Abstract
Objective
To compare cognitive profiles and dementia severity among older patients with atrial fibrillation (AF), with and without ischemic stroke (IS), and to evaluate the contribution of vascular burden to global cognitive status.
Methods
This cross-sectional clinical study included 124 patients aged ≥55 years who were stratified into three groups: AF without IS (n = 50), AF with IS (n = 25), and IS without AF (n = 49). Global cognitive status was assessed using ordinal categories derived from the Mini-Mental State Examination (MMSE). Attention and working memory were additionally evaluated using the Information-Memory-Concentration (IMC) test derived from the Blessed Dementia Scale. Vascular burden was assessed using the Hachinski Ischemic Score (HIS). Group differences were analyzed using appropriate statistical tests, and predictors of worse cognitive status were examined using ordinal logistic regression.
Results
Patients with combined AF and IS demonstrated a trend toward a less favorable cognitive profile and higher vascular burden compared with patients with AF alone or IS alone. The proportion of female participants differed significantly across groups (p = 0.022), whereas age category and educational level were comparable. In multivariable ordinal logistic regression analysis, higher Hachinski Ischemic Score independently predicted worse global cognitive status (OR 1.79, 95% CI 1.42–2.25; p < 0.001) after adjustment for age, sex, education, and major vascular risk factors.
Conclusions
Vascular burden plays an important role in cognitive impairment among older patients with atrial fibrillation, particularly when accompanied by ischemic stroke. Incorporating vascular burden assessment into routine clinical evaluation may facilitate earlier recognition and characterization of cognitive impairment in aging populations.
Keywords
Introduction
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia in older adults, with prevalence increasing substantially with advancing age. 1 Beyond its well-established role as a major risk factor for ischemic stroke (IS), growing evidence suggests that AF is also associated with cognitive impairment and dementia, even in the absence of clinically overt cerebrovascular events.2–6 As populations continue to age worldwide, understanding the cognitive consequences of AF has become an increasingly important clinical and public health priority.
Several mechanisms have been proposed to explain the association between AF and cognitive decline. These include chronic cerebral hypoperfusion, recurrent microembolization, silent cerebral infarctions, systemic inflammation, and progression of cerebral small vessel disease.7–9 Epidemiological studies have demonstrated that AF is associated with an increased risk of both vascular cognitive impairment and Alzheimer-type dementia, suggesting a complex interaction between cardiovascular pathology and neurodegenerative processes.6,10,11
Ischemic stroke remains one of the leading causes of long-term cognitive impairment in older adults. Post-stroke cognitive deficits frequently involve attention, executive function, and working memory, domains that are essential for daily functioning and independence.12,13 Patients with concomitant AF and IS may represent a particularly vulnerable subgroup, as the combined effects of arrhythmia-related cerebral hypoperfusion and overt cerebrovascular injury may contribute to more heterogeneous cognitive profiles. 14
From a geriatric neuropsychiatric perspective, cognitive impairment related to AF and cerebrovascular disease is clinically important not only because of its impact on cognitive functioning but also because of its association with functional decline, behavioral symptoms, and reduced quality of life. 15 However, cognitive evaluation in routine clinical practice often relies on brief global screening instruments such as the Mini-Mental State Examination (MMSE), which may have limited sensitivity for detecting domain-specific deficits commonly observed in vascular cognitive disorders.16,17
The Hachinski Ischemic Score (HIS) was developed as a clinical instrument to estimate the vascular contribution to cognitive impairment and remains a useful tool for differentiating vascular and degenerative cognitive profiles.16,17 When combined with brief cognitive screening and targeted neuropsychological assessment, HIS may help clinicians better characterize dementia severity and the potential vascular contribution to cognitive decline in patients with AF and cerebrovascular disease.
Despite increasing recognition of the relationship between AF, ischemic stroke, and cognitive impairment, comparative data examining cognitive profiles and dementia severity across patients with AF alone, AF with concomitant ischemic stroke, and ischemic stroke without AF remain limited. Improved understanding of these patterns may have important implications for clinical risk stratification, patient management, and follow-up strategies in geriatric neurology and clinical neuroscience.
Accordingly, the aim of the present study was to compare cognitive profiles and dementia severity in older patients with atrial fibrillation, with and without ischemic stroke, and to examine the association between vascular burden and global cognitive status using clinically applicable assessment tools.
Methods
Study design and participants
This cross-sectional clinical study included consecutive patients aged ≥55 years who were recruited between August 2024 and July 2025 at the University Clinical Center Tuzla, Bosnia and Herzegovina. The reporting of this study conforms to the STROBE guidelines for observational studies. 18
Participants were classified into three groups according to clinical diagnosis: atrial fibrillation without ischemic stroke (AF), atrial fibrillation with ischemic stroke (AF + IS), and ischemic stroke without atrial fibrillation (IS). The diagnosis of atrial fibrillation was established based on electrocardiographic documentation, while ischemic stroke was confirmed using standard neuroimaging criteria, including computed tomography (CT) or magnetic resonance imaging (MRI) of the brain.
Patients with severe sensory deficits, acute delirium, previously diagnosed dementia, or other conditions that could preclude valid cognitive assessment were excluded from the study.
Cognitive assessment
Global cognitive status was assessed using the Mini-Mental State Examination (MMSE), a widely used screening instrument for cognitive impairment that evaluates orientation, memory, attention, language, and visuospatial abilities. 15
MMSE scores were categorized into ordinal levels reflecting increasing severity of cognitive impairment as follows: severe cognitive impairment (≤10), moderate impairment,5,13–21 mild impairment,6,22–24 borderline cognitive status (26), and normal cognition (27–30).
In addition to global cognitive screening, attention and working memory were evaluated using the Information-Memory-Concentration (IMC) test derived from the Blessed Dementia Scale, which assesses three cognitive domains: information, memory, and concentration.
Assessment of vascular burden
Vascular contribution to cognitive impairment was assessed using the Hachinski Ischemic Score (HIS), a clinically validated instrument designed to estimate the likelihood of vascular cognitive impairment.16,17 Higher HIS values indicate greater vascular burden and a stronger contribution of cerebrovascular pathology to cognitive dysfunction.
Clinical and demographic variables
Demographic variables included age category, sex, and education category. Clinical variables included hypertension, diabetes mellitus, and hyperlipidemia, obtained from medical records and patient history.
Age and education were analyzed as ordinal categorical variables based on predefined clinical categories.
Statistical analysis
Descriptive statistics were calculated for all variables and presented as mean ± standard deviation or number (percentage), as appropriate. Group comparisons were performed using analysis of variance (ANOVA) for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables, as appropriate.
To examine factors associated with worse global cognitive status, ordinal logistic regression analysis was performed, with MMSE category as the dependent variable.
Independent variables included age category, sex, education category, hypertension, diabetes mellitus, hyperlipidemia, and Hachinski Ischemic Score.
Results are presented as regression coefficients, odds ratios (OR) with 95% confidence intervals (CI), and p-values. A two-sided p-value < 0.05 was considered statistically significant.
All statistical analyses were conducted using SPSS software.
Ethics approval
The study was approved by the Ethics Committee of the University Clinical Center Tuzla (Approval No. 02-09/2-86-1/24, issued on July 16, 2024). The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024).
Written informed consent was obtained from all participants or their legally authorized representatives (LARs) prior to study participation. In cases where consent was provided by a LAR, participants were considered to lack sufficient decisional capacity to provide informed consent independently due to cognitive impairment. Participants who provided consent themselves were considered to have preserved decision-making capacity.
Results
Demographic and clinical characteristics
Demographic and clinical characteristics of patients by study group.

AF – atrial fibrillation; IS – ischemic stroke; SD – standard deviation. Continuous variables are presented as mean ± standard deviation and categorical variables as number (percentage). Group comparisons were performed using analysis of variance (ANOVA) for continuous variables and chi-square or Fisher’s exact tests for categorical variables, as appropriate.
Age category did not differ significantly between groups (p = 0.332), nor did educational level (p = 0.842). The proportion of female participants differed significantly across groups, with a higher representation of women in the AF + IS group compared with the AF and IS groups (p = 0.022). The prevalence of hypertension, diabetes mellitus, and hyperlipidemia was high across all groups and did not differ significantly between them.
Cognitive status and vascular burden
Hachinski Ischemic Score and global cognitive status by study group.
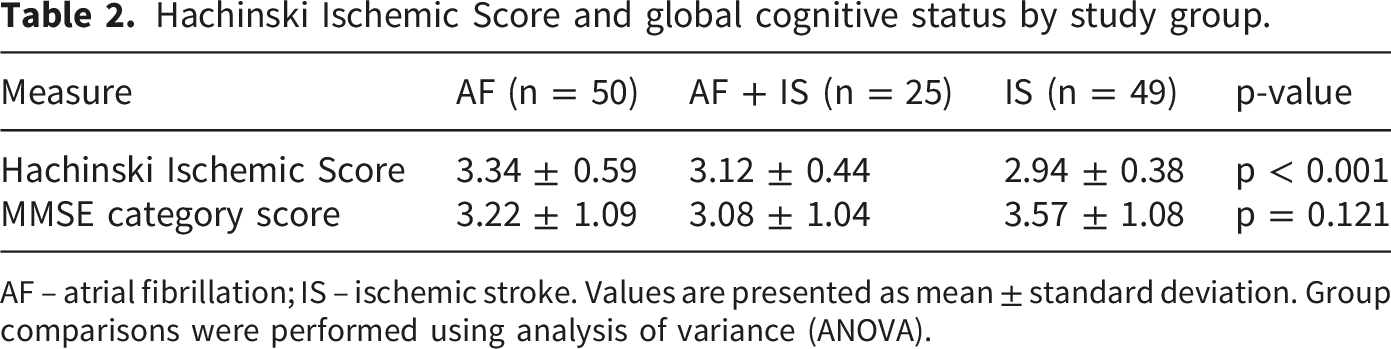
AF – atrial fibrillation; IS – ischemic stroke. Values are presented as mean ± standard deviation. Group comparisons were performed using analysis of variance (ANOVA).
Adjusted group comparisons
Adjusted global cognitive performance (MMSE category) across study groups.
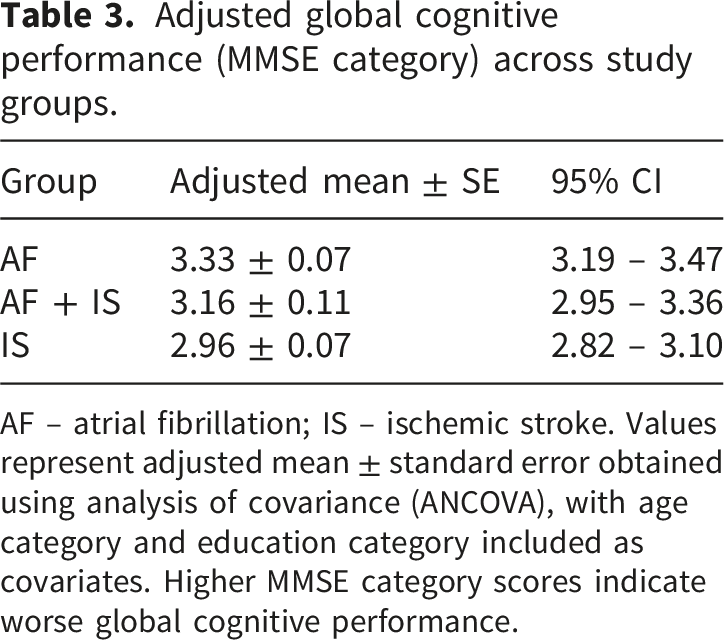
AF – atrial fibrillation; IS – ischemic stroke. Values represent adjusted mean ± standard error obtained using analysis of covariance (ANCOVA), with age category and education category included as covariates. Higher MMSE category scores indicate worse global cognitive performance.
Predictors of worse global cognitive status
Ordinal logistic regression analysis of factors associated with worse global cognitive status (MMSE category).
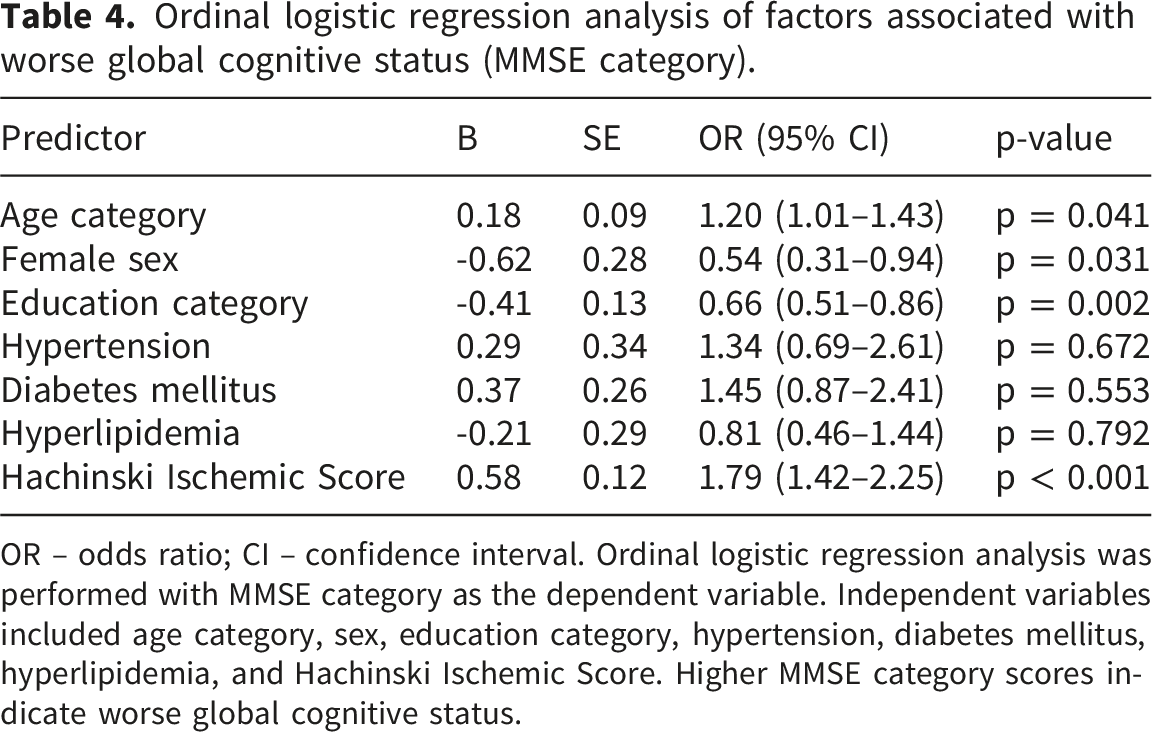
OR – odds ratio; CI – confidence interval. Ordinal logistic regression analysis was performed with MMSE category as the dependent variable. Independent variables included age category, sex, education category, hypertension, diabetes mellitus, hyperlipidemia, and Hachinski Ischemic Score. Higher MMSE category scores indicate worse global cognitive status.
Importantly, higher Hachinski Ischemic Score was independently associated with worse global cognitive status (OR 1.79, 95% CI 1.42–2.25; p < 0.001), after adjustment for age category, sex, education category, and major vascular risk factors. Hypertension, diabetes mellitus, and hyperlipidemia were not independently associated with cognitive status in the adjusted model.
Discussion
In this study, we compared cognitive profiles and dementia severity among older patients with atrial fibrillation (AF), with and without ischemic stroke (IS), and examined the association between vascular burden and global cognitive status. Our findings suggest that greater vascular burden, as reflected by the Hachinski Ischemic Score (HIS), is associated with worse global cognitive status in patients with AF and cerebrovascular disease.
Previous studies have consistently demonstrated that AF is associated with an increased risk of cognitive impairment and dementia, even in the absence of clinically overt stroke.2–4,6,22,24 Several mechanisms may contribute to this association, including chronic cerebral hypoperfusion, recurrent microembolization, and progression of cerebral small vessel disease.7–9 These processes may contribute to cumulative cerebral injury and progressive cognitive decline in older individuals with AF.
Ischemic stroke represents an additional major risk factor for cognitive impairment in aging populations. Post-stroke cognitive impairment frequently affects executive function, attention, and memory, and is associated with increased disability and reduced quality of life.12,13,23 Patients with concomitant AF and IS may therefore represent a particularly vulnerable subgroup in which both arrhythmia-related hemodynamic factors and overt cerebrovascular injury contribute to cognitive dysfunction.
In the present study, global cognitive status assessed using the MMSE did not demonstrate large differences across diagnostic groups. This finding may reflect several factors. First, the MMSE is a global screening instrument that may be less sensitive to subtle domain-specific deficits commonly observed in vascular cognitive impairment.16,17 Second, the relatively small size of the AF + IS subgroup may have limited statistical power to detect subtle between-group differences. The use of the IMC test provided additional domain-specific insight into attention and working memory deficits, supporting and complementing the global cognitive categorization based on MMSE.
An additional finding of our analysis was the association between female sex and lower odds of worse cognitive status in the regression model. Although many epidemiological studies report a higher prevalence of dementia in women, sex differences in vascular cognitive impairment are complex and may reflect differences in vascular risk profiles, survival bias, and healthcare utilization. 6 Therefore, this finding should be interpreted cautiously and requires confirmation in larger cohorts.
The Hachinski Ischemic Score demonstrated a significant association with global cognitive status in the regression analysis. HIS was originally developed to differentiate vascular dementia from degenerative cognitive disorders and remains a clinically useful tool for identifying vascular contributions to cognitive impairment.19,20 Several items included in the HIS reflect clinical features that may overlap with cognitive symptoms, which introduces a potential degree of circularity when interpreting associations with cognitive outcomes. However, HIS is not a direct measure of cognitive performance but rather an index of vascular contribution to cognitive impairment. The persistence of its independent association with cognitive status after adjustment for demographic and vascular risk factors supports its clinical relevance as a marker of vascular burden rather than a purely circular construct. Therefore, while conceptual overlap cannot be entirely excluded, the consistent independent association observed in adjusted analyses supports the interpretation that HIS captures vascular burden rather than cognitive status itself.
Several limitations of this study should be acknowledged. First, the cross-sectional design precludes conclusions regarding causality between atrial fibrillation, vascular burden, and cognitive decline. Second, the relatively small size of the AF + IS subgroup may have limited statistical power for detecting subtle group differences. Third, global cognitive assessment relied primarily on screening instruments rather than comprehensive neuropsychological testing, which may limit sensitivity for detecting domain-specific cognitive deficits.
Despite these limitations, the present study contributes to the growing body of evidence highlighting the importance of vascular factors in cognitive impairment among older adults with atrial fibrillation. From a clinical perspective, routine assessment of vascular burden using simple clinical instruments such as the Hachinski Ischemic Score may assist clinicians in identifying patients at higher risk of cognitive decline.
Future research should include larger prospective cohorts and incorporate neuroimaging markers of vascular brain injury, including white matter hyperintensities and silent cerebral infarctions. Such approaches may provide deeper insight into the mechanisms linking atrial fibrillation, cerebrovascular disease, and cognitive decline.
Conclusion
The findings of this study indicate that vascular burden is significantly associated with cognitive impairment in older patients with atrial fibrillation, particularly when ischemic stroke is also present. Although global cognitive differences between diagnostic groups were modest, higher Hachinski Ischemic Score values were associated with worse global cognitive status.
These results highlight the potential clinical value of incorporating vascular burden assessment into routine evaluation of older patients with atrial fibrillation. Early identification of patients at increased risk of cognitive impairment may support more targeted monitoring and management strategies.
Further prospective studies with larger cohorts and more comprehensive neuropsychological assessment are needed to clarify the mechanisms linking atrial fibrillation, cerebrovascular disease, and cognitive decline.
Footnotes
Acknowledgements
The authors thank the clinical staff of the University Clinical Center Tuzla for their assistance during data collection. The authors used ChatGPT (OpenAI) to assist with language editing.
Author contributions
M.S.S. contributed to study design, data collection, and manuscript drafting.
Z.D. contributed to study conception, supervision, and critical revision of the manuscript.
A.D. contributed to data collection and analysis.
D.S. contributed to data interpretation and manuscript revision.
O.Ć.I. contributed to study design and clinical oversight.
D.S. (Salihović) contributed to data analysis and manuscript preparation.
All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
