Abstract
Objective
Endovascular recanalization is the preferred treatment for peripheral vascular diseases. This study evaluated the short-term outcomes of endovascular therapy for transplant renal artery stenosis (TRAS) administered based on anatomical foundations and three-dimensional digital subtraction angiography (3D-DSA).
Methods
Patients with TRAS treated with endovascular therapy at a single center between October 2022 and October 2023 were retrospectively analyzed. The primary endpoint was the 12-month primary patency rate, defined as uninterrupted vessel patency without reintervention. Secondary endpoints included secondary patency rate (patency maintained after reintervention), blood pressure, and serum creatinine (Scr) levels.
Results
The study included 23 patients (mean age: 41.1 ± 11.5 years; 9 females). All patients underwent successful plain old balloon angioplasty (POBA); 13 underwent POBA plus balloon-expandable bare-metal stent (BMS) deployment. The procedure-related complication rate was 13.04%. Technical success was defined as <30% residual stenosis with restoration of antegrade flow at completion angiography and was achieved in all cases (100%). The median follow-up was 14.9 ± 2.3 months (range11–19 months). The 12-month primary and secondary patency rates were 86.96% and 100%, respectively. The primary patency rates significantly differed between the POBA + BMS (100%) and POBA-only (70%) groups (P = 0.032). The systolic and diastolic blood pressure, pulse pressure, and Scr levels significantly differed pre- and post-operatively. No stent-related complications occurred.
Conclusions
Endovascular treatment of TRAS based on anatomical foundations and 3D-DSA is safe and effective with a high patency rate. POBA with BMS might be a better solution.
Introduction
Transplant renal artery stenosis (TRAS) is a severe vascular complication following renal transplantation that affects graft function and patient outcomes, occurring in 1–23% of renal transplant recipients. TRAS has been associated with hypertension, graft dysfunction, and an increased risk of allograft loss. 1 Endovascular therapy is the preferred treatment for TRAS owing to its minimally invasive nature and high technical success rate. 2 However, patient-specific anatomical variations and the complexity of lesion morphology require advanced imaging techniques for an accurate diagnosis and optimal treatment planning. 3
Three-dimensional digital subtraction angiography (3D-DSA) has superior diagnostic accuracy compared to conventional angiography, allowing for precise visualization of the transplant renal artery anatomy. 4 Its ability to reconstruct vessel orientation and guide C-arm angulation is particularly useful in TRAS, where standard two-dimensional DSA often underestimates lesion severity or misrepresents the anastomotic angle. The impetus for incorporating 3D-DSA in our study was the recognition that misinterpretation of lesion morphology can lead to improper stent deployment or vessel injury. Thus, our hypothesis was that 3D-DSA-guided intervention would facilitate safer device navigation, reduce complications, and improve long-term patency compared with standard approaches.
Therefore, this study aimed to evaluate the clinical and anatomical characteristics of TRAS and to assess the outcomes of endovascular therapy performed under 3D-DSA guidance. Specifically, the primary objective was to determine the technical success and 12-month primary patency after endovascular treatment. The secondary objectives were to compare outcomes between plain old balloon angioplasty (POBA) alone and POBA combined with bare-metal stent (BMS) deployment, and to investigate whether 3D-DSA–based anatomical assessment facilitates procedural planning and post-intervention outcomes.
Materials and methods
Study population
A retrospective review of the efficacy of endovascular therapy for TRAS was conducted at Xiamen Branch of Zhongshan Hospital of Fudan University between October 2022 and October 2023. All patients underwent preoperative Doppler ultrasonography as part of the standard diagnostic protocol. The baseline patient demographics, lesion characteristics, operative details, and follow-up outcomes were retrospectively collected and analyzed (Table 1). The primary objective of this study was to evaluate technical success and 12-month primary patency following 3D-DSA–guided endovascular therapy for TRAS. The secondary objectives included (1) comparing clinical and imaging outcomes between patients treated with POBA alone versus POBA plus BMS, and (2) assessing the role of 3D-DSA in delineating arterial anatomy to support procedural decision-making.
Baseline clinical characteristics.

Note. Clinical presentation: 0: none; 1: anuria or oliguria; 2: increased serum creatinine; 3: hypertension; 4: edema; TRA: transplanted renal artery.
The inclusion criteria for endovascular treatment were I) graft dysfunction, such as anuria, oliguria, increased Scr levels, hypertension, and edema, and II) TRAS indicated by Doppler ultrasound. Hemodynamically significant TRAS on Doppler ultrasonography was defined as a peak systolic velocity (PSV) ≥ 200 cm/s. One patient did not exhibit overt graft dysfunction but demonstrated hemodynamically significant TRAS on Doppler ultrasound and 3D-DSA. Specifically, this patient was asymptomatic but had hemodynamically significant TRAS on imaging. Because this finding represents a clear indication for endovascular intervention, this patient was included. The exclusion criteria were I) graft dysfunction without TRAS indicated by Doppler ultrasound or confirmed by 3D-DSA, and II) loss to follow-up.
Technical success was defined as <30% residual stenosis with restoration of antegrade flow. Primary patency was defined as vessel patency without restenosis or reintervention during follow-up. Secondary patency was defined as vessel patency restored after reintervention. Procedural complications were defined as dissection, rupture, or thrombotic occlusion occurring intraoperatively or within 30 days. The reporting of this study conforms to the STROBE guidelines. 5
Statistical analyses
Statistical analyses were performed using SPSS 22.0 and GraphPad Prism 9. Continuous data were assessed for normality (Kolmogorov-Smirnov test). Normally distributed continuous variables were compared using paired Student's t-test; non-normally distributed variables were compared using the Mann–Whitney U test. Categorical variables were compared with chi-square or Fisher's exact test. Survival curves (primary and secondary patency) were analyzed by Kaplan–Meier method, with group comparisons by log-rank test. Exact p-values are reported; significance was set at P < 0.05.
Follow-up protocol
Follow-up assessments were scheduled at 1 day, and at 1-, 3-, 6-, and 12-months post-procedure. These included Doppler ultrasonography, office blood pressure measurement, and kidney function tests (Scr, eGFR). All patients received dual antiplatelet therapy with aspirin (100 mg/day) and clopidogrel (75 mg/day) for at least 6 months, unless contraindicated.
Ethics statement
This retrospective study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024). The Institutional Review Board of Xiamen Branch of Zhongshan Hospital confirmed that formal ethical approval was not required due to the retrospective nature of the study, and all patient data were fully de-identified prior to analysis.
Results
Patient selection
A total of 27 patients with suspected TRAS were screened between October 2022 and October 2023. Among them, 3 patients were excluded due to no evidence of TRAS on Doppler ultrasonography or 3D-DSA, and 1 patient were excluded because of loss to follow-up. Finally, 23 patients met the inclusion criteria and were enrolled in the analysis (Figure 1).

Flow diagram of patient selection, exclusion, and final inclusion in the study.
Patient demographics and clinical presentation
This study included 23 patients with TRAS, comprising 9 females (39.10%) and 14 males (60.90%) with an average age of 41.1 ± 11.5 years. The median duration from kidney transplantation to the diagnosis of TRAS was 9.2 ± 9.2 months (range: 1–34 months). In total, 21 had right-sided and 2 had left-sided TRAS (Table 2). One patient (4.35%) presented with acute thrombosis.
Lesion characteristics.

Note. I: Internal iliac artery-Trunk-Proximal; II: Internal iliac artery-Trunk-Proximal; III: Internal iliac artery-Trunk-Midpiece; IV: External iliac artery-Trunk-Proximal; V: External iliac artery-branch; TRA: Transplanted Renal Artery.
Lesion characteristics
Lesions were categorized as follows (Table 2): (I) internal iliac artery-trunk proximal (Figure 2A); (II) internal iliac artery-trunk proximal (Figure 2B); (III) internal iliac artery-trunk midpiece (Figure 2C); (IV) external iliac artery-trunk proximal (Figure 2D); and (V) external iliac artery-branch (Figure 2E). These categories correspond to the site of anastomosis and lesion location: “trunk” denotes the main stem of the transplanted renal artery; “midpiece” indicates the mid-portion between anastomosis and bifurcation; “branch” refers to distal segmental branches. Notably, in our center, both internal and external iliac arteries were used as recipient sites depending on vessel size and prior surgical considerations, which explains the proportion of internal iliac artery anastomoses.

Transplant renal artery stenosis types. (A) Internal iliac artery-trunk proximal; (B) Internal iliac artery-trunk proximal; (C) Internal iliac artery-trunk midpiece; (D) External iliac artery-trunk proximal; (E) External iliac artery-branch.
Outcomes
The ipsilateral common femoral artery approach, contralateral common femoral artery access, and combined paths were used in 5, 13, and 5 patients, respectively. Technical success was achieved in all patients (100%). Furthermore, 15 balloon-expandable bare stents were used in 11 patients, including three Express LD (Boston Scientific, Bally Brit Business Park, Galway, Ireland), two Precise (Cordis Corporation, Miami Lakes, Florida, USA), eight Palmaz Blue (Cordis Cashel, Clonmel Tipperary, Ireland), and two coronary artery stents (Table 3). Two minor non–flow-limiting dissections occurred during the index procedures (Table 4).In addition, one case of transplant renal artery rupture occurred during reintervention and required coil embolization, which was classified as a major complication. The results of univariate analysis confirmed that sex, age, coexisting medical conditions, clinical presentation, lesion side, and TRAS type were not significantly correlated with reintervention (p > .05) (Table 5).
Details of endovascular treatment.
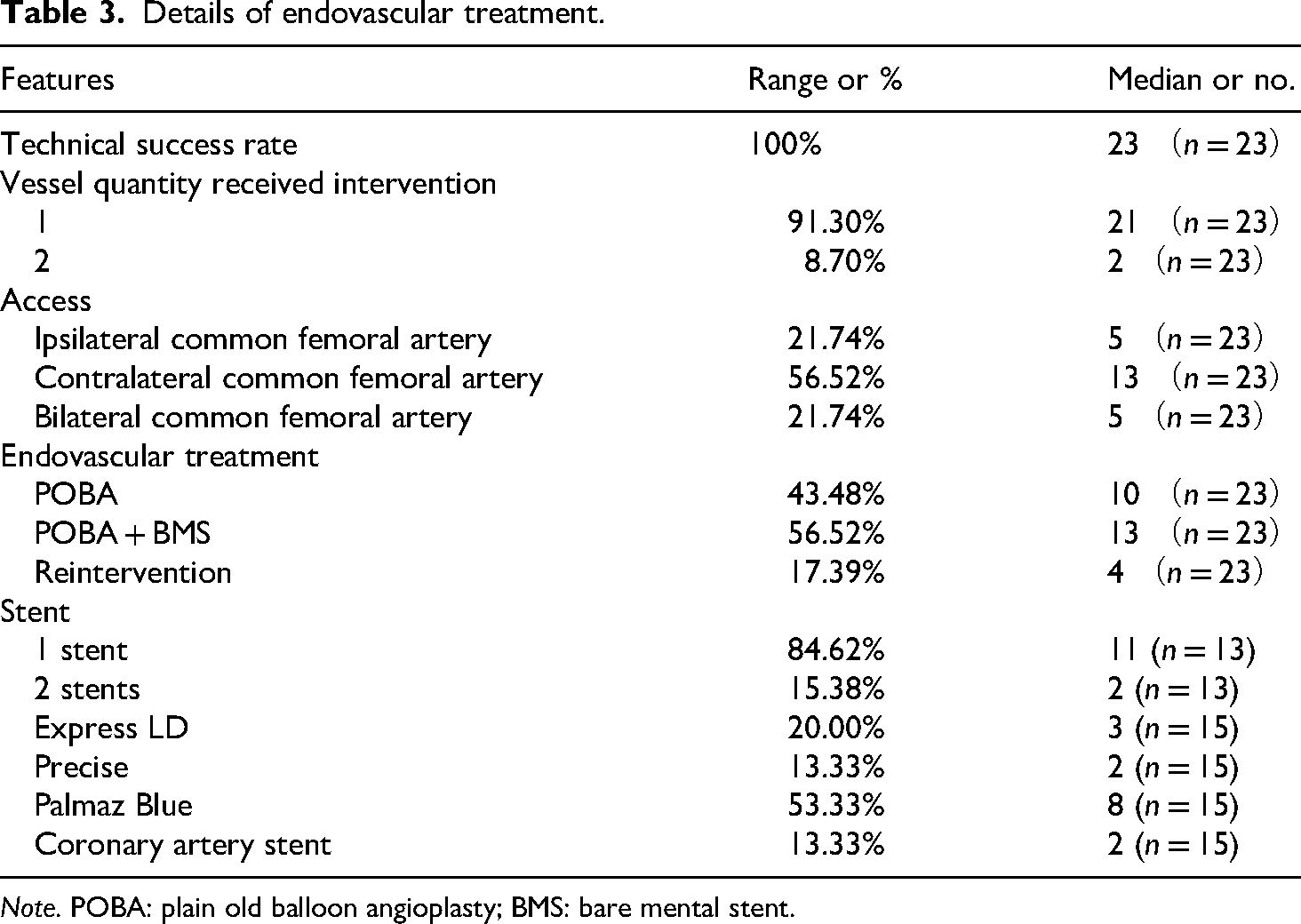
Note. POBA: plain old balloon angioplasty; BMS: bare mental stent.
Details of post intervention and complications.

Univariate analysis for reintervention.
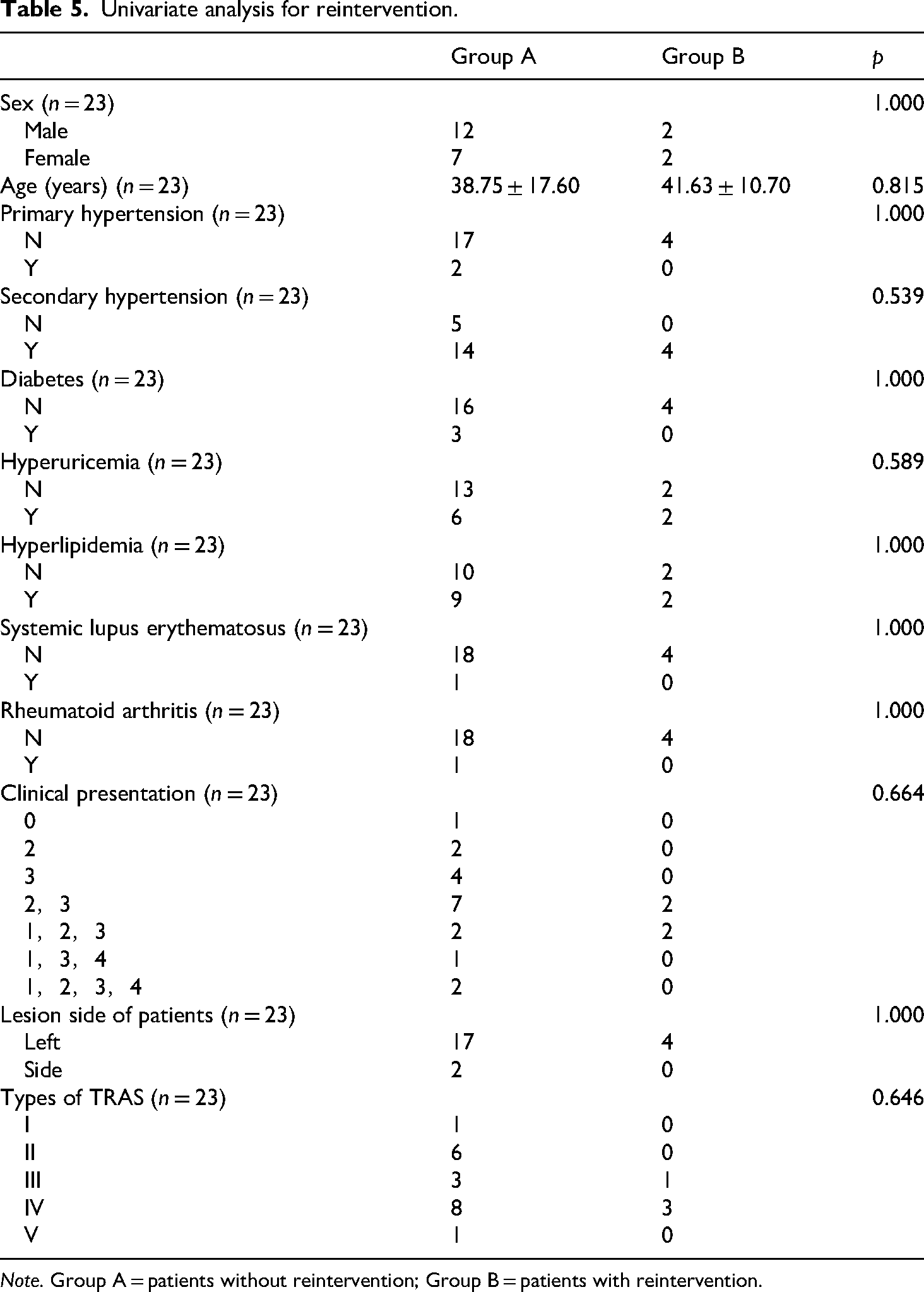
Note. Group A = patients without reintervention; Group B = patients with reintervention.
Long term follow-up
The median follow-up was 14.9 ± 2.3 months (range: 12–19 months). Four patients underwent a secondary intervention with three balloon-expandable BMS deployments, two of which were newly discovered lesions. In one patient with two transplanted renal arteries, the remaining vessel underwent BMS deployment during reintervention, as detailed in Table 4.
The 12-month primary and secondary patency rates were 86.96% and 100%, respectively (Figure 3A). The primary patency rates significantly differed between the POBA + BMS (100%) and POBA-only (70%) groups (p < .05) (Figure 3B). Blood pressure and renal function showed notable improvement following endovascular treatment. Systolic blood pressure (SBP) decreased from 152.3 ± 16.9 mmHg preoperatively to 126.5 ± 17.8 mmHg on postoperative day 1 (p = .006), 128.8 ± 8.6 mmHg at 3 months (p = .023), 127.5 ± 9.7 mmHg at 6 months (p = .0115), and further to 123.5 ± 7.5 mmHg at 12 months (p = .0003) (Figure 4A). Diastolic blood pressure (DBP) also improved from 82.1 ± 14.7 mmHg preoperatively to 75.2 ± 8.7 mmHg at 6 months (p = 0.0163) (Figure 4B). Pulse pressure (PP) consistently declined from 70.3 ± 23.7 mmHg preoperatively to 52.5 ± 12.3 mmHg on postoperative day 1 (p = .0034), 53.1 ± 10.1 mmHg at 3 months (p = .0002), 52.3 ± 8.5 mmHg at 6 months (p < .0001), and 51.2 ± 9.7 mmHg at 12 months (p < .0001) (Figure 4C). Scr improved markedly, declining from 256.0 ± 230.2 μmol/L preoperatively to 187.7 ± 118.6 μmol/L on postoperative day 1 (P = .0029), 174.3 ± 136.8 μmol/L at 3 months (P = .0181), 157.1 ± 100.1 μmol/L at 6 months (P = .0002), and 140.9 ± 80.7 μmol/L at 12 months (P < 0.0001), with a further significant reduction between 3 and 12 months (P = .0167) (Figure 4D). Although eGFR increased from 43.0 ± 26.5 to 59.2 ± 28.1 mL/min/1.73 m2 over 12 months, this change did not reach statistical significance (Figure 5). No stent-related complications, such as fractures or migration, were observed during the follow-up period.
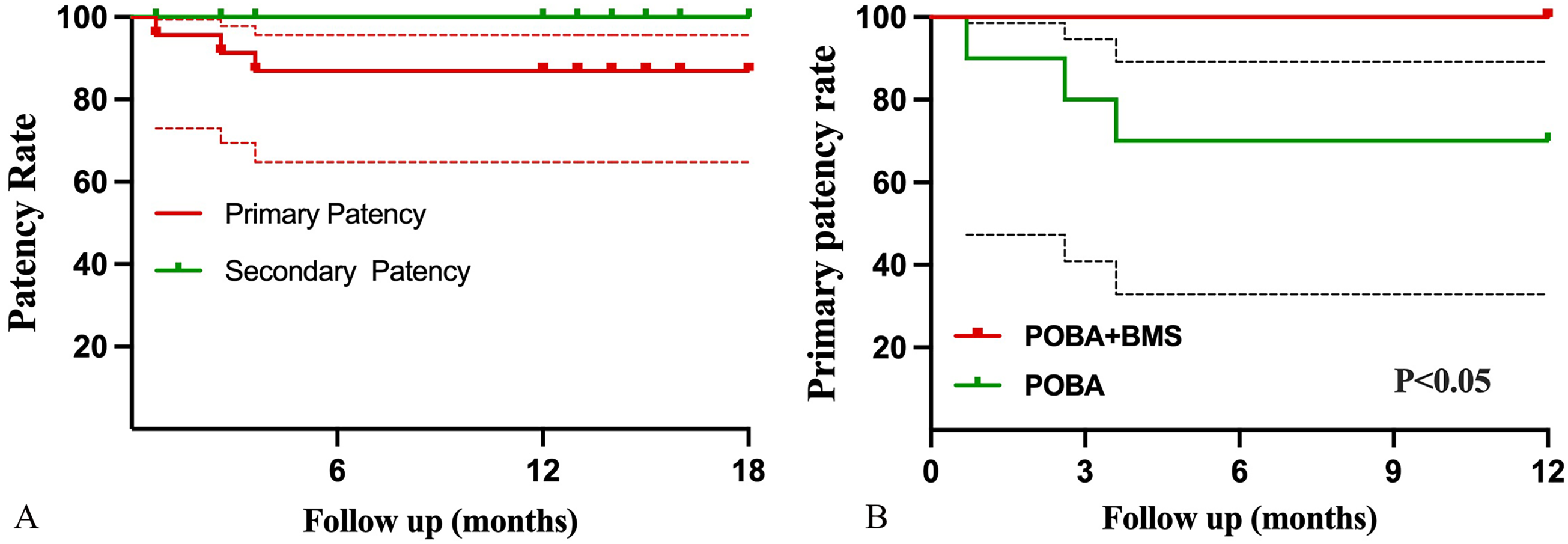
(A) Kaplan–Meier analysis of primary and secondary stent patency rates; (B) Kaplan–Meier analysis of the primary patency rate for patients undergoing plain old balloon angioplasty with balloon-expandable bare-metal stent deployment.

Pre- and post-operative changes in systolic blood pressure (A), diastolic blood pressure (B), pulse pressure (C), and serum creatinine (D) at multiple follow-up timepoints. X-axis labels indicate the measurement timepoints (preoperative, postoperative day 1, 3 months, 6 months, and 12 months). Exact p-values are shown for each comparison, with very small values reported as (p < .0001).
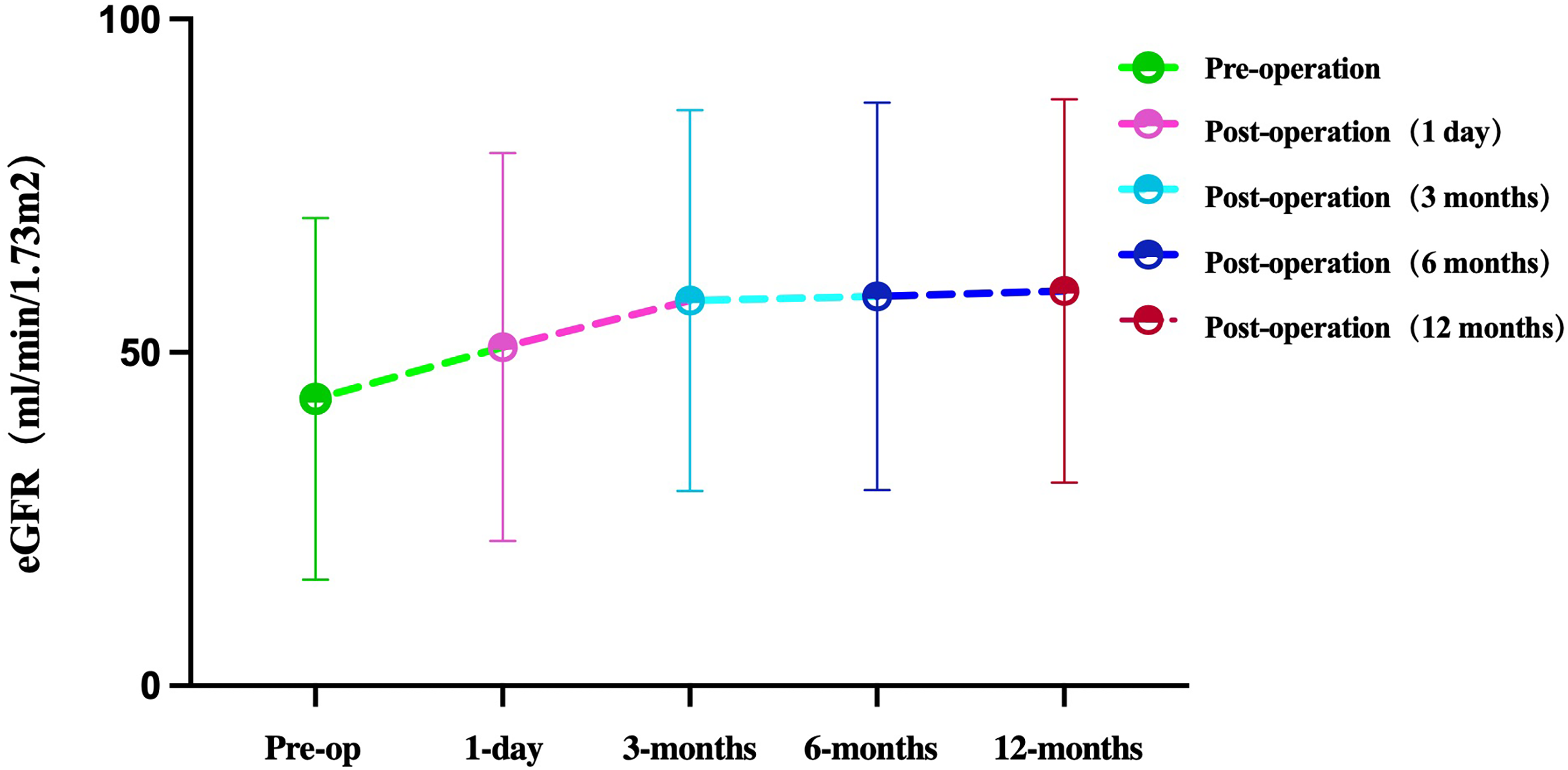
Pre- and post-operative estimated glomerular filtration rates (p > .05).
Discussion
This study investigated the immediate clinical efficacy of minimally invasive interventions for transplant renal artery narrowing using detailed anatomical insights coupled with advanced 3D-DSA imaging.
Clinical implications of endovascular therapy for TRAS
The 12-month primary and secondary patency rates of approximately 87% and 100%, respectively, consistent with previous reported outcomes.6–8 Specifically, the PTA plus BMS group had a primary patency rate of 100%, which was significantly better than the 70% rate in the PTA-only group, highlighting the critical role of stent placement in reducing re-narrowing and sustaining vascular function.9–11
Our findings demonstrate that endovascular therapy improves SBP, DBP, PP, and Scr, consistent with previous studies.11–13 However, eGFR did not significantly improve, likely reflecting irreversible chronic parenchymal injury despite restored perfusion, which is physiologically plausible and aligns with prior reports.14,15 In addition, most patients in our cohort had relatively preserved baseline renal function, leaving limited room for measurable improvement in eGFR. Only a small subset of patients demonstrated true graft dysfunction prior to intervention, reflected by markedly elevated serum creatinine levels, and these cases generally showed stabilization rather than dramatic improvement in eGFR following treatment.
Role of 3D-DSA in optimizing endovascular outcomes
3D-DSA enhances diagnostic precision over conventional DSA by enabling detailed lesion mapping and optimal angulation during intervention.16,17 In our cohort, 3D-DSA substantially improved anatomical visualization by enabling precise superimposition of live fluoroscopic images with 3D vascular reconstructions—a mechanism validated for enhancing complex anatomical identification in neuro-allowing accurate identification of the anastomotic angle, focal or tandem lesions, and the spatial relationship between the transplant renal artery and the iliac vessels.18,19 Our findings reinforce the utility of 3D-DSA in cases of complex TRAS, particularly in patients with complex anatomy or multiple renal arteries (Figure 6A‒E).These features facilitated wire passage, balloon positioning, and stent landing zone selection, particularly in cases with complex anatomy. These advantages were particularly helpful in anatomically complex cases, where accurate visualization and planning are essential for safe and effective intervention. Although our retrospective design does not allow direct comparison of imaging modalities, the consistently high technical success rate in this cohort supports the practical utility of 3D-DSA as an adjunctive tool for procedural planning in TRAS. Regarding cost considerations, 3D-DSA requires additional imaging acquisition, but the improved procedural efficiency may offset this extra cost in selected patients.

(A) 0°: the lesion overlaps with the external iliac artery, resulting in unclear visualization. Both the anastomosis and the bifurcation arteries require positioning at different projection angles; (B, D) Right anterior oblique (RAO) 40°, Caudal 6°: 3D-DSA reconstruction demonstrates the optimal display of the anastomosis; (C, E) Left anterior oblique (LAO) 20°, Caudal 0°: 3D-DSA reconstruction demonstrates the optimal visualization of the lesion in both preoperative and postoperative images.
Reintervention and complications
Although 17.4% of the patients required reintervention, subsequent procedures successfully restored complete patency at the 12-month follow-up. This rate is consistent with the published data on TRAS recurrence following endovascular therapy.20,21 Procedure-related complications were minimal; only two instances of minor, nonobstructive dissection occurred, both of which resolved without further intervention. Importantly, stent fracture or migration did not occur, suggesting that BMSs are reliable for TRAS treatment.22,23
Comparisons with alternative treatment strategies
Compared with open surgical revision, which carries morbidity rates up to 20% and graft loss risk, our minimally invasive approach yielded lower complication rates and higher patency.24–26 Compared with self-expanding stents, our balloon-expandable BMS achieved 100% 12-month patency, consistent with reports favoring balloon-expandable stents for anastomotic lesions.27–29 The cost-effectiveness of using 3D-DSA in all TRAS cases remains an important consideration. Although 3D-DSA requires additional imaging acquisition and post-processing, it may reduce overall procedural complexity by minimizing catheter exchanges, improving wire navigation, and decreasing contrast use in selected patients. These procedural advantages have the potential to offset the additional imaging cost. Future prospective studies are needed to quantify the economic impact and to determine which patient subgroups may benefit the most from routine 3D-DSA application.
Limitations and future directions
Several limitations should be noted. First, this study's retrospective, single-center design may limit the generalizability of our findings, and a median follow-up of 14.9 months might be insufficient to fully assess long-term durability. In addition, several potentially relevant baseline variables—including smoking status, history of thrombosis, anticoagulation use, number of antihypertensive medications, donor type, and detailed donor demographics—were not uniformly available because many transplant surgeries were performed at external centers. Detailed reconstruction data were unavailable because all kidney transplant surgeries were conducted at external centers, and operative records were not accessible for review. The incomplete nature of these retrospective records limited our ability to perform more granular risk-factor analyses. Additionally, the small sample size limits the statistical power of the study and may reduce the precision and generalizability of the observed outcomes. Future studies incorporating prospective multicenter designs and extended follow-up periods are warranted to validate these results and further optimize the management of TRAS.
Conclusions
Endovascular treatment of TRAS, particularly when supported by detailed anatomical assessment and 3D-DSA guidance, may provide favorable technical success and short-term clinical improvement. Combined POBA and BMS may offer more sustained patency than POBA alone. Further studies are needed to confirm these findings and to evaluate long-term durability.
Footnotes
Acknowledgements
We thank the patients and clinical staff involved in this study.
Authors’ contributions
Conceptualization: Y.H. and W.L.; analysis and interpretation: X.X., Y.L. and X.H.; data collection: Y.H., X.X., and Y.L.; writing the article: Y.H., X.X., and Y.L.; statistical analysis: X.X.; funding acquisition: Y.H. and X.X.; overall responsibility: W.L. All authors have read and agreed to the published version of the manuscript.
Informed consent
The authors obtained written informed consent from the patient for the publication of the case information and any images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Xiamen Medical and Health Guidance Project [grant number 3502220214201088 and 3502220214201062].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and analyzed during the current study are not publicly available due to institutional restrictions on patient data but are available from the corresponding author upon reasonable request.
