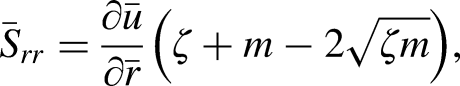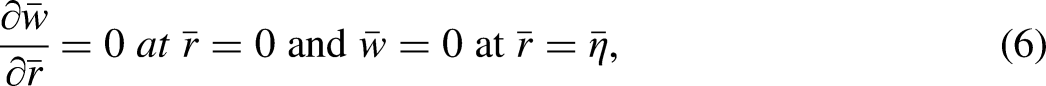Abstract
This research article interprets the computational fluid dynamics analysis on blood flow inside a symmetric stenosed artery. The current problem models the blood flow inside the left coronary artery as having a symmetric stenosis in the central region. A comprehensive physiological examination of coronary artery disease is numerically evaluated by using the computational fluid dynamics toolbox Open-Field Operation And Manipulation. There are no assumptions of mild stenosis taken into account since the considered stenosis has an exactly measured length, height and position, etc. The blood flow problem is modeled for the non-Newtonian Casson fluid with unsteady, laminar, and incompressible flow assumptions. The underlying problem is solved numerically in its dimensional form. A thorough graphical analysis is provided on the blood flow simulations, pressure profile, velocity line graphs, pressure line graphs, and streamlines for the left coronary artery having a symmetric stenosis formation. The considered artery is divided into three sections, i.e. pre-stenosis, post-stenosis, and stenosis region, and the velocity and pressure line graphs are plotted for these considered regions. The graphical illustrations provide a detailed analysis of how the blood flow is affected inside the left coronary artery due to coronary artery disease. These pre- and post-stenosis velocity line graphs reveal two intriguing results: In the pre-stenosis zone, the velocity increases with increasing axial coordinate length, whereas in the post-stenosis region, the velocity decreases with rising axial coordinate length. It is evident that as the flow moves toward the stenosis region, the flow profile rises; yet, after passing through the stenosis zone, the flow profile begins to fall as the flow moves away from the stenosis region.
Introduction
Stenosis mostly occurs in the left coronary artery due to plaque collection on the inside walls of the artery. The prime reasons for plaque collection are lack of physical activities like exercise, eating fats and oils, smoking and high cholesterol, etc. This is termed coronary artery disease (CAD). CAD can cause blood clots and stenosis inside the artery that can result in a stroke or heart attack in severe cases. The treatments used for CAD include balloon/laser angioplasty, atherectomy, and stent placement. Heart disease is a prime reason for death all around the world. The statistical data
1
shows that cardiovascular disease is the major death cause in the United States and one patient dies every 34 s due to CAD. In 2020,
2
the statistical data shows that CAD is the most common heart disease, and more than 20 million adults have CAD. United States
3
has spent more than 229 billion dollars on cardiovascular diseases from 2017 to 2018. The left coronary artery has a diameter of 3–4 mm which is slightly smaller as compared to the drinking straw width and it has a length of 10.5
Haldar 7 modeled the arterial blood flow by considering the distinct formations of stenosis with their effect on flow resistance. He revealed in his study that the highest flow resistance is observed for the symmetric formation of stenosis. Ku 8 presented a theoretical model of blood flow that reveals the unsteady flow in arteries with turbulence phenomenon near stenosis regions. In most of the cases, many researchers have considered the non-Newtonian models to discuss the blood flow in arteries, since the non-Newtonian models provide a more realistic approach to model hemodynamics. Mandal 9 modeled the arterial blood flow issue with a mild formation of stenosis. He interpreted numerical solutions for this blood flow issue by considering the non-Newtonian Power law fluid model. Siddiqui et al. 10 mathematically modeled the arterial blood flow issue for a stenosed artery by using the Casson non-Newtonian model. Venkatesan et al. 11 numerically interpreted the non-Newtonian Casson model as blood flow inside a narrow stenosed region of an artery in comparison to the non-Newtonian Bingham and Herschel Bulkley fluid. Carvalho et al. 12 modeled the review issue on the blood flow analysis in a coronary artery. Some recent studies on the blood flow analysis for stenosed arteries are referred to as.13–15 Further, some more relevant scientific literature on blood flow through a stenosed segment with endoscopic catheter applications are provided.16–22 Akhtar et al. 23 already modeled the steady Casson fluid blood flow problem for a multiple stenosed artery considering cases of both symmetric addition to non-symmetric stenosis and provided an exact solution for the problem. In the current study, we have modeled the unsteady Casson fluid blood flow problem for a single symmetric formation of stenosis and provided numerical solutions for the problem.
This issue provides the blood flow analysis for the left coronary artery having a symmetric formation of stenosis at the center. The problem is modeled for the non-Newtonian Casson fluid which provides a better approximation to consider the non-Newtonian nature of blood. There is no assumption of mild stenosis formation used in this study as the considered stenosis has a fixed length, height and position, etc. The problem is modeled for an unsteady, laminar, incompressible, non-Newtonian Casson fluid flow. The present problem is solved numerically by using the computational fluid dynamics (CFD) toolbox Open Field Operation And Manipulation (FOAM) which interprets the governing partial differential equations in their dimensional form. This is a theoretical study model, and it is the first research article that models the numerical simulation work on blood flow inside a symmetric stenosed coronary artery. There is no experimental study present on this topic to provide a comparative analysis. We are already working on some more similar models. In future work, we will provide a comparative study of this theoretical model. An intensive graphical analysis is provided on the blood flow profile, and pressure profile for this stenosed left coronary artery. The flow iterations are added for a detailed analysis that incorporates an extensive graphical analysis for
Mathematical formulation
The present problem is modeled for an unsteady, incompressible, laminar, non-Newtonian blood flow inside a stenosed left coronary artery. The geometry of the model is given in Figure (1a) and (1b) shows the pre, post, and main stenosis regions with

(a) Geometrical model of left coronary artery with a symmetric stenosis at center. (b) Pre-stenosis, stenosis and post-stenosis locations with different
The wall of the artery with a symmetric formation of stenosis in the central region is considered by the following mathematical expression given as
The existing blood flow problem is modeled for the non-Newtonian Casson fluid since it is the most extensively used model for blood flow and we can have more realistic simulation results. The governing equations for a non-Newtonian flow problem in cylindrical coordinates are given as
23
The boundary conditions are set for an axially symmetric flow with no slip at walls. The mathematical equations used for this set of boundary conditions are as follows.
Numerical solution
The partial differential equations that govern this blood flow problem are solved numerically. The basic working principle of this program is the finite volume Method in which the entire domain volume is discretized into small volume elements. Then the considered differential equation is integrated over these small-volume element cells. The present problem case is coded for an unsteady, incompressible, non-Newtonian Casson fluid using the C + + toolbox in Open-FOAM. The partial differential equations are solved in their dimensional form. The mesh diagrams of the left coronary artery are given by Figures (2a) to (2c). Figure 2(a) shows the three-dimensional (3D)-mesh diagram of the considered artery. More precise refinement of mesh is used in the central stenosed region and around the corners of the artery. Figure 2(b), the two-dimensional (2D) slice mesh diagram also shows a more precise meshing of the central stenosed region and corners as well. Figure 2(c) is the clip or side view of the arterial mesh diagram. The case is set up for

(a) 3D-Mesh representation of symmetric stenosis. (b) 2D-Mesh representation of symmetric stenosis (a slice view). (c) Mesh representation of symmetric stenosis (a clip view). 3D: three-dimensional; 2D: two-dimensional.
Finite volume method
The prime physical models and their practical engineering applications require the handling of complex partial differential equations numerically. Open-FOAM provides the facility to solve such complex models numerically by utilizing the finite volume method. To solve complex PDE's, the FVM is a reasonable choice. FVM provides a better approach to deal with non-linear conservation laws, to handle complex curve geometries, etc. The discretization via finite volume method is provided in detail by Jasak and Tukovic, 24 in which the complex set of partial differential equations that models the physical problem is transformed into a set of algebraic equations at the discrete set of small-scale volume elements in time and space. An arbitrary polyhedral control volume (CV) is given in Figure 1(c).
Point P is assumed to be located at the centroid of CV. We consider the following equation over a CV as
Results and discussion
The partial differential equations are evaluated numerically using the CFD toolbox in Open-FOAM. The graphical simulations are presented here to interpret the blood flow inside the left coronary artery having symmetric shape stenosis in the central region. The governing equations, flow simulations, and geometries are provided for a 2D flow profile. Figures (2a) and (2c) are just the 3D mesh diagram views of the considered geometry. Figure (3) depicts the graphical simulations of the blood flow profile for data ranging from

(a)

(a) Pressure profile at t = 1. (b) Pressure profile at t = 2. (c) Pressure profile at t = 3. (d) Pressure profile at t = 4. (e) Pressure profile at t = 5. (f) Pressure profile at t = 6. (g) Pressure profile at t = 7. (h) pressure profile at t = 8. (i) Pressure profile at t = 9. (j) Pressure profile at t = 10.

(a) Pre-stenosis region velocity profile at t = 10. (b) Post-stenosis region velocity profile at t = 10. (c) Stenosis region velocity profile at t = 10.

(a) Pre-stenosis pressure profile at t = 10. (b) Post-stenosis pressure profile at t = 10. (c) Stenosis region pressure profile at t = 10.

(a) Streamlines at t = 1. (b) Streamlines at t = 3. (c) Streamlines at t = 8. (d) Streamlines at t = 10.
In Figures 3(a) to (j), the blood flow simulations inside the stenosed left coronary artery are presented for time
In Figures 4(a)–(j), the pressure profile for this flow inside the left coronary artery is provided for time
Figures 5(a) to (c) provide the line graphs of the velocity profile for pre-stenosis, post-stenosis, and stenosis regions, respectively. These results are presented for the final time value
Figure 6(a) to (c) present the pressure profile line graphs for pre-stenosis, post-stenosis, and stenosis regions respectively. In Figure 6(a), the pre-stenosis pressure profile line graphs are plotted against
Figure 7(a) to (d) provide streamlined graphs for blood flow inside the stenosed left coronary artery. The streamline graphs are plotted for time values
Conclusions
The blood flow issue is modeled for the left coronary artery featuring a symmetric stenosis formation due to plaque collection at the central region. The non-Newtonian nature of blood is tackled via the non-Newtonian Casson model in this study. The major outcomes are given as
The interpretation of blood flow numerical simulations on this arterial stenosis segment provides the necessary data for the surgical purposes of these stenosis segments like the location of stenosis, shape, and formation of stenosis can be evaluated through this study. Once the severeness, shape, location, and formation of the stenosis segment is known then the method that can be used for the removal of this CAD can be decided like whether an angioplasty or a bypass is required. Angioplasty is good for a mild case of stenosis, otherwise a bypass is required for a severe stenosis. The flow problem is modeled for a laminar non-Newtonian model, but a mild turbulent phenomenon can be seen in the streamlined graphs. This turbulence is occurring just ahead of the stenosis segment. The reason for this turbulence flow occurrence is the presence of a stenosis segment. Since a slight turbulence phenomenon is observed in real-life blood flow problems of stenosed arteries. Thus, our results also verify this real-life turbulence phenomena for blood flow through the stenosis segment. Since the stenosis in the center region under consideration is symmetrical in shape, all these blood flow simulations display an axially symmetric blood flow. When compared to the pre- or post-stenosis sections, a greater velocity range is seen inside the stenosis zone. Only the velocity magnitude within the stenosis region increased as a result of the stenosed walls; nevertheless, the overall flow profile decreased as a result of the constricted artery walls. When compared to the pre- or post-stenosis regions, the pressure profile is lowest inside the stenosis region.
Footnotes
Nomenclature
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The researcher is demonstrating that they have adhered to the accepted ethical standards of a genuine research study.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia through project number (IFPIP- 666-135-1443) and King Abdulaziz University, DSR, Jeddah, Saudi Arabia.
Author biographies
Salman Akhtar has his part in the mathematical modeling of physical problem, finite volume solution section, Open-FOAM software and graphical simulations, diagrams, writing original draft, and interpretation of data used for the article.
Zahir Hussain has his role in the supervision, analysis, review, and validation of results.
Sohail Nadeem has his part in supervision, analysis, review, and validation of results.
I. M. R Najjar has his role in analysis, critical revision of manuscript, validation of results, and interpretation of data for the article.
A.M Sadoun has his role in the critical revision of manuscript, validation of results, and methods, analysis, and interpretation of data.