Abstract
Neurofibromas are benign peripheral nerve sheath tumors that occur in individuals with neurofibromatosis type 1. In pediatric patients, giant neurofibromas pose significant management challenges due to their large size and potential to cause functional impairments. Given their characteristic radiation resistance, indolent growth, and limited chemosensitivity, surgical intervention is frequently required to alleviate tumor-related symptoms, restore function, and mitigate complications. The two primary surgical difficulties are the high recurrence rates resulting from the infiltrative nature of giant neurofibromas and the risk of massive intraoperative hemorrhage. This case report details the surgical management of a pediatric giant neurofibroma, highlighting the multidisciplinary strategy and operative techniques employed to achieve optimal results.
Introduction
Neurofibromatosis type 1 (NF1) is an autosomal dominant genetic disorder characterized by neurofibroma development, which may cause severe disfigurement and functional impairment. 1 In pediatric patients, giant neurofibromas can lead to significant morbidity because of their large size and the potential to compress vital structures. Within NF1, nonplexiform and plexiform neurofibromas (PN) constitute clinically distinct entities. Plexiform neurofibromas, however, presents greater management challenges secondary to its infiltrative behavior, risk of neurological compromise, and potential for malignant transformation. 2 Given their characteristic radiation resistance, indolent growth, and limited chemosensitivity, 3 surgical intervention is frequently required to alleviate tumor-related symptoms, restore function, and mitigate complications. 4 However, giant neurofibromas present formidable surgical challenges due to extensive tissue involvement and hypervascularity with friable vessels. Although preoperative embolization, specialized techniques, and hemostatic devices are recommended to reduce intraoperative hemorrhage, the persistent risk of life-threatening bleeding complicates surgical resection. Beyond perioperative bleeding management, postoperative wound repair and reconstruction remains critical. To restore both appearance and function, numerous repair techniques have been developed, including skin grafting, free flap transfer, local flap transfer, and other innovative approaches. 2
Recent extensive research on NF1 management has established multiple therapeutic modalities, including watchful waiting, surgery, medical therapy (e.g. MEK inhibitors), or combined approaches. 5 Given the heterogeneous clinical presentations of NF1, particularly giant neurofibromas, surgical intervention remains an indispensable standard treatment for many patients. This case report details multidisciplinary management of a pediatric giant neurofibroma, focusing on surgical strategies to control intraoperative bleeding and reconstructive techniques for optimal aesthetic outcomes.
Case presentation
The reporting of this study conforms to CARE guidelines. 6 A preadolescent child was admitted to Zibo Central Hospital, Zibo, China, with a rapidly enlarging sacrococcygeal mass in August 2021. Over 9 months, the mass expanded to 25 cm in diameter, causing pain and functional impairment (Figure 1(a)–(d)). Physical examination revealed diffusely distributed subcutaneous neurofibromas predominantly over the trunk and thighs. These lesions, elevated above the skin surface, measured 0.5–1.5 cm in diameter. Additionally, >6 café-au-lait macules (CALMs) (>5 mm diameter) were diffusely distributed. Imaging studies, including MRI, demonstrated a giant PN (Figure 1(e)–(g)). Family history: Mother with NF1 fulfilling NIH diagnostic criteria, complicated by dystrophic scoliosis. Patient history: Cutaneous neurofibromas were first noted at age 3 months over the trunk, sacrum, and thighs, exhibiting progressive enlargement. No prior surgical or medical interventions were documented. The patient fulfilled NIH diagnostic criteria for NF1 by meeting ≥2 features: (1) ≥6 CALMs >5 mm, (2) multiple cutaneous neurofibromas, and (3) first-degree relative with confirmed NF1. 7
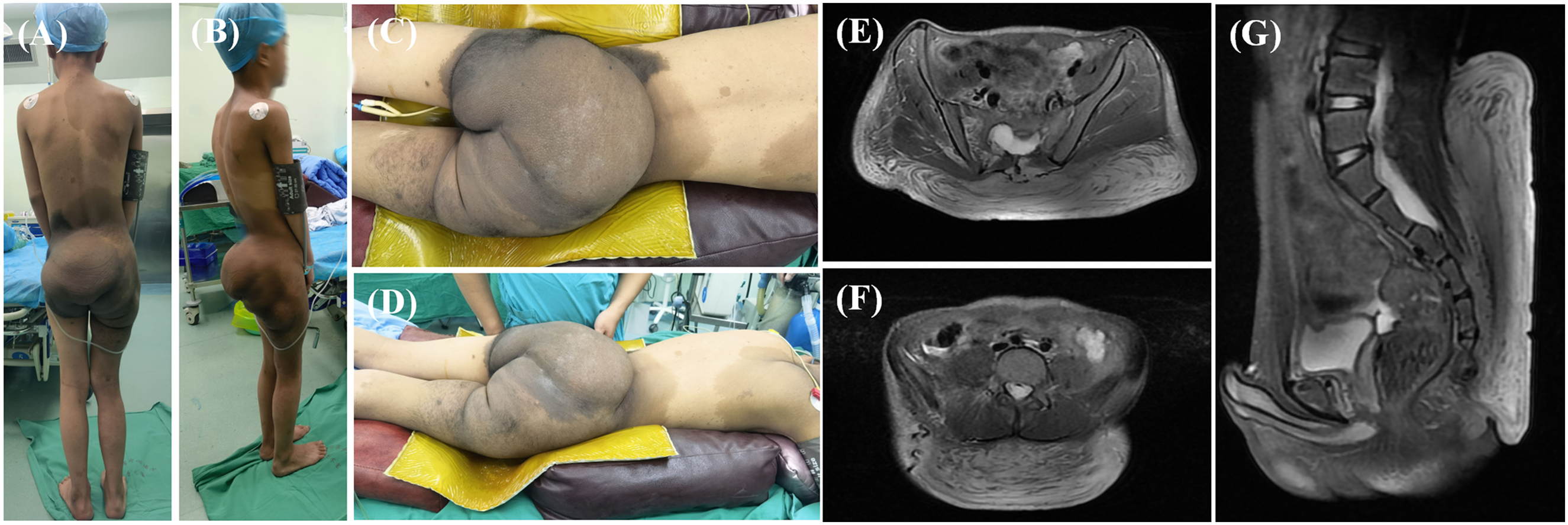
The clinical presentation and preoperative imaging of the patient. (a–d) Preoperative physical examination findings; (e–g) Preoperative MRI studies.
Before the surgery, the patient was referred to a multidisciplinary team (MDT) composed of pediatric surgeons, neurologists, oncologist, and plastic surgeons for evaluation and management consultation. Due to the tumor's giant size (25 cm), accelerated growth rate, and progressive neurological deficits, the MDT consensus indicated surgical resection as the definitive management strategy. Preoperative planning involves detailed imaging to assess the extent of the tumor and its relationship to surrounding structures. The surgery was carried out under general anesthesia with the patient in a prone position. The two primary intraoperative challenges were: (1) controlling massive hemorrhage risk from hypervascular friable vessels, and (2) achieving complete resection despite the infiltrative nature of giant neurofibromas. 2 Firstly, we used the method of making two concentric purse-string sutures 3 cm and 5 cm peripheral to the tumor margin to compress collateral vasculature and reduce intraoperative hemorrhage (Figure 2(a) and (b)). Simultaneously, controlled hypotension (mean arterial pressure maintained at 60–65 mmHg) and tumescent solution injection (epinephrine 1:500,000 in normal saline) were implemented to minimize blood loss through vasoconstriction and reduced perfusion pressure. Subsequently, en bloc resection was performed through a circumferential incision while preserving sacral nerve roots under intraoperative neuromonitoring (Figure 2(c)–(e)). The incision was placed between the two concentric purse-string sutures (4 cm from the tumor margin), enabling dual hemostatic compression: The outer suture compressed perforating vessels in the subcutaneous plexus; The inner suture occluded feeder vessels penetrating the tumor pseudocapsule. Then, the tumor was meticulously dissected from surrounding tissues using blunt dissection combined with bipolar electrocoagulation for hemostasis. Major feeding vessels at the tumor base were identified, doubly ligated with 2-0 silk sutures, and divided between ligatures to ensure secure vascular control. Eventually, the giant neurofibroma was successfully excised from the surrounding tissues, with special attention paid to preserving the function of the adjacent nerves and vessels. Secondly, the extensive wound post-resection could not be closed primarily. We used skin grafting to cover the wound to minimize damage to the patient. We prepared partial-thickness skin grafts from the tumor skin tissue for reimplantation to repair the wound. This approach provided adequate tissue volume for large-area defect coverage, optimized functional limb restoration, and minimized donor-site morbidity. Negative pressure wound therapy (NPWT) was applied to the wound to improve graft take percentages. Negative pressure wound therapy dressings were changed on postoperative day 5 and discontinued after 14 days. The skin graft demonstrated complete take without evidence of necrosis or infection (Figure 2(f)–(h)).

Surgical resection and postoperative follow-up. (a and b) Placement of double concentric purse-string sutures (3 cm and 5 cm from the tumor margin) to control intraoperative hemorrhage; (c–e) Circumferential incision and tumor excision; (f) Postoperative appearance on day 5; (g) Postoperative appearance at 1 month; (h) Postoperative appearance at 2 years (without recurrence).
The patient's postoperative recovery was uncomplicated. A multimodal analgesic protocol effectively controlled pain, and early mobilization was encouraged to prevent complications such as deep vein thrombosis. Multimodal Analgesia Protocol: (1) Provide psychological support to the child preoperatively and postoperatively. (2) Administer continuous intravenous analgesia (IV) via patient-controlled analgesia (PCA) pump for 48 h postoperatively (Dexmedetomidine 0.05 μg·kg−¹·h−¹ + Fentanyl 0.2 μg·kg−¹·h−¹; total solution volume: 100 mL; background infusion: 2 mL/h; bolus dose: 1.5 mL per activation). (3) After discontinuation of the IV PCA pump, administer oral Ibuprofen Suspension (200 mg/dose; maximum daily dose <800 mg) for pain control. Postoperative pathology confirmed a diffuse cutaneous neurofibroma in the sacrococcygeal region, exhibiting focal cellular hyperplasia and myxoid degeneration. Hematoxylin and eosin staining revealed predominantly bland spindle cells with pale eosinophilic cytoplasm and indistinct borders. The background stroma contained abundant mucinous matrix forming amorphous eosinophilic deposits, accompanied by collagenous fibrosis (Figure 3(a)). Immunohistochemistry revealed the following profile: S-100 (+), BCL-2 (+), Vimentin (+), CD99 (+), CD34 (+), GFAP (-), NF (-), CKAE1/E3 (-), SMA (-), desmin (-), CD117 (-), DOG1 (-), ALK (-), Myoglobin (-), MoyD1 (-), EMA (-), and STAT6 (-). The Ki-67 proliferative index was 3% (Figure 3). The patient was discharged on postoperative day 14 with scheduled quarterly follow-up. At 24-month surveillance, clinical demonstrated no local recurrence in the sacrococcygeal region (Figure 2(h)).
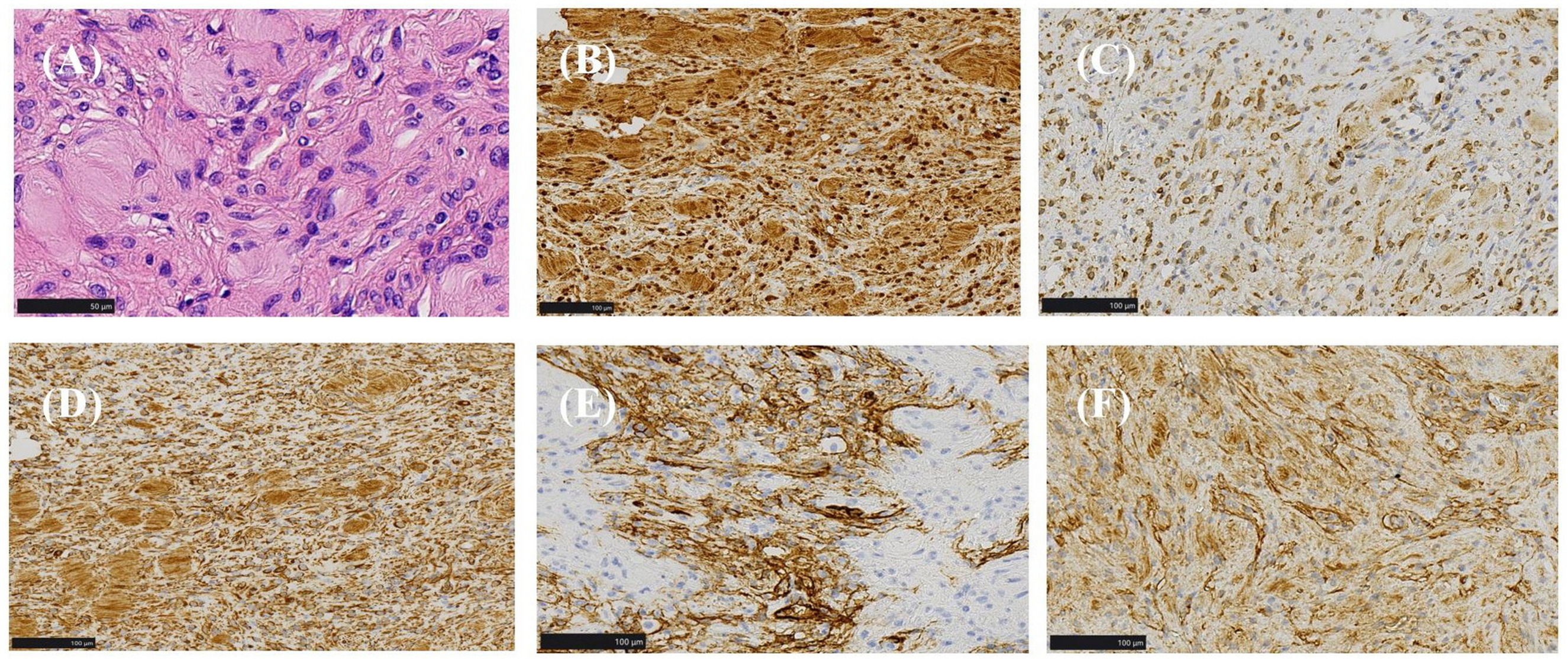
Histopathological findings of the surgical specimen. (a) Hematoxylin and eosin (H&E) staining; (b–f) Immunohistochemical staining: (b) S-100 (+), (c) BCL-2 (+), (d) Vimentin (+), (e) CD34 (+), and (f) CD99 (+).
Discussion
Neurofibromatosis type 1, once known as von Recklinghausen's disease, is a neurocutaneous disorder inherited in an autosomal dominant pattern. Its incidence is approximately 1:3000 live births worldwide. 8 Neurofibromatosis type 1 manifests as two distinct subtypes: nonplexiform and PN. Plexiform neurofibromas demonstrate aggressive growth kinetics, frequently developing large tumor burdens. These lesions exert mass effect on adjacent structures, causing significant functional impairment and compromising quality of life. Tumors exceeding 20 cm in maximal dimension are classified as giant neurofibromas.3,9 Plexiform neurofibromas typically present congenitally. Initial manifestations include hyperpigmented skin macules and subcutaneous tissue hypertrophy along nerve distributions. With progressive growth throughout childhood and adolescence, these tumors often exceed 10 cm in diameter, culminating in clinically significant complications: neuropathic pain, functional deficits, and malignant transformation to malignant peripheral nerve sheath tumor in 8–15% of cases. 10 Surgical intervention for NF1 primarily focuses on the resection of neurofibromas. The guiding principle of surgery is to achieve as complete tumor removal as possible, aiming to minimize recurrence and improve patient quality of life. However, resection of larger tumors often results in significant tissue defects, necessitating repair with grafts from extensive donor sites. Furthermore, the abundance of abnormal vasculature within these tumors poses a substantial risk of life-threatening hemorrhage during surgery. Although selective embolization of tumor-feeding arteries prior to resection has been reported by some researchers to facilitate a more complete excision of giant neurofibromas, this approach is associated with a notably high incidence of postoperative hematoma formation. 11 During preoperative planning, the option of preoperative embolization of the tumor's main feeding vessels via an interventional approach was also considered. However, given the patient's age and the family's preferences, we opted for the double concentric purse-string suture technique instead of selective embolization of the tumor-feeding arteries.
To minimize intraoperative blood loss and achieve maximal tumor resection, we developed a sequential treatment strategy comprising the following steps: (1) Extended tumor resection with hemostasis optimization: Guided by preoperative imaging, the double concentric purse-string suture technique was employed to reduce blood loss. This was combined with intraoperative controlled hypotension and tumescent solution injection to further minimize bleeding. (2) Wound reconstruction using overlying skin grafts: The skin overlying the tumor was harvested and prepared as partial-thickness skin grafts for immediate reimplantation. This approach provided ample tissue coverage for the large defect, maximized functional limb restoration, and minimized secondary donor-site morbidity and scarring. (3) Application of NPWT: Following resection, NPWT was applied over the grafted site to enhance skin graft survival. 12 Negative pressure wound therapy enhances skin graft viability through multiple mechanisms. By creating a negative-pressure environment, it improves local blood flow, thereby augmenting oxygen and nutrient delivery while facilitating waste removal. Negative pressure wound therapy also reduces edema, preventing vascular compression and maintaining adequate perfusion. The therapy secures the graft interface, minimizing shear-related disruptions. Furthermore, NPWT controls infection through continuous exudate and bacterial removal, while simultaneously stimulating the proliferation and migration of wound-healing cells. Collectively, these actions establish an optimal microenvironment for graft survival, improving overall success rates. In this case, postoperative follow-up revealed no instances of wound infection or skin graft necrosis.
Conclusion
In conclusion, the management of giant neurofibromas in pediatric NF1 patients presents significant challenges due to lesion size and potential functional impairment. In this case, a multidisciplinary approach facilitated comprehensive preoperative planning and adaptive intraoperative decision-making. Precise surgical technique enabled complete tumor resection while significantly minimizing complication risks. Postoperatively, early mobilization and a multimodal analgesic strategy contributed to an uncomplicated recovery. Long-term follow-up remains imperative to monitor for tumor recurrence and the development of new lesions.
Footnotes
Acknowledgements
The authors appreciate all attending physicians working in our hospital for providing their full support for this study.
Ethical considerations and consent to participate
The study was approved by the Human Research Ethics Committee of Zibo Central Hospital (Approval No. 2025Y015). Written informed consent for treatment and publication of this case report was obtained from the patient's legal guardian.
Author contributions
The authors confirm their contribution to the paper as follows: study conception and design: Pidong Liu and Yanwei Sun; data collection: Pidong Liu and Yanwei Sun; draft manuscript preparation: Pidong Liu and Yanwei Sun; supervision: Yanwei Sun. All authors reviewed the results and approved the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data were available within the article.
