Abstract
Ovarian cancer (OC), one of the most common gynecological cancers, is usually diagnosed at an advanced stage. High-grade serous ovarian cancer (HGSOC) is a histological subtype of OC that accounts for approximately 70% of all OC cases. erb-b2 receptor tyrosine kinase 2 (ERBB2) overexpression commonly occurs in HGSOC, accounting for 12.5% of stage III and IV HGSOCs. Currently, there is no established treatment strategy for OC patients with ERBB2 amplification or ERBB2 overexpression. Whether these patients will benefit from ERBB2 inhibitors remains unclear. Herein, we report a patient with metastatic HGSOC carrying ERBB2 amplification and ERBB2 overexpression. Following progression to platinum-based chemotherapy and subsequent antiangiogenic therapies, the patient benefited from late-line pyrotinib treatment, achieving stable disease with a progression-free survival of 9.5 months. Our study provides preliminary clinical evidence supporting pyrotinib as a feasible treatment option for metastatic HGSOC patients with ERBB2 amplification and/or ERBB2 overexpression. Prospective trials of pyrotinib should be conducted in appropriately selected OC populations.
Introduction
Ovarian cancer (OC), one of the most common gynecological cancers, represents the leading cause of death from cancers of the female reproductive system.1,2 Approximately 90% of OC cases are classified as epithelial OC (EOC), typically diagnosed at advanced stages and constituting the most lethal gynecological cancer subtype.3,4 According to a dualistic model, EOCs are classified into two pathogenic types: Type I tumors are genetically stable, arise from recognizable precursor lesions (e.g. endometriosis), and exhibit indolent behavior; Type II tumors are biologically aggressive, characterized by early metastatic and frequent TP53/BRCA mutations.5,6 High-grade serous OC (HGSOC), a Type II subtype, accounts for approximately 70% of OC cases.3,4 Despite standard treatment involving cytoreductive surgery and platinum-based adjuvant chemotherapy, HGSOC patients exhibit a 5-year survival rate of only 5% to 30%. 7 Although novel therapeutic strategies, including poly(ADP-ribose) polymerase inhibitors (PARPis), immune checkpoint inhibitors, and their combinations, show promise in subsets of OC, 8 effective systemic therapy for advanced or recurrent HGSOC remains an unmet need.
Erb-b2 receptor tyrosine kinase 2 (ERBB2/HER2), a transmembrane growth factor receptor of the ErbB receptor tyrosine kinase subfamily. Abnormal activations of ERBB2, such as ERBB2 mutations, amplification, and ERBB2 overexpression, activate RAS/RAF/MEK/ERK and PI3K/AKT/mTOR pathways and promote carcinogenesis.9,10 Dysregulation of these pathways frequently contributes to chemotherapy resistance, disease progression, and poor survival in OC.11–13 ERBB2 amplification is observed in 2% to 10% of OC patients, depending on the detection method and cutoffs for defining ERBB amplification.14,15 Pyrotinib, an irreversible dual pan-ErbB receptor tyrosine kinase inhibitor, is conditionally approved in China for use in combination with capecitabine in the treatment of ERBB2-positive, advanced, or metastatic breast cancer previously treated with anthracycline or taxane chemotherapy. 16 Whether patients with ERBB2-amplified HGSOC benefit from pyrotinib treatment is not yet well established. Here, we report clinical evidence of the efficacy of pyrotinib in an HGSOC patient with ERBB2 amplification and ERBB2 overexpression.
Case presentation
In September 2019, a Chinese woman in her late 60s was admitted to the Fifth Medical Center of PLA General Hospital (Beijing, China). The patient was initially diagnosed with stage IV HGSOC according to the International Federation of Gynecology and Obstetrics (FIGO) staging system (Figure 1). Multiple metastases to the liver and peritoneum were observed (Figure 2(a) and (c)). Notably, liver cirrhosis was present at the initial diagnosis. Due to concurrent thrombocytopenia and Child-Pugh B liver function (score 8), she received primary platinum-based doublet chemotherapy (paclitaxel 270 mg plus carboplatin 400 mg, intravenously [IV] on day 1 of a 3-week cycle for one course) in November 2019 instead of debulking surgery (Figure 1(a)). Post-cycle enhanced abdominal pelvic computed tomography (CT) scans showed a partial response (PR) in the primary ovarian and metastatic lesions (Figure 2(a)), with obviously decreased cancer antigen 125 (CA-125) (from 1215 at baseline to 222 U/mL) and human epididymis protein 4 (HE4) (from 1276 to 118 pmol/L) (Figure 1(c)). Due to grade IV myelosuppression, she subsequently received paclitaxel plus cisplatin (paclitaxel 180 mg plus cisplatin 90 mg, IV on day 1 of a 3-week cycle for one course) but was intolerant of grade III gastrointestinal reaction and continuous grade IV myelosuppression. Figure 1(a) illustrates the treatment procedure.
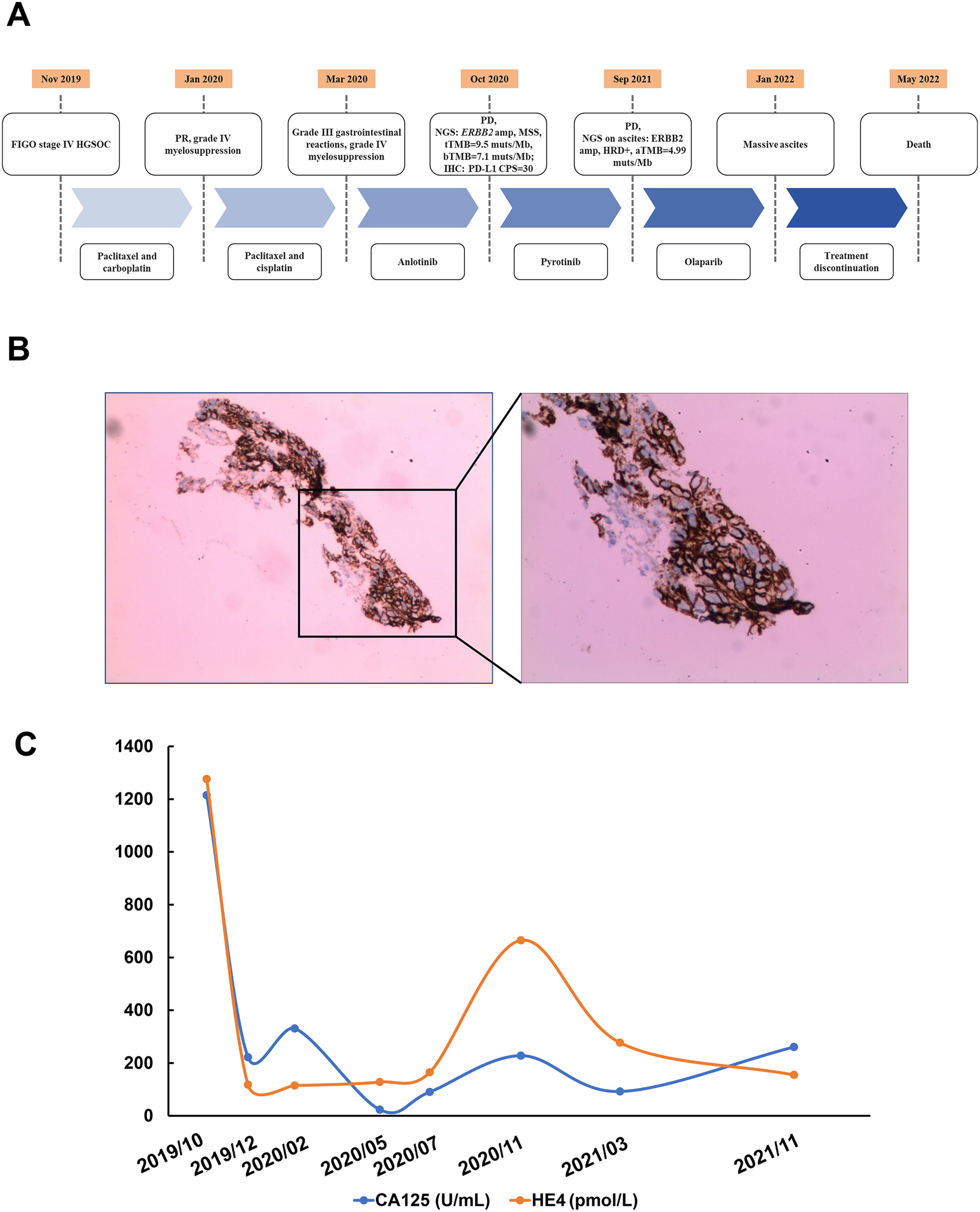
A summary of the patient's treatment history. (A) The timeline of treatment procedure; (B) immunohistochemistry staining on the metastatic peritoneal lesion for ERBB2 protein; (C) the changes of CA-125 and HE4 levels during treatment.

Magnetic resonance imaging (MRI) of the primary and metastatic lesions at treatment milestones. (A) MRI of primary ovarian lesions at baseline; (B) MRI of primary ovarian lesions during treatment; (C) MRI of metastatic lesions at baseline; (D) MRI of metastatic lesions during treatment.
In March 2020, she was administered anlotinib (12 mg once daily [q.d.]) for 8 months and achieved stable disease (SD) for 7 months. By October 2020, she developed progressive disease (PD) in the presence of ascites and was subsequently administered bevacizumab (a single intraperitoneal injection of 500 WU) and intraperitoneal hyperthermic perfusion. Biopsy of the metastatic peritoneal lesion and plasma samples underwent capture-based next-generation sequencing (NGS) using a panel consisting of 520 cancer-related genes (OncoScreen Plus, Burning Rock Biotech, Guangzhou, China). NGS results revealed an ERBB2-amplified (tumor-based copy number [CN] = 19.8, plasma-based CN = 2.8), microsatellite stable tumor with a high tissue-based tumor mutational burden (TMB, 9.5 mutations/megabase [muts/Mb]), and a plasma-based TMB of 7.1 muts/Mb. Immunohistochemical (IHC) staining of the metastatic peritoneal lesions revealed overexpression of ERBB2 (3+, Figure 1(B)) and high programmed cell death ligand-1 (PD-L1) expression (tumor proportion score = 30%, combined positive score = 30). Concurrently, increased levels of CA125 and HE4 were observed (Figure 1(C)). Table 1 lists the molecular alterations detected in samples.
Results from NGS-based molecular analysis before pyrotinib treatment and after resistance to pyrotinib.

NGS: next-generation sequencing; PD: progressive disease; TMB: tumor mutational burden; MSI: microsatellite instability; MSS: microsatellite stability; NA: not applicable/not tested; ND: not detected; CN: copy number; muts/Mb: mutations/megabase; fs: frameshift; HRD+: homologous recombination deficiency-positive.
Based on the presence of ERBB2 amplification, in November 2020, the patient was administered pyrotinib (400 mg q.d.), an irreversible dual pan-ErbB receptor tyrosine kinase inhibitor developed for the treatment of ERBB2-positive advanced solid tumors. 16 The best response to pyrotinib treatment was SD. In August 2021, abdominal CT revealed increased ascites, and the treatment response to pyrotinib was evaluated as PD. The progression-free survival (PFS) of 9.5 months (Figure 2(B) and (D)). Ascites samples obtained after pyrotinib treatment failure were subjected to NGS, which revealed ERBB2 amplification with a CN of 11.7, ascites-based TMB of 4.99 muts/Mb, and homologous recombination deficiency-positive (HRD+) without BRCA1/2 mutations. The patient has been treated with olaparib (300 mg q.d.), a PARPi, since September 2021 for 3 months. Olaparib was discontinued in January 2022 because of massive ascites. In May 2022, the patient died from ascites, cirrhosis, and peritonitis.
The report in this case conforms to the CARE guidelines. 17 Signed consents for treatment and publication were obtained from the patient. All patient details were de-identified.
Discussion
ERBB2 as an oncogenic driver is a well-established therapeutic target in both breast and gastric cancer. ERBB2 amplification/ERBB2 overexpression is observed in 10% to 20% of breast cancers and 8% to 30% of gastric cancers.18,19 The introduction of ERBB2-targeted therapies (such as trastuzumab, pertuzumab, ado-trastuzumab emtansine [T-DM1], lapatinib, and neratinib) has markedly improved outcomes in these malignancies. Although ERBB2 overexpression is frequently observed in OC, including HGSOC,15,20 the clinical efficacy of ERBB2-targeted therapies for OC remains inconclusive. Trastuzumab and pertuzumab monotherapies show limited objective response rates (ORRs) in ERBB-overexpressing OC.21,22 While isolated reports describe durable responses (>36 months) to trastuzumab plus pertuzumab in ERBB2-amplified HGSOC with a late-line trastuzumab–pertuzumab combination, 14 robust evidence from large-scale clinical trials is lacking. In ovarian mucinous carcinomas, exceptional cases with ERBB2-amplification/ERBB2 overexpression exhibit sustained disease control or post-surgery complete response with trastuzumab-based regimens.23–25 Nevertheless, similar outcomes have rarely been documented in other epithelial OC subtypes. For HER2-directed antibody–drug conjugates (ADCs) including T-DM1, SYD985, and trastuzumab deruxtecan (T-DXd), preclinical studies have indicated potent antitumor activity against ERBB2-overexpressing OC models.26–28 The phase II DESTINY-PanTumor02 trial reported an ORR of 45% (18 out of 40) with T-DXd in ERBB2-overexpressing OC. 29 Notably, the HER2 IHC 3+ subgroup within this trial achieved a higher ORR of 63.6%. 29 Emerging ADCs, such as SHR-A1811 and IBI354, have shown promising activity in early-phase trials,30,31 though phase III validation is needed.
Pyrotinib, an irreversible pan-ErbB inhibitor, has been approved for the treatment of recurrent and metastatic breast cancer. 16 Promising efficacy and acceptable safety of pyrotinib have been observed in ERBB2-amplified advanced non-small cell lung cancer (ChiCTR1800020262) and ERBB2-positive metastatic breast cancer (NCT03691051).32,33 In addition, pyrotinib monotherapy or combination regimens demonstrated antitumor activity in ERBB2-expressing colorectal and gastric cancer across early-phase trials.34–37 Case reports have indicated that pyrotinib may confer clinical benefits in ERBB2-altered OC. Fang et al. 38 reported a durable clinical benefit with pyrotinib in a patient with ERBB2-amplified recurrent mucinous ovarian carcinoma progressing after trastuzumab therapy. In a patient with HER2-positive recurrent ovarian clear cell carcinoma, pyrotinib monotherapy achieved a PFS of 28 months. 39 To our knowledge, the present case is the first documentation of pyrotinib efficacy in ERBB2-amplified HGSOC. In this study, the metastatic HGSOC patient with high-level ERBB2 amplification (ERBB2 CN of 11.7) and ERBB2 overexpression (3+ positivity) had progressed on platinum-based doublet chemotherapy and subsequent antiangiogenic therapies. This patient achieved a PFS of 9.5 months on late-line pyrotinib. In platinum-ineligible patients, this PFS outcome significantly exceeds the historical benchmark of ≤6 months typically achieved with non-platinum chemotherapy. 40 It also compared favorably with other HER2-targeted therapies: trastuzumab (median PFS [mPFS] 2 months, 95% CI unshown) 21 ; pertuzumab (mPFS 4.9 months, 95% CI: 2.6 to 5.7 months) 22 ; and T-DXd (mPFS 5.9 months, 95% CI: 4.0 to 8.3 months). 29 These data suggest that pyrotinib may be an effective treatment option in advanced OC with ERBB2 amplification/ERBB2 overexpression. Nevertheless, resistance to anti-HER2 therapy remains a significant challenge in OC. The resistance may arise from tumor heterogeneity in ERBB2 expression or activation of alternative signaling pathways (e.g. PI3K/AKT/mTOR). CCNE1 amplification was detected in the presented case, potentially dysregulating the cell cycle. Elevated TMB reflects genomic instability, which could facilitate the emergence of treatment-resistant subclones. These molecular features may underlie the secondary resistance to pyrotinib, though precise mechanisms require further investigation.
Liquid biopsy has emerged as a new diagnostic concept that provides the opportunity to monitor cancer in various body fluids instead of in tissue samples, such as plasma, cerebrospinal fluid, and pleural effusion.41,42 However, the feasibility of using ascites as a liquid biopsy sample to detect actionable alterations has not yet been established. In this study, ERBB2 amplification and HRD positivity were detected in the ascites after progression to pyrotinib. The patient was administered the PARPi olaparib based on the presence of HRD+ and achieved a PFS of 4 months. Our study indicates that ascites may be an alternative tool for detecting actionable alterations and expanding therapeutic options for OC patients with insufficient tumor samples for molecular testing.
This study has some limitations associated with this work. First, only one patient was included in the study. Larger cohorts and clinical trials are needed to further evaluate the efficacy of pyrotinib against ERBB2 alterations in OC patients. Second, the concordance of genomic profiling between ascites and paired tissue samples derived from OC patients should be investigated in a large cohort study. Third, due to data-sharing regulations in our region, we are unable to deposit the raw NGS data in public repositories. However, the analyzed NGS results presented in this article are available upon reasonable request.
Patient perspective
The patient mentioned that pyrotinib treatment made her feel better with reduced pain, and the discomfort caused was acceptable.
Conclusions
We revealed clinical evidence that a FIGO stage IV HGSOC patient carrying ERBB2 amplification and ERBB2 overexpression achieved SD with a PFS of 9.5 months upon late-line pyrotinib treatment. Our data shed light on the treatment options for previously treated metastatic HGSOC patients with ERBB2 amplification/ERBB2 overexpression.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Bing Qi, Haiwei Du, and Tengfei Zhu from Burning Rock Biotech, Guangzhou, China, for their assistance. The AI tools DeepSeek-R1 and Kimi k1.5 were used for language improvement.
Ethical considerations
Ethical review committee approval was waived for this case report, following the regulations of the Fifth Medical Center of the PLA General Hospital. This study was conducted in accordance with the World Medical Association Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from the reported patient.
Consent to publication
Written consent was obtained from the patient for publication of this case.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Key Research and Development Program of China (grant no. 2020YFC0122302).
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The analyzed NGS results are presented in the manuscript, and raw data are available from the corresponding author on reasonable request. Other data and materials have been provided in this manuscript.
