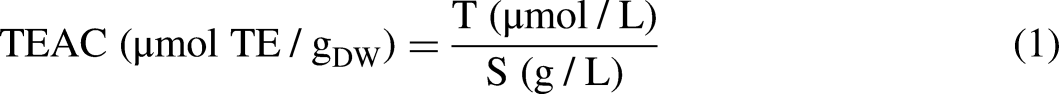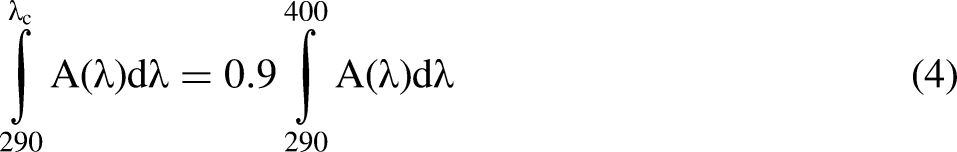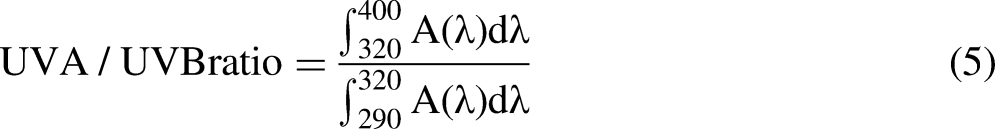Abstract
UV filters in current sunscreen formulations can have negative effects on human health, such as endocrine disruption and allergic reactions, as well as on the environment, including bioaccumulation and coral health toxicity. As a result, there is a need to find alternative compounds that serve as safer and more ecofriendly active ingredients. This study successfully isolated actinomycetes from the octocoral Eunicea fusca and assessed their potential as producers of photoprotective compounds. The use of bio-based chemical agents, particularly natural products, has been a highly effective strategy for discovering bioactive compounds, especially in marine invertebrates and their associated microbiota. Eighteen bacterial isolates were obtained and subsequently employed to prepare raw methanolic extracts from seven-day submerged cultures in Zobell marine broth. The resulting extracts were screened for 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging capacity and characterized by total phenolic and flavonoid content measurements. After screening, the Gordonia hongkongensis EUFUS-Z928-derived raw extract exhibited the best antioxidant profile, i.e. DPPH and ABTS radical scavenging of 4.93 and 6.00 µmol Trolox per gram of extract, respectively, and selected for further photoprotection-related analysis. Thus, this extract demonstrated a UV-absorbing capacity of 46.33% of the in vitro sun protection factor calculated for 30 µg/mL oxybenzone but did not exhibit any cytotoxicity on human dermal fibroblasts (HDFa cell line) at concentrations up to 500 µg/mL. The liquid chromatography-mass spectrometry chemical characterization of this extract showed compounds with structural features associated with free radical scavenging and UV absorption (i.e. photoprotection-related activities). These findings highlighted the potential of the microbiota associated with E. fusca and confirmed the feasibility of exploiting its metabolites for photoprotection-related purposes.
Introduction
Exposure to ultraviolet (UV) radiation has important human health benefits; nevertheless, prolonged exposure can produce adverse effects such as skin photodamage (e.g. irregular pigmentation, collagen fibrils deterioration, DNA lesions 1 ). According to the wavelength, UV light is divided into UV-A (320–400 nm), UV-B (290–320), and UV-C (100–290). Since UV-C is filtered by the ozone layer, UV-A and UV-B are the main types of radiation affecting human health, UV-A being the main type of radiation. 2 UVB induces erythema, injuring the epidermis, while UVA is related to oxidative stress in the dermis layer. 3 Chronic lesions like those mentioned can lead to skin cancer, including basal cell carcinoma, squamous cell carcinoma, and melanoma. For instance, between 1990 and 2017, squamous cell carcinoma cases increased by 310%, while melanoma cases have increased by 4–6% annually. 4
The use of sunscreen is an essential approach for photoprotection.3,5 However, there are concerns about several UV filters’ safety and ecotoxicity issues. 6 For instance, oxybenzone and octyl-methoxycinnamate (the most widely used sunscreening agents) have been reported as endocrine disruptors. 7 In addition, they have also been described as bioaccumulating agents in aquatic environments, disturbing these important ecosystems. 6
Photoprotection effectiveness is also another factor that deserves attention. Individuals select sunscreens based on the sun protection factor (SPF) values. 8 Nevertheless, the SPF is a reliable indicator of UV-B protection; however, it is inadequate for UV-A protection.8,9 Given that oxidative damage represents a critical molecular mechanism for the detrimental effects of UV-A radiation on the skin,10,11 it is evident that antioxidant agents are necessary components of such products.8,9,12
Oxidative stress is a physiological phenomenon that plays a role in the development of several adverse health conditions and diseases, including photodamage. 13 Hence, the concept of antioxidants as chemopreventive agents is well-established in the scientific literature, with numerous studies supporting this notion.14–16 Consequently, the investigation of antioxidant agents is generating considerable interest, with important implications for the food, pharmaceutical, and cosmetic industries. The rising prevalence of skin cancers and the documented role of photooxidative damage in photocarcinogenesis 17 underscore the urgency of identifying compounds with photoprotective properties, including UV-absorbing and antioxidant capabilities.
Historically, natural products and their related structures significantly influenced the pharmaceutical industry. 18 Recently, microorganisms, particularly those associated with marine invertebrates, have emerged as highly promising sources of bioactive compounds. 19 Although plant-derived metabolites offer the potential for discovering natural antioxidants, manufacturing commercial products based on these compounds is impeded by major obstacles, such as limited availability due to synthesis constraints and low yields during isolation processing.20–22 In contrast to macroorganisms (e.g. plants, sponges, corals), microbial biotechnology offers a promising alternative to overcome these limitations. For instance, metabolic engineering can improve the production of target compounds in microorganisms, enabling their application on a mass production level. 23
A further aspect to emphasize about microorganism-derived natural products is that microorganisms become renewable bioresources. This fact has particular relevance to meeting the sustainable development goals (SDGs). 24 Among the diverse goals that microbial biotechnology can contribute to, economic development (SDG8) and good health and well-being (SDG3) are critical scenarios worth highlighting. The first is through the development of bio-based economies, promoting diversification and the demand and retention of a highly qualified workforce. 25 Regarding SDG3, bio-based products (such as those derived from microorganisms) represent an ecofriendly way of obtaining high-value-added substances. 26 The latter is particularly relevant in skincare products since several UV filters have become potential environmental toxicants. 6
In light of the aforementioned benefits, our study focuses on identifying actinomycetes as bioresources of bioactive compounds. In particular, we describe photoprotection-based screening of raw extracts obtained from culturable actinobacteria isolated from the octocoral Eunicea fusca. The antioxidant capacity was tested by radical scavenging assays, and the ability to absorb UV radiation was measured by spectrophotometric techniques (total phenolics and flavonoids were also examined). The most active strain was identified by 16S ribosomal gene sequencing, and the cytotoxic potential of its methanolic extract was explored and characterized by high-resolution mass spectrometry. This work contributes to an understanding of the potential of host-associated bacteria as producers of exploitable high-value-added metabolites that could be further employed as active principles in photoprotective products.
Materials and methods
Sample collection and actinobacteria isolation
The octocoral sampling was carried out by scuba diving at 10 m depth in Bahía de Santa Marta, Punta de Betín (11°15'02.1"N 74°13'16.0"W), in Santa Marta, Magdalena, Colombia. The sample was rinsed with sterile seawater (SSW), cut into small pieces, and crushed in 10 mL of SSW. Serial dilutions were plated in Zobell medium (1.25 g of yeast extract, 3.75 g of peptone, 18 g of NaCl, 2 g of MgCl2, 0.525 g of KCl, 0.075 g of CaCl2 and 15 g of agar dissolved in 1 L of distilled water 27 ). The isolation media were prepared with nalidixic acid (50 μg/mL) to prevent the growth of Gram-negative bacteria, and with cycloheximide (100 μg/mL) to avoid fungi contamination. The plates were incubated at 30° C for four weeks and the colonies showing actinobacterial-like morphology were picked up and transferred to a new plate until pure strains were obtained. The pure putative actinobacteria strains were stored at −80° C in their respective isolation medium at 30% (v/v) glycerol.
Extract preparation
Each isolated strain was independently grown in Zobell medium for seven days at room temperature. Then, a bacterial plug (55.81 mm2) was used as inoculum in 3 mL of liquid Zobell (i.e. without agar), and incubated for seven days at 200 r/min on a rotary shaker. After this, to establish the fermentation culture, 1 mL of the seed culture was transferred to 9 mL of the fresh Zobell broth and was incubated for an additional seven days at the same seed culture conditions. Afterward, the whole fermented culture was lyophilized until dryness to be subsequently extracted through an ultrasound-assisted procedure (frequency: 25 kHz; power effective: 200 W; temperature: < 30 °C) using methanol to afford the respective crude extract, following the methodology proposed by Khan et al. 28 Finally, the solvent was removed using a rotary evaporator to afford the raw extracts. The resulting microbial extracts were stored at −20°C before using them in biological and chemical assays.
Radical scavenging assays
The antioxidant capacity of each bacterial extract was explored by 2,2-diphenyl-1-picrylhydrazyl (DPPH, Sigma) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS, Sigma) radical scavenging assays, which are well known and widely reported elsewhere. 29 The DPPH assay was used to measure the scavenging capacity of this radical dissolved in methanol. DPPH was prepared at a concentration of 0.20 mM, and 100 µL of each extract was mixed with 100 µL of DPPH solution. The mixture was then left to react for 30 min at room temperature and protected from light. Absorbance was measured at λ = 515 nm using an iMarkTM microplate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA), with Microplate Manager® Software v6.3 (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The final concentration of each extract was adjusted to 5 mg/mL. Radical scavenging measurements were performed in triplicate, and standard curves using Trolox (ranging from 30.00 to 1.56 µM) were constructed and used to interpolate the antioxidant capacity results.
Regarding ABTS assay, the ABTS●+ radical cation was first prepared by its oxidation with potassium persulfate 16 h prior to the assay execution. The ABTS solution was then adjusted to 0.70 ± 0.02 absorbance units at λ = 735 nm by dilutions with deionized water. The assay was performed by mixing 190 µL of ABTS solution with 10 µL of each extract (or Trolox) for 10 min at room temperature and protected from light. Absorbance was measured at λ = 735 nm using the same microplate reader and software as mentioned earlier. The tests were also conducted in triplicate. The concentration of the bacterial extracts and Trolox was prepared to achieve the same final concentrations as used in the DPPH assays.
The results of antioxidant capacity in both DPPH and ABTS assays were expressed as Trolox Equivalents Antioxidant Capacity (TEAC), according to the following equation:
Quantification of total phenolics and flavonoids content
Total phenols and flavonoids content (TPC and TFC, respectively) were measured according to widely used methodologies reported elsewhere. 30 For TPC, we followed the methodology reported by Magalhães et al. 31 Briefly, Folin-Ciocalteu reagent (MilliporeSigma, St Louis, MO, USA) was diluted 1:5 in deionized water and a standard curve with a gallic acid (GA; MilliporeSigma) was constructed. First, 50 µL of GA or the sample was mixed with 50 µL of Folin-Ciocalteu reagent. Then, 100 µL of sodium hydroxide solution (0.35 M) was added. This mixture was left to react for 3 min and then the absorbance was measured at 750 nm. The results are expressed as mg GAE/100 gDW.
Regarding TFC assays, the methodology described by Buitrago et al. was followed. 32 Briefly, 50 µL of ethanol was mixed with 10 µL of aluminum chloride (10%), then 10 µ L of sodium acetate (0.1 M) was added and finally 70 µL of the sample/standard to be evaluated. After 40 min, protected from light and at room temperature, the absorbance was measured at 415 nm. Absorbance was measured using the iMarkTM microplate reader.
Cytotoxicity
Cytotoxicity was measured by 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay using human dermal fibroblasts (HDFa, ATCC (American Type Culture Collection)® PCS-201-012™, Primary Dermal Fibroblast; Normal, Human, Adult) cells. First, 2 × 104 cells in 100 µL per well were placed in 96-well microplates and incubated overnight to allow adequate adherence. Then, the treatments (i.e. EUFUS-Z928 and oxybenzone or actinobacteria-derived extracts) were added completing a final volume of 200 µL. The concentrations evaluated for the crude extract were 500, 50, and 5 µg/mL and for oxybenzone were 400, 200, 100, 50, 25, 12.5, and 6.25 µg/mL. The cells with the different treatments were incubated for 24 h at optimal conditions. After this, the supernatants were replaced with fresh medium with MTT at 5 mg/mL. Then, the media were removed after one more incubation for 4 h. Finally, 50 µL of DMSO was added to each well to dissolve the formazan crystals. Absorbance at 570 nm was recorded using an Elx800™ microplate reader (BioTek Instruments Inc., Winooski, VT, USA) and the Gen5™ Reader Control Software (BioTek Instruments Inc.). The following equation was employed for viability calculations:
Measurement of UV-absorbing capacity
To estimate the UV-absorbing properties, the absorbance spectrum of the crude extract (1 mg/mL) was measured in the region of 290 to 320 nm in 1 nm steps in a quartz cuvette using a GENESYS™ 10S UV-Vis spectrophotometer (Thermo Fisher Scientific Inc., Waltham, MA, USA). Oxybenzone (30 µg/mL) was used as a benchmark UV filter. The in vitro SPF (SPFi) was calculated from the absorbance values at 290, 295, 300, 305, 310, 315 and 320 nm by applying the Mansur equation
33
:
Phylogenetic analysis by 16S rRNA gene sequence
Identification of strain EUFUS-Z928 was established by phylogenetic analysis of the 16S rRNA gene sequence using the universal primers 27F/1492R. The sequence was deposited in GenBank under accession number OM439643. Highly similar sequences were searched and retrieved using the Basic Local Alignment Search Tool. The phylogenetic tree was inferred using the Neighbor-Joining method by applying the Kimura 2-parameter. The degree of support for internal branches of the tree was assessed by bootstrapping (1000 replicates). The analyses were conducted in MEGA v11.0.10. 36
LC/MS-based characterization
A chemical characterization of the EUFUS-Z928-derived extract was performed by liquid chromatography–mass spectrometry (LC/MS) using an Agilent 6545 LC/Q-TOF (Agilent, Santa Clara, CA, USA). Chromatographic separation was conducted on a C18 column (InfinityLab Poroshell 120 EC-C18; 100 × 3.0 mm, 2.7 μm). The mobile phase consisted of 0.1% (v/v) formic acid in Milli-Q water (Phase A) and 0.1% (v/v) formic acid in acetonitrile (Phase B). The column temperature was maintained at 30°C. Samples were injected with a volume of 5 µL at a flow rate of 0.4 mL/min. The gradient profile was as follows: 0 min—15% B, 3 min—15% B, 8 min—40% B, 10 min—40% B, 14 min—70% B, 17 min—70% B, 21 min—100% B, 25 min—100% B, 29 min—15% B, and 32 min—15% B. Detection was performed using a dual AJS ESI source with a Vcap of 3500 V. The drying gas temperature was set to 8 L/min, the gas temperature to 325°C, the nebulizer pressure to 50 psi, the sheath gas temperature to 350°C, and the sheath gas flow to 11 L/min. MS analysis was performed using a qToF MS with a fragmentor voltage of 175 V, a skimmer voltage of 65 V, and an OCT RF Vpp of 750 V. A methanolic extract obtained from an uninoculated seven-day Zobell broth (subjected to the same experimental conditions) was used as a blank. The features detected in both crude extract and blank were not considered for further analyses. MS detection was performed in positive ESI mode in full scan from 70 to 1100 m/z. Throughout the analysis, two reference masses were used for mass correction: m/z 121.0509 (C5H4N4) and m/z 922.0098 (C18H18O6N3P3F24). Table 1 summarizes the optimized chromatographic and MS conditions. Detected features were annotated using StreptomeDB v3.0 37 and Natural Products Atlas v2.0 38 databases.
UV-absorbing profile of the Z928 extract.

SPFi: in vitro sun protection factor.
Oxybenzone.
in-vitro sun protection factor.
critical wavelength.
Data analysis
Data are presented as the mean and standard deviation from triplicate assays (M ± SD). For inferential analyses, the normal distribution of the residuals was evaluated by D'Agostino-Pearson and Shapiro–Wilk tests, before conducting the ANOVA (analysis of variance) and calculating Pearson's correlation coefficients. In case of failure to pass normality tests, nonparametric tests (i.e. Kruskal–Wallis and Spearman correlation) are applied. Multiple comparisons were made including actual multiplicity-adjusted p-values (Tukey–Kramer test). The analyses were run in the software GraphPad Prism v9.0.0 (GraphPad Software, LLC, San Diego, CA, US).
Results
Considering that oxidative stress is a primary mechanism of UV-induced damage, we first examined the free radical scavenging capacity of crude methanolic extracts of the 18 actinomycetes isolated from the octocoral E. fusca. The antioxidant capacity shown by the methanolic extracts of the bacterial isolates varied significantly (p < .0001) and ranged from 1.16 ± 0.03 to 4.93 ± 0.01 TEAC for DPPH and 2.49 ± 0.30 and 6.00 ± 0.06 TEAC for ABTS (Figure 1). No correlation (Spearman r = 0.261, p = .295) was found between the results obtained by the DPPH and ABTS radical scavenging assays. Interestingly, bacterial extracts showed stronger quenching activity against ABTS●+ than DPPH● (p < .0001). Considering that phenolic-type compounds are well known for their antioxidant potential, we measured their content in the methanolic extracts. TPC varied between 40.81 ± 15.16 and 111.64 ± 6.49 mg GAE/100 gDW, while no detectable levels of flavonoids were found (detection limit: 3.13 µg/mL). To observe a plausible relationship between antioxidant action and phenolic content for the studied extracts, we correlated TPC and the observed radical scavenging capacity. Only a marginally significant correlation (p = .049), but of low magnitude (r = 0.444), was found between TPC and ABTS results (Figure 1(b)). Since the Z928 extract was the most active, and the only one that quenched above 50% of both radicals (i.e. DPPH● and ABTS●+), we selected the EUFUS-Z928 isolate for further evaluation of its photoprotective potential.

Antioxidant capacity of methanolic extracts obtained from actinomycete-like isolates derived from Eunicea fusca. Free radical scavenging capacity was measured with DPPH and ABTS assays, statistically significant differences were determined by one-way ANOVA, and differences between groups were tested by Tukey's multiple comparisons test (a). Correlation of antioxidant capacity with phenolic content (b). The mean and standard deviation of triplicate assays are plotted. The results of DPPH and ABTS assays are expressed as TEAC (μmol TE/gDW), and those of TPC assays are expressed as mg GAE/100gDW.
UV radiation absorption is the primary mechanism used by the current sunscreens; therefore, in the search for new photoprotective agents, UV absorption capacity is a crucial property. The Z928 extract shows an SPFi of 6.00 ± 0.24, representing 46.33% of that shown by oxybenzone (Table 2). Unlike oxybenzone, the Z928 extract showed enhanced UVA absorbing properties, evidenced by the UVA/UVB ratio and λC values (Table 1).
Mass spectra data of the annotated features in Gordonia hongkongensis EUFUS-Z928.

Feature number ID.
Retention time (min).
quasi molecular ion [M + H]+, acetonitrile adduct [M + ACN + H]+.
Experimental.
e Calculated.
Identification confidence levels according to Schymanski et al.41.
We next investigated the cytotoxic activity of Z928 extract on HDFa cells. Oxybenzone, widely employed in the manufacture of sunscreens and known for its toxic effects, 39 was used as a reference UV filter. As shown in Figure 2, oxybenzone inhibited HDFa viability below 75% at the concentrations tested (i.e. 50, 100, and 200 µg/mL). On the other hand, the Z928 extract did not showed a toxic effect on HDFa, even at the maximum concentration evaluated (i.e. 500 µg/mL) (Figure 2).

Cytotoxic evaluation of oxybenzone and Z928 extract. Human dermal fibroblasts (HDFa cell line) were exposed for 24 h and then cell viability was quantified by colorimetric MTT assay. The mean and standard deviation of triplicate assays are plotted. Statistically significant differences were determined by one-way ANOVA and differences between groups were tested by Tukey's multiple comparisons test.
Given the photoprotective potential shown by isolate Z928, we identified the strain by sequencing the 16S ribosomal gene. The strain belongs to the genus Gordonia (family Gordoniaceae, order Corynebacteriales, class Actinomycetia, and phylum Actinomycetota). The closest Gordonia species were Gordonia didemni (NR146040; 97% coverage and 99.85% identity), Gordonia lacunae (NR044120; 100% coverage and 98.91% identity), Gordonia terrae (NR037022; 100% coverage and 99.93% identity), and Gordonia hongkongensis (NR152022; 100% coverage and 100% identity). The strain was classified as G. hongkongensis strain EUFUS-Z928 based on the percentage of identity (the 16S ribosomal gene sequence was deposited in the GenBank under accession number OM439643). This result is consistent with the clade formed between these species and strain EUFUS-Z928 in the phylogenetic tree (Figure 3). Furthermore, the identification was confirmed by whole-genome sequencing. 40

Phylogenetic tree of the bioactive actinomycete isolate. The phylogram was built using the sequence of the 16S rDNA gene. The optimal tree is shown. The accession code of the blasted strains is shown in parentheses. The percentage (for those that were >50) of replicate trees with associated taxa clustered together in the bootstrap test (1000 replicates) is displayed next to the branches. All ambiguous positions were removed for each sequence pair (pairwise deletion option). Nocardia farcinica DSM 43665 was used as an outgroup.
We also conducted an LC-MS-based characterization of the Z928 extract. A total of 92 features were detected (detailed information provided in Supplemental Table S1). The m/z values ranged from 72.94 to 1086.62, with a mean of 475.43 ± 293.43 and a median of 400.32 (Q1 = 226.13 and Q3 = 695.78). After querying the microbial-derived natural product databases, 26 compounds were putatively identified in the extract (Table 2) at confidence levels 3 and 4 according to Schymanski et al. 41
The names and biological activities described for the 13 compounds identified at level 3 are presented in Table 3. Compound
Bioactivity records of the main identified compounds at level 3 in Gordonia hongkongensis EUFUS-Z928.

Calculated logarithm of the partition coefficient between n-octanol and water.
Similarity analysis based on the FragFp descriptor (this descriptor uses a predefined database of substructures to group similar compounds together
54
) was performed to define the diversity of structural features in the isomers associated with the compounds identified at level 4 (i.e.
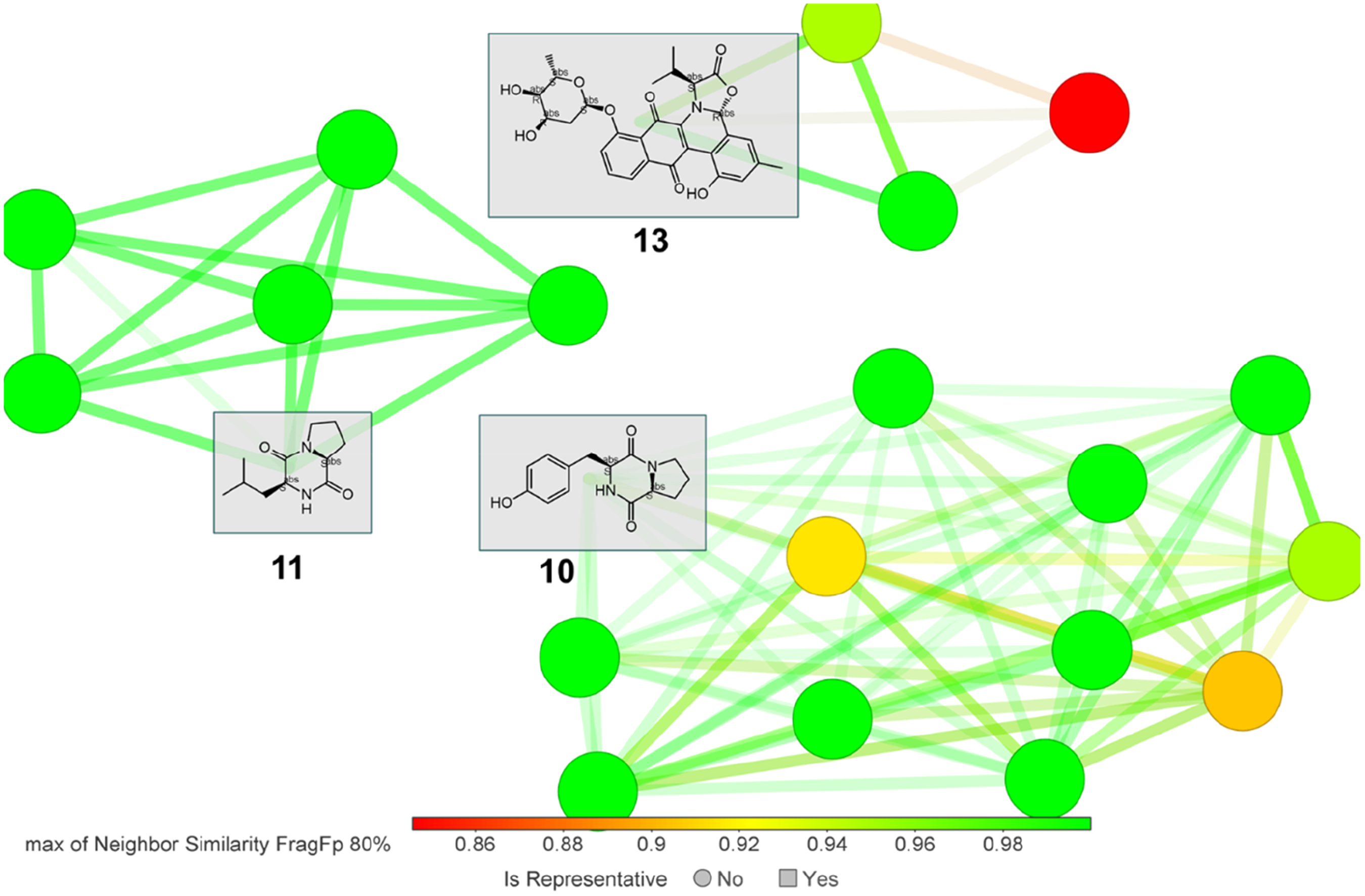
Structure similarity analysis between isomers associated with features
Discussion
UV-induced lesions result from damage caused directly by UV radiation and indirectly by oxidative bursts. A photoprotective effect can be achieved through the action of free radical scavengers, antioxidants and, naturally, the reduction of radiation load by UV absorbers. The production of metabolites with these properties in culturable microorganisms could serve as the basis for identifying renewable sources of ingredients for the development of sun protection products.
Photoprotective potential of E. fusca-derived actinomycetes
The phylum Actinomycetota is the most diverse in specialized metabolism, which is associated with a wide range of bioactive compounds of commercial interest, 55 including photoprotection-related activities. 56 In our search for culturable marine invertebrate-associated actinomycetes producing bioactive metabolites, we have evaluated the photoprotective potential of actinobacterial isolates (n = 18) from the Caribbean octocoral E. fusca. Octocorals (including E. fusca) are known for their content of bioactive compounds, many originally derived from their associated microbiota. To our knowledge, there is only one report on the composition of the E. fusca microbiome, with a 2.5% content of Actinomycetota, 57 which was consistent with the relatively small number of isolates achieved.
Among the evaluated isolates, the most bioactive (i.e. with higher antioxidant and UV-absorbing capacities) strain (EUFUS-Z928) was identified as G. hongkongensis, a rare actinomycete—i.e. non-Streptomyces—(Figures 1(a) and 4), and its isolation from E. fusca is reported for the first time. Although the genus Streptomyces is the most prolific producer of bioactive compounds among the phylum Actinomycetota, 55 recently, a growing body of literature has shown that rare actinomycetes (non-Streptomyces) are a promising source of bioactive metabolites. 58 Interestingly, no streptomycetes were identified in the E. fusca microbiome. 57 This occurrence of rare actinomycetes has also been reported in other corals (including soft corals). 59 Thus, considering our findings, E. fusca could be an interesting source of bioactive rare actinomycetes.
Antioxidant, UV-absorbing and cytotoxic effects of G. hongkongensis EUFUS-Z928
The antioxidant capacity shown by G. hongkongensis EUFUS-Z928 is comparable to that reported for other actinomycetes in DPPH and ABTS radical scavenging assays.60–65 Interestingly, in all these, the ability to quench the ABTS●+ cation-radical was superior to that of DPPH● and was independent of the solvent used in obtaining the extract (i.e. methanol or ethyl acetate). In the ABTS assay, approximately half of the strains exhibited similarly high values (Figure 1(a)), indicating comparable levels of antioxidant activity among them. Consequently, the results did not provide sufficient discriminatory power to identify strains with unique or particularly potent antioxidant compounds, possibly by the nature of this assay since ABTS can react with both hydrophilic and lipophilic antioxidants. The inability of the assay to differentiate between strains suggests that the compounds with ABTS●+ radical neutralizing capacity are widespread or occur in similar concentrations across multiple isolates. Conversely, the DPPH● test revealed that EUFUS-Z92 strain exhibited a significant difference from the others, indicating a markedly superior free radical scavenging ability, mainly related to the fact that the reaction is primarily performed in organic solvents like methanol or ethanol, making it more suitable for lipophilic antioxidants. This particular antioxidant capacity makes it a promising candidate for further investigation as a source of photoprotective compounds. Furthermore, the best performance in the DPPH assay suggests that this strain may contain unique or highly effective lipophilic antioxidant compounds, which contribute to its photoprotective potential.
The differences between DPPH and ABTS assay results could be attributed to the solubility favored in each system (i.e. hydrophilicity in ABTS vs. lipophilicity in DPPH), the stereoselectivity against each radical, and the mechanism (hydrogen atom transfer vs electron transfer) that dominates the quenching of radical. 66 Steric hindrance also plays a key role, especially against DPPH●, which is why it usually shows a lower scavenging efficiency than against ABTS●+. 67
Additionally, considering the phenolic content shown by the bacterial extracts (Figure 1(b)), compounds of a nonphenolic nature are relevant contributors to the total antioxidant capacity exhibited by EUFU-Z928. This fact has been reported for other actinomycete extracts (including rare actinomycetes), 65 and highlights the potential of EUFUS-Z928 as a renewable bioresource of novel antioxidative agents. In terms of photoprotective potential, the incorporation of antioxidants in sunscreens and skin care products has been shown to play a crucial role in the anti-UV effect; hence their demand has been growing.68,69
UV absorption is the direct mechanism by which photodamage is prevented and is therefore mainly used in sunscreens. As shown in Table 2, compounds with UV-absorbing properties were also found in the methanolic extract of EUFUS-Z928. Although SPFi was lower than that of oxybenzone (a broad-spectrum UV filter, i.e. against UV-A and UV-B), the λC and UVA/UVB ratio results suggest a preferential anti-UV-A capability (Table 2). It is crucial to recognize that SPF values serve as a proxy for the capacity to block UV-B radiation, yet they do not account for the ability to absorb UV-A radiation. This phenomenon has already been observed in other sunscreens when the ingredients are more effective in protecting against UV-A radiation. 70 It is reasonable to conclude that the high SPF values observed in this crude extract are not representative of the true efficacy of the active compounds, which are present in very low concentrations and therefore unable to contribute significantly to UV-B absorption. This is the reason why these values can be found in the analysis of natural products that are not entirely pure or enriched.71,72 Furthermore, the integration of natural extracts with commercially available synthetic ingredients represents a significant avenue of investigation, with the objective of maintaining optimal levels of photoprotection while minimizing adverse effects.73,74
Considering the aforementioned results and those of antioxidant capacity, the metabolites in the methanolic extract of EUFUS-Z928 would provide direct and indirect protection against UV-A radiation. This fact is fundamental considering that there are few (i.e. 37.74%) UV filters against UV-A, 75 whose deleterious effect is mediated through oxidative stress.10,11 Hence, the discovery of renewable sources to obtain these metabolites offers promising avenues for the improvement of sunscreen formulations and mitigate the harmful effects of UV-A exposure.
In the search for ideal/improved photoprotective agents, it is crucial to reject/avoid adverse effects. We investigated the potential cytotoxic effects on human dermal fibroblasts (using the HDFa cell line) of EUFUS-Z928 extract. Compared to oxybenzone, the results of the EUFUS-Z928 extract are promising and suggest a harmless behavior against a relevant skin cell population. Although these results and those of photoprotective potential are encouraging, it should be noted that they are based on in vitro assays and therefore must be further confirmed in in vivo assays. Nevertheless, they are an excellent starting point to explore the potential of G. hongkongensis EUFUS-Z928 as a renewable source of agents with application in the field of cosmeceuticals.
MS-based characterization of methanolic extract of G. hongkongensis EUFUS-Z928
Accurate mass measurements are an incredibly useful tool for structure-based analysis of crude extracts. 76 However, the availability and size of databases represent a major bottleneck, especially for microbial extracts. 76 For instance, most of the features detected in the methanolic extract of EUFUS-Z928 were unknown (n = 66, i.e. 71.74%). This is also partially explained by the fact that Gordonia is a rarely studied actinomycete genus. 65 Nevertheless, the experimental data allow enhanced analyses when related to biological activity results.
In the search for agents for topical application (as intended for the most photoprotectors), molecular masses >330 Da represent a desired characteristic due to their low dermal bioavailability. We found that 58.70% of the detected features were above 330 m/z. In the group of compounds whose structural information was more delimited (i.e.
Given the potential shown by G. hongkongensis EUFUS-Z928 we have sequenced its genome.
40
Interestingly, it contains a large content of specialized metabolite biosynthetic gene clusters (BCGs) (i.e. 14 clusters
40
). Particularly, terpenoids, polyketides, nonribosomal peptides, and hybrids among them, which could be involved in the biosynthesis of compounds such as
The advent of biotechnological tools presents a suitable avenue for the elucidation and exploitation of bacterial biosynthetic pathways. In this context, the rare actinomycete species G. hongkongensis emerges as a compelling subject of inquiry, harboring the potential to furnish sustainable photoprotective agents. Nevertheless, translating laboratory discoveries into industrial-scale production mandates a contemplation of scalability. The optimization of fermentation processes, downstream processing, and yield enhancement strategies assumes paramount importance in this endeavor. Challenges inherent to large-scale production, including ensuring product consistency, mitigating production costs, and adhering to regulatory standards, necessitate meticulous consideration.
The scalability of bacteria cultures offers multifaceted advantages, encompassing environmental sustainability, renewable sourcing of raw materials, and the prospect of developing novel, eco-friendly sunscreen formulations. Moreover, this pursuit aligns with the ethos of the SDGs, advocating for innovation while safeguarding ecological integrity. The isolation and characterization of G. hongkongensis EUFUS-Z928 exemplify a convergence of scientific inquiry and technological innovation intended to advance sustainable sunscreen production. However, this research primarily focuses on the in vitro stages of bacterial isolation and characterization. To fully realize the potential of G. hongkongensis EUFUS-Z928 in commercial sunscreen applications, extensive further research is necessary. This includes large-scale cultivation studies, comprehensive safety assessments, and the evaluation of long-term environmental impacts.
Conclusions
In conclusion, our study highlights the promising potential of actinomycetes, especially G. hongkongensis, as a source of photoprotective compounds. The methanolic extracts from octocoral-associated actinomycetes were subjected to analysis, during which it was discovered that G. hongkongensis EUFUS-Z928 exhibited significant antioxidant and UV-absorbing capabilities. The extract demonstrated promising results in scavenging free radicals, which is likely mediated by a variety of N-containing bioactive compounds. Additionally, it exhibited UV-absorbing properties, particularly in the UV-A range, and demonstrated no cytotoxic effects on human dermal fibroblasts, indicating its safety for cosmetic applications. Our findings highlight the potential of marine actinomycetes for developing ecofriendly photoprotective agents, which aligns with global sustainability goals.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241272454 - Supplemental material for Photoprotection-related properties of a raw extract from Gordonia hongkongensis EUFUS-Z928: A culturable rare actinomycete associated with the Caribbean octocoral Eunicea fusca
Supplemental material, sj-docx-1-sci-10.1177_00368504241272454 for Photoprotection-related properties of a raw extract from Gordonia hongkongensis EUFUS-Z928: A culturable rare actinomycete associated with the Caribbean octocoral Eunicea fusca by Jeysson Sánchez-Suárez, Luisa Villamil, Ericsson Coy-Barrera and Luis Díaz in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504241272454 - Supplemental material for Photoprotection-related properties of a raw extract from Gordonia hongkongensis EUFUS-Z928: A culturable rare actinomycete associated with the Caribbean octocoral Eunicea fusca
Supplemental material, sj-docx-2-sci-10.1177_00368504241272454 for Photoprotection-related properties of a raw extract from Gordonia hongkongensis EUFUS-Z928: A culturable rare actinomycete associated with the Caribbean octocoral Eunicea fusca by Jeysson Sánchez-Suárez, Luisa Villamil, Ericsson Coy-Barrera and Luis Díaz in Science Progress
Footnotes
Acknowledgments
We thank Sven Zea for coral sampling and Juan Armando Sánchez for coral identification. We would like to thank the staff of Marine Bioprospecting Laboratory—LabBIM—at “INVEMAR” for logistic support, especially Javier Gómez-León for facilitating our research activity. In addition, we thank Jorge Rodríguez for his assistance with laboratory procedures. The samples used by this research were of Colombian origin, and they were obtained according to Amendment No. 5 to ARG Master Agreement No. 117 of 26 May 2015, granted by the Ministry of Environment and Sustainable Development, Colombia. Thanks to the Department of Norte de Santander (Colombia) for the JSS PhD scholarship funding through the Minciencias Bicentenario grant.
Author contribution statement
Conceptualization: J.S.-S.; methodology: J.S.-S.; software: J.S.-S.; validation: J.S.-S. and E.C.-B.; formal analysis: J.S.-S.; investigation: J.S.-S.; resources: L.V., E.C.-B., and L.D.; data curation: J.S.-S. and E.C.-B.; writing—original draft preparation: J.S.-S.; writing—review and editing, L.V., E.C.-B., and L.D.; visualization: J.S.-S.; supervision: L.V. and L.D.; project administration: L.V. and L.D.; funding acquisition: L.V. and L.D. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministerio de Ciencia, Tecnología e Innovación, Universidad de La Sabana, General Research Directorate (Grant No. 80740-168-2019, Project ING-175-2016).
Ethics approval and informed consent
This study did not involve any local people or patients. Thus, the authors did not have ethics approval nor informed consent.
Supplemental material
Supplemental material for this article is available online.
Author biographies
Jeysson Sánchez-Suárez was born in Cucuta, Norte de Santander, Colombia. He received his master's degree from Universidad Nacional de Colombia and his doctorate from Universidad de La Sabana, Colombia. Currently, he is employed at the Schepens Eye Research Institute, Harvard Medical School, and also serves as a researcher in the Research Group in Ecology and Biogeography at Universidad de Pamplona, Colombia. His research interests encompass natural product exploration, pharmacology, microbiology, and bioinformatics.
Luisa Villamil is a Marine Biologist from Universidad Jorge Tadeo Lozano in Colombia. She has a PhD in Biological Sciences and Aquaculture from the Universidad de Vigo in Spain. She was a postdoctoral researcher at the University of Rhode Island and a visiting Professor at the Woods Hole Oceanographic Institute. She is currently the director of the PhD program in Biosciences at Universidad de La Sabana.
Ericsson Coy-Barrera, born in Bogotá, Colombia, completed his undergraduate studies in Chemistry at Universidad Nacional de Colombia. Subsequently, he pursued his master's and PhD in Organic Chemistry at the same university. Additionally, he worked on organic synthesis during a research stay at the University of Potsdam, Germany. He was subsequently appointed as a professor of Organic Chemistry at MU Nueva Granada, Colombia, where he founded the Bioorganic Chemistry Laboratory. Here, he initiated his own research program focused on lead finding from nature, employing holistic and rational approaches based on metabolomics, chemoinformatics, molecular modeling, and organic synthesis.
Luis Díaz was born in Bogotá, Colombia. He received his undergraduate degree in chemistry and doctorate in chemistry from Universidad Nacional de Colombia. He is currently an Associate Professor at the Universidad de La Sabana, Faculty of Engineering, and the leader of the Bioprospecting Research Group. Senior Research of the Minister of Science and Technologies. His research interests include exploring natural products, pharmacology, microbiology, and biochemistry.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.