Abstract
Purpose
It is currently controversial whether renal artery stenting is beneficial for the clinical outcomes of atherosclerotic renal artery stenosis (ARAS) patients. The study aims to evaluate whether blood pressure (BP) and renal function improve in ARAS patients after stenting.
Methods
A retrospective study was conducted on 78 ARAS patients who underwent renal artery stenting at the First Affiliated Hospital of Chongqing Medical University from January 2013 to June 2024. BP, the number of oral antihypertensive medications, renal function, and adverse events were analyzed before and after stenting.
Results
A total of 90 stents were implanted. The median follow-up time was 1.59 years. From 24 hours post-operation, significant decline in BP were observed. During follow-up, systolic BP decreased by 24.27 mmHg (95% CI: 16.54–32.00; P < 0.0001) and diastolic BP by 12.27 mmHg (95% CI: 7.25–17.29; P < 0.0001), but the reduction in the number of oral antihypertensive medications was not significant (P > 0.05). Serum creatinine also showed a significant decrease of 8.50 μmol/L at follow-up (P < 0.05). Among the four subgroups, 30 patients with mild renal impaired [eGFR ≥ 60 mL/(min·1.73 m2) and <90 mL/(min·1.73 m2)] showed an increase of 10.68 mL/(min·1.73 m2) in eGFR during follow-up (P < 0.01), while the eGFR of the remaining 48 patients with other renal function stages showed no significant change before and after stenting. Moreover, 24 patients experienced cardiovascular or/and renal adverse events.
Conclusions
Renal artery stenting might significantly reduce BP and improve renal function in ARAS patients, particularly those with mild renal impairment, which still requires prospective multicenter studies to confirm.
Keywords
Introduction
Renal artery stenosis (RAS) is one of the common causes of secondary hypertension. 1 RAS can be categorized into three main types. The majority of cases are attributed to atherosclerosis, referred to as atherosclerotic RAS (ARAS), with lesions predominantly located at the proximal third of the renal artery or the ostium.2,3 The prevalence of ARAS is approximately 90% of RAS cases in the European countries, 4 and over 80% in China, 5 with a particularly significant presence among the elderly population. 6 Risk factors for increased prevalence include patient age, gender, smoking, medication history, coronary heart disease, diabetes, peripheral artery disease, and other comorbidities. 7 The other two types are fibromuscular dysplasia and Takayasu arteritis, accounting for about 10% of RAS patients. 3 ARAS can be classified based on the luminal stenosis into mild (<50%), moderate (≥50% and <70%), and severe (≥70%).8,9 Mild ARAS is usually asymptomatic and can often self-regulate through renal mechanisms to maintain dynamic equilibrium. Moderately severe ARAS, resulting from inadequate renal perfusion and reduced renal blood flow, can cause a rapid decline in estimated glomerular filtration rate (eGFR) of more than 30%, leading to worsening renal function and activating the renin–angiotension–aldosterone system (RAAS). This activation results in water and sodium retention, as well as elevating blood pressure (BP). Consequently, it increases the risk of cardiovascular and renal adverse events including resistant hypertension, recurrent heart failure, renal fibrosis and even renal atrophy.10–13
Current treatment options for ARAS mainly include guideline-directed medical treatment and revascularization. Renal artery stenting is a minimally invasive strategy that can restore blood flow through the stenosed vessel, with high procedural success rates and relatively few complications. 14 However, observational studies and randomized controlled trials have failed to show any clinical advantages of renal artery stenting in ARAS patients, leaving the choice between stenting and medical therapy still controversial. 15 Therefore, this study performed a retrospective analysis of ARAS patients to assess changes in BP, the number of oral antihypertensive medications, renal function, and long-term adverse events following renal artery stenting, aiming to evaluate clinical benefits of stenting for ARAS patients.
Materials and methods
Study design and participants
A retrospective research method design was used to observe and analyze patients. A systematic search was conducted for severe RAS patients who underwent renal angiography at the First Affiliated Hospital of Chongqing Medical University from January 2013 to June 2024. After identifying a total of 426 patients who had undergone renal artery stenting, those with non-ARAS or severe baseline data deficiencies were excluded. Patients who were lost to follow-up or had missing outcome data were further excluded. Ultimately, 78 ARAS patients were included in the study, as shown in Figure 1. Among them, 65 patients provided the complete data on the number of oral antihypertensive medications. The study was conducted in accordance with the Declaration of Helsinki of 1975 as revised in 2013 and was approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (no. K2024-087-01) on March 1, 2024, with the need for written informed consent waived. All patient details have been de-identified. The reporting of this study conforms to STROBE guidelines. 16
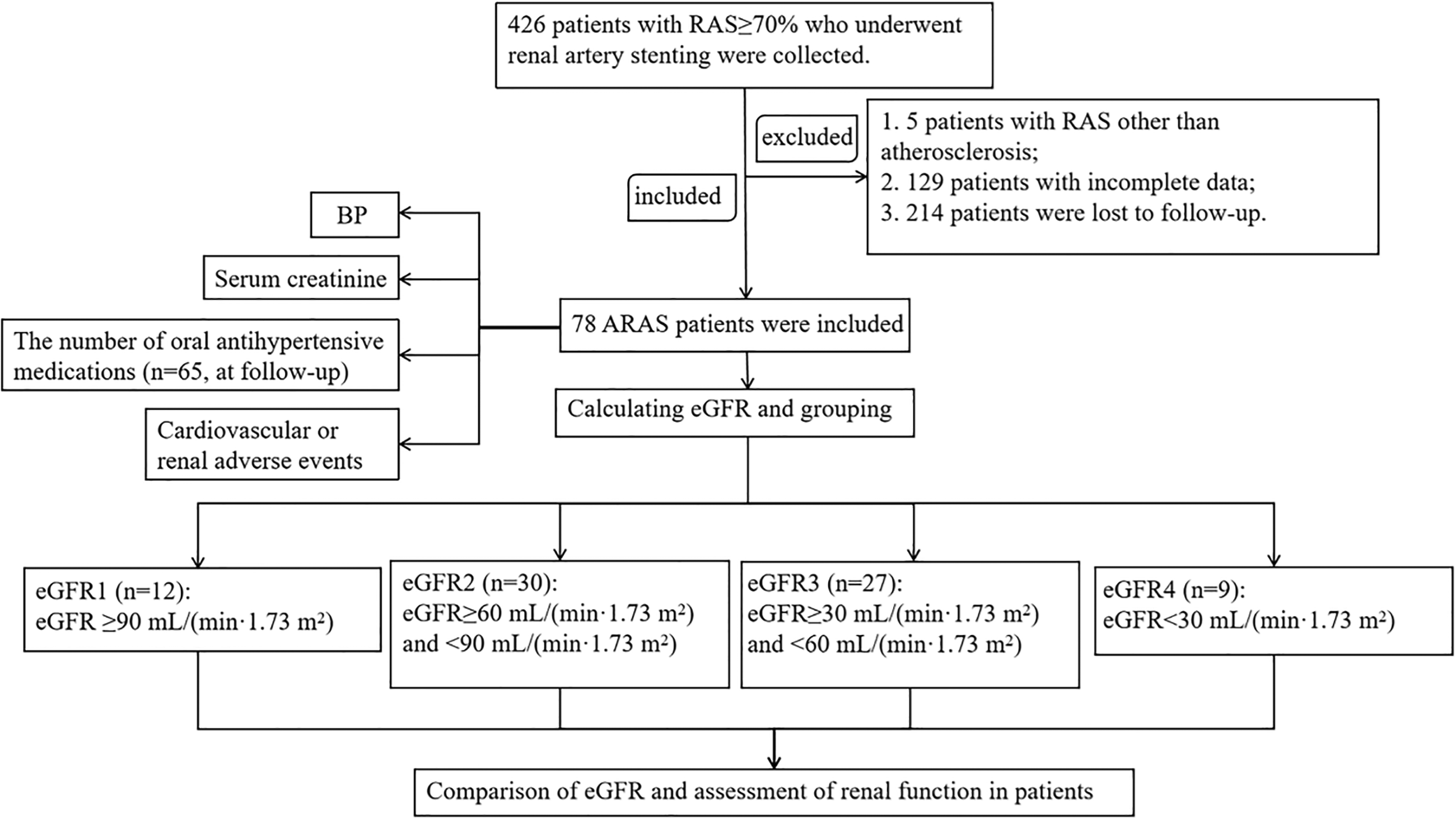
ARAS patients screening process diagram. RAS, renal artery stenosis; ARAS, atherosclerotic renal artery stenosis; BP, blood pressure; eGFR, estimated glomerular filtration rate.
Inclusion and exclusion criteria
Inclusion criteria included: ① RAS with luminal narrowing ≥70% confirmed by renal angiography; ② patients who voluntarily underwent renal artery stenting.
Exclusion criteria included: ① patients with non-ARAS caused by Takayasu arteritis, fibromuscular dysplasia, or other etiologies; ② patients with severe data deficiencies, including missing major baseline characteristics, BP or creatinine; ③ patients lost to follow-up.
Data collection and follow-up
This study collected the following baseline characteristics for patients with ARAS: age, gender, active smoking, drinking, blood lipids (including total cholesterol, triglycerides, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol), comorbidity (coronary heart disease, diabetes, stroke, peripheral artery disease, and chronic kidney disease), medication history (aspirin, clopidogrel, statin, and antihypertensive agents including angiotensin-converting enzyme inhibitor, angiotensin receptor blocker, angiotensin receptor-neprilysin inhibitor, calcium-blocker, beta-blockers, diuretic and other types of antihypertensive medications), number of left or right RAS, details of renal artery stenting, and cardiovascular adverse events during hospitalization (including acute heart failure, unstable angina pectoris, and myocardial infarction). The endpoint data included systolic BP (SBP) and diastolic BP (DBP) measured pre-operation, 1 hour post-operation, 24 hours post-operation, and during follow-up. Additionally, the number of oral antihypertensive medications, serum creatinine and eGFR were recorded pre-operation, within 1 week post-operation, and during follow-up. Any cardiovascular or renal adverse events occurring during the follow-up period were also documented.
eGFR was calculated using the 2009 Chronic Kidney Disease Epidemiology Collaboration equation. 17 According to the eGFR value calculated before stenting, patients were categorized into four subgroups as follows: (a) normal renal function group (eGFR1): eGFR ≥ 90 mL/(min·1.73 m2); (b) mild renal function impairment group (eGFR2): eGFR ≥ 60 mL/(min·1.73 m2) and <90 mL/(min·1.73 m2); (c) moderate renal function impairment group (eGFR3): eGFR ≥ 30 mL/(min·1.73 m2) and <60 mL/(min·1.73 m2); (d) severe renal function impairment group (eGFR4): eGFR < 30 mL/(min·1.73 m2).
Outcomes and definitions
Primary outcomes included: (a) changes in BP; (b) changes in the number of oral antihypertensive medications; (c) changes in renal function including serum creatinine and eGFR. eGFR was divided into three levels based on the difference between the follow-up period and baseline comparison: ① improvement: an increase in eGFR ≥ 20%; ② worsening: a decrease in eGFR ≥ 20%; ③ stability: eGFR changes were less than 20%. 18
Secondary outcomes included cardiovascular or renal adverse events that occurred during the follow-up period. Cardiovascular adverse events were defined as rehospitalization due to acute congestive heart failure or acute coronary syndrome. Renal adverse events were defined as the necessity of renal replacement therapy.
Statistical analysis
The data analysis was conducted using SPSS 26.0 software. Continuous variables that followed a normal distribution were presented as mean ± standard deviation and analyzed using repeated measures ANOVA for correlated samples or one-way ANOVA for independent samples. Continuous variables that followed a skewed distribution were presented as median with interquartile range and analyzed using non-parametric tests such as the Friedman test for correlated samples or the Kruskal–Wallis test for independent samples. Bonferroni correction was applied to adjust significance values. Categorical variables were presented as numbers and percentages and analyzed using the Kruskal–Wallis test for ordinal data with multiple groups or Fisher's exact test for binary variables. By convention, a P-value less than 0.05 was considered statistically significant.
Results
A total of 78 patients were included in this study, comprising 56 males and 22 females, with ages ranging from 41 to 86 years and an average age of 64.87 ± 11.04 years. The median follow-up time was 1.58 (0.58, 4.21) years (range: 0.08–10.58 years). The baseline characteristics of all patients were shown in Table 1. Among the 78 patients, 95 renal arteries exhibited severe stenosis, with 90 stent implantations performed. Specifically, 66 patients accepted unilateral stenting, while 12 underwent bilateral stenting. Among 5 patients with bilateral ARAS, 2 were deemed unsuitable for stenting due to severe renal atrophy on one side and 3 failed to open the affected artery due to severe stenosis and unfavorable vascular conditions on one side, resulting in unilateral stent implantation for these 5 patients. No complications were reported during the procedures. During hospitalization, 15 patients experienced cardiovascular adverse events, including 5 cases of acute heart failure and 10 cases of unstable angina. No patients experienced myocardial infarction. All patients improved and were discharged after receiving active treatment.
Baseline characteristics.

Values are presented as number (%), mean ± standard deviation.
Comparison of blood pressure before and after renal artery stenting
BP was compared in 78 patients at pre-operation, 1 h post-operation, 24 h post-operation and at follow-up (Figure 2). There was no significant difference in BP between pre-operation and 1 hour post-operation. However, from 24 hours post-operation to the final follow-up, both SBP and DBP showed significant reductions. During the follow-up period, SBP significantly decreased by 24.27 mmHg (95% CI: 16.54–32.00; P < 0.0001), and DBP significantly decreased by 12.27 mmHg (95% CI: 7.25–17.29; P < 0.0001) compared to pre-operation levels. The results indicated that stenting could significantly reduce BP.

Comparison of blood pressure, the number of oral antihypertensive medications and serum creatinine before and after renal artery stenting. Continuous variables including SBP, DBP and the number of oral antihypertensive medications followed a normal distribution, presented as mean (above the bar) ± standard deviation (in the bar). Continuous variable of serum creatinine followed a skewed distribution, presented as median (above the bar) with interquartile range (in the bar). aIndicates a significant difference compared to pre-operation. bIndicates a significant difference compared to 1 hour post-operation. cIndicates a significant difference compared to 24 hours post-operation. SBP, systolic blood pressure; DBP, diastolic blood pressure.
Comparison of the number of oral antihypertensive medications before and after renal artery stenting
The analysis of the number of oral antihypertensive medications was conducted at three time points: pre-operation, within 1 week post-operation and follow-up period. Due to the retrospective study, 13 elderly patients failed to provide the current number of oral antihypertensive medications during the follow-up period, and there were no related records available in the system. Consequently, data from 65 patients were obtained at follow-up. As shown in Figure 2, the changes in the number of antihypertensive medications before and after operation were not significant (P = 0.167). Among these 65 patients, 11 had discontinued their antihypertensive medications by the time of follow-up.
Comparison of serum creatinine before and after renal artery stenting
Statistical analysis of serum creatinine was conducted on all patients at three different time points, and the data showed statistical significance (P = 0.022), as shown in Figure 2. The results demonstrated that a significant decrease of 8.50 μmol/L in serum creatinine at follow-up compared to pre-operative levels (P = 0.017), which showed a potential improvement effect on the long-term prognosis of renal function.
Subgroup analysis based on eGFR
All patients were divided into four subgroups according to the pre-operative eGFR, indicating different renal function stage. Among them, 12 patients with eGFR1 [eGFR ≥ 90 mL/(min·1.73 m2)] had normal renal function. Thirty patients with eGFR2 [eGFR ≥ 60 mL/(min·1.73 m2) and <90 mL/(min·1.73 m2)] had mild impairment. Twenty-seven patients with eGFR3 [eGFR ≥ 30 mL/(min·1.73 m2) and <60 mL/(min·1.73 m2)] had moderate impairment, and 9 patients with eGFR4 [eGFR < 30 mL/(min·1.73 m2)] had severe impairment. To assess the comparability between the four subgroups, baseline comparisons were conducted, as shown in Table 2. The severity of renal impairment was significantly associated with age (P = 0.006). Given the close relationship between eGFR values and impaired renal function, significant disparities in the prevalence of chronic kidney disease were observed across the four subgroups. Additionally, there were significant differences in the use of beta-blocker, aspirin, clopidogrel, and statins among the groups. It was worth mentioning that the number of patients receiving these medications was higher in the eGFR3 and eGFR4 groups, which might be related to the higher prevalence of coronary heart disease in these groups, although it revealed no significance in the prevalence of coronary heart disease among the four subgroups.
Comparison of baseline characteristics among four subgroups of patients.

presented as P < 0.05.
eGFR, estimated glomerular filtration rate.
Comparison of eGFR before and after renal artery stenting
In order to observe the changes in renal function stages of four subgroups, eGFR was compared as shown in Table 3. The results indicated a significant variation in eGFR2 between pre and post-operation (P = 0.001), with an increase of 10.68 mL/(min·1.73 m2) in eGFR at follow-up compared to pre-operation (P = 0.006). No significant change in eGFR was observed in patients with eGFR1, eGFR3, and eGFR4, with P values greater than 0.05. This finding suggested that the renal function of patients with mild impairment might be reversed during long-term follow-up, while renal function might remain unchanged in patients with moderate to severe damage. This discrepancy might be linked to the glomerular survival rate in the kidneys.
Comparison of eGFR before and after renal artery stenting in four subgroups of patients.

Continuous variables followed an abnormal distribution, presented as median with interquartile range.
Indicated a significant difference compared to pre-operation.
eGFR, estimated glomerular filtration rate.
Comparison of the efficacy of renal function after renal artery stenting
The changes in renal function between pre-operative and follow-up periods were compared again, divided into three different degrees: improvement, stability, and worsening. During the follow-up, 26 patients showed improved renal function, 35 had stable renal function, and 17 experienced further worsening. Kruskal–Wallis test showed a non-significant change in the stratification of post-operative renal function treatment among the three groups (P = 0.647), as shown in Table 4.
Comparison of renal function treatment effects in patients after renal artery stenting.
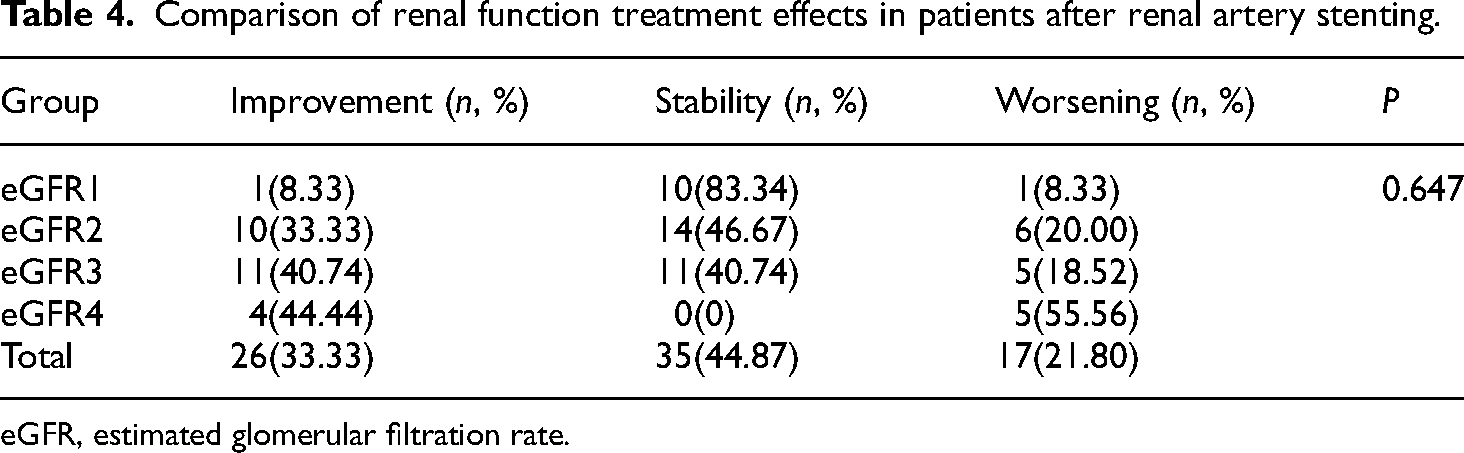
eGFR, estimated glomerular filtration rate.
Adverse events after renal artery stenting
Finally, 24 (30.77%) patients experienced cardiovascular or renal adverse events at follow-up, including 14 cases with only cardiac adverse events, 8 cases with only renal adverse events, and 2 cases with both adverse events. Of the 24 patients, 13 had coronary heart disease, 11 had diabetes, 22 had peripheral artery disease, 5 had experienced a stroke, and 13 had chronic kidney disease. Of the 10 patients who progressed to renal replacement therapy, 9 (90%) had a pre-operative eGFR of less than 45 mL/(min·1.73 m2). These comorbidities significantly contributed to the progression of ARAS. As indicated in the baseline analysis of patient comorbidities presented in Table 1, the failure to actively perform stenting might lead to a higher incidence of adverse events among patients with chronic conditions.
Discussion
This study retrospectively analyzed the efficacy of renal artery stenting in 78 ARAS patients. The results showed that renal artery stenting had certain benefits, although the changes in the number of oral antihypertensive agents were non-significant pre- and post-operation. The BP of ARAS patients significantly decreased from 24 hours post-operation, with SBP decreasing by 24.27 mmHg (95% CI: 16.54–32.00) and DBP decreasing by 12.27 mmHg (95% CI: 7.25–17.29) during follow-up. Similarly, serum creatinine also decreased by 8.50 μmol/L during follow-up. In subgroup analysis, ARAS patients in renal function with eGFR ≥ 60 mL/(min·1.73 m2) and <90 mL/(min·1.73 m2) showed improvement after stenting. In addition, 24 patients were observed to have experienced cardiovascular or/and renal adverse events.
The ACC/AHA guidelines recommended that patients requiring renal artery revascularization should have significant hemodynamic changes due to ARAS and exhibit at least one of the following symptoms: resistant hypertension, rapid deterioration of renal function, or cardiac destabilization syndromes. 19 In addition to these criteria, optimal medical therapy should be the preferred treatment option for other ARAS patients. 20 Although the potential indications for revascularization in ARAS patients have been outlined, many researchers continue to conduct extensive clinical trials to investigate the benefits of percutaneous renal artery angioplasty and stent implantation, owing to the persistent controversies and uncertainties in this field. Therefore, the primary objective of this study was to explore the clinical value of renal artery stenting. This study indicated that after stenting, the BP of ARAS patients not only significantly decreased in the short term, but also achieved long-term effective control. Although the short-term impact on renal function was not significant, it showed long-term stable enhancement over time, especially in patients with mild renal impairment where improvement was more pronounced. In addition, there were still some ARAS patients who experience cardiovascular or renal adverse events after stenting.
There have been multiple studies exploring the relationship between renal artery stenting and BP, among which the two most famous large prospective randomized controlled trials were the CORAL study and the ASTRAL study.21,22 Both studies randomly divided ARAS patients into a medical therapy group and a combination of revascularization and medical therapy group, and the results showed that revascularization had an advantage in reducing BP. Although the sample sizes of these two study populations were substantial, there were still some limitations. For instance, the majority of ARAS patients included in the study exhibited moderate stenosis and mild conditions, which might not be ideal candidates for renal revascularization. 23 Therefore, many researchers have successively explored the necessity of stenting for ARAS patients. A study by Reinhard et al. included 102 patients with ARAS ≥ 70% and significant hemodynamic changes who were prospectively followed up for 2 years after stenting, and evaluated using 24 hours dynamic BP monitoring. The results showed a decrease in SBP of 25.7 mmHg (95% CI: 20.6–30.8; P < 0.001) and DBP of 9.2 mmHg (95% CI: 6.8–11.7; P < 0.001). 24 Similarly, a research team in China investigated BP changes before and after stenting in 125 ARAS patients over a 2-year period. The results demonstrated that stenting effectively reduced BP and antihypertensive agents required (P < 0.05). 18 In addition, some small non-randomized controlled trials have also demonstrated the above results.25,26 Moreover, high baseline BP had a better reduction effect after intervention compared to low baseline. 27 This observation aligned with our study on BP, which revealed a significant decrease in both SBP and DBP after stenting, showed during both short-term and long-term follow-up (P < 0.0001). In this study, the number of oral antihypertensive medications changed minimally, which contrasted with previous research findings and potentially correlated with the high incidence of poor prognosis (30.77%). Overall, this might indicate that, with a consistent number of antihypertensive agents pre- and post-operation, renal artery stenting in BP management held significant clinical value.
The potential for renal artery stenting to improve renal function remains an area requiring further research, as it is both a controversial and intriguing topic. Bax et al. compared stent placement and medical therapy in ARAS patients accompanied by impaired renal function, but no significant improvement was observed in renal function after stenting. 28 The RADAR study was conducted to assess the impact of stenting on renal function in ARAS patients with significant hemodynamic changes. The results showed that after 1 year, the eGFR of the stent group increased by 4.3 mL/(min·1.73 m2) compared to baseline, while the best medical treatment group increased by 3.0 mL/(min·1.73 m2), but the difference was not significant, indicating that the renal function benefit of stenting was not superior to that of the medical treatment. 29 In addition, some retrospective studies have also denied the benefits of stenting on renal function.15,25 However, some studies have also drawn conclusions opposite to the above. A study showed that eGFR increased by 7.8 mL/(min·1.73 m2) (95% CI: 4.5–11.1; P < 0.001) post-stenting, compared to baseline. 24 In our study, after a median follow-up of 1.6 year, improvement in renal function was also observed with a decrease in serum creatinine of 8.50 μmol/L.
The controversial aspect concerning renal function is linked to the pathophysiological changes after RAS. Severe luminal stenosis reduces renal perfusion, which activates the RAAS system, further leading to remodeling of the renal vascular structure and promoting renal fibrosis. This finally contributes to recurrent cardiovascular relevant complications, including congestive heart failure and pulmonary edema. At this stage, stent placement is unlikely to reverse renal function or diminish the occurrence of such complications. 30 However, performing revascularization before permanent fibrosis of renal blood vessels can promptly alleviate luminal stenosis and inhibit the activation of the RAAS system, which may potentially reverse renal function and reduce the incidence of cardiovascular disease. Catena et al. found that timely stenting in ARAS patients with significant hemodynamic changes could improve the function and structure of the left ventricle. 31 But in the ASTRAL trial, the average eGFR of enrolled patients was 40 mL/(min·1.73 m2) with a median follow-up time of 4.7 years, and no overall benefit of stenting on cardiovascular and renal outcomes was observed. 32 The CORAL subgroup analysis also noted that chronic kidney disease patients who had undergone stenting exhibited no significant differences in renal function deterioration during follow-up compared to those receiving medical treatment. 33 This raises an important question: should we refine the analysis of renal function to better understand at which stage it can provide greater clinical benefits after stent implantation? In the subgroup analysis of this study, it was observed that patients experiencing mild renal impairment were more potentially beneficial from stenting, with a follow-up eGFR increase 10.65 mL/(min·1.73 m2), compared to baseline (P = 0.006). This might be associated with a certain level of glomerular survival, as these patients had not yet reached a stage of renal fibrosis. Furthermore, these findings aligned with another study, which emphasized that individuals with eGFR > 30 mL/(min·1.73 m2) before stenting were more likely to experience improvements in renal function during subsequent follow-up. 27 This investigation established predictive scores to identify which patients might experience renal function improvement from stenting, underscoring the strong relationship between renal function enhancement and event-free survival.
Studies indicated that individuals in chronic kidney disease stages 3B-5 had a greater risk of renal events following stenting. 34 Ninety percent of the 10 patients who ultimately required renal replacement therapy in this study had eGFR ≤ 45 mL/(min·1.73 m2) before stenting, suggesting that aggressive treatment struggled to improve outcomes. Additionally, diabetes is a crucial factor. Effective glycemic control could delay renal function progression and cardiovascular events post renal artery stenting. 35 Among the 24 patients in our study experiencing end-stage events, 11 had diabetes, possibly related to poor long-term glycemic control or existing diabetic nephropathy. Unfortunately, further details about their glucose levels and urine albumin-to-creatinine ratio were not available. Therefore, clinicians must meticulously evaluate such patients to ascertain whether renal impairment results from diabetes or ARAS, select optimal intervention timing, and intensify glycemic management rather than solely concentrating on localized renal artery treatment. 36
Our study reported some positive outcomes through a self-controlled comparison before and after renal artery stenting, but it also had several limitations: (a) as a retrospective study, the indications for stenting could not be determined, lacking randomized patient selection, leading to selection bias, and also lacking a control group for medical therapy; (b) due to a higher number of missing data cases and fewer patients with systematic follow-up records, the final available sample size was relatively small, especially when the sample was grouped by eGFR values, resulting in relatively inadequate sample size within each group. Further enlargement of the sample size or prospective clinical controlled studies are needed to continue exploring the impact of stenting on renal function in ARAS patient subgroups; (c) glycemic and urine albumin levels were not included in the current study analysis. Future research should focus on the influence of these biological indicators on renal function recovery after ARAS stenting and their predictive response analysis.
Conclusion
Atherosclerosis is a progressive disease that can affect the entire arterial system. When it involves the renal artery lumen, it is referred to as ARAS. Without early intervention, severe ARAS can lead to a range of complications. Therefore, early intervention and the selection of an appropriate treatment strategy are crucial for the effective management of this disease. Our study indicated that ARAS patients experienced long-term benefits in both BP and renal function after stenting. Notably, while improvements in BP were observed in the early post-operative stages, significant changes in renal function were only evident later. Furthermore, ARAS patients with mild renal impairment appeared to derive greater clinical benefits from stenting, although further investigation is needed to evaluate the long-term outcomes across different levels of renal impairment. This study may provide valuable insights for ARAS patients considering stenting.
Footnotes
Abbreviations
Authors’ contribution
Research design and methods, statistical analysis and manuscript writing: Qiuyue Shi. Data collection and data curation: Xin Li and Yanwei Li. Revising the article critically for important intellectual content: Jing Chang and Min Mao. All authors have read and approved the final manuscript.
Consent to participate
In this retrospective study, formal written informed consent from patients was not required, patient information was anonymized, and all data were kept confidential.
Consent for publication
The authors have gone through the final draft of the manuscript and agreed to the submission/publication.
Data availability statement
The data was available from the corresponding authors upon the reasonable requests.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (no. K2024-087-01) on March 1, 2024.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
