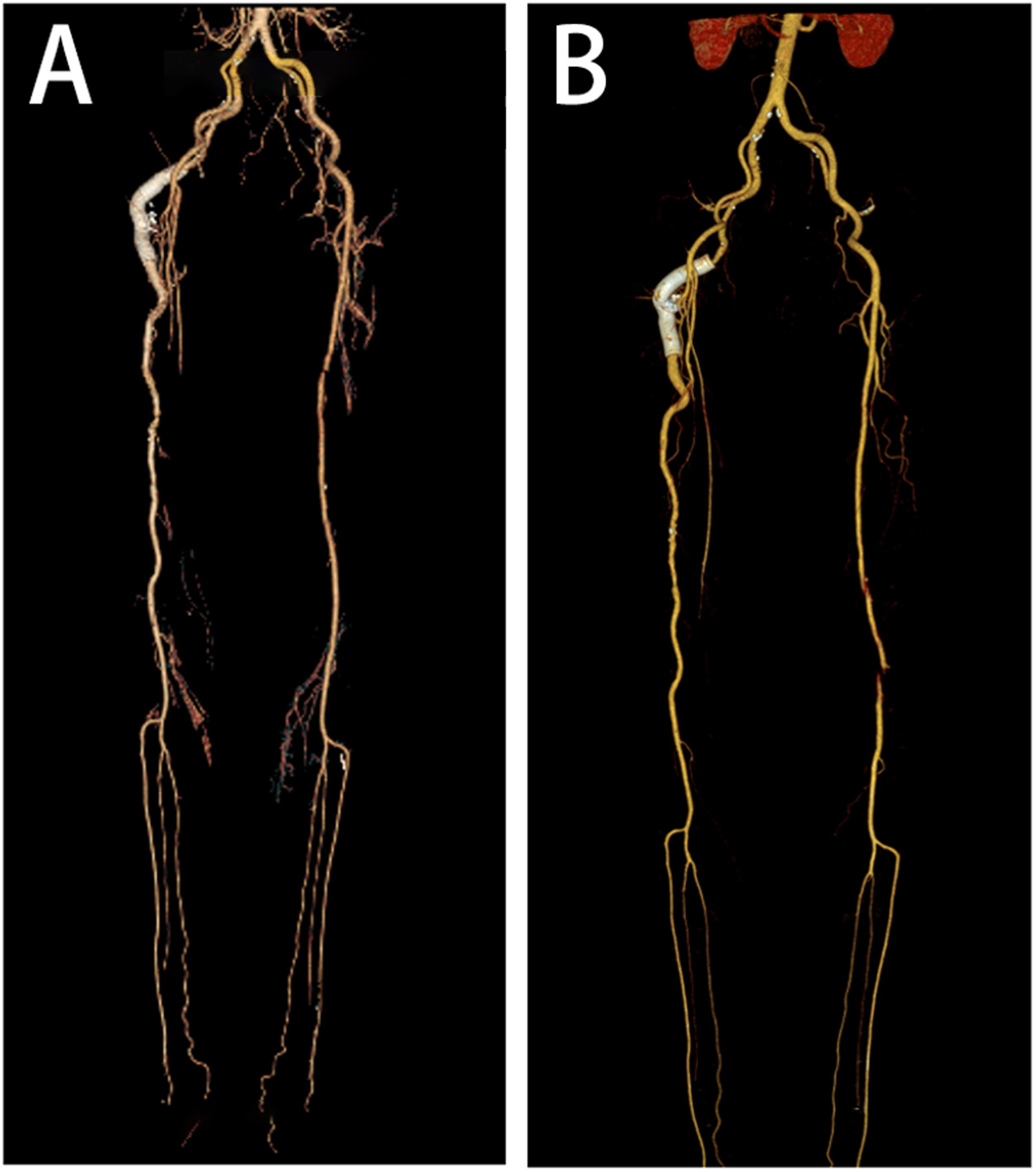
(a) CTA of both lower limbs at 1 month after operation and (b) CTA of both lower limbs 1 year after operation. The stents remained patent, and blood flow to both lower limbs was adequate. CTA: computed tomographic angiography.
This case report was prepared in accordance with the CARE guidelines. 6 The study was conducted in compliance with the Declaration of Helsinki. Prior to endovascular intervention, the patient was fully informed of the treatment plan, potential risks, and alternative options, and provided written informed consent. Before manuscript submission, the patient was apprised of the intent to publish clinical details, imaging findings, and follow-up outcomes; all personally identifiable information was anonymized, and written informed consent for publication was obtained.
Discussion
PSA is a rare congenital vascular malformation with an estimated incidence of 0.025–0.040%. 1 It results from failure of embryonic sciatic artery regression, typically presenting with either lower extremity ischemia (31–63% of cases) or aneurysm formation (48% of cases).7–9 Yet awareness of PSA (including its neurocompressive manifestations and Pillet-Gauffre classification) remains limited among clinicians, often leading to misdiagnosis as lumbar spinal disease or common peripheral artery disease. 9
In the initial diagnostic workup, the patient was initially suspected of having internal iliac artery branch aneurysm. However, detailed CTA analysis revealed two key diagnostic hallmarks of PSA: marked femoral artery asymmetry (right SFA hypoplasia) and disproportionate dilation of the right internal iliac artery. These findings raised clinical suspicion for PSA and prompted DSA confirmation—highlighting the value of multimodal imaging in diagnosing rare vascular anomalies. Doppler ultrasound served as a valuable initial screening tool, identifying the buttock mass and raising aneurysm suspicion, but its diagnostic accuracy was limited by intestinal gas interference and difficulty in visualizing the proximal sciatic artery. 10 CTA complemented ultrasound by providing comprehensive anatomical details (aneurysm size, vessel diameters, SFA hypoplasia) and enabling bilateral comparison, which is critical for PSA classification. 11 DSA, while invasive, confirmed the vascular supply pattern and PSA type, guiding treatment planning. 12 This multimodal approach aligns with the principle that no single imaging modality suffices for PSA diagnosis, and underscores the role of cross-sectional imaging in distinguishing PSA from other pelvic/buttock masses.
Treatment options for PSA aneurysms include open surgery, endovascular repair, and hybrid treatment. Open surgery is associated with significant trauma, prolonged postoperative recovery, and a notable risk of complications, 13 whereas endovascular therapy offers minimally invasive advantages, improved safety, and faster recovery.14,15 Hybrid treatment has also been reported in the literature,8,16 with the largest aneurysm in these series reaching approximately 8.5 cm in diameter. A recent systematic review 5 confirmed high technical (100%) and clinical success rates of covered stents for PSA, with acceptable midterm patency (81.5% at 1 year, 67.6% at 2 years). Notably, the review reported a median aneurysm diameter of 3.5 cm (range 1.7–7.3 cm), with sparse documentation of endovascular treatment for aneurysms >5 cm. Our case adds valuable data to this rare subgroup: the patient's 6.5 cm PSA aneurysm—among the largest reported to undergo endovascular repair—achieved nearly complete sac resolution and unobstructed blood flow at 1-year follow-up. This further validates the feasibility and efficacy of endovascular therapy for large PSA aneurysms (>5 cm), supplementing critical real-world evidence for this underreported clinical scenario.
Endoleak, defined as persistent blood flow into the aneurysm sac or around the stent-graft following endovascular aneurysm repair, is classified into five types based on its etiology and anatomical characteristics. 17 Type III endoleaks (arising from incomplete stent-graft component apposition or material defects) are high-risk subtypes with an 8.9% 1-year rupture risk, mandating intervention per Society for Vascular Surgery and European Society for Vascular Surgery guidelines. In our case, a small type III endoleak occurred at the stent overlap, likely due to the stenotic aneurysm neck and insufficient stent graft overlap, leading to suboptimal stent apposition.
Consistent with contemporary management principles,17–20 we individualized the intervention based on endoleak characteristics, anatomical context, and device-related risks. While balloon angioplasty or additional overlapping covered stents are common endovascular strategies for type III endoleaks,17–20 these approaches were avoided in our case: the minimal leak volume, combined with the PSA weight-bearing anatomical location, raised concerns about potential vascular wall injury, or stent deformation from excessive mechanical manipulation. Instead, we adopted aneurysm sac coil embolization to promote intrasac thrombosis, a strategy aligned with the core goal of “targeted flow interruption” for type III endoleak treatment.17,21 This approach effectively resolved the endoleak while minimizing invasiveness, and follow-up imaging confirmed sac regression, meeting the definition of clinical success. 17 A stent overlap of at least 15 mm is recommended. Although the overlap in this case was 20 mm, a type III endoleak still occurred, likely due to the use of stents of the same diameter and the influence of the stenotic aneurysm neck. Whenever possible, a longer overlap is desirable.
This case has several limitations. First, the initial clinical presentation of a pulsatile mass in the hip led to a CT scan that was limited to the pelvic region. Expanding the imaging to include CTA of the lower extremities would have provided more comprehensive information, facilitating earlier diagnosis, classification, and treatment planning. Second, a minor type III endoleak occurred at the stent overlap site. Although the leakage was minimal, selecting a more suitable stent during the procedure might have prevented this complication. Finally, long-term follow-up (>2 years) is needed to confirm sustained stent patency and sac regression, given the PSA's mechanical stress environment.
Conclusions
PSA exhibits characteristic imaging findings that facilitate timely diagnosis. This case demonstrates that endovascular repair with covered stents, combined with coil embolization for type III endoleaks, is safe and effective for large PSA aneurysms (>6 cm)—a subgroup with limited existing data. Over 1-year follow-up, the patient achieved complete symptom resolution, aneurysm sac regression, and unobstructed lower limb perfusion, reinforcing that endovascular therapy is a viable first-line option for PSA aneurysms, even in large diameters. Future studies should focus on long-term outcomes of endovascular treatment for large PSA aneurysms, as well as optimal strategies to prevent type III endoleaks in this anatomically challenging setting.
Footnotes
Acknowledgments
We express our gratitude to the patient for consenting to the publication of this case report.
Author contributions
Bohui Lin: drafting the original manuscript, critical revision of the manuscript, and final approval of the version to be published.
Jianmin Liang: data collection and revision of the manuscript for important intellectual content.
Shugui Huang: supervision of the study design, review of clinical treatment details, and final approval of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
