Abstract
Objective
Ischemic stroke, resulting from the interruption of blood supply to the brain, leads to cerebral ischemia, hypoxia, and necrosis, and is a prevalent disease characterized by high incidence, mortality, and disability rates. This study investigates the protective effects of ischelium on cerebral ischemia-reperfusion injury in a rat model, along with the potential mechanisms of action.
Methods
We established a rat cerebral ischemia-reperfusion model and administered different doses of ischelium as intervention across treatment groups. Multiple techniques were used to evaluate brain tissue damage, oxidative stress markers, inflammatory cytokine levels, and apoptosis, while also examining the expression of key proteins in the Nrf2/HO-1 antioxidant pathway and the HMGB1/TLR4/RAGE/NF-κB pro-inflammatory pathway.
Results
The results indicate that ischemia-induced injury causes severe brain tissue damage and activates both the Nrf2/HO-1 antioxidant pathway and the HMGB1/TLR4/RAGE/NF-κB pro-inflammatory pathway, subsequently leading to oxidative stress, inflammation, and apoptosis. Treatment with ischelium significantly alleviated the brain tissue damage induced by cerebral ischemia-reperfusion, promoted the Nrf2/HO-1 antioxidant pathway, and suppressed the HMGB1/TLR4/RAGE/NF-κB inflammatory pathway. Ischelium also reduced MDA levels and increased SOD, CAT, and GSH levels, while inhibiting the production of pro-inflammatory cytokines IL-6, IL-1β, TNF-α and apoptosis.
Conclusions
These findings suggest that ischelium confers neuroprotective effects against cerebral ischemia-reperfusion injury, likely by regulating oxidative stress and neuroinflammation. Further investigations into the exact molecular mechanisms and clinical translations of ischelium may lead to novel therapeutic approaches for ischemic stroke.
Introduction
Stroke, particularly ischemic stroke, has alarmingly risen to become the second leading cause of death globally. 1 This health crisis is characterized by a high propensity for recurrence and a significant mortality rate. 2 Ischemic stroke primarily results from the formation of blood clots within the cerebral vessels, leading to the necrosis of brain tissue and focal neuronal damage. 3 The escalating incidence and mortality rates of stroke on a worldwide scale present formidable challenges to public health infrastructures, highlighting the critical demand for more effective therapeutic interventions. 4
Ischelium, with its active constituent methanesulfonic acid dihydroergotamine, not only functions as a vasodilator but also possesses noteworthy antioxidant properties. 5 This dual functionality positions Ischelium as a potentially protective agent against the detrimental effects of oxidative stress and neuroinflammation in stroke. 6 The primary functions of this compound include improving neuronal metabolism, inhibiting ATPase and adenylate cyclase activity, reducing ATP degradation, enhancing energy balance in brain cells, and promoting the shift from anaerobic to aerobic glucose metabolism; it simultaneously improves neural transmission, increases cerebral blood flow and oxygen utilization, and ameliorates abnormal electroencephalogram patterns and cerebral/peripheral vascular conditions in elderly patients. 7 Consequently, Ischelium effectively improves vascular cognitive impairment manifested as sensory, emotional, and behavioral changes, as well as declining cognitive abilities and memory function caused by cerebrovascular diseases and/or their risk factors. The Alzheimer's Working Group in the United States has already employed this drug as a cerebral metabolic enhancer in clinical practice, with the U.S. FDA (Food and Drug Administration) evaluating it as an “effective” nootropic medication. 8 Despite these promising applications, comprehensive investigations into the specific roles and mechanisms of Ischelium in mitigating brain ischemia-reperfusion injury remain relatively unexplored and warrant further scientific inquiry.
In light of this knowledge gap, our study endeavors to explore and elucidate the protective effects of Ischelium on brain ischemia-reperfusion injury in a rat model. We employed a multifaceted approach, encompassing behavioral assessments, measurement of cerebral infarction rates, and evaluation of oxidative stress markers, to comprehensively assess the therapeutic potential of ischelium. By delving into these aspects, we aim to contribute to the development of innovative and effective strategies for the management and treatment of ischemic stroke. This research is driven by the overarching goal of providing new avenues to alleviate the profound and often devastating consequences of stroke, thereby improving patient outcomes and quality of life.
Materials and methods
Experimental animals and grouping
30 male SD rats weighing 260–300 g were purchased from Shanghai Slac Laboratory Animal Co. Ltd (license number: SCXK (Hu) 2022–0004). They were housed in the SPF barrier environment of the Zhejiang University Laboratory Animal Center (license number SYXK (Zhe) 2022–0037). The rats had free access to food and water, acclimatized for 7 days in an environment with a temperature of 20–24 °C, humidity of 40–70%, and a 12 h light/12 h dark cycle. The air change rate was 10∼20 times/hour. The rats were randomly assigned to five groups: a sham group (sham), a model group (operation), and three ischelium-treated groups. Each group consisted of six rats.
Anesthesia was induced by intraperitoneal injection of sodium pentobarbital (35 mg/kg). Following anesthesia, the neck vessels were exposed, and a nylon filament was inserted from the carotid bifurcation into the internal carotid artery (ICA), blocking the origin of the middle cerebral artery (MCA) and all its branches, thus creating focal ischemia in the MCA territory. The sham and operation group received PBS, while the treatment groups received high-dose (5 mg/kg), medium-dose (1 mg/kg), or low-dose (0.2 mg/kg) ischelium for 7 consecutive days. At the end of the experiment, animals were deeply anesthetized with 3% sodium pentobarbital (50 mg/kg, intraperitoneal) and then euthanized by intravenous injection of excess 10% chloral hydrate (300 mg/kg). Operators confirmed death by verifying the absence of spontaneous respiration, heartbeat, pupillary light reflex, and corneal reflex. The entire procedure was performed in a dedicated laboratory by trained personnel to ensure animal welfare and minimal suffering. This study complies with the ARRIVE 2.0 guidelines and the Guide for the Care and Use of Laboratory Animals.9,10
Ischelium, batch number 2210008, was obtained from Polifarma (Nanjing) Co., Ltd, China.This compound comprises Dihydroergocornine Mesylate at 33.3%, Dihydro-α, β-ergocryptine Mesylate at 33.2%, and Dihydroergocristine Mesylate at 33.5%. The ratio between Dihydro-α-ergocryptine Mesylate and Dihydro-β-ergocryptine Mesylate stands at 2:1. The purity of ischelium is an impressive 99.63%.
Neurological scoring
After treatment, the rats were scored according to the Longa grading method. A score of 0 indicated no neurological injury; 1 point indicated mild neurological injury with the contralateral forelimb flexing when lifting the tail; 2 points indicated moderate neurological injury with circling towards the contralateral side when crawling; 3 points indicated severe neurological injury with falling towards the contralateral side when standing or crawling; 4 points indicated no spontaneous activity accompanied by consciousness disorders.
2,3,5-Triphenyltetrazolium chloride (TTC) staining
After treatment, 6 rats from each group were anesthetized, decapitated, their brains removed and quickly frozen. Brain slices were then incubated in 0.5% TTC solution at 37°C for 20 min in the dark to ensure even staining. After staining, the slices were fixed in 4% paraformaldehyde and photographed using the built-in camera of an iPhone 15. Images were then analyzed to calculate the cerebral infarction rate, which was determined as (cerebral infarction volume / total brain volume) × 100%.
Blood-brain barrier permeability assessment
To evaluate the effect of cerebral ischemia-reperfusion injury on blood-brain barrier permeability, 2% Evans Blue dye (4 ml/kg) was administered via tail vein injection 2 h before the end of the experiment. After injection, animals were maintained under standard conditions for an additional 2 h. Subsequently, rats were deeply anesthetized and perfused with saline through the left ventricle until the effluent was clear. The intact brain tissue was quickly removed, gently wiped with wet gauze, and the brainstem and cerebellum were discarded. The remaining brain tissue was weighed and dried to a constant weight in a 37°C oven. The dried brain tissue was homogenized in formamide (1 ml/100 mg) and incubated at 60°C for 24 h to extract the Evans Blue dye. The samples were then centrifuged at 15,000 g for 10 min, and the supernatant was collected for absorbance measurement at 620 nm using a spectrophotometer. Dye content was calculated using a standard curve, and results were expressed as g/g dry weight. Evans Blue fluorescence leakage was photographed using an iPhone 15 camera.
Blood index testing
After treatment, blood indices were tested according to the instructions. Complete blood count, coagulation tests, whole blood viscosity, erythrocyte aggregation index (EAI), and hematocrit (Hct) were measured using a blood biochemistry analyzer and hemorheological analyzer (Roche). All measurements were performed following the manufacturer's protocols to ensure accuracy and reliability of the results.
TUNEL staining for apoptosis in rat brain tissue
In this study, TUNEL (Terminal deoxynucleotidyl transferase dUTP Nick End Labeling) assay was utilized to detect apoptosis in tissue sections. We employed a commercial TUNEL kit, which included all necessary reagents. Tissue sections were first prepared by deparaffinization and rehydration, followed by permeabilization with proteinase K. The TUNEL reaction was carried out according to the kit's protocol, with sections incubated in a labeling solution containing terminal deoxynucleotidyl transferase and fluorescein-dUTP. After the reaction, sections were examined under a fluorescence microscope. Apoptotic cells were identified by their red fluorescent signal. The TUNEL kit is made by Beyotime Company, product number is C1089.
Immunofluorescence detection
Brain tissue was paraffin-embedded, sectioned, dewaxed and rehydrated, followed by antigen retrieval in EDTA buffer (pH 8.0). After PBS washing, endogenous peroxidase activity was blocked using 3% hydrogen peroxide for 25 min at room temperature. Sections were circled with a hydrophobic pen, blocked with 3% BSA, and incubated with anti-NF-κB primary antibody (Amyjet Scientific, PSI-60–327, 1:200 dilution) overnight at 4°C. The next day, HRP-conjugated goat anti-rabbit secondary antibody (haokebio, HKI0029, 1:500 dilution) was applied for 50 min, followed by Flare570 signal amplification to label NF-κB. Nuclei were counterstained with DAPI for 8 min. After mounting with anti-fade medium, sections were examined and imaged using a Nikon (ECLIPSE C1) inverted fluorescence microscope.
Enzyme activity measurement in rat brain tissue
At the end of the experiment, rat brain tissues from each group were collected, homogenized, and centrifuged to obtain the supernatant. Levels of superoxide dismutase (SOD), catalase (CAT), glutathione (GSH), and malondialdehyde (MDA) were measured strictly according to the kit instructions. All enzyme activity assay kits were purchased from Nanjing Jiancheng Bioengineering Institute, with catalog numbers: SOD (A001–1–1), CAT (A007-1-1), GSH (A006-2-1), and MDA (A003-2-1).
ELISA for inflammatory cytokines
Brain tissues were homogenized in RIPA lysis buffer, ultrasonicated for complete tissue disruption, and centrifuged at high speed. The supernatant was carefully collected and used for the determination of inflammatory cytokines (TNF-α, IL-1β, IL-6) and oxidative stress-related proteins (Nrf2 and HO-1) using enzyme-linked immunosorbent assay (ELISA) kits. All procedures were performed strictly according to the manufacturer's instructions. The ELISA kits were purchased from Shanghai Xuanya Biotechnology with the following catalog numbers: IL-6 (XYR1876A), IL-1β (XYR1618A), TNF-α (XYR1272A), Nrf2 (XYR2292A), and HO-1 (XYR3453A). Protein concentrations were determined using a BCA protein assay kit (Beyotime, P0011) prior to ELISA analysis to ensure equal loading. Absorbance was measured using a microplate reader, and the final concentrations were calculated based on standard curves.
qRT-PCR for gene expression in rat brain tissue
Total RNA from rat brain tissue was extracted using the Trizol method and reverse transcribed into cDNA. The amplification was performed using the SYBR Green (TAKARA, RR820A) method with β-actin as an internal control. The reaction conditions included an initial denaturation at 95 °C for 30 s, followed by 40 cycles of 95 °C for 5 s, and 60 °C for 30 s. The relative expression levels of the target genes were calculated using the 2−ΔΔCT formula. The PCR primer sequences are listed in Table 1.
Details of the primer sequence used for qPCR.

Statistical methods
Data were analyzed using SPSS 18.0 software. Quantitative data were expressed as mean ± SD. Normally distributed data were analyzed using one-way ANOVA, and comparisons between two groups were conducted using the t-test.
Results
Protective effect of ischelium on the brain
The protective effect of ischelium was first evaluated using neurological scoring and TTC staining. Compared with the sham group, the infarct area in the operation group reached 24.6% of the total brain volume, indicating successful replication of the cerebral ischemia-reperfusion injury model. The neurological function score in the Ischelium group at 5 mg/kg was reduced by 1.7 points compared to the operation group, and the cerebral infarction rate decreased to 5.8% (Figure 1A-C).

Neuroprotective effects of ischelium on rats with cerebral ischemia-reperfusion. (A) Representative images of TTC staining showing infarct areas (white) in brain sections from sham (normal control rats), operation (cerebral ischemia-reperfusion injury), and ischelium-treated groups (0.2 mg/kg, 1 mg/kg and 5 mg/kg). (B) Quantification of brain infarction rate expressed as percentage of total brain volume (Mean ± SEM, n = 6). (C) Neurological deficit scores assessed using a 5-point scale (0-4) where higher scores indicate more severe functional impairment (Mean ± SEM, n = 6). *P < 0.05, **P < 0.01 compared with the operation group.
Effect of ischelium on blood indices in ischemic rats
To further clarify the mechanism by which ischelium regulates cerebral ischemia through blood-related indices, we examined its effect on complete blood count and hemorheology in experimental animals. As shown in Figure 2, there were no significant changes in white blood cells, red blood cells, and platelets among the sham, operation, and Ischelium groups. Additionally, no significant differences in hemorheological parameters were observed between the groups.
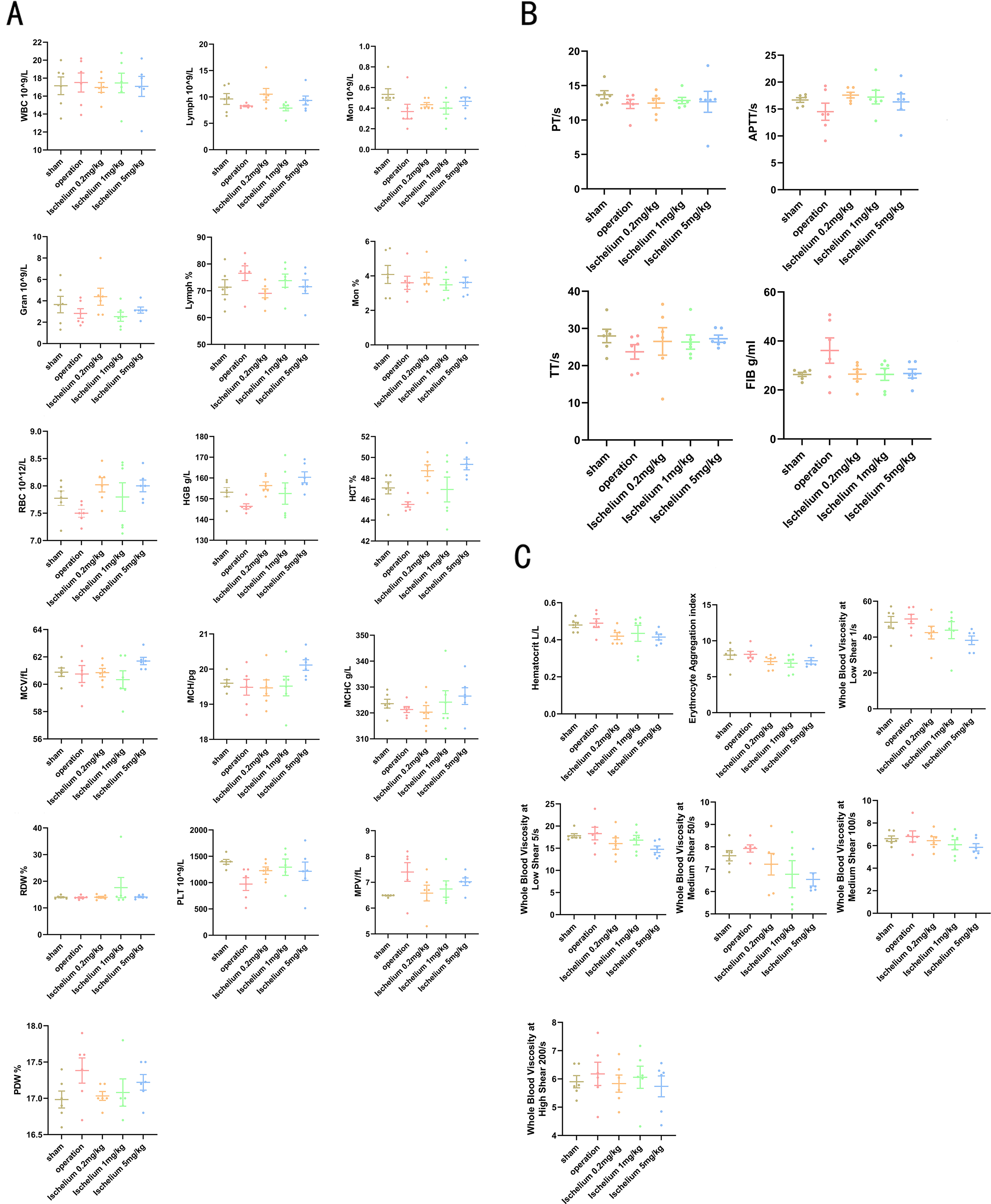
Examination of blood-related parameters. (A) Routine blood test. (B) Coagulation profile testing. (C) Hemorheology analysis. WBC: white blood cells; Gran: neutrophilicgranulocyte; RBC: red blood cells; MCV: Mean Corpuscular Volume; RDW: Red Cell Distribution Width; PDW: Platelet distribution width; LYMPH: Lymphocyte; HGB: Hemoglobin; MCH: Mean Corpuscular Hemoglobin; PLT: Platelet; Mon: Monocyte; HCT: hematocrit; MCHC: mean corpusular hemoglobin concentration; MPV: Mean platelet volume; PT: Prothrombin time; TT: Thrombin Time; APTT: Activated partial thromboplastin time; FIB: Fibrinogen.
Evans blue
The Evans Blue experiment results indicate that Ischelium has an effect on restoring the blood-brain barrier. As shown in Figure 3, the operation group exhibited the most severe disruption of the blood-brain barrier, demonstrated by extensive Evans Blue extravasation into brain tissue. This increased permeability reflects the substantial damage to tight junctions between endothelial cells caused by ischemia-reperfusion injury. In contrast, treatment with Ischelium (5 mg/kg) markedly reduced Evans Blue leakage, suggesting its protective effect on blood-brain barrier function.

Evans blue extravasation assay for blood-brain barrier integrity assessment. (A) Representative images of Evans Blue dye distribution in brain sections from sham (normal control rats), operation (cerebral ischemia-reperfusion injury), and ischelium treatment groups. (B) Quantification of Evans Blue dye content (g/g tissue) in brain tissue as a measure of blood-brain barrier permeability. Data presented as Mean ± SEM (n = 6). *P < 0.05, **P < 0.01 compared with the Model group.
Enzyme activity and inflammatory cytokine levels in rats
After the occurrence of brain ischemia-reperfusion, a large amount of MDA was produced in the brain tissue of the operation group, while the levels of SOD, GSH, and CAT decreased, leading to peroxidation reactions. After treatment with Ischelium, MDA levels decreased, and the levels of SOD, GSH, and CAT increased. Simultaneously, the levels of IL-6, IL-1β, and TNF-α significantly increased in the model group, while the ischelium group showed reduced levels of inflammatory cytokines (Figure 4A-B). Additionally, Ischelium at 5 mg/kg significantly increased the expression of Nrf2 and HO-1 proteins compared to the operation group (Figure 5).
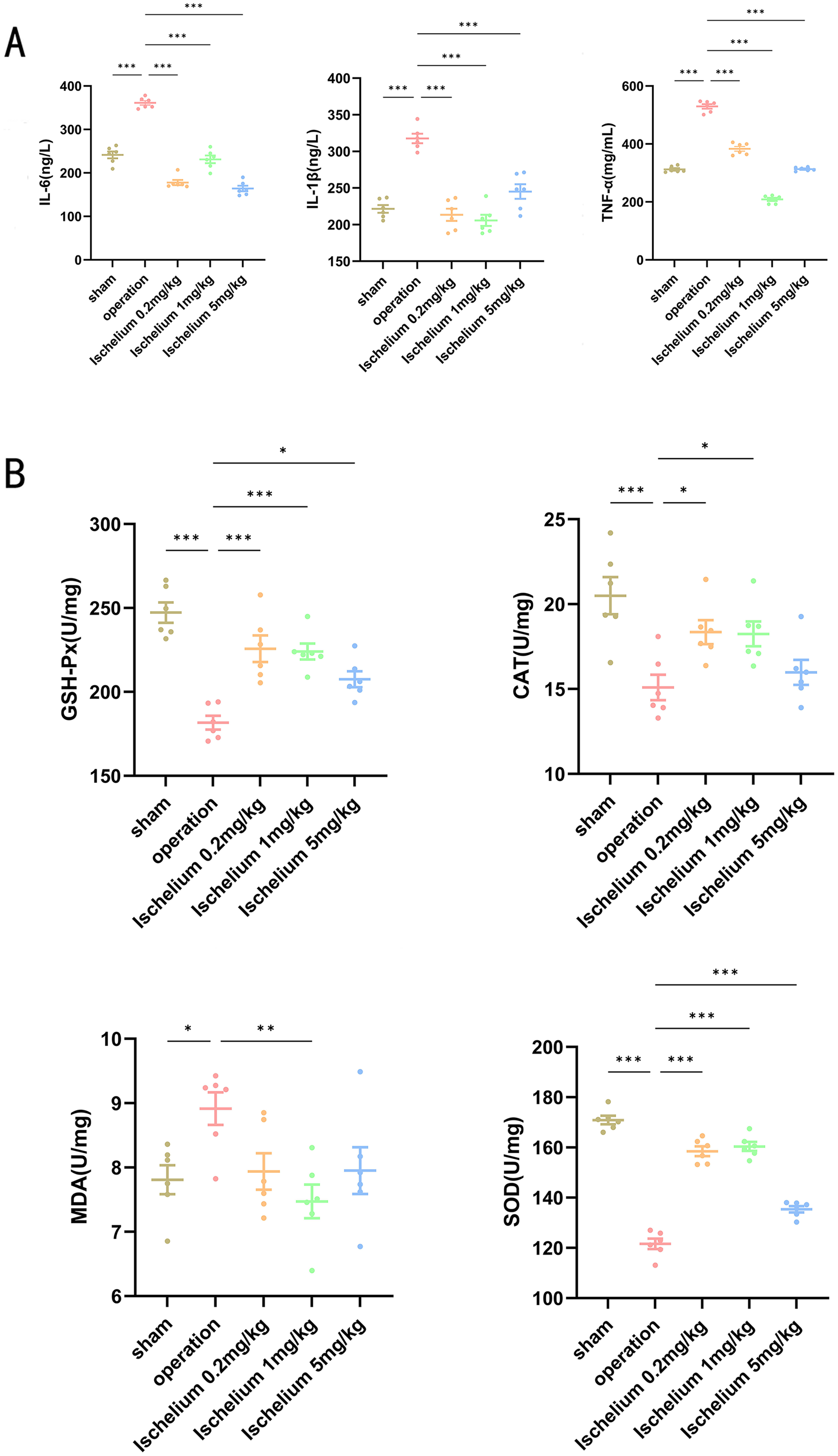
Inflammation and oxidative stress assessment in rat brain tissue. (A) Inflammatory factor analysis showing levels of IL-6, IL-1β, and TNF-α in brain tissue from sham (normal control rats), operation, and ischelium-treated groups. (B) Enzyme activity assay displaying MDA (malondialdehyde), SOD (superoxide dismutase), GSH (glutathione), and CAT (catalase) levels across all experimental groups. Results are presented as Mean ± SEM (n = 6). Comparisons between groups showed statistical significance at *P < 0.05, **P < 0.01, and ***P < 0.001.

Expression of Nrf2/HO-1 protein content in brain tissue. The expression levels of Nrf2 and HO-1 proteins in brain tissue of rats from each group were detected by ELISA method. Data are presented as Mean ± SEM (n = 6). *P < 0.05, **P < 0.01 compared with the Model group.
Comparison of cell apoptosis in different groups
Compared with the sham group, the rate of brain tissue apoptosis in the model group increased significantly. In contrast, the ischelium-treated groups showed a marked decrease in apoptosis rates, especially noticeable in the high-dose ischelium group (Figure 6).

TUNEL staining for apoptosis detection in rat brain tissue. Representative fluorescence microscopy images of TUNEL staining in brain sections from sham (normal control rats), operation (cerebral ischemia-reperfusion injury), and ischelium treatment groups. TUNEL-positive cells (red) indicate apoptotic cells. Quantification of apoptotic cells is shown as percentage of TUNEL-positive cells relative to total cell count. Data presented as Mean ± SEM (n = 6). Scale bar = 50 μm. *P < 0.05, **P < 0.01 compared with the Model group.
Activation of brain tissue pathways in rats
In exploring the mechanisms of cerebral ischemia-reperfusion (CIR) injury, the roles of the Nrf2/HO-1 pathway and the HMGB1/TLR4/RAGE/NFкB pathway are particularly significant. As shown in Figure 7, the gene expression of the HMGB1/TLR4/RAGE/NFкB pathway in the ischelium group was significantly lower compared to the operation group. Immunofluorescence and immunohistochemistry results in Figure 8 show a reduction in NF-κB protein expression.

qPCR analysis of inflammatory pathway genes in rat brain tissue. Relative mRNA expression levels of HMGB1, TLR4, RAGE, and NF-κB in brain tissue from sham (normal control rats), operation, and ischelium treatment groups. Gene expression was normalized to GAPDH and presented as fold change relative to the sham group. Results are presented as Mean ± SEM (n = 6). Comparisons between groups showed statistical significance at *P < 0.05, **P < 0.01, and ***P < 0.001.

Immunofluorescence staining of NF-κB in rat brain tissue. Representative confocal microscopy images showing NF-κB expression (red, Flare570 fluorescence) and cell nuclei (blue, DAPI) in brain sections from sham (normal control rats), operation(cerebral ischemia-reperfusion injury), and ischelium-treated groups. The merged images demonstrate the colocalization pattern and nuclear translocation of NF-κB. Data shown as Mean ± SEM (n = 6). Scale bar = 50 μm. *P < 0.05, **P < 0.01 compared with the Model group.
Discussion
This study aimed to explore the potential effectiveness of ischelium in protecting against cerebral ischemia-reperfusion (CIR) induced brain injury in rats. The study began by inducing CIR in experimental animals and administering different doses of ischelium. The protective effects of ischelium against CIR were assessed through various indicators, including TTC staining, which confirmed brain tissue damage post-CIR surgery, consistent with previous reports. 11 Treatment with ischelium improved neurological scores and reduced infarct size in rats, demonstrating its protective effect on the brain.5,12
In investigating the mechanisms of ischelium's protective effects against CIR, several potential biological pathways were examined. The study indicated that patients with acute cerebral infarction exhibit abnormally high blood viscosity, characterized by a thick, sticky, coagulated, and aggregated state.13,14 Blood rheology indicators in patients with acute cerebral infarction were significantly higher than those in the sham group, particularly manifesting as high-viscosity blood syndrome and hypercoagulability.15,16 This state of high blood viscosity and coagulation leads to the continuous deterioration of local microcirculation, inducing neuronal apoptosis - a crucial mechanism in infarct expansion and delayed neuronal injury, and a risk factor for recurrent cerebral death. 17 Therefore, the treatment of acute cerebral infarction should not only aim to restore blood flow in occluded vessels and protect neurological functions but also address high blood viscosity to prevent the expansion of infarction and neuronal injury, as well as the recurrence of cerebral infarction.18,19 This study examined blood routine indicators in rats, finding that ischelium did not directly affect platelet count. Consequently, further examination of ischelium's impact on blood rheology and coagulation function in rats was conducted. The results showed that, compared to the model group, Ischelium had no significant effect on prothrombin time, thrombin time, fibrinogen content, whole blood viscosity, erythrocyte aggregation index, and hematocrit.
During the process of cerebral ischemia-reperfusion, a large number of oxygen free radicals and peroxides, such as MDA and peroxynitrite, are produced.20–23 MDA, a major peroxide, reflects the level of lipid peroxidation and indirectly indicates the extent of cell damage.24,25 The Keap1/Nrf2/HO-1/NQO-1 pathway can resist oxidative stress.26,27 Under normal conditions, Keap1 (Kelch-like ECH-associated protein 1) inhibits the activity of Nrf2 (nuclear factor erythroid 2-related factor 2), leading to the degradation of Nrf2 in the cytoplasm28–30 . Under oxidative stress, Nrf2 dissociates from Keap1, enters the nucleus, and activates the transcription of antioxidant response genes, including HO-1 (heme oxygenase-1) and NQO1 (NAD(P)H:quinone oxidoreductase 1). 31 HO-1 and NQO1, as antioxidant enzymes, can clear excess reactive oxygen species (ROS), reducing the damage caused by oxidative stress. 32 This study found that ischelium activates this pathway, increasing the levels of SOD, CAT, and GSH, and reducing MDA levels, thereby demonstrating strong antioxidant capacity.
Increasing evidence suggests that the HMGB1/TLR4/RAGE/NF-κB pathway is a key signaling pathway in ischemic injury.33,34 HMGB1 (high mobility group box 1) is a nuclear protein that, when released extracellularly, can bind to TLR4 (Toll-like receptor 4) and RAGE (receptor for advanced glycation end-products), activating the NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) signaling pathway.35–37 The activation of NF-κB leads to the upregulation of inflammatory cytokines (such as IL-1β, IL-6, TNF-α), thereby promoting the inflammatory response. 38 In this study, animals under cerebral ischemia-reperfusion (CIR) conditions showed significantly elevated levels of these inflammatory factors, accompanied by a marked increase in cerebral infarct area. Our experimental results demonstrated that Ischelium treatment significantly reduced the expression levels of pro-inflammatory cytokines IL-1β, IL-6, and TNF-α, as confirmed by ELISA. Additionally, Ischelium improved the activity of key enzymes related to oxidative stress and inhibited the activation of the NF-κB signaling pathway. Through TTC staining and image analysis, we observed that the cerebral infarct area in the Ischelium treatment group was notably smaller than that in the sham group, showing a good correlation with the reduction in inflammatory cytokine expression. This dose-dependent protective effect suggests that Ischelium may alleviate brain tissue damage and reduce infarct area by inhibiting the HMGB1/TLR4/RAGE/NF-κB inflammatory pathway and decreasing the release of pro-inflammatory factors. These results collectively demonstrate the neuroprotective effect of Ischelium in cerebral ischemia-reperfusion injury, providing scientific evidence for its clinical application.
The experimental results did not demonstrate a dose-dependent effect of ischelium, which may be related to insufficient sample size. Smaller sample sizes increase random fluctuations and reduce statistical power. Additionally, the dose design might not have been optimized, failing to cover the complete dose-response range. Notably, the absence of a clear dose-dependent effect does not necessarily indicate that the drug is ineffective. Future research should consider increasing sample size, optimizing dose design, employing more sensitive biomarkers, and using appropriate statistical models to more comprehensively evaluate the pharmacological properties of ischelium.
Through the modulation of these two major pathways, Ischelium demonstrated its protective effect on brain tissue in the CIR model. It not only reduced oxidative stress but also alleviated inflammation induced by CIR, potentially through the regulation of multiple biological pathways, including but not limited to the Nrf2/HO-1 and HMGB1/TLR4/RAGE/NF-κB pathways. These actions suggest that Ischelium could be an effective treatment for brain injury caused by CIR.
Conclusion
This study demonstrated the potential efficacy of ischelium in alleviating brain injury induced by cerebral ischemia-reperfusion in rats. By activating the Nrf2/HO-1 antioxidant pathway and mitigating the HMGB1/TLR4/RAGE/NF-κB inflammatory pathway, ischelium significantly protected brain tissue from ischemic damage. Furthermore, ischelium improved oxidative stress status and alleviated inflammation and apoptosis by reducing whole blood viscosity and improving coagulation function. These findings contribute new insights into the pharmacological actions of ischelium, highlighting its potential as a protective agent in cerebral ischemia/reperfusion and related conditions.
Footnotes
Ethical considerations
The experiment was approved by the Zhejiang University Laboratory Animal Ethics Committee (Ethics Committee of Zhejiang University, Hangzhou, China; Approval Number: ZIU20230263; Application Number: 26455; Date of Approval: July 05, 2023).
Author contributions
Yanhong Chen: Conceptualization, Writing - original draft, Writing - review & editing; Wei Cheng: Methodology, Visualization, Investigation, Writing - original draft; Boneng Xiao: Software, Writing - original draft; Jia Tang: Validation, Formal Analysis, Writing - original draft; Ming Kuang: Funding Acquisition, Writing - review & editing; Liaoyi Xu: Resources, Writing - review & editing; Xiaosa Xu: Data Curation, Writing - original draft; Hongying Liu: Project Administration, Writing - review & editing, Supervision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The experiment was approved by the Zhejiang University Laboratory Animal Ethics Committee (approval number: ZJU20230263), (grant number ZJU20230263). This research project was supported by Hangzhou Kang Ming Information Technology Co., Ltd and grants from the National Natural Science Foundation of China (No. 32120103007).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data produced or examined in the course of this study are present within this published article. Datasets utilized and/or analyzed during the current study can be provided by the corresponding author upon reasonable request.
