Abstract
This study tested the protective effect of Rumex nervous (R. nervosus) methanol extract against streptozotocin (STZ)-mediated type 1 diabetes mellitus (T1DM)-induced nephropathy in rats and examined if this protection involves activating the nuclear factor erythroid 2 related factor-2 (Nrf2). Rats were divided into control, R. nervous (300 mg), STZ (T1DM), STZ + R. nervosus (100, 200, or 300 mg/kg), and STZ + R. nervosus (300 mg/kg) + brusatol (an Nrf2 inhibitor). With no effect on fasting glucose and insulin levels, R. nervosus methanol extract preserved kidney histological structure and alterations kidney function markers (e.g. albumin, creatinine, and urine volume) in the STZ-diabetic rats. R. nervosus also reduced levels of reactive oxygen species (ROS), malondialdehyde (MDA), tumor necrosis factor-α (TNF-α), and interleukine-6 (IL-6), nuclear levels of the nuclear factor kappa beta (NF-κB), and mRNA of caspase-3 and Bax in the kidneys of these diabetic rats. Concomitantly, it stimulated renal mRNA levels of Bcl2 and Nrf2, cytoplasmic and nuclear levels of Nrf2, and levels of glutathione (GSH), catalase (CAT), and superoxide dismutase (SOD). All these effects were dose-dependent, with the maximum effect seen with the 300 mg/kg dose, all prevented by brusatol. Also, these effects occurred without any alteration in the transcription of the Kelch-like ECH-associated protein 1 (keap-1). Similar effects on levels of GSH, SOD, CAT, and NF-κB, as well as expression of Nrf2, were also observed in the kidney of control + R. nervous-treated rats. In conclusion, R. nervosus prevents diabetic nephropathy in rats by upregulating and activating Nrf2.
Introduction
Type 1 diabetes mellitus (T1DM) is a major threat to the health and economic systems due to the hyperglycemia-associated dreadful repercussions and multi-organ damage. 1 Diabetic nephropathy (DN) remains the early major event associated with T1DM and is a leading cause of increased morbidity and mortality among affected subjects by promoting end-stage renal disease (up to 40%) and other cardiovascular disorders. 2 Yet, oxidative stress, inflammation, fibrosis, and apoptosis are major mechanisms mediating DN.3–6 Among all, it is currently well-established that overproduction of reactive oxygen species (ROS) in response to hyperglycemia is the leading upstream mechanism that promotes all the other damaging pathways in the kidneys of diabetic animals.3,7,8 In support, antioxidant therapy alone was sufficient to prevent DN in experimental animals, with promising data to be depicted in some clinical trials.6,8
The nuclear factor erythroid 2 related factor-2/Kelch-like ECH-associated protein 1 (Nrf2/ keap1) Axis is a unique cellular system that stimulates cell survival by suppressing oxidative stress, inflammation, fibrosis, and apoptosis.9,10 In the cytoplasm and under normal physiological conditions, keap-1 binds Nrf2, causing its proteasome degradation. 11 However, under oxidative stress, modification of the critical cysteine residues of keap-1 by ROS and electrophiles inhibits the activity of the Keap1-Cul3-Rbx1 E3 ubiquitin ligase complex, therefore weakening the biding of keap1 with Nrf2. 11 This leads to the nuclear translocation of Nrf2, dimerization with musculoaponeurotic fibrosarcoma proteins, and then binding to the antioxidant response element (ARE) to initiate the transcription process. Accordingly, Nrf2 mainly stimulates the transcription of several antioxidant genes such as the glutamate-cysteine ligase, glutathione s-transferase, catalase (CAT), superoxide dismutase (SOD), heme-oxygenase-1 (HO-1), and NAD(P)H: quinone oxidoreductase 1. 12 Besides, Nrf2 inhibits cell inflammation, fibrosis, and mitochondria-mediate (intrinsic) cell apoptosis by suppressing the transcription/levels of nuclear factor kappa beta (NF-κB), NRLP3 inflammasome, and transforming growth factor-β1, as well as upregulating the Bcl2 anti-apoptotic protein.10,13–15
Of interest, recent advances in the field have indicated that low expression/activation of Nrf2 is considered a major leading mechanism that mediates DN.9,13 In this context, we have, and others have previously shown a reduction in the transcription Nrf2 with a concomitant increase in levels of keap1 and degradation of Nrf2 in the kidney diabetic animal.9,16–20 However, in all these studies, the activation of Nrf2 by modulating the transcription and expression of Nrf2/keap-1 signaling pathways was an effective strategy to prevent renal damage and dysfunction in these animals by suppressing oxidative stress, inflammation, fibrosis, and apoptosis, suggesting that Nrf2 is a novel target to prevent or alleviate symptoms and kidney function during the development and progression of DN. 16
Several novel phytochemical active compounds with antioxidant and anti-inflammatory powers have been isolated from plants and documented to fight DN. 21 The Rumex plants are known to be the big family of Polygonaceae that contains more than 250 species. 22 Roots, leaves, and stems of the different Remex family members are currently used in traditional medicine to treat a variety of disorders, including hypertension, DM, inflammatory disorders, constipation, wounds, diarrhea, and are characterized by their analgesic, anti-microbial, anti-parasitic, antioxidant, anti-inflammatory, and diuretics potentials.22,23 Rumex nervosus (R. nervosus) is the most common species found in the Arabian and African countries, where the leaves are usually consumed for their medicinal values.24,25 In Africa, extracts obtained from R. nervosus leaves are used to treat burns, skin rashes, gastritis, snake bites, and cancer. 26 In Saudi Arabia and Yemen, the leaves of R. nervosus are used to fasten the healing of new burns and injuries, as well as treat inflammatory and skin disorders.25,27,28 Various extracts (i.e. water, ethanol, and methanol) of R. nervosus were demonstrated to exert anti-obesity, anti-diabetic, hypolipidemic, anti-inflammatory, and radical scavenging activities.29,30 The chronic treatment with the leaf powder of R. nervosus Vahl suppressed hyperglycemia, hyperlipidemia, and weight gain in rats fed a high-fat diet (HFD). 31 In vitro studies have shown the potential of this plant species’ leaves and stem extracts to chelate iron and scavenge free radicals.29,30 In addition, the anti-inflammatory potential of the flower extract of R. nervosus was demonstrated in vitro by suppressing NF-κB, IL-1, nitric oxide synthase (NOS), and cyclooxygenase-2. 29
Despite these findings, the systemic protective effects of R. nervosus are not well characterized in animal models. Besides, the mechanisms of protection are largely unknown. Therefore, in this study, we tested the nephroprotective effects of leaves of R. nervosus against streptozotocin (STZ)-induced DN in rats and investigated whether this protection involves an independent activation of Nrf2 signaling.
Materials and methods
Animals
Adult male and female Wistar rats (220 ± 15 g) and aged 9 weeks old were obtained from and maintained in the animal house at King Saud University, Riyadh, KSA. During the whole period of the study, the animals were housed under ambient conditions of 21 ± 1°C, the humidity of 61%, and the 12 h dark-light cycle and had free access to their normal show (Cat D12450B, 3.85 Kca/g 20%, 10%, and 70% protein, fat, Research Diets, Inc) and drinking water. All procedures, protocols, treatments, sampling, and euthanasia were approved by the Official Review Board at Princess Nourah University, Riyadh, KSA (IRB Number 20-0478), Riyadh, KSA.
Plant collection and identification
The fresh leaves of R. nervosus were harvested from the south of Saudi Arabia during the cultivation phase. The plant was authenticated by the herbarium of the Pharmacognosy Department, College of Pharmacy, King Saud University (KSU), Riyadh, Saudi Arabia. The plant specimen was retained in the herbarium with voucher specimen No (15374).
Plant methanol extraction preparation
The extraction procedure was conducted according to Omodanisi et al. 32 The green leaves were washed with tap water and then air-dried in the dark. They were then blended to powder and extracted (1 kg) via continuous stirring for 24 h in n-hexane. The resulted residue was then re-extracted in 80% (v/v) methanol at 23°C for another 24 h. The solvent was evaporated using a rotatory evaporator and stored at −4°C until use. The dried material was reconstructed in distilled water during the experiment and used for treatment.
Acute toxicity study
An acute oral toxicity study was performed as recommended by testing the acute toxicity of some plant extracts up to 5000 mg/kg.33,34 In brief, three groups of rats were assigned as 1 control received distilled water as a vehicle and 2 treated rated with the extract of R. nervosus at doses of 2000 and 3000 mg/kg, respectively, for 14 days. Survival rate, food, and water intake, gross behavioral and neurological abnormality, and liver and kidney function test, as well as the pathological examination of the liver, kidney, heart, and brain tissues, were determined during this proposed period.
Induction of T1DM
T1DM was induced in the selected groups of rats as previously described by Wu et al. 35 In brief, STZ (Cat 142155, Sigma Aldrich, MO, USA) was dissolved in freshly prepared 0.5 M citrate buffer and administered intraperitoneally (i.p) to each rat at a final dose of 65 mg/kg. After 3 days’ blood glucose was measured in all rats using a glucometer, and any rat with a fasted blood glucose of more than 300 mg/dl was considered diabetic 36 and included in the further experiments 7 days later. In addition, T1DM was confirmed by the histological destruction of the beta-cells.
Experimental design
This protocol was conducted based on the acute toxicity test results, which have shown no toxicity of the R. nervosus methanolic extract up to 3000 mg/kg. Rats were divided into 7 groups as follows: (1) control: non-diabetic rats which were orally administered distilled water as a vehicle, (2) control + R. nervosus (300 mg/kg): non-diabetic rats which were orally administered the extract of R. nervosus at a final dose of 300 mg/kg, (3) T1DM: diabetic rats which were orally administered distilled water, (4–6) T1DM + R. nervosus (100, 200, 300 mg/kg): were diabetic rats which orally administered the extract of R. nervosus at a final dose of 100, 200, or 300 mg/kg, respectively, and (7) T1DM + R. nervosus (300 mg/kg) + brusatol: were diabetic rats which were administered the extract of R. nervosus at a final dose of 300 mg/kg and concomitantly i.p. administered brusatol (an Nrf2 inhibitor) at a final concentration of 2 mg/kg. The extract and the vehicle administration were given using a gavage cannula daily for a total period of 56 days (8 weeks). The in vivo treatment with brusatol was given twice per week as described by other authors to block the cellular nuclear translocation of Nrf2 in the kidneys and livers of rats.37,38 Body weights were recorded weekly, and food consumption was recorded every day. Treatments were Experiments were stopped 12 h after day 56.
The 24 h urine collection
On the last day of the experiment (12 h after the last treatment on day 56), all animals were placed in metabolic cages, and their urine samples were collected over 24-h. All animals had free access to water and food. Urine samples were collected and centrifuged at 11,200 x g for 15 min. Urine volume was measured for each sample, and all samples were then kept at −20°C until further measurements.
Plasma, serum, and tissue collection
Directly after urine collection, all rats were anesthetized with ketamine/Xylazine hydrochloride mix (90:10 mg/kg, i.p). Blood (1 mL samples) was withdrawn in either EDTA or plain tubes and centrifuged at 1500 x g (10 min/room temperature) to collect plasma and serum. All samples were preserved at −20°C until further analysis. Then, all rats were killed by cervical dislocation, and their kidneys were removed, washed with ice-cold phosphate buffer saline (PBS) (pH = 7.4), and cut into smaller parts on ice. Parts of the kidneys’ collected tissues were snap-frozen in liquid nitrogen and stored at −80°C. Other parts were preserved in 10% buffered formalin for 24 h and then used for the histological evaluation.
Assessment of kidney function
The urea levels in the serum were measured using an assay kit (# DIUR-100 BioAssay Systems, CA, USA). Serum levels of blood urea nitrogen (BUN) levels were analyzed using assay kits (# EIABUN, ThermoFisher Scientific, Germany). An assay kit (# MBS841754, MyBioSorces, CA, USA) was used for the determination of creatinine (Cr) and albumin levels and the urinary albumin/Cr ratio (UACR). Cr execration (per 24 h) was calculated by the following formula [CrE (mg) = urinary Cr (mg/dl) × urine volume in 24 h (dl)]. 39 Cr clearance (CrCl) (ml/min), a marker of the glomerular filtration rate, was measured based on the following formula: [CrCl = urine creatinine (mg/dl) × urine flow (ml/min)/serum creatinine (mg/dl) where urine flow = urine volume (ml)/1440 (min/24 h). 39 All procedures were performed per the manufacturer's instructions.
Biochemical measurements in the renal homogenates
Samples (100 mg) of the frozen renal tissues were homogenized in 0.5 ml ice-cold PBS (pH = 7.4), then centrifuged at 1200 x g to isolate the supernatant containing the tissue homogenates. All supernatants were frozen at −80°C and used later to measure levels of some oxidative stress and inflammation-related markers. At the same time, the nuclear extracts were prepared from other kidney samples using a special kit (# Ab113474, Abcam, Cambridge, USA). The following parameters were measured in the tissue homogenates: MDA (# 10009055, Cayman, MI, USA), GSH (# orb782371, Biorbit, MO, USA), SOD (# MBS036924, MyBioSource, CA, USA); tumor necrosis factor-alpha (TNF-α) (# BMS622, ThermoFisher), interleukine-6 (IL-6) (# R6000B, R&D System, MN, USA), and IL-1β (Ab100768, Abcam, Cambridge, USA). The levels of NF-κB p65 and Nrf2 in the total cell homogenates and the nuclear extracts were measured using ELISA kits (Cat. No. MBS2505513; Cat. No. MBS752046; MyBioSource, CA, USA). All protocols were conducted per each kit's instructions.
Real-time PCR (qPCR)
All primers used for this part (ThermoFisher Scientific) are shown in Table 1. Total RNA was prepared using Trizol reagent (# T9424, Sigma Aldrich, MO, USA). First-strand cDNA was synthesized using the iScript preparation kit (# 1708891, Bio-Rad, USA). Amplification was conducted in CFX 348 qPCR machine in a 20 µl reaction using the Ssofast Evergreen Supermix (# 172-5200, BioRad, USA). With the following ingredients: master mix reagent (10 µl), forward primer (0.2 μl/500 nM), reverse primer (0.2 μl/500 nM); template cDNA (2 μl/50 ng), nuclease-free water (7.2 µl). qPCR steps were heating (1 cycle/98°C/30 s), denaturation (40 cycles/98°C/5 s), annealing (40 cycles/60°C/5 s), and melting (1 cycles/95°C/5 s/step). The mRNA expression levels were normalized to β-actin, and the expression ratio was calculated using the ΔΔCT method using the provided software.
Primers characteristics of the real-time PCR.

Histopathological evaluation
Kidney-formalin preserved sections were rehydrated in descending ethanol concentrations (100%, 90%, and 70%) and then embedded in wax. All sections were then at a thickness of 3-5 µM. and then stained with Harris hematoxylin (H)/glacial acetic acid solution. All sections were then de-stained with 1:400 v/v HCL/ethanol (70%) solution. After this, the sections were stained with Eosin (E) and dehydrated again with ethanol and xylene. A mounting media was added, and the tissue was covered with a coverslip. All tissue was examined under a Nikon-light microscope and photographed at 200X.
Statistical analysis
GraphPad Prism analysis software (Version 8) was used for the statistical analysis of all data. Kolmogorov-Smirnov test was utilized to test the normality. Analysis was performed using the 1-way ANOVA test. The levels of significance were determined using Tukey's test as post hoc (p < 0.05) All data were expressed in the results as means ± standard deviation (SD).
Results
Changes in body weight, food intake, and levels of glucose and insulin
Final body weights, weekly food intake, and fasting plasma glucose levels were significantly increased, whereas levels of fasting insulin were significantly decreased in STZ-induced T1DM rats as compared to control rats (Table 2). No significant variation in the levels of all these markers was seen between the control and R. nervous-treated rats, which received the extract at the dose of 3000 mg/kg (Table 2). In the same, weekly food intake, final body weights, and fasting glucose and insulin levels were not significantly varied between T1DM-treated rats, T1DM + R. nervosus of all doses (100, 200, 300 mg/kg), and with T1DM + R. nervosus + brusatol (Table 2). In addition, no significant differences in the levels of all these parameters were seen when a comparison was made between all treated groups (T1DM + R. nervosus 100, 200, 300 mg/kg).
R. nervosus failed to attenuate the loss in body weight, hyperphagia, hyperglycemia, and hyperinsulinemia in streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) rats.
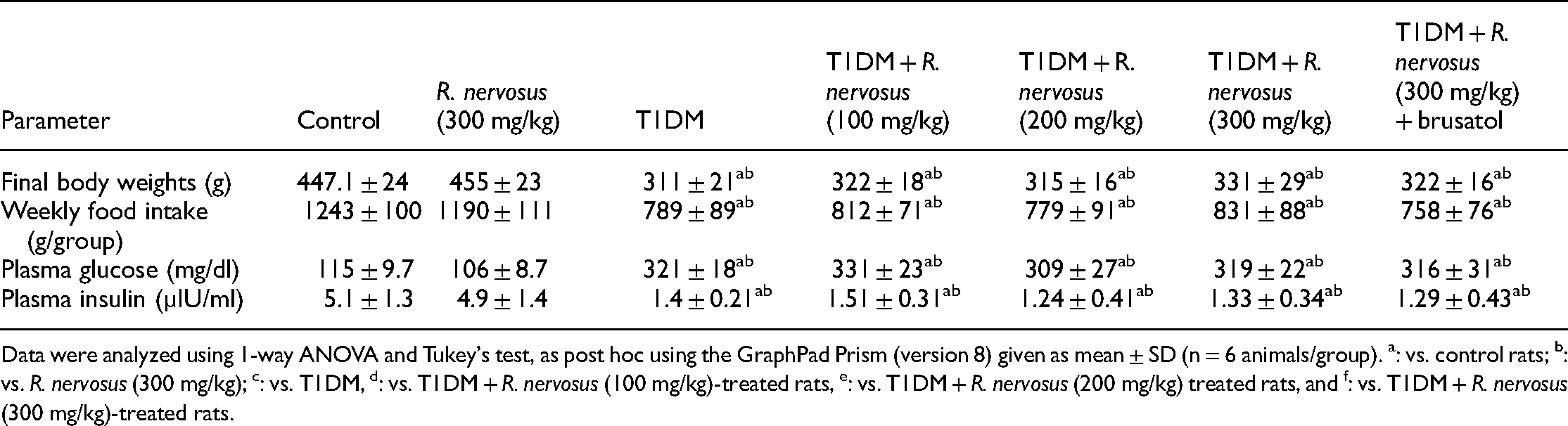
Data were analyzed using 1-way ANOVA and Tukey's test, as post hoc using the GraphPad Prism (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats.
Changes in kidney function test
All measured renal-related parameters were not significantly different between the control and R. nervosus-treated rats (300 mg/kg) (Table 3). On the other hand, serum albumin levels, urinary output and flow, urinary creatinine, and Cr clearance were significantly decreased, but serum urea and creatinine, as well as urinary albumin and albumin/Cr ratio, were significantly increased in T1DM-treated rats as compared to control rats (Table 3). The levels of all these markers were significantly reversed in a dose-dependent manner in T1DM + R. nervosus-treated rats as compared to T1DM-treated rats (Table 3). The maximum significant improvement in the levels of all these kidney function markers was seen in T1DM + R. nervosus (300 mg/kg) as compared to the other doses (100 and 200 mg/kg) (Table 3). However, serum albumin levels, urinary output and flow, urinary creatinine, and Cr clearance were significantly lower but whereas serum levels of urea and creatinine, as well as urinary albumin and albumin/Cr ratio, were significantly higher in T1DM + R. nervosus (300 mg) + brusatol-treated rats as compared to all T1DM + R. nervosus-treated groups (100, 200, and 300 mg/kg) (Table 3). Of note, no significant variation in the levels of all these biochemical endpoints was seen between the T1DM + R. nervosus (300 mg) + brusatol treated rats as compared to T1DM model rats (Table 3).
R. nervosus improves renal function markers in streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) rats in dose-dependent and an Nrf2-dependent manner.

Data were analyzed using 1-way ANOVA and tukey's test, as post hoc using the GraphPad prims (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats. CrCl: creatinine clearance.
Histological findings
Kidneys obtained from the control and R. nervosus (300 mg/kg)-treated rats showed normal architectures where intact glomeruli, glomerular membranes, and glomerular mesangial mass, as well as well-preserved proximal and distal convoluted tubules, were seen (Figure 1(a) and (b)). Kidneys obtained from T1DM model rats showed a reduction in glomerular mesangial mass and severe damage in the glomerular membranes, PCT, and DCT (Figure 1(c)). On the other hand, a significant progressive improvement in the structure of glomeruli, PCT, and DCT was observed in T1DM-rats, which were treated with R. nervosus (100, 200, and 300 mg/kg) with almost normal features were observed with the highest dose (300 mg/kg) (Figures 1(d) and 2(a–c)). On the other hand, a similar mirror image of the changes observed in the T1DM was also observed in T1DM + R. nervosus (300 mg/kg) + brusatol-treated rats (Figure 2(d)).

Histological sections of the kidenys of some groups of rats as stained by hematoxylin and eosin (H&E) staining. (a and b) Were taken from control and R. nervosus-treated rats (300 mg/kg) and show normal renal morphology with intact glomerulus containing normal mesangial mass (long thin arrow) and surrounded by an intact membrane (short thin arrow). All the proximal and distal convoluted tubules (PCT and DCT) appeared normal and intact (short and long thick arrows, respectively). (c) Was taken from T1DM-model rats and showed obvious damage in the glomerular membrane (short thin arrow) and a reduction in the glomerular mesangial mass (long thin arrow) with severe damage in the PCT and DCT (short and thick short arrows, respective) in large number. (d) Was taken from T1DM + R. nervosus-treated rats (100 mg/kg) and showed some little improvements in the structure of the PCT and DCT (thin and thick curved arrows), as well as in the glomerular mesangial mass (long thin arrow), and glomerular membrane (short thin arrow). However, there is still an increased number of damaged PCT and distal tubules (short and long thick arrows, respectively). H& E: 200X.

Histological sections of the kidneys of other groups of rats as stained by hematoxylin and eosin (H&E) staining. (a) Was taken from T1DM + R. nervosus-treated rats (200 mg/kg) and showed many improvements in the structure of the PCT and DCT (thin and thick curved arrows) with almost normal glomerular mesangial mass (long thin arrow) and glomerular membrane (short, thin arrow). However, some damage in the PCT and DCT is still seen in a few numbers (short and long thick arrows, respectively). (b and c) were taken form were taken from T1DM + R. nervosus-treated rats (300 mg/kg) and showed almost normal features like those seen in the control group. In this group of rats, most of the PCT and DCT (short and long thick arrows) appeared normal. Also, the glomerular mass appeared normal (long thin arrow) with the intact glomerular membrane (short thin arrow). However, in some sections, some vacillation is still seen in a few PCT (thin curved arrow) and DCT (thick curved arrow). (d) Was taken from T1DM + R. nervosus (300 mg/kg) and brusatol and showed similar morphological changes to those in T1DM model rats. Herein, reduced glomerular mesangial mass (long thin arrow) with severe and increased damage in the PCT (short thick arrow) and DCT (long thick arrow) were dominant. H& E: 200X.
Changes in renal levels /activation of Nrf2/keap1 axis
mRNA levels of keap-1 were not significantly varied, but mRNA, cytoplasmic, and nuclear levels of Nrf2 have significantly increased in R. nervosus-treated rats as compared to control rats (Figure 3(a–d)). mRNA levels of Keap1 were significantly increased, but mRNA, as well as cytoplasmic and nuclear levels of Nrf2, were significantly decreased in T1DM-treated rats as compared to control rats (Figure 3(a–d)). With no significant differences in the mRNA levels, T1DM + R. nervosus-treated rats of all tested doses (100, 200, and 300 mg/kg) showed a significantly progressive and dose-dependent increase in the mRNA, cytoplasmic, and nuclear levels of Nrf2 as compared to T1DM-treated rat, with the maximum significant increase to be seen in the group that received the highest dose (300 mg/kg) (Figure 3(a–d)). Administration of brusatol to T1DM + R. nervosus-treated rats didn't affect mRNA levels of keap-1 but reduced the mRNA, cytoplasmic, and nuclear levels of Nrf2 as compared to R. nervosus-treated rats (100, 200, and 300 mg/kg), to levels which are not significantly different when compared to T1DM-treated rats (Figure 3(a–d)).

R. nervosus increases the mRNA and total cytoplasmic and nuclear levels of the nuclear factor erythroid 2 related factor-2 (Nrf2) but without affecting the high mRNA levels of Kelch-like ECH-associated protein 1 (keap-1) in the kidney of the control and streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) rats in a dose and Nrf2-dependent mechanisms. Data were analyzed using 1-way ANOVA and Tukey's test, as post hoc using the GraphPad Prism (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats.
Changes in renal levels of oxidative stress, antioxidant, and inflammatory markers
Renal total levels of ROS and MDA, TNF-α, and IL-6, as well as nuclear levels of NF-κB p65, were significantly increased, but the total levels of GDH and MDA were significantly decreased in the T1DM-treated rats as compared to control rats (Figures 4(a–d) and 5(a–c)). The levels of GSH and SOD were significantly increased, and the levels of ROS and MDA, TNF-α, and IL-6, as well as nuclear levels of NF-κB p65, were significantly decreased in the kidneys of R. nervosus (300 mg/kg)-treated rats as compared to control rats and in the kidneys of T1DM + R. nervosus-treated rats (100, 200, and 300 mg/kg) as compared to T1DM-treated rats (Figures 4(a–d) and 5(a–c)). All these effects observed with these increasing doses of R. nervosus in T1DM-treated rats were in a dose-response manner with the effects to be associated with the highest dose (300 mg/kg), all of which were significantly reversed after co-administration of brusatol (T1DM + R. nervosus (300 mg/kg) + brusatol) (Figure 4(a–d) and 5(a–c)). Interestingly, the renal levels of all these oxidants, antioxidants, and inflammatory markers were not significantly different between the T1DM and (T1DM + R. nervosus (300 mg/kg) + brusatol-treated rats (Figure 4(a–d) and 5(a–c)).

R. nervosus inhibits the levels of reactive oxygen species (ROS), and malondialdehyde (MDA) in the kidneys of streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) rats but stimulates levels of total reduced glutathione (GSH) and superoxide dismutase in the kidneys of both the control and STZ-T1DM rats, in a dose and Nrf2-dependent manners. Data were analyzed using 1-way ANOVA and Tukey's test, as post hoc using the GraphPad Prism (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats.

R. nervosus suppressed total levels of tumor necrosis factor-α (TNF-α) and interleukine-6 (IL-6), as well as the nuclear levels of nuclear factor-kappa beta p65 (NF-κB p65) in the kidneys of streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) in a dose and Nrf2-dependent manner. Data were analyzed using 1-way ANOVA and Tukey's test, as post hoc using the GraphPad Prism (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats.
Changes in levels of markers of intrinsic cell apoptosis
R. nervosus-treated rats (300 mg/kg) showed no significant variations in the mRNA levels of Bcl2, Bax, and caspase-3 as compared to control rats (Figure 6(a–d)). The mRNA levels of Bcl2 were significantly decreased, but the mRNA levels of Bax and caspase-3, as well as the ratio of Bax/Bcl2, were significantly increased in the kidneys of T1DM-treated rats as compared with the control rats (Figure 6(a–d)). Treatment with the various doses (100, 200, and 300 mg/kg) of methanolic extract of R. nervous to T1DM-rats resulted in a dose response-reduction in the mRNA levels of Bax and caspase-3 and an increase in the levels of Bcl2 (Figure 5(a–d)). These effects associated with the high dose of R. nervosus (300 mg/kg) were prevented by co-administration of brusatol (Figure 6(a–d)).

R. nervosus attenuates the increase in mRNA levels of Bax and caspase-3 and upregulates mRNA levels of Bcl2 in the kidneys of streptozotocin (STZ)-induced type 1 diabetes mellitus (T1DM) in a dose and Nrf2-dependent manner. Data were analyzed using 1-way ANOVA and Tukey's test, as post hoc using the GraphPad Prism (version 8) given as mean ± SD (n = 6 animals/group). a: vs. control rats; b: vs. R. nervosus (300 mg/kg); c: vs. T1DM, d: vs. T1DM + R. nervosus (100 mg/kg)-treated rats, e: vs. T1DM + R. nervosus (200 mg/kg) treated rats, and f: vs. T1DM + R. nervosus (300 mg/kg)-treated rats.
Discussion
During the last decades, increasingly renewed interest has been given to identifying novel ingredients from herbs to prevent and alleviate DN. In this study, we are listing R. nervosus as a potential protective safe plant that could prevent the progression of DN, even without possessing any hypoglycaemic effect. Indeed, our data confirm the safety of this plant even at very high doses (2000 and 3000 mg/kg). Also, the chronic administration of the methanol extract of R. nervosus was not only able to reduce the generation of ROS and lipid peroxides, but also to stimulate antioxidant expression, suppress NF-κB activation and inflammatory cytokines production, and inhibit markers of intrinsic cell death. In addition, our data show a potent stimulatory effect of the methanol extract of R. nervosus on the nuclear activation of Nrf2, which seems to be mediated by stimulating the transcription of Nrf2 and inhibiting its degradation by decreasing the transcription of keap-1. Interestingly, and using brusatol as an Nrf2 inhibitor, the data in our hand suggest that activation of Nrf2 is a major mechanism underlying the nephroprotective effect of R. nervosus in this diabetic animal model.
Streptozotocin remains the best drug of choice to induce T1DM in mice and rats due to its ability to cause specific oxidative-induced necrosis in the pancreatic β-cells, leading to hypoinsulinemia and hyperglycemia, which induces multi-organ damage. 35 In addition, the rats model of STZ-induced diabetes develops clinical signs that are very close to those seen in humans, including polyphagia, weight loss (muscle and fat store wasting), polyuria, and polydipsia.40,41 In addition, STZ-induced diabetes can lead to renal damage and dysfunction as it reduces the glomerular filtration rate, Cr clearance, urine output, uremia, micro/macroalbuminuria, and glomerulo¬sclerosis, and severe necrosis and apoptosis in the renal tubules. 20 The majority of the morphological and clinical manifestations were also confirmed in STZ-induced rats in this study, thus being the strongest evidence for validating this animal model. However, after 8 weeks of administration of a high dose (300 mg/kg) or increasing doses (100, 200, and 300 mg/kg) of R. nervosus (100 mg/kg and 300 mg/kg) to either the control or T1DM-treated rats has no effects on rats’ food intake, blood glucose, and insulin levels, it seems reasonable that the observed neuroprotection afforded by the methanolic extract of the plant is not due to any hypoglycaemic or insulin-sensitizing effects. Supporting these data, and during the acute toxicity test examination, we also found no significant change in blood glucose and insulin levels 14 days after administering 2000 and 3000 mg/kg of the extract (data not shown). However, the hypoglycemic effect of R. nervosus was poorly investigated in the literature. In a single study, and contradicting our findings, treatment with leaf powder of R. nervosus (1% in diet) reduced fasting glucose levels in HFD-fed rats. Such variations between our data and these data could be explained by the difference in the animal model treatment (source, extraction, and period). Supporting our results, some authors reported no alterations in blood glucose levels in subjects using leaves and stems of R. nervosus for treatment. 42
On the other hand, the role of oxidative stress and inflammation in the pathogenesis of DN has gained increasing research attention in recent years, being the key mediator of the progression of the disease. 5 Markers of oxidative stress and inflammation are significantly upregulated in the diabetic kidneys of both humans and animals.5,20 In the diabetic kidneys and other tissues, oxidative stress and inflammation act in a self-perpetuating vicious cycle. ROS activates inflammation by several mechanisms, including the activation of NF-κ B.5,43,44 In turn, inflammation leads to oxidative damage by several mechanisms, including increasing the generation of ROS from the infiltrating and activated resident macrophages.43,44
On the other hand, both ROS and inflammatory cytokines can induce cell apoptosis by activating intrinsic (mitochondria-mediated) and extrinsic cell apoptosis. We and others have previously shown that hyperglycemia-induced ROS is the upstream event leading to renal inflammation and cell death during DN and is associated with increased activation of NF-κB and intrinsic cell death characterized by high expression of Bax and low expression of Bcl2.7,8,20 Mechanisms by which hyperglycemia induces ROS are well-explained in literature and include impairing the mitochondria oxidative phosphorylation, scavenging antioxidants, glucose autoxidation, induction of inflammation, and activating the ANGII/NADPH oxidase, advanced glycation end productions (AGEs), protein kinase C (PKC), hexosamine, and polyol pathways.45,46 In this regard, several free radicals species include superoxide radicals (O2−), singlet oxygen (1O2), highly reactive hydroxyl free radical (·OH), hydrogen peroxide (H2O2), and peroxynitrite (ONOO−) were reported to mediate DN. 47
Similar to the above-mentioned studies, oxidative stress, inflammation, and intrinsic cell apoptosis were also evidenced in the kidneys of STZ-induced rats as indicated by the increase in the levels of ROS, MDA, inflammatory cytokines (i.e. TNF-α and IL-6), mRNA levels of Bax and caspase-3, and the activation of NF-κB, as well as low expression of Bcl2. In addition, we have also found a significant reduction in the levels of antioxidant GSH, CAT, and SOD. These findings support many previous studies in STZ-treated rats.5,20,44 On the other hand, administration of increasing doses of R. nervosus methanol extract to STZ-diabetic rats resulted in a dose-response increase in the levels of all tested antioxidants, as well as in dose-response reduction in the generation of ROS, lipid peroxides, TNF-α and IL-6, and activation of NF-κB. Interestingly, levels of GSH, CAT, and SOD, as well as activities of NF-κB were also increased in the kidneys of rats which administered the high dose of R. nervosus extract indicating the emerging role of this extract to act as an antioxidant and anti-inflammatory through stimulating the expression of enzymatic and non-enzymatic antioxidant genes and inhibiting NF-κB. Although not yet well characterized, a few pre-existing studies may support our data. Indeed, the antioxidant and anti-inflammatory potential of R. nervosus has been documented in multiple in vitro studies and was attributed to its high content of polyphenols and flavonoids, which can scavenge ROS, chelated iron, upregulate antioxidants, and suppress NF-κB.31,48 In addition, treatment with R. nervosus reduced levels of TNF-α and IL-6 in caecal goblet cells of chicken infected with Eimeria tenella. 23
Further supporting evidence is documented in other Rumex species. Within this view, the ethanolic extract of the roots of R. patientia prevented ferric nitrilotriacetate-induced hepatic oxidative stress by reducing the generation of ROS and restoring levels of SOD, GSH, GPx, and catalase. 49 Extracting the leaves of R. hastatus and R. tingitanus leaf extracts attenuated CCL4-hepatic damage and fibrosis mainly by upregulating antioxidants.50,51 Also, water extract of R. crispus leaves inhibited osteoclastogenesis by suppressing NF-κB. 51 On the same page, the total extract of R. japonicus attenuated atopic skin dermatitis by inhibiting NF-κB and the generation of TNF-α. 18
Nonetheless, accumulating data have indicated that activation of Nrf2 is crucial for maintaining the redox potential of the renal cells and is a major mechanism for protection against DN by suppressing oxidative stress, inflammation, fibrosis, and apoptosis.9,16 The apparent reduction in the transcription, total levels, and nuclear accumulation of Nrf2 and the concomitant upregulation of keap-1 in the kidneys of STZ-induced rats of this study follows many previous similar studies.9,17,19,20,44,52 However, reversal of these effects after administration of the methanolic extract of R. nervosus indicates its ability to stimulate Nrf2 mainly by stimulating its transcription of Nrf2, even in the absence of any effect on levels keap-1. These data dissipate the degradation of Nrf2 from the observed result and suggest that its cytoplasmic levels and nuclear accumulation are secondary to the increase in its transcription. Interestingly, R. nervosus also stimulated mRNA, as well as the cytoplasmic and nuclear levels of Nr2, with no effects on mRNA levels of keap1 in the kidney of control rats too. This effect was described in a dose-dependent manner in the kidneys of STZ-diabetic rats.
To confirm our data, we have treated the rats with a pre-established dose of brusatol, a selective inhibitor that reduces the nuclear translocation of Nrf2 at the posttranslational levels and is independent of modulating the expression of Keap-1, the degradation systems, and protein kinase signaling, and transcription of Nrf2. 53 Interestingly, the antioxidant, anti-inflammatory, and anti-apoptotic effects of R. nervosus were completely disappeared in the STZ-treated rats, which showed severe deterioration in the structure and function of their kidneys like those seen in the model rats. Based on these observations, we concluded that Nrf2 is indispensable for the protection afforded by R. nervosus and is a key mechanism underlying its action.
Since Nrf2's major function is to regulate the redox potential of the cell by upregulating GSH and antioxidant enzymes, it seems reasonable that previously discussed increase in the levels of GSH, SOD, and CAT in the kidneys of both the control and STZ-diabetic rats are attributed to the parallel activation of Nrf2. On the other hand, As R. nervosus only inhibited cell apoptotic markers (i.e. Bax, caspase-3) and improved levels of Bcl2 in the kidney of STZ rats alone but not in the control rats, an effect that was reversed by brusatol, these data indicate that the anti-apoptotic effect of this plant is secondary to its antioxidant potential. On the other hand, several studies have shown the ability of R. nervosus to inhibit apoptosis by inhibiting NF-κB. 31 However, the mechanism behind this is still unclear. In this study, we are also showing that this effect could be Nrf2-dependent, as administration of brusatol also increased the nuclear accumulation of NF-κB in the kidneys of treated diabetic rats to the same levels seen in STZ-model rats. Nrf2 can also NF-κB by suppressing the generation of ROS (trigger), and activation of HO-1-which inhibits the degradation of the inhibitor of NF-κB, a major suppressor of NF-κB. 54 To the best of our knowledge, this is the first study that demonstrates that activation of Nrf2 is one major mechanism behind the antioxidant and anti-inflammatory effects of R. nervosus. These data can be supported by other studies using other species. In this view, the antioxidant and anti-inflammatory effects of the species R. cripsus in HepG2-C8 cells were related to its ability to phase II antioxidant enzymes through upregulating Nf2 and stimulating it and activation. 55 In the same manner, the cytoprotective effect of R. aquaticus against H2O2 induced oxidative stress in AGS cell lines is mediated by PI3 K/Akt-induced upregulation and activation of Nrf2. 56
Conclusions
These data demonstrated a potent-dose-dependent ability of the methanolic extract of R. nervosus to alleviate STZ-induced DN in rats and suggest that activation of the Nrf2/antioxidant axis is a major protective mechanism. Such findings encourage further examination and moving to subclinical and translational studies. However, some limitations should be resolved before this. Importantly, further studies in Nrf2 knocked out animals or cultured mesangial cells are needed to confirm our data further. Unfortunately, this was not available and will be considered in future studies. In addition, several studies have been conducted previously to examine the phytochemical composition of the leaves of R. nervosus and showed plenty of active compounds such as fatty acids (e.g. Palmitoleic Acid and Palmitic acid), vitamins, and other polyphenols (i.e. glucoside, quercetin, Quercetin rutinoside and isomers, Hesperetin, Apigenin 7-(O-6-acetyl)-glycoside, etc.) which possess antioxidant and anti-inflammatory effects.25,32 Further studies identifying the active ingredients responsible for the upregulation and activation of Nrf2 should be considered in future studies, which may present a novel Nrf2 activator that can treat diabetic complications and other disorders.
Footnotes
Acknowledgments
The authors are grateful to the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Funding Program (RFP-1442-6), for funding this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The authors are grateful to the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Funding Program (RFP-1442-6), for funding this work.
Ethics approval
All procedures, protocols, treatments, sampling, and euthanasia were approved by the Official Review Board at Princess Nourah University, Riyadh, KSA (IRB Number 20-0478), Riyadh, KSA.
Author biographies
Lujain A AlMousa Assistant professor of Nutrition , Department of Physical Sport Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Nora A AlFaris Professor of Nutrition and Food Science, Department of Physical Sport Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Ghedeir M Alshammari Associate professor in Department of Food Science and Nutrition, College of Food and Agricultural Sciences, King Saud University, Riyadh, Saudi Arabia.
Muneer M Alsayadi Associate professor in Department of Food Science and Technology, Faculty of Agriculture and Food Science, Ibb University, Ibb, Yemen.
Jozaa Z ALTamimi Associate professor of Nutrition, Department of Physical Sport Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Reham I Alagal Assistant professor of Nutrition, Department of Physical Sport Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Mohammed Abdo Yahya Ph.D. in Department of Food Science and Nutrition, College of Food and Agricultural Sciences, King Saud University, Riyadh, Saudi Arabia.
