Abstract
Objective
Parent artery and hemodynamics are related to intracranial aneurysms (IAs) rupture. Although proximal parent artery stenosis (PPAS) can both change parent artery configuration and hemodynamics, correlation between PPAS and IAs rupture has not been clearly clarified in large sample size. This study aims to explore the correlation between PPAS and IAs rupture in a large Chinese cohort.
Methods
A retrospective cross-sectional study was conducted. Imaging and clinical data were collected from consecutive patients with IAs diagnosed by CTA between January 2013 and December 2022. Two neuroradiologists independently assessed the characteristics of the IAs. To minimize the effect of confounding variables, propensity score matching (PSM) was applied.
Results
After PSM, no significant differences were found between groups for interference factors. However, IAs in the PPAS group had a significantly lower proportion of ruptures compared to the non-PPAS group (p = 0.031). Additionally, unruptured aneurysms had a significantly higher prevalence of PPAS compared to ruptured aneurysms (p = 0.011). There was no significant difference in rupture rates between non-PPAS IAs with other intracranial artery stenosis (IAS) and those without IAS (p = 0.596). And ruptured non-PPAS IAs showed a significantly higher incidence of IAS compared to unruptured non-PPAS IAs (p = 0.010).
Conclusion
Non-PPAS may be associated with a higher risk of aneurysm rupture. In contrast, PPAS may be associated with unruptured aneurysm.
Keywords
Introduction
Intracranial aneurysms (IAs) are a common cerebral disease with low rupture rate(≤1% per year). 1 Once ruptured, the mortality and morbidity are high. 1 Surgery and interventional preventive treatment both have their own risks, so it is crucial to more accurately assess the risk of aneurysm rupture. Proximal parent artery stenosis (PPAS) has been suggested as a potential high-risk factor for rupture due to its effects on hemodynamics. 2 Hemodynamics play an important role in the formation, growth, and rupture of IAs. 3 Narrowed artery lumen can lead to increased inflow velocity and wall shear stress (WSS) in downstream aneurysms. 2
However, high WSS is typically associated with aneurysm formation, while low WSS is more often linked to rupture. 4 Low WSS can trigger an inflammatory response in the endothelium, leading to instability in the aneurysm wall. 3 Therefore, it is too simplistic to assume that increasing WSS will automatically raise rupture risk. In fact, it seems that elevated WSS might even lower the risk of rupture. Obviously, treating it as a high-risk factor can have disastrous consequences.
In addition, many factors linked to aneurysm rupture may overlap with those associated with intracranial artery stenosis (IAS). Previous studies2,5 often overlooked this due to limited sample sizes. Furthermore, IAS can signal undeniable damage to the arterial walls, which may also heighten the risk of rupture and should not be ignored or incorrectly attributed to hemodynamic changes.
To better understand the rupture risk in cases of proximal parent artery stenosis, we conducted a large-scale, cross-sectional retrospective study. In this study, we controlled the most widely recognized risk factors to minimize interference, and analyzed the relationship between maternal proximal artery stenosis, intracranial artery stenosis and aneurysm rupture, in order to provide more references for the treatment of aneurysms complicated with artery stenosis.
Materials and methods
The study protocol was reviewed and approved by the local Ethics Committee (Beijing Tiantan Hospital, Capital Medical University; KY-2020-016-02; 2020.05.10). All procedures strictly adhered to the ethical principles of the Declaration of Helsinki and relevant national regulations. Due to the retrospective cross-sectional study design, informed consent was waived, and participant who included in this study have signed informed consent forms, authorizing the use of information for future scientific research. Retrospective data analysis followed the ethical guidelines applicable at the time of the original data collection (Declaration of Helsinki, 2013 revision). Participant confidentiality was protected through data anonymization, with all imaging and clinical data stored in password-protected hospital information systems. In addition, all data have de-identified all patient details. And the reporting of this study conforms to STROBE guidelines. 6
Subjects
The retrospective cross-sectional study consecutively included imaging and clinical baseline data from 2575 patients diagnosed with saccular intracranial aneurysms (IAs) at Beijing Tiantan Hospital between January 2013 and December 2022. Patients with traumatic or inflammatory IAs, cerebral vascular malformations, or Moyamoya disease were excluded. Those with multiple aneurysms were also excluded due to potential bias in determining the site of rupture and probability of discovering unruptured aneurysms. Patients with a history of intracranial hemorrhage (including subarachnoid hemorrhage, SAH) were excluded to avoid bias from identifying pre-bleeding status and incomplete previous medical records. Aneurysms located in the circle of Willis and basilar artery were also excluded due to the complexity of collateral blood flow in those areas. Patient demographic data, including age, sex, blood lipids, blood glucose, blood pressure, and smoking history, were recorded.
Feature acquisition
All data were anonymized before processing. Two neuroradiologists, with 12 and 13 years of experience respectively, independently assessed each aneurysm's origin, determined whether it was a bifurcation aneurysm, checked for the presence of a daughter sac, and evaluated for proximal parent artery stenosis using the reconstructed three-dimensional CTA (GE Discovery CT750 HD or GE Revolution CT or Philips iCT 256) datasets. A 50% reduction in arterial diameter is defined as arterial stenosis. Proximal stenosis is defined as the CTA data of the participants at the time of visit indicating that the parent artery upstream of the aneurysm is narrowed (>50%), and the distance between the stenosis and the neck of the aneurysm is less than 9 mm. 2 In cases of disagreement, a third neuroradiologist with 21 years of experience resolved the discrepancies. Aneurysm size was measured as the maximum diameter, and aneurysms were classified as either ruptured or unruptured. Patient demographic data, including age, sex, blood lipid levels, blood glucose, blood pressure, and smoking history, were gathered from clinical records.
Grouping
IAs were classified into two categories based on the condition of the proximal parent artery: the PPAS group (with proximal parent artery stenosis) and the non-PPAS group (without proximal parent artery stenosis). The non-PPAS group was further divided into the IAS group (with intracranial artery stenosis) and the non-IAS group (without intracranial artery stenosis).
Furthermore, IAs were categorized into ruptured and unruptured groups based on their status. Within the non-PPAS group, those that were ruptured were labeled as non-IAS-R, while those that were unruptured were labeled as non-IAS-U. Aneurysms were also classified into small size (size ≤ 7 mm) and large size (size > 7 mm). 7 And participants were classified into elder (age > 70 years) and younger (age ≤ 70 years). 7
Statistical analysis
The chi-square test, continuity correction, and Fisher's exact test were used to evaluate the distribution of factors between groups and to assess the correlation between risk factors and ruptured aneurysms. Propensity score matching (PSM) (at a 1:1 ratio) was employed to minimize the influence of other factors with significant differences. Differences were considered statistically significant at p < 0.05. All data were analyzed using the SPSS software (version 23.0; SPSS Inc., Chicago, IL, USA).
Results
A total of 3345 aneurysms (662 ruptured; 2683 unruptured) from 2575 participants were included, while 1320 aneurysms from 550 participants were excluded because they were multiple aneurysms and 948 aneurysms from 948 participants were excluded because of located at Willis circle. Ultimately, 1077 aneurysms (128 ruptured; 949 unruptured) from 1077 participants were analyzed in this study. Some cases are shown in Figure 1.

Some cases of aneurysms and arterial stenosis (A. ruptured internal carotid artery aneurysm combined with parent artery stenosis; B. unruptured middle cerebral artery aneurysm combined with parent artery stenosis; C. ruptured anterior cerebral artery aneurysm combined with middle cerebral artery stenosis) (red arrow: aneurysm; yellow arrow: arterial stenosis) (CTA-3D: the three-dimensional reconstruction results of computed tomography angiography; CT: the scan result of computed tomography).
Comparison between PPAS and non-PPAS group
Before PSM, there were 154 in the PPAS group and 923 aneurysms in the non-PPAS group. The distribution of several factors showed significant differences between the two groups: female (p < 0.001), bifurcation (p = 0.002), ruptured aneurysms (p = 0.012), smoking history (p = 0.003) and large size (p < 0.001). After PSM, each group contains 112 aneurysms with no significant differences in the distribution of including factors. However, as shown in Figure 2, the non-PPAS group still had a higher proportion of ruptured aneurysms (p = 0.031). More details were shown in Table 1.

Comparison results between each group after propensity score matching (A. ruptured aneurysms distribution between non-PPAS group and PPAS group; B. proximal parent artery stenosis distribution between ruptured group and unruptured group; C. ruptured aneurysms distribution between non-IAS group and IAS group; intracranial artery stenosis distribution between non-PPAS-R group and non-PPAS-U group).
Comparison results of baseline data between PPAS group and non PPAS group, IAS group and non IAS group.

Comparison between IAS and non-IAS group
Before PSM, there were 395 aneurysms in the IAS group and 528 in the non-IAS group. Significant differences in the distribution of the following factors were observed: female (p = 0.001), bifurcation (p < 0.001), sac (p = 0.033), hypertension (p = 0.017), ruptured aneurysms (p = 0.006), and elder age (p = 0.002). After PSM, there were 344 aneurysms in each group and there were no significant differences in the distribution of including factors between two groups. And as shown in Figure 2, there was no significant difference in the proportion of ruptured aneurysms between the two groups (p = 0.596). More details were shown in Table 1.
Comparison between ruptured and unruptured group
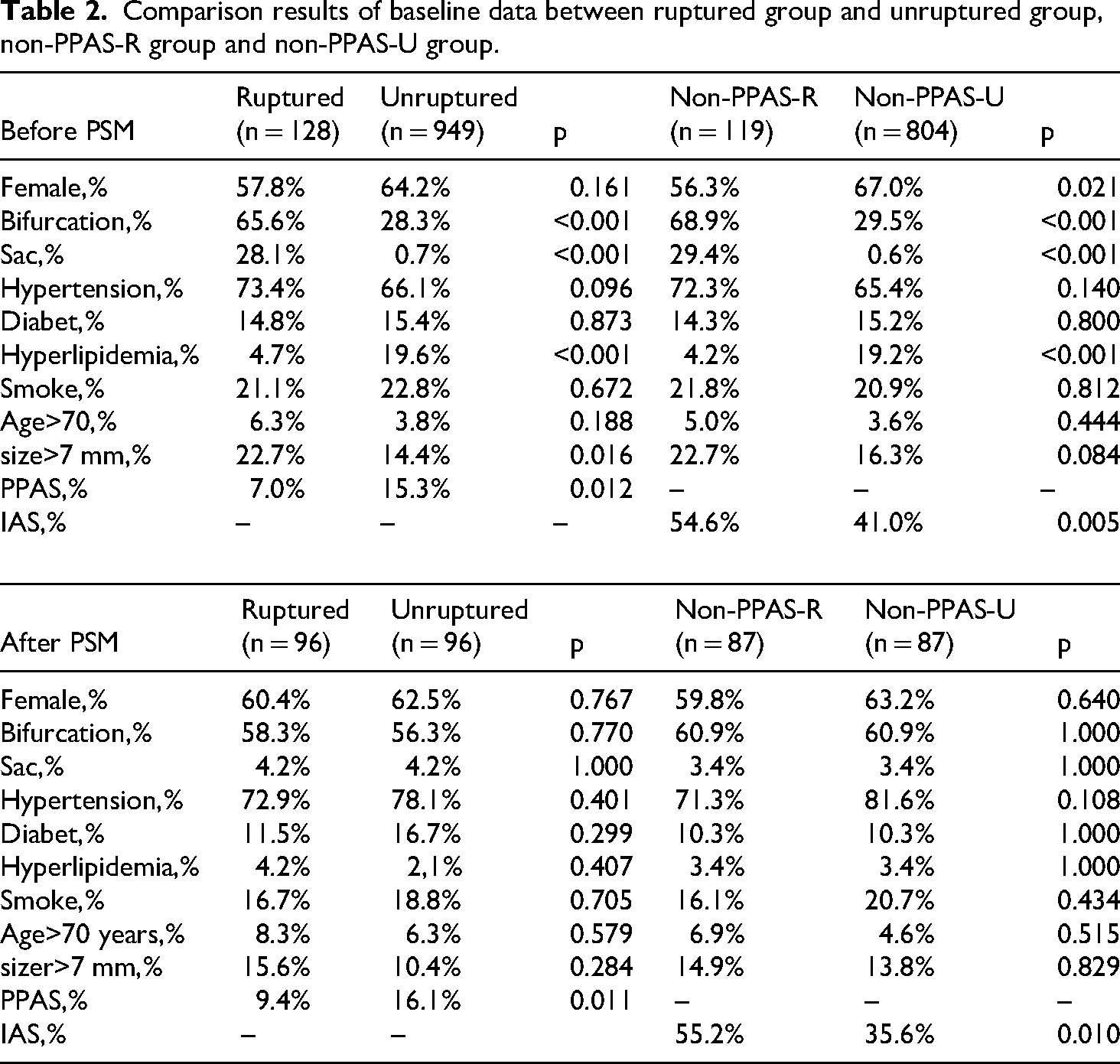
Before PSM, there were 128 aneurysms classified as ruptured and 949 as unruptured. Significant differences were found in the distribution of bifurcation (p < 0.001), sac (p < 0.001), hyperlipidemia (p < 0.001), PPAS (p = 0.012), and large size (p = 0.016) between the two groups. After PSM, each group contained 96 aneurysms, with no significant differences in the distribution of these factors. However, as shown in Figure 2, the ruptured group still had a lower PPAS ratio than the unruptured group (p = 0.011). More details are available in Table 2.
Comparison results of baseline data between ruptured group and unruptured group, non-PPAS-R group and non-PPAS-U group.
Comparison between non-PPAS-r and non-PPAS-u group
Before PSM, there were 119 aneurysms in the non-PPAS-R group and 804 in the non-PPAS-U group. Significant differences were observed in the distribution of female (p = 0.021), bifurcation (p < 0.001), sac (p < 0.001), hyperlipidemia (p < 0.001), and IAS (p = 0.011) between the two groups. After PSM, there were 87 aneurysms in each group, with no significant differences in the distribution of these factors. Notably, as shown in Figure 2, the non-PPAS-R group had a higher IAS ratio than the non-PPAS-U group (p = 0.005). More details can be found in Table 2.
Discussion
Although PPAS has received limited research in previous studied we have reviewed, it plays a significant role in assessing rupture risk and treatment planning. Feng et al. 8 analyzed 507 IAs and found that severity of atherosclerosis stenosis is associated with ruptured aneurysms, which is similarly with our results. However, no significant association between parent artery stenosis and rupture was observed, which might be limited by the size of the data. Tallarita et al. 9 retrospectively reviewed 53 carotid artery stenosis participants with 63 concomitant unruptured intracranial aneurysms (UIAs) and found that carotid artery revascularization can be performed safely without an increased 30-day or late-term risk of rupture. However, ignoring the distance between the stenosis and the aneurysm, the data volume was not persuasive enough and the bias was not controlled.
In this study, we found that the ratio of PPAS is lower in the unruptured group compared to the ruptured group. Additionally, the proportion of ruptured aneurysms is lower in the PPAS group compared to the non-PPAS group. This suggests that proximal parent artery stenosis may reduce the rupture risk of IAs. PPAS usually leads to increased WSS in aneurysms.2,5 Lu et al. 5 Analyzed 8 aneurysms with PPAS and found that both eccentric and concentric stenosis resulted in increased average WSS and average blood flow velocity at the neck of the aneurysm, correlating with the severity of the stenosis. However, our study did not investigate the impact of varying degrees of PPAS on the risk of aneurysm rupture, a larger prospective study for further research. Moreover, Antonov et al.2 established a model and proven that WSS usually increased in aneurysms with PPAS.
WSS usually plays an important role in aneurysms formation, development and rupture. 3 Previous research indicates that high WSS is usually associated with aneurysm formation 10 and low WSS is usually associated with aneurysm rupture. 4 High WSS can injury arterial wall, decreased arterial tone, promote aneurysms formation and maintain their development. 3 Low WSS can cause inflammation, gradually thicken the aneurysm wall and finally leading to rupture. 3 And aneurysms can transfer from mechanisms dominated by high WSS to those influenced by low WSS, especially when their size increases. 11 Therefore, appropriately increasing WSS could potentially reduce the risk of rupture, particularly in aneurysms operating under a low-WSS mechanism (usually in larger aneurysms). This may explain the lower proportion of ruptured aneurysms observed in those with PPAS. Furthermore, based on this theory, the high WSS caused by artery stenosis may also promote the formation of aneurysms. Héman et al. 12 prospectively collected a series of participants with ICA stenosis and found that the proportion of aneurysms in the collected participants was higher than that in the general population.
Inflammation serves as a critical pathophysiological component throughout the progression of atherosclerosis, 13 a process that shares mechanistic similarities with aneurysm formation. 11 Obviously, intracranial aneurysms have a non-negligible association with vascular stenosis. Particularly, aneurysms with PPAS demonstrate anatomical proximity to stenotic inflammatory foci and may exhibit heightened inflammatory activity compared to their counterparts in other locations. While these pathophysiological characteristics suggest a potential influence on aneurysm initiation and progression, the precise impact of such inflammatory microenvironments on aneurysm incidence and rupture risk needs systematic research.
Clinically, symptomatic intracranial artery stenosis may inadvertently enhance the detection probability of unruptured aneurysms. This observational bias was partially mitigated through comparative analysis between IAS and non-IAS group. However, the complex interactions among hemodynamic pressure, inflammation and the pathobiology of aneurysms require further research and clarification.
Participants with artery stenosis may receive some treatments, such as aspirin, to prevent stroke—treatments that can also reduce the risk of aneurysm rupture 14 —this does not imply that the lower rupture risk associated with PPAS is solely due to these treatments. After PSM, we found that aneurysms in the IAS group, which typically receive similar treatments, did not exhibit a lower proportion of ruptured aneurysms. In fact, aneurysms in the non-PPAS-R group even showed a higher proportion of IAS. Thus, in the absence of unique changes, PPAS appears to correlate with a higher rupture risk, which aligns with our intuition.
Moreover, PPAS is a very important factor not only for treatment planning but also for surgical strategies. Cebral et al. 15 analyzed the hemodynamics of aneurysms rupture after flow diverter treatment and found that the combination of proximal stenosis reduction and flow diverter implantation can lead to dramatic hemodynamics change, potentially increasing the risk of rupture. In addition, abnormal high WSS can also contribute to rupture. Zhang et al. 16 analyzed 162 participants harboring anterior communicating artery (ACoA) aneurysms and found that high wall shear stress beyond a certain range in the parent artery could predict the risk of rupture. Cebral et al. 17 analyzed 9 aneurysms and found that 8 aneurysms ruptured at abnormal high WSS region. Such phenomenon is not difficult to understand. For the aneurysm area under a high-WSS mechanism, higher WSS means more severe damage. Obviously, PPAS should be evaluated with other factors together rather than only considered as a low risk factor.
Limitation
This study has several limitations. First, it was a single-center, retrospective cross-sectional study with limited sample size, and most of the participants included were local residents, particularly those with ruptured aneurysms. This limited population may affect the generalizability and accuracy of the findings. Second, most aneurysms were treated shortly after discovery, and the lack of long-term follow-up makes it difficult to verify the results. Third, because of strict inclusion and exclusion criteria, the conclusions may not be applicable to all aneurysms, such as those that are multiple or located in the circle of Willis. Finally, this study lacks in vivo experimental verification, and the mechanism of the phenomenon can only be inferred based on existing research. A multicenter prospective cohort study with larger sample size incorporating pathological detection experiments will enhance the credibility of the research results.
Conclusion
Non-PPAS may be associated with ruptured aneurysms. Ruptured aneurysms exhibit a lower proportion of PPAS, while non-PPAS aneurysms have a higher proportion of ruptured cases in this study.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251346013 - Supplemental material for Association between intracranial artery stenosis and aneurysm rupture risk: Proximal stenosis as a potential protective factor
Supplemental material, sj-docx-1-sci-10.1177_00368504251346013 for Association between intracranial artery stenosis and aneurysm rupture risk: Proximal stenosis as a potential protective factor by Yudi Tang, Jian Lv, Peike Chen, Junqiang Feng, Xiaoyan Wang, Dachao Wei, Siming Gui, Jia Jiang, Zhengkun Xu, Yan Zhao, Yuhua Jiang, Youxiang Li and Peng Liu in Science Progress
Footnotes
Acknowledgements
In the writing manuscript, the artificial intelligence tool DeepSeek (R1) is used to check for grammar errors. The author gratefully acknowledges both the AI system and its development team for language improvement.
Author contributions
Yudi Tang: Conceptualization, Methodology, Investigation, Writing-Original Draft, Formal analysis; Jian Lv: Data Curation, Investigation; Peike Chen: Investigation; Junqiang Feng: Data Curation, Formal analysis; Xiaoyan Wang: Data Curation, Investigation, Formal analysis; Dachao Wei: Formal analysis, Investigation; Siming Gui: Data Curation, Investigation; Jia Jiang: Data Curation; Zhengkun Xu: Formal analysis, Software; Yan Zhao: Formal analysis; Yuhua Jiang: Methodology, Data Curation; Youxiang Li: Funding acquisition, Data Curation, Project administration, Resources; Peng Liu: Writing-Original Draft, Methodology, Writing-Review & Editing, Supervision. Agreement to be accountable for all aspects of work: Peng Liu.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Beijing Municipal Science and Technology Project (grant number Z231100004823009).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
