Abstract
Objective
The aim is to enhance clinician awareness of the disease, reduce the risk of misdiagnosis, and analyze the risk factors for the progression of Chlamydia psittaci infection to severe pneumonia.
Methods
We conducted a retrospective analysis of the clinical data from patients infected with Chlamydia psittaci at our hospital.
Results
Thirty-three patients diagnosed with Chlamydia psittaci pneumonia were included in this study. Among them, 23 (72.2%) were male and 10 (27.8%) were female, with a mean age of 59.1 ± 11.1 years. Twenty-four patients (72.7%) had a clear history of poultry exposure. The main clinical manifestations were high fever (n = 30, 90.9%), cough (n = 28, 84.8%), chill (n = 24, 72.7%), expectoration (n = 22, 66.7%), fatigue (n = 20, 60.6%), poor appetite (n = 20, 60.6%), dyspnea (n = 13, 39.4%), and myalgia (n = 10, 30.3%). The lymphocyte count in severe pneumonia group was significantly lower than that in non-severe pneumonia group. C-reactive protein, erythrocyte sedimentation rate, and lactate dehydrogenase levels in the severe group were remarkably higher than those in the non-severe group. The common imaging findings included flake high-density shadows (n = 29, 87.9%), consolidation (n = 20, 60.6%), pleural effusion (n = 17, 51.5%), bronchial inflation signs (n = 15, 45.5%), and ground-glass exudation (n = 20, 60.6%). The majority of patients received treatment with either doxycycline alone, quinolones alone, or a combination of doxycycline and quinolones.
Conclusion
History of contact with birds or poultry, repeated high fever, and flake high-density shadows with consolidation in Chest computed tomography can serve as important indicators for diagnosing C. psittaci pneumonia. Lower lymphocyte counts were identified as the sole risk factor associated with severe C. psittaci pneumonia. Quinolones and doxycycline are effective treatments for C. psittaci pneumonia.
Keywords
Introduction
Chlamydia psittaci (C. psittaci) pneumonia is a zoonotic disease caused by C. psittaci, which is a gram-negative, obligate intracellular bacterium.1,2 Infection with C. psittaci is more common in individuals who have direct contact with infectious birds, poultry, or their feces and respiratory secretions. Additionally, exposure to contaminated aerosols or dust containing C. psittaci can also lead to infection.3–5 Furthermore, recent studies have demonstrated human-to-human transmission of C. psittaci,1,6,7 horses have also been demonstrated as a novel source of infection,8,9 but the mechanism is not clear. Clinicians often exhibit a significant lack of awareness regarding C. psittaci pneumonia, a condition that tends to be overlooked or misdiagnosed. This oversight can primarily be attributed to the non-specific nature of the symptoms associated with the disease, alongside the limited availability of effective testing options. When a diagnosis is either missed or delayed, patients are at risk of developing serious complications, including severe pneumonia, multiorgan failure, and, in the worst-case scenario, death. This underscores the critical importance of timely and accurate diagnosis for improving patient outcomes.10–12
Therefore, given the pressing need to address this issue, it is crucial to conduct comprehensive clinical analyses that delve into the epidemiology and clinical traits of C. psittaci pneumonia. In light of this necessity, our study undertook a retrospective analysis of the clinical data concerning 33 patients diagnosed with C. psittaci pneumonia, utilizing targeted next-generation sequencing (tNGS) for confirmation. This thorough examination encompassed a wide array of factors, including demographic variables, presenting symptoms, relevant laboratory findings, co-infecting pathogens, chest computed tomography (CT) imaging results, treatment approaches, and patient outcomes. Such a detailed investigation aims to foster a deeper understanding of the disease, thereby enhancing clinical awareness and informing better diagnostic and treatment strategies. The purpose of this report is to enhance clinicians’ awareness of the disease, improve the accuracy and timeliness of its identification, provide evidence to inform the diagnosis and treatment of C. psittaci infection. Our study also analyzed the risk factors associated with the progression of C. psittaci infection into severe pneumonia using multivariate logistic regression analysis, a relatively rare approach in existing literature. This analysis aids clinicians in accurately assessing disease severity and progression.
Material and methods
Participants
This is a retrospective study. This study was conducted by gathering data from the medical records of patients who were confirmed to have infections caused by Chlamydia psittaci at The First Hospital of Changsha (The Affiliated Changsha Hospital of Xiangya School of Medicine) between October 2021 and December 2024. The patients in the study were selectively enrolled. All patient details have been de-identified. C. psittaci infection was confirmed by tNGS in bronchoalveolar lavage fluid (BALF) or blood. The study was conducted in accordance with the revised Declaration of Helsinki in 2024. The reporting of this study conforms to STROBE guidelines. 13 The research study received ethical approval from the Institutional Review Board (IRB) of The First Hospital of Changsha (Grant number: 2024 (06) V. 1.0). The study specifically focused on participants who met the criteria as follows: 1) patients who met the diagnostic criteria for community acquired pneumonia, 2) C. psittaci fragments were identified through tNGS and discharged with a diagnosis of C. psittaci pneumonia, and 3) patients with complete clinical information. Exclusion criteria: 1) patients with incomplete data; 2) patients who have recently participated in other interventional trials; 3) pregnant or lactating women.
Targeted next-generation sequencing
Sample preparation
A volume of 650μL of the sample was liquefied by combining it with an equal volume of 80 mmol/L dithiothreitol in a 1.5 mL centrifuge tube. The mixture was homogenized for 15 s using a vortex mixer. Meanwhile, a positive control and a negative control from the Respiratory Pathogen Detection Kit (KS608-100HXD96, KingCreate, Guangzhou, China) were set up to monitor the whole experiment process of tNGS.
Nucleic acid extraction
Subsequently, 500μL of the homogenate was utilized for total nucleic acid extraction and purification via the MagPure Pathogen DNA/RNA Kit (R6672-01B, Magen, Guangzhou, China), following the manufacturer's protocol.
Library construction and sequencing
The library was constructed using the Respiratory Pathogen Detection Kit, and a no template control was set up to monitor the library construction and sequencing process. This process encompassed two rounds of PCR amplification. The sample nucleic acid and cDNA were employed as templates, and a set of 153 microorganism-specific primers were selected for ultra-multiplex PCR amplification to enrich the target pathogen sequences, spanning bacteria, viruses, fungi, mycoplasma, and chlamydia. After the amplification, PCR products underwent purification with beads, followed by amplification using primers containing sequencing adapters and distinct barcodes. The quality and quantity of the constructed library were evaluated using the Qsep100 Bio-Fragment Analyzer (Bioptic, Taiwan, China) and Qubit 4.0 fluorometer (Thermo Scientific, Massachusetts, United States), respectively. Generally, the library fragments exhibited sizes within the approximate range of 250–350 bp, and the library concentration was maintained at a minimum of 0.5 ng/μL. The concentration of the mixed library was reassessed and subsequently diluted to a final concentration of 1 nmol/L. Subsequently, 5μL of the mixed library was mixed with 5μL of freshly prepared NaOH (0.1 mol/L). Following brief vortexing and centrifugation, the library was incubated at room temperature for 5 min. The diluted and denatured library was subsequently subjected to sequencing on an Illumina MiniSeq platform using a universal sequencing reagent kit (KS107-CXR, KingCreate, Guangzhou, China). On average, each library yielded approximately 0.1 million reads, with a sequencing read length of single-end 100 bp.
Bioinformatics analysis
Sequencing data were analyzed using the data management and analysis system (v3.7.2, KingCreate). The raw data underwent initial identification via the adapter. Reads with single-end lengths exceeding 50 bp were retained, followed by low-quality filtering to retain reads with Q30 > 75%, ensuring high-quality data. The single-ended aligned reads were then compared using the Self-Building clinical pathogen database to determine the read count of specific amplification targets in each sample. The reference sequences used for read mapping were a database curated from different sources, including Genbank database, Refseq database, and Nucleotide database from NCBI.
Data collection
Patients’ clinical data included age, sex, underlying diseases, poultry or avian contact history, disease onset time, smoking history, treatment protocol, outcomes, and length of hospital stay. In addition, data on clinical symptoms, laboratory examination results, co-infection of principal pathogens in BALF, and chest CT imaging characteristics were collected. Laboratory data were collected before treatment or on day one after admission. The data were as follows: white blood cell (WBC) count, neutrophil count, lymphocyte count, platelet count, hemoglobin, C-reactive protein (CRP), procalcitonin (PCT), erythrocyte sedimentation rate (ESR), lactate dehydrogenase (LDH), creatine kinase (CK), CK-myocardial band, aspartate aminotransferase (AST), alanine transaminase (ALT), and D-dimer.
Statistical analysis
Data analysis and mapping were conducted utilizing GraphPad Prism, version 10. In this analysis, Categorical data was presented as counts and percentages, and continuous data were expressed as mean ± SD if the data were normally distributed, or as medians with their interquartile range. Means for continuous variables were compared using independent group Students t tests when the data were normally distributed and the Mann–Whitney test in cases of non-normal distribution. Proportions for categorical variables were compared using the χ2 test. Additionally, for the analysis of categorical variables, Fisher's exact test was utilized, which is particularly effective for small sample sizes. Using multivariate logistic regression analysis to identify risk factors associated with severe C. psittaci pneumonia. P < 0.05 in univariate analysis was considered for the multivariate model. The analyses used a two-tailed test, and statistical significance was indicated by p < 0.05.
Results
Clinical information of patients
Of the 33 patients, 23 were male (72.2%) and 10 were female (27.8%), with an average age of 59.1 ± 11.1 years; the mean length of hospital stay was 10.4 ± 3.2 days. Thirty patients had clear fever symptoms (90.9%, 30/33), with a peak value median of 39.5 °C(range 38.9–40.5 °C), cough (n = 28, 84.8%), chill (n = 24, 72.7%), expectoration (n = 22, 66.7%), fatigue (n = 20, 60.6%), poor appetite (n = 20, 60.6%), dyspnea (n = 13, 39.4%), myalgia (n = 10, 30.3%), headache (n = 6, 18.2%), vomiting and diarrhea (n = 5, 15.2%). Dyspnea emerged as the sole clinical symptom that exhibited a statistically significant difference between the groups of patients diagnosed with severe pneumonia and those with non-severe pneumonia (p<0.01). This finding underscores the importance of dyspnea as a distinguishing feature in the clinical assessment of pneumonia severity. The clinical manifestations observed in the enrolled patients can be found detailed in Table 1.
The common clinical manifestations of patients with C. psittaci pneumonia.

Bold values indicate statistically significant differences between the two groups (P-value less than 0.05).
Epidemiological data
Twenty-four of 33 patients (72.7%) had a clear history of avian exposure (including keeping parrots, raising or slaughter pigeons, chickens, and ducks). Seven patients in severe cases of C. psittaci pneumonia, 17 patients in non-severe pneumonia group, the remaining patients did not show a clear epidemiological history.
Smoking history and underlying diseases
Fifteen patients had a history of smoking (n = 15; 45.5%). The underlying diseases were Type 2 diabetes (n = 12, 36.4%), hypertension (n = 11, 33.3%), chronic liver disease (n = 9, 27.3%), chronic obstructive pulmonary disease (n = 7, 21.2%), coronary heart disease (n = 7, 21.2%), and bladder cancer postoperation (n = 1, 3.03%), as shown in Figure 1. The length of stay in hospital and age showed a significant difference between the severe and non-severe group. The sex distribution and smoking history did not differ significantly between the severe and non-severe group (Table 1).
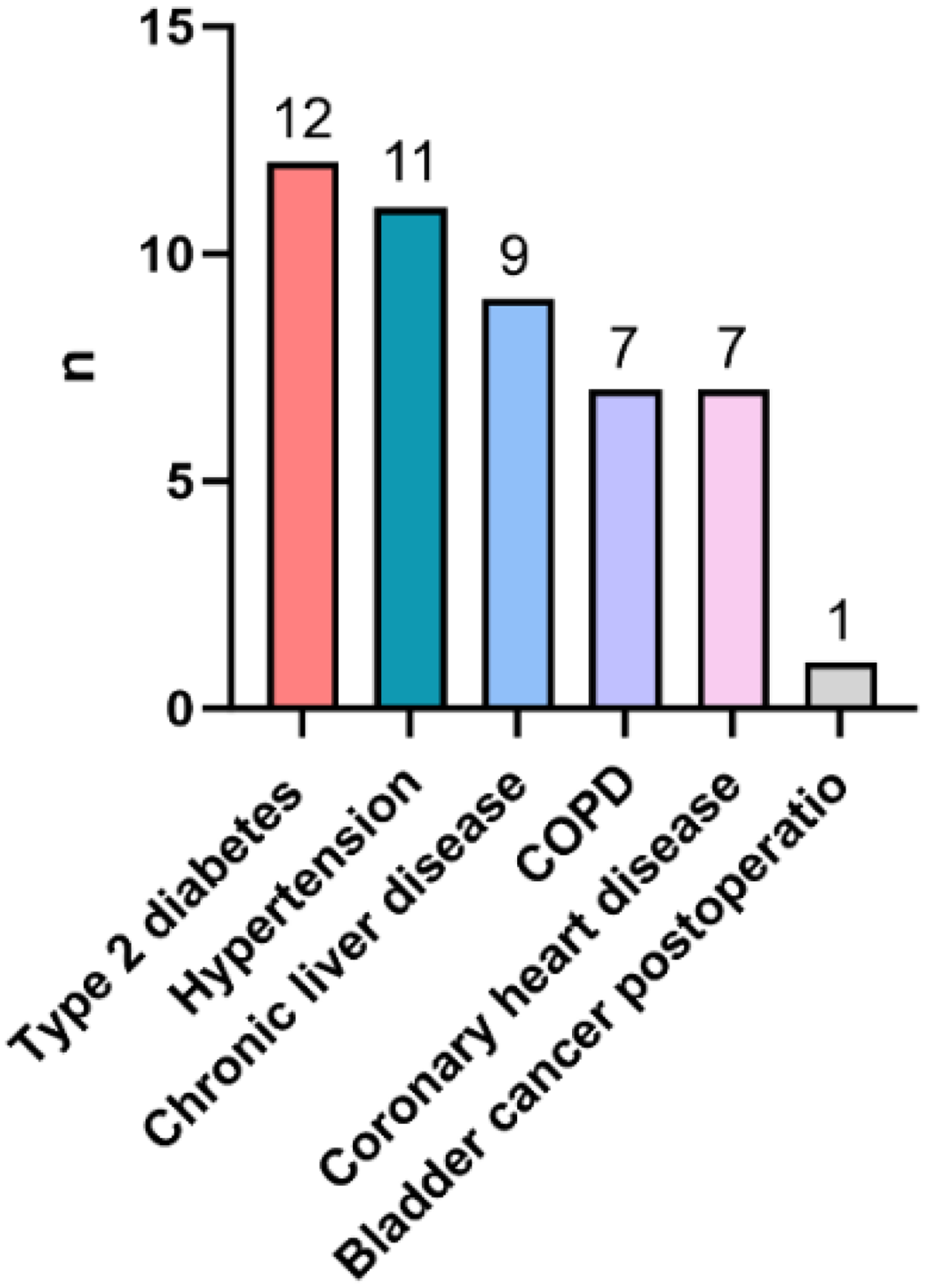
The common underlying disease in C. psittaci pneumonia.
Complications and laboratory findings
Common complications included electrolyte disorders (51.5% [17/33]), acute liver injury (45.5% [15/33]), type I respiratory failure (36.4% [12/33]), hypoalbuminemia (27.3% [9/33]), sepsis (15.2% [5/33]), and MODS (6.1% [2/33]) (Figure 2).
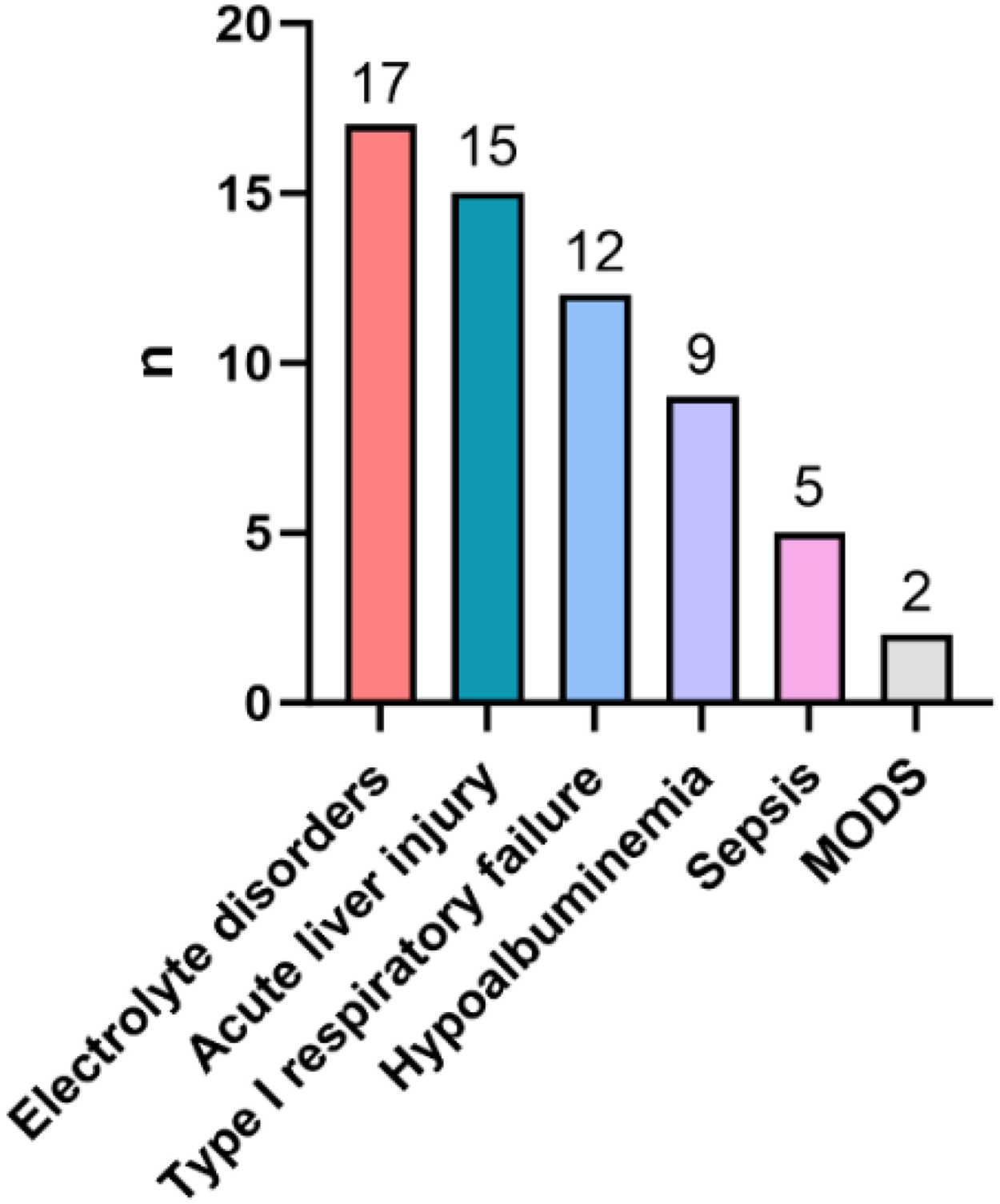
The common complications in C. psittaci pneumonia.
The results of the laboratory assessments conducted on the two groups are summarized in Table 2. Within the cohort, five patients exhibited elevated WBC counts, 27 cases displayed normal WBC counts, and one case presented with decreased WBC counts (in the severe pneumonia group) during admission. CRP levels and D-dimer levels were elevated in all patients, PCT levels were elevated to >0.05 ng/mL in 27 patients, of which two showed an elevation of >2 ng/mL. Furthermore, 29 patients presented with elevated LDH levels, 10 patients with elevated ALT levels, and 15 patients with elevated AST levels. The lymphocyte (LYM) count in the severe pneumonia group was significantly lower than that in non-severe pneumonia group (p<0.05) (Figure 3A). The CRP, ESR, and LDH levels were significantly higher in the severe group than in the non-severe group (p<0.05) (Figure 3 B, C, D).
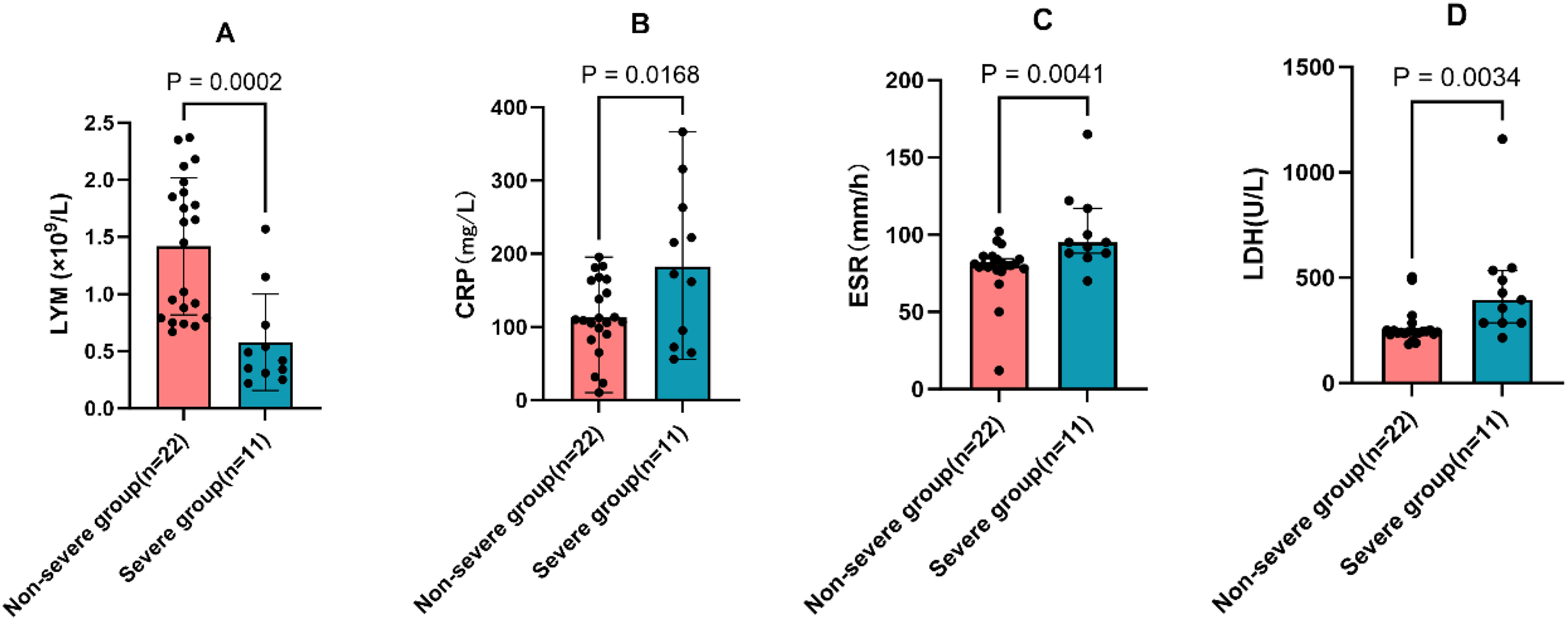
(A) The lymphocyte (LYM) count in the severe group was significantly lower than that in non-severe group (p<0.001). (B, C, D) The levels of CRP, ESR and LDH were significantly higher in the severe group compared with the non-severe group (p<0.05). CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; LDH: lactate dehydrogenase.
Laboratory data of patients with C. psittaci pneumonia.

WBC: white blood cell; NE: neutrophil; PLT: platelet; LYM: lymphocytes; ALB: albumin; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; PCT: procalcitonin; CK: creatine kinase; CK-MB: CK-myocardial band; LDH: lactate dehydrogenase; AST: aspartate aminotransferase; ALT: alanine transaminase.
Risk factors associated with severe C. Psittaci pneumonia
Multivariate analysis revealed that lower lymphocyte counts (OR 0.8048, 95% CI 0.7245–0.9125, p = 0.045) were the only risk factor for progression to severe pneumonia (Table 3).
Multivariate analysis of the risk of severe pneumonia.

Chest CT images
In this study, every patient underwent chest CT scans, revealing various noteworthy findings. The most frequently observed manifestations included flake high-density shadows (n = 29, 87.9%), consolidation (n = 18, 54.5%), pleural effusion (n = 17, 51.5%), bronchial inflation signs (n = 15, 45.5%), and ground-glass exudation (n = 20, 60.6%). The distribution of the lesions was as follows: right lung (12/33), left lung (11/33), and both lungs (10/33). Furthermore, pleural effusion was noted in 18 patients; of these, 13 were found to have unilateral pleural effusion, while four patients demonstrated bilateral pleural effusion, highlighting the complexity of the observed conditions.
Co-detected microbiota distribution in C. psittaci pneumonia
Out of the 33 BALF samples, the tNGS fastq data from four patients was lost due to improper data storage, resulting in their exclusion from our statistical analysis. Therefore, our analysis focused on the remaining 29 patients. Among these samples, Chlamydia psittaci was identified as the sole pathogen in five BALF samples. The most frequently co-detected pathogen with C. psittaci was Candida albicans, followed by the Epstein-Barr virus.
Treatment and outcome
Most patients were treated with quinolones (24/33, 72.7%) or doxycycline (12/33, 36.4%). Among these patients, 44.4% patients had a history of empirical antibiotic treatment. Among the patients receiving empirical antibiotic treatment, 55.6% altered their antibacterial drug regimen based on the tNGS results. Among the 33 patients, 2 were prescribed doxycycline alone, 14 received quinolones alone (Levofloxacin 0.5 g Qd, or Moxifloxacin 0.4 g, Qd), 5 with doxycycline (200 mg, Qd) and moxifloxacin in combination, 3 received a combination of doxycycline and meropenem, 2 received a combination of doxycycline and piperacillin/tazobactam, 2 were prescribed a combination of moxifloxacin and meropenem, and 3 patients were treated with a combination of piperacillin/tazobactam and moxifloxacin. Among the 33 patients, three patients were treated with invasive mechanical ventilation, two patients received high-flow nasal cannula therapy, and four patients were treated with non-invasive ventilation. A higher proportion of severe pneumonia patients (9/11, 81.8%) received combination therapy compared to non-severe pneumonia patients (8/22, 36.4%). Severe patients were more likely to receive invasive ventilation than non-severe patients (p = 0.0302). Sepsis was observed in five patients with severe infection in the severe group, Table 4. Ultimately, all patients were successfully treated and discharged without any fatalities during their hospitalization.
Treatment and outcomes of patients with C. psittaci pneumonia.

Discussion
Chlamydia psittaci is a gram-negative and obligate intracellular bacterium that can lead to C. psittaci pneumonia. Infection with C. psittaci is more common through the inhalation of aerosols containing urine, feces, or respiratory secretions from birds or poultry. 14 Adults, particularly those in direct contact with birds and poultry, are more susceptible to C. psittaci.15,16 In our study, 24 patients (72.7%) had a clear history of avian exposure history, which is consistent with a previous study. 17 Therefore, a history of exposure to poultry or pigeons can serve as a crucial indicator for diagnosing C. psittaci infection.
In the past, the diagnosis of C. psittaci infection mainly relied on clinical manifestations and traditional testing methods. Therefore, the clinical incidence of C. psittaci pneumonia may be greatly underestimated.
Traditional diagnostic methods for Chlamydia psittaci include pathogen isolation and culture, serological testing, and fluorescence quantitative PCR. However, these culture and serum detection methods frequently entail long processing times, demonstrate low positivity rates, and require specialized laboratory support. Consequently, missed diagnoses and misdiagnoses are common.18–21 Additionally, the atypical symptoms of this infection further complicate diagnosis. Fortunately, with the advancements in tNGS, the identification of rare pathogens has become more feasible.22–24 This method is less influenced by prior antibiotic exposure, offering not only a rapid and accurate diagnosis but also minimizing the risk of ineffective treatment resulting from misdiagnosis. Although the cost of this testing method is only one-sixth that of metagenomic next-generation sequencing, it remains relatively high compared to traditional methods. Consequently, its widespread adoption in clinical practice continues to face challenges.
In this study, we categorized 33 patients diagnosed with C. psittaci pneumonia into severe pneumonia and non-severe pneumonia groups, conducting a retrospective analysis of their clinical characteristics. The findings indicate that C. psittaci pneumonia predominantly affects middle-aged and elderly individuals, with older patients demonstrating a higher propensity for developing severe pneumonia. Notably, there is no significant difference in gender distribution between the two groups. In our study, common clinical manifestations include high fever, cough, expectoration, fatigue, poor appetite, and dyspnea, which align with findings from existing studies.25,26 Notably, our results indicate that the severe pneumonia group is more likely to experience symptoms of dyspnea compared to the non-severe group, with this difference being statistically significant. This finding contrasts with previous studies. 10
In our study, CRP levels and D-dimer levels were elevated in all patients. Additionally, PCT levels were recorded as exceeding 0.05 ng/mL in 27 patients (81.8%), with five individuals showing levels greater than 2 ng/mL. This increase in PCT values suggests the presence of a mixed infection involving multiple pathogens. The CRP, ESR, and LDH levels in the severe C. psittaci pneumonia group were remarkably higher than those in the non-severe group, 17 and complications were frequent in the severe group in this study. In contrast, lymphocyte count levels were significantly lower in the severe group compared to the non-severe group (p < 0.05). Multivariate analysis indicated that lower lymphocyte counts emerged as the sole risk factor for progression to severe pneumonia. This finding indicates that lymphocyte counts may serve as valuable markers for assessing disease severity and prognosis. Furthermore, since lymphocyte levels reflect the body's immune status, we speculate that the occurrence and severity of the disease may be closely related to the patient's immune status. Therefore, in future studies, the analysis of CD4 T cells, CD8 T cells and cytokines in these patients can be included to enhance our understanding of the role of the immune-inflammatory response in this disease.
Additionally, acute liver injury is also prevalent in patients with C. psittaci pneumonia, which is consistent with previous studies,27–30 in the current study, nearly half of the patients experienced acute liver injury. Furthermore, electrolyte disorders, acute respiratory failure, hypoalbuminemia, and sepsis were also commonly observed in our study, aligning with prior research. Our study also indicated that severe cases were more likely to experience Type I respiratory failure, sepsis, and septic shock.
Our research indicates that chest CT is crucial in the diagnosis of pneumonia caused by C. psittaci. The primary imaging features associated with this type of pneumonia include the presence of high-density shadows indicative of consolidation, alongside pleural effusion and signs of bronchial inflation. Additionally, ground-glass exudation is also observed in these patients. Notably, we did not find evidence of cavitary lesions or tree-in-bud patterns in our imaging results. This absence of certain signs can serve as a valuable differentiating factor, aiding clinicians in distinguishing C. psittaci infections from other types of pneumonia. 31
The results of tNGS validated the occurrence of mixed infections within our investigation, revealing that 82.8% of the patients displayed this condition. The predominant co-infections identified were linked to Candida albicans and EBV. It was noted that patients suffering from C. psittaci pneumonia who also had co-infections might experience more intense infections and complications, underscoring the importance of additional research in this area.
Tetracyclines, macrolides, and quinolones are widely recognized as the standard treatments for pneumonia caused by C. psittaci, with tetracyclines being considered the first-line pharmacological agents. 1 In the present study, it was observed that over half of the patients selected quinolone therapy, either alone or in combination with other antimicrobials, as their initial treatment approach. Notably, most of these patients exhibited positive favorable therapeutic outcomes from this regimen.
However, a subset of patients initially displayed suboptimal responses to the treatment, which led to necessary adjustments based on precise pathogen identification achieved through tNGS. When these patients transitioned to quinolone or a regimen that included doxycycline, every individual demonstrated marked improvement in their condition. The results of our investigation indicate that both doxycycline and quinolones are effective in managing cases of C. psittaci pneumonia. Furthermore, the findings underscore the significant therapeutic efficacy of quinolones in treating this type of pneumonia. This highlights the importance of tailoring antimicrobial therapy based on patient response and pathogen identification to optimize treatment outcomes for individuals suffering from this infection.
However, our study has several limitations. First, the relatively small sample size of only 33 patients restricts our ability to comprehensively examine all relevant characteristics of C. psittaci pneumonia. Second, our study did not perform sample size calculations, thus the limited sample size may affect the statistical significance of the results. In addition, a small number of cases can undermine statistical power by increasing random errors, reducing the ability to detect effects, and exacerbating model instability, leading to the omission of genuine risk factors, particularly for rare outcomes. Third, our study did not include a control group of patients with non-psittacosis pneumonia or other types of severe pneumonia. Comparing clinical features, laboratory findings, and outcomes with such patients may help to more clearly distinguish psittacosis pneumonia and validate the identified risk factors. Fourth, as our study involved analyzing the medical records of patients previously hospitalized for psittacosis pneumonia, these cases only documented the diagnostic results from NGS but lacked specific detailed high-throughput sequencing data. Fifth, as a single-center retrospective study, there is a potential for selection bias among participants. In future studies, we will expand the sample size and conduct multi-center studies to verify the findings of this study.
Conclusion
In conclusion, this study evaluates the clinical characteristics of C. psittaci pneumonia, along with the treatment and outcomes for C. psittaci pneumonia patients, which may enhance our understanding of the disease and reducing the incidence of missed and misdiagnosed cases. Notably, lower lymphocyte counts were identified as the sole risk factor associated with severe C. psittaci pneumonia. Respiratory quinolones and doxycycline demonstrate significant efficacy in treating C. psittaci pneumonia. Furthermore, clinicians should consider this disease in cases of repeated high fever, chest CT scans showing high-density shadows with consolidation, or bronchial inflammation, particularly in patients with a history of contact with birds or poultry. Early testing with tNGS may aid in achieving an accurate diagnosis and enhancing treatment outcomes.
Footnotes
Acknowledgments
We thank the patients and their families for their support of this study. We thank the Changsha King Med Center for Clinical Laboratory for their assistance in detection methodology.
Ethical considerations
The study was approved by the Institutional Ethics Board of the Affiliated Changsha Hospital of Xiangya School of Medicine (The First Hospital of Changsha; Grant number: 2024 (06) V. 1.0). The date of ethical approval is 4 June 2024. Written informed consent has been obtained. This study was conducted in accordance with the principles of the Declaration of Helsinki.
Consent to participate
Participant consent to participate in the study.
Consent for publication
The participants consented to publication.
Author contributions
ZH participated in the data sorting, analysis and drafting of the manuscript. YT and XC participated in the data analysis and revised the manuscript. RC, YL, and XH participated in data collection, sorting and analysis. All authors approved the submitted version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Youth Innovation Fund of The First Hospital of Changsha (2025).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
According to institution's policy on patient privacy and confidentiality, we are unable to publicly share patient information. However, the authors will provide the corresponding data upon reasonable request.
