Abstract
Objective
To assess medication appropriateness using the medication appropriateness index (MAI) criteria, the prevalence of potentially inappropriate medication (PIM) prescribing, and factors associated with increased PIM use in elderly ambulatory patients with cirrhosis.
Methods
A cross-sectional study of 70 elderly ambulatory patients with cirrhosis (≥65 years old), from January to December 2021. Two clinical pharmacists evaluated medication appropriateness using the MAI tool. Data were analyzed using SPSS version 27.0. Descriptive statistics were used to present patient demographics, clinical characteristics, and MAI responses. Regression analysis was used to identify predictive factors for PIM prescribing. The kappa statistic was used to assess interrater agreement.
Results
All participants had at least one medication with ≥ 1 inappropriate MAI criterion, with the most common issue being incorrect treatment duration (18%). Of 610 evaluated medications, 44.1% were classified as PIMs. The mean MAI scores per patient and per medication were 15.97 (±10.48) and 1.83 (±3.18), respectively, consistent with other studies. However, the prevalence of PIMs was higher, likely due to the specific disease population studied, as this is the first study to evaluate medication appropriateness in cirrhotic patients. Good interrater agreement was observed (kappa = 0.74), indicating good interrater reliability. Increased inappropriate prescribing was associated with the number of medications, age and severe renal impairment.
Conclusions
PIMs are common in elderly patients with cirrhosis, highlighting the need for better prescribing practices to ensure medication safety. Involving clinical pharmacists with geriatrics expertise and using medication appropriateness tools can reduce PIMs and drug-related problems. Further, healthcare team training is essential to improve prescribing practices. Assessing PIMs in this population could enhance clinical outcomes, reduce adverse drug reactions, and lower healthcare costs. Incorporating comprehensive medication management into routine care for elderly cirrhotic patients is a key strategy to improve patient safety and quality of life.
Keywords
Introduction
Polypharmacy, the use of five or more medications 1 is a prescribing challenge, particularly for older adults with multiple comorbidities. 2 In addition, this population is underrepresented in clinical trials, limiting our understanding of the medications impact on older adults. 3 Changes in pharmacokinetics and pharmacodynamics that occur with aging are usually associated with unfavorable adverse effects and drug interactions. 4
Polypharmacy in the elderly has doubled during the past two decades 5 and is associated with increased use of potentially inappropriate medications (PIMs), 6 including patients with liver cirrhosis, the end result of chronic liver disease. Liver cirrhosis, a leading cause of mortality, has a global mortality rate increase of 47.17% from 1990 to 2017. 7 The essential drivers of morbidity and cost in cirrhotic patients are complications of decompensated disease. 8 Moreover, healthcare providers prescribe a wide variety of medications to treat these complications. 9 Thus, optimizing medication regimens is an essential perspective for older adults, especially those presented with polypharmacy. Therefore, some studies emphasize the importance of personalized medication regimens in elderly populations to improve their health-related quality of life. 10
PIM, defined as medications that have been prescribed to patients whose risks outweigh benefits. 11 Prescribing pertains to adverse health outcomes such as deterioration of physical status, preventable hospitalization, and death.12,13 To address medication inappropriateness, several explicit and implicit criteria have been introduced based on experts consensus to screen for “PIMs” in older individuals. 3 The explicit criteria primarily focus on drugs and evaluating whether they meet a predetermined set of criteria, based on strict standards. Implicit criteria, on the other hand, entail the judgment of healthcare professionals to evaluate medication appropriateness. 14 Implicit criteria help the evaluation process by benefiting from clinical knowledge and judgment. 14 One of the early efforts to standardize this approach was the advancement of an authoritative adverse drug reaction causality algorithm. 15 However, the medication appropriateness index (MAI) is considered the most valid tool to assess medication appropriateness. 16 The validity of the MAI has been studied in the literature in different patient settings. We and others have previously shown that it is valid to detect PIMs in elderly patients with chronic diseases.17–19
The MAI was introduced to improve prescribing quality based on clinical pharmacist interventions in the context of randomized clinical trials. 20 It consists of 10 questions with three-point Likert-type rating choices: “A” being appropriate, “B” being marginally appropriate, and “C” being inappropriate. 20 According to this instrument, the drug is recognized as inappropriate once a single item is evaluated as inappropriate. While item-level evaluation can be informative, it is believed that the MAI utilization would be improved by calculating a single summated score per medication. 21 Therefore, several studies report the MAI score as a summated score per patient.22–24 The weight of each criterion ranges from 1 to 3 (3 = definitely important, 1 = moderately important). 16 The originator of the scale expected a three-point change to be qualitatively significant. 16
Patients with cirrhosis are usually prescribed multiple medications for therapeutic and prophylactic purposes. 25 However, polypharmacy is associated with poor adherence and medication discrepancies, 26 which contribute to patient harm or reduced therapeutic efficacy. 27 Understanding the causes of medication inappropriateness in outpatient clinics can help physicians optimize medication prescribing, permitting optimizing the treatment of different diseases from bioethical and economic points of view. This study did not collect information about how prescribing physicians considered the degree of hepatic impairment when prescribing a medication. While previous studies extensively studied PIM and polypharmacy in elderly population, there is a lack of studies that specifically address outpatient cirrhotic population. This study aims to assess the prevalence of PIM in elderly patients with chronic liver diseases and on polypharmacy using MAI criteria, to provide insights into optimizing pharmacological treatment for this group.
Methods
Study population and setting
This retrospective cross-sectional study was conducted at gastroenterology outpatient clinics at King Abdullah University Hospital (KAUH) in Jordan. Medical records of 70 elderly patients diagnosed with cirrhosis and had been under follow-up between January and December 2021 were reviewed and included in the study. The included patients were all older adults (≥65 years old) with polypharmacy (defined as taking ≥5 medications for at least three months), who had visited the gastroenterology outpatient clinics. Subjects younger than 65 years old or those with incomplete data were excluded from the study. The 70 patients were randomly selected using the “random” function in SPSS. This study was conducted at a single site, which may restrict the generalizability of its findings to larger populations. However, this focused approach allows for more controlled conditions and deeper insights into the specific context of the study.
Data collection
Patients’ medical records, including electronic and paper, were retrospectively reviewed to collect the required data. Patients’ demographic and clinical data were collected. Demographic data included age, gender, and body weight (as available). Clinical data included: relevant past medical history and diagnoses during the last three years, current diagnoses (list of diseases), relevant treating physician's comments/medical notes (as available), relevant laboratory data from the last two years, and medications list (including name, dose, strength, frequency, directions, route of administration, and duration of treatment) (Appendix 1).
Evaluation of prescribing appropriateness
After they received proper training on the tool, two clinical pharmacists with three years of experience applied MAI criteria to each medication in this study. In this study, we excluded the 10th criterion related to cost. Therefore, the maximum summated score per medication became 17. The reason for excluding the cost criterion was because most patients at this hospital had governmental insurance. Medications are typically sourced through government tenders, meaning the actual cost may not accurately reflect the appropriateness of the medication in this context. Prior to the actual study, a pilot study was conducted on three patients who had been prescribed a total of 22 medications. This helped to better understand the process and minimize discrepancy in evaluation process. All necessary changes were made based on the results of the pilot study.
The two raters initially evaluated each medication using the nine criteria: indication, effectiveness, dosage, correct directions, practical directions, drug–drug interactions, drug–disease interaction, therapeutic duplication, and duration. 28 Each item was rated on a three-point Likert-scale as described above. The points represent prescribing within medical standards (appropriate “score = 0”), marginally within medical standards (marginally appropriate “score = 0.5”) or outside of medical standards (inappropriate “score = 1”). 28 The answers were guided by an approved guideline “UpToDate” or standard textbooks. Each rater's scores were sent to a third person who performed the appropriate analyses on them. Then, both raters met to clarify discrepancies, and reach a consensus final score for each criterion. Before reaching the consensus score, the two raters rated each medication independently, they met to resolve any discrepancies. The reached consensus score was based on their discussion and after reviewing each medication together. It took into consideration each patient's characteristics, then reached a mutual understanding to agree on the final consensus score for each medication. The MAI score for each medication was weighted out of 17 (0 = no prescription error, 17 = most prescribing problems). 28 The weighted score for each patient was calculated by summing scores across all medications prescribed to that patient. The number and percentage of patients with at least one medication with an inappropriate MAI criterion were also calculated. In addition, the summated weighted MAI score was also calculated for each patient.
Statistical analysis
Data were analyzed using Statistical Package for the Social Sciences (IBM SPSS statistics for Windows, Version 27.0). Descriptive statistics were used to summarize patients’ demographics and clinical characteristics, most frequent illnesses and prescribed medication classes, number of medications and patients with appropriate, marginally appropriate, and inappropriate MAI responses, and distribution of prescribed medications according to their weighted MAI score. Data were checked for normality of distribution using the Shapiro–Wilk's test (p-value >.05) with visual inspection of histograms. Categorical variables were presented as frequency (percentage) while continuous variables with normal distribution were presented as M ± SD, while continuous variables without normal distribution were presented as median (interquartile range (IQR)).
Since the dependent variable (summated MAI score) was not normally distributed, quartile regression analysis was used to identify predictive factors associated with PIM prescribing. In the initial analysis, the following factors were analyzed individually: age, gender, number of medications, number of chronic diseases and glomerular filtration rate (GFR). Then variables with a p-value < .250 were included in the regression model. Statistical significance was set at a p < .05.
The agreement, the chance-adjusted agreement (kappa statistic),11,22 and the proportion of appropriate (Ppos) and inappropriate (Pneg) ratings from the two raters were calculated for each one of the nine MAI questions. The Kappa statistic was used to calculate the overall interrater agreement between the two raters.11,22 A Kappa value of 0.40–0.75 was considered to have good reproducibility.11,22
The sample size of 70 patients was selected based on this type of research in literature. In addition, since this type of patient is on large number of medications, the number of evaluated medications was large enough based on this kind of research. The selected sample size (n = 70) was able to detect a large effect size (0.267) with a power of 0.80 and an α = 0.05. 29 An effect size of 0.010 is classified as small, 0.090 is medium, and 0.250 is large. 29
The reporting of this study conforms to STROBE guidelines. 30
Ethical approval
The study was approved by the Institutional Review Board (IRB) committee at King Abdullah University Hospital (Approval No. 43/141/2021) before any study related activities started. This research was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2013. Patient details that may identify the patient were deidentified in the study. All collected data was treated in a confidential manner with patient privacy being protected, and no patient confidential information disclosed.
Results
Demographic and clinical characteristics of patients
A total of 1735 patients with liver cirrhosis visited the gastroenterology outpatient clinic. Of the 170 patients who were considered eligible, 70 patients were randomly selected (Figure 1). The median (IQR) age was 73.00 (69.00–78.25) years (age range 65–90), all were Caucasian. Approximately two-thirds of the patients (64.3%) were females. As shown in Table 1, less than half (45.7%) of the patients were presented with three to four chronic illnesses, 11.4% with one to two and 42.9% had five or more chronic illnesses. Half of the patients (50.0%) were taking 9–12 medications, 45.7% on five to eight medications, and only 4.3% were taking 13 or more medications daily (medications range 5–14). The median summated weighted MAI score (IQR) per patient and per medication were 13.5 (8.0–21.0) and 0.00 (0.00–2.00), respectively.
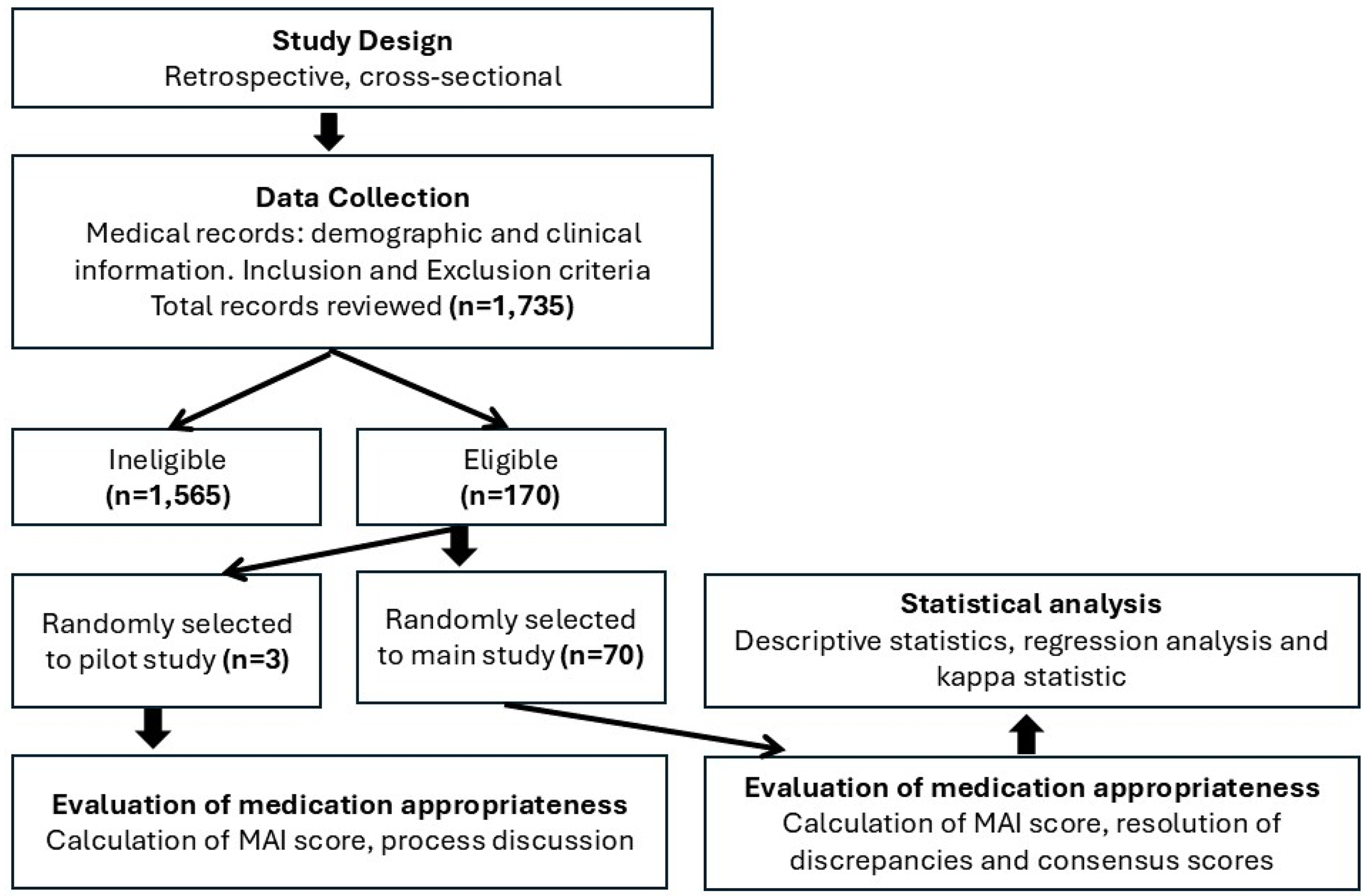
Flowchart of patient selection and study methodology.
Demographic and clinical characteristics of participants, and weighted MAI score per patient and per medication.

SD: standard deviation; IQR: interquartile range; GFR: glomerular filtration rate; DMRD: The original Modification of Diet in Renal Disease; MAI: appropriateness index.
Based on the GFR (M ± SD = 55.03 ± 29.24 mL/min), 12.9% of the patients had normal renal function, whereas 24.3%, 44.3%, 12.9%, and 5.7% had mild, moderate, severe renal impairment and end-stage renal disease, respectively. A detailed description of patients’ demographic and clinical characteristics is presented in Table 1. The association of patient variables with medication inappropriateness as evaluated by MAI score and number of PIMs is discussed below.
The digestive system pathogenesis (24.3%) was the second after circulatory diseases (31.9%) in terms of the most frequently present illnesses (Table 2). Cardiovascular medications were the most frequently prescribed medication classes (25.9%), followed by diuretics and gastrointestinal medications (17.9% and 15.9%, respectively). The least frequently encountered medications were bisphosphonates (1.0%; Table 2).
The top 10 present illnesses, most frequently prescribed medication classes along with their corresponding proportion of PIMs and median summated MAI score per medication class.

MAI: medication appropriateness index; PIM: potentially inappropriate medication; IQR: interquartile range.
Prevalence of PIMs and MAI scores
As illustrated in Table 3, all study participants had PIMs. Less than one fifth of the patients (18.5%) had 1–2 PIMs, and 22.9%, 30%, and 28.5% presented with three, four, and five or more PIMs, respectively. The median (IQR) number of PIMs per patient was 4.00 (3.00–5.00) (range 1–10). More than two-thirds (44.1%) of the medications presented with at least one inappropriate MAI criterion.
Number and percentage of patients with potentially inappropriate medications, number of potentially inappropriate medications per patient, and total number of medications with potentially inappropriate criteria.

PIMs: potentially inappropriate medications; MAI: medication appropriateness index; SD: standard deviation; IQR: interquartile range.
Our results show that the medication class with the highest percentage of PIMs was cardiovascular medications (29.4%), followed by gastrointestinal medications (26.4%) and vitamins and minerals (13.8%) (Table 2). In contrast, the medication classes with the lowest percentages of PIMs were bisphosphonates, respiratory medications, and pain and antiinflammatory drugs (<1.0%). Additionally, dermatologic, ophthalmologic, and immune medications had no PIMs (0.0%) (Table 2).
Medications used for gastrointestinal diseases had the highest median (IQR) summated MAI score among all classes 4.0 (0.0–4.0), followed by cardiovascular medications 0.5 (0.0–2.0). The rest of the medication classes had a median of 0.0 and IQR ≤2.0. Medications used for pain and inflammation and genitourinary had a median of 0.0 and IQR: 0.0–0.0 (Table 2). In summary, cardiovascular and gastrointestinal medication classes had the highest prevalence of PIMs and MAI scores among other medication classes, while some other medication classes had no PIMs.
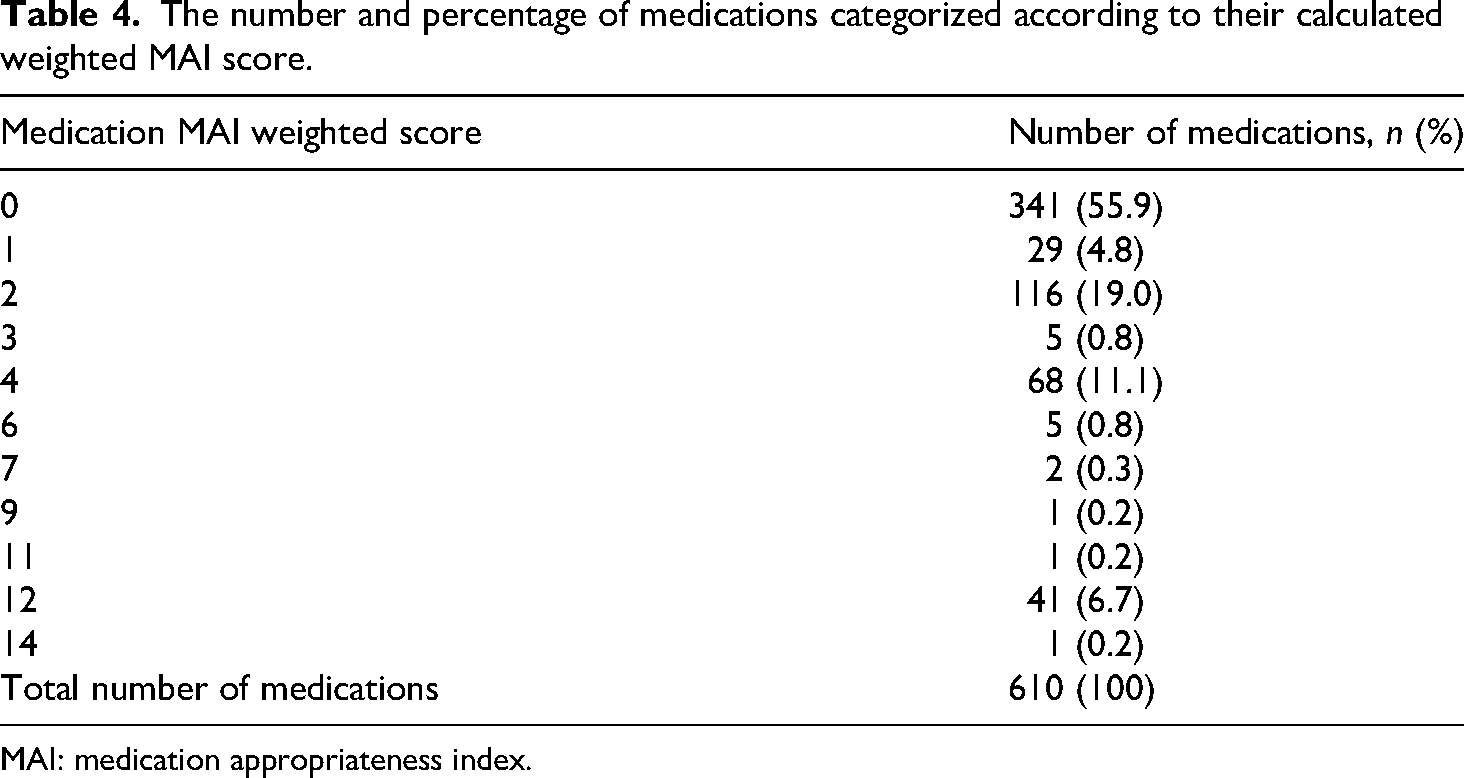
Table 4 describes the distribution of prescribed medications according to their calculated weighted MAI score. A total of 610 prescriptions were assessed by two clinical pharmacists. More than half of the medications, 341 (55.9%), were found to be appropriate. While 269 (44.1%) had one or more inappropriate ratings in the nine criteria. Only 6.9% of the medications had a weighted score of ≥12, suggesting more potential for prescribing errors (Table 4).
The number and percentage of medications categorized according to their calculated weighted MAI score.
MAI: medication appropriateness index.
MAI responses and interrater reliability
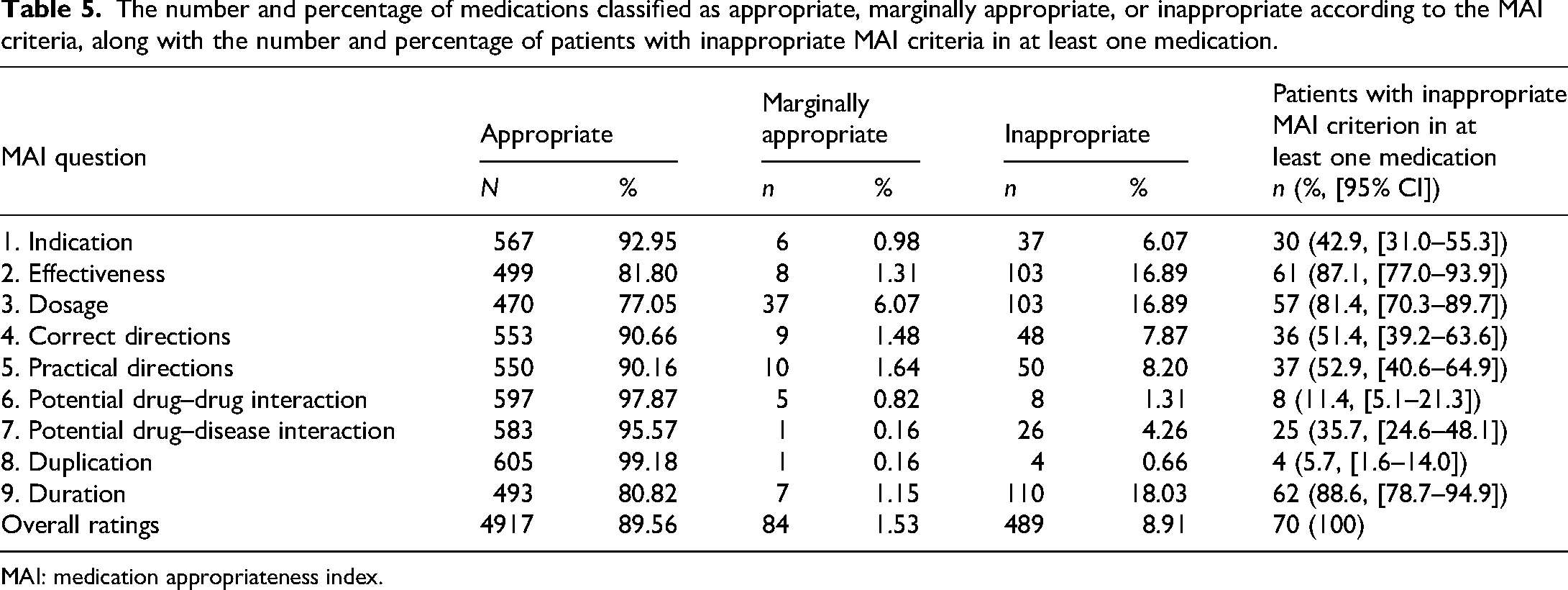
Table 5 reports the proportion of patients with appropriate and inappropriate medications based on the evaluated MAI criteria. For indication, 92.95% of the prescriptions had appropriate indications, 0.98% were marginally appropriate, and 6.07% were inappropriate. Our study revealed that 18.3% of responses related to medication duration were inappropriate prescribing, followed by treatment dosage and effectiveness (16.89%; Table 5). In contrast, drug duplication had the lowest percentage (0.66%).
The number and percentage of medications classified as appropriate, marginally appropriate, or inappropriate according to the MAI criteria, along with the number and percentage of patients with inappropriate MAI criteria in at least one medication.
MAI: medication appropriateness index.
On the other hand, most of the study participants (88.6%; 95% CI [78.7–94.9]) presented with inappropriate MAI criteria (duration) in at least one medication. The proportion of patients with inappropriate criteria for medication effectiveness, dosage, practical and correct directions were 87.1%, 81.4%, 52.9%, and 51.4% respectively (Table 5). However, only 11.4% and 5.7% of patients had inappropriate MAI criteria for potential drug–drug interaction and medication duplication, respectively.
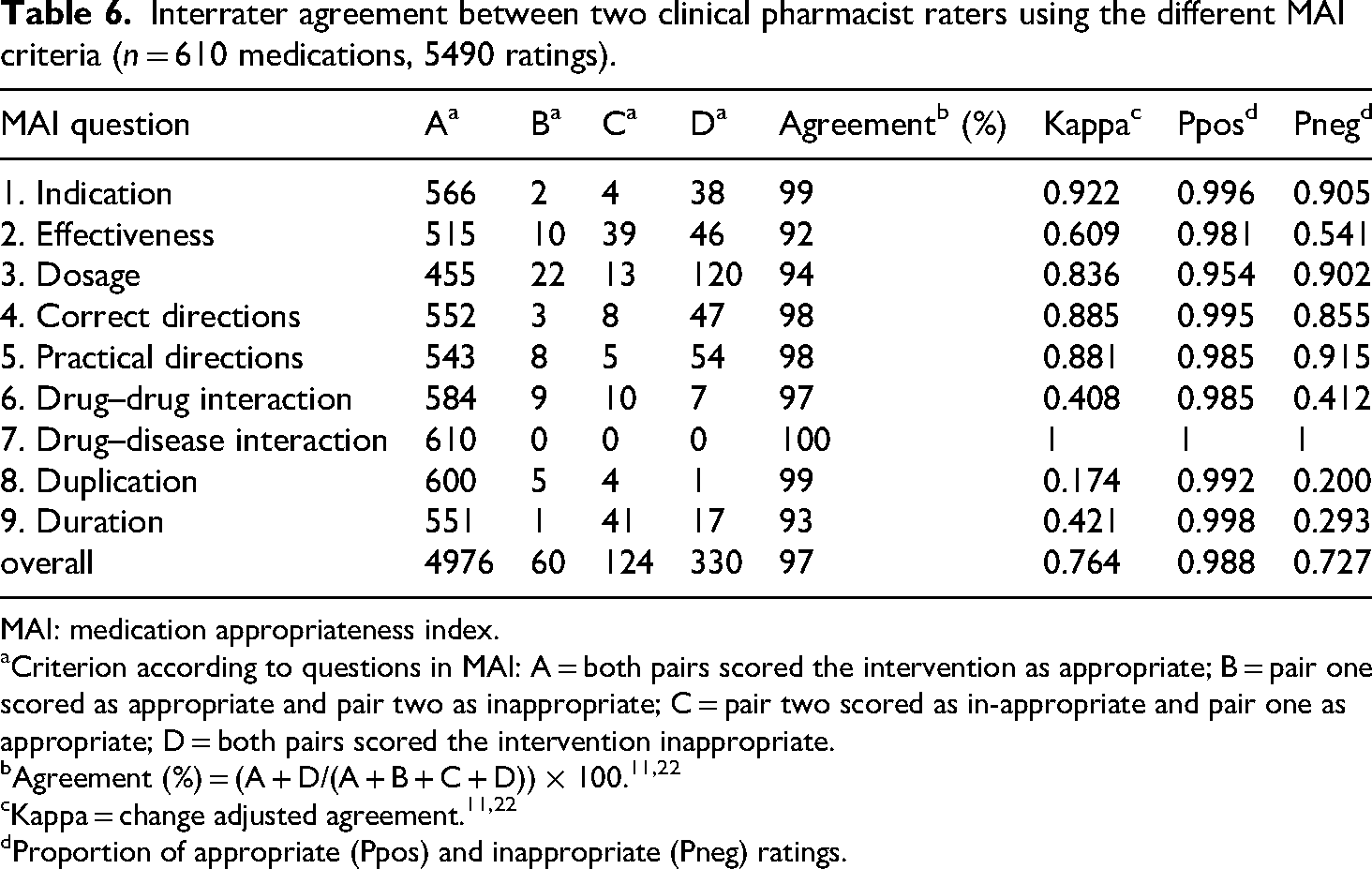
Table 6 outlines the interrater agreement between the two pharmacist raters. The level of agreement was ≥92% for all criteria with an overall agreement of 97% and good chance-adjusted agreement (kappa 0.764).
Interrater agreement between two clinical pharmacist raters using the different MAI criteria (n = 610 medications, 5490 ratings).
MAI: medication appropriateness index.
Criterion according to questions in MAI: A = both pairs scored the intervention as appropriate; B = pair one scored as appropriate and pair two as inappropriate; C = pair two scored as in-appropriate and pair one as appropriate; D = both pairs scored the intervention inappropriate.
Proportion of appropriate (Ppos) and inappropriate (Pneg) ratings.
As summarized in Table 6, the interrater agreement for each of the nine different MAI criteria was ≥ 0.954 for the appropriate rating (Ppos) and ≥0.200 for inappropriate prescribing (Pneg).
Factors associated with inappropriate prescribing as determined by MAI criteria
The variables that may affect inappropriate prescribing as determined by MAI criteria were evaluated, including age, gender, renal function, number of medications, and number of illnesses. Based on the multivariable quartile regression analysis of the summated MAI score, only the number of medications and renal function (GFR) were independently associated with higher summated MAI scores (Table 7). For every drug taken, the MAI score increased by 2.195 (95% CI [0.902–3.489], p = .001) points. As shown in Table 7, patients receiving 9–12 medications (OR = 7.750, 95% CI [2.220–13.280], p = .007) were more prone to have higher summated MAI scores compared to the reference group (five to eight medications). On the other hand, increased GFR was associated with lower MAI scores (OR = −0.104; 95% CI [−0.195–−0.013]; p = .025). Compared to normal renal function patients (reference), patients with end-stage renal disease had higher MAI scores (OR = 22.000; 95% CI [9.661–34.339]; p = .001). Patients with mild and severe renal impairment tend to have lower MAI scores, while patients with moderate renal impairment tend to have decreased MAI scores, however, in either case the change was statistically significant (Table 7). On the other hand, the patient's age, gender, and number of illnesses were not associated with higher summated MAI scores. Our results show that the association between patients’ age and gender, and summated MAI score was not significant. There was a tendency to have lower summated MAI score as number of illnesses increases, however, the difference was not statistically significant. Patients who had three to four and ≥ 5 chronic diseases tend to have lower MAI scores (OR: −11.571, 95% CI [−20.234–−2.908]; and OR: −9.679, 95% CI [−19.201–−0.156], respectively) compared to those who had one to two chronic diseases (Table 7).
Patient factors associated with increased summated weighted MAI score according to the different MAI criteria.

MAI: medication appropriateness index; OR: odds ratio; CI: confidence interval; GFR: glomerular filtration rate.
*: significant (p < .05).
Discussion
Liver cirrhosis is the end stage of a group of chronic liver diseases that have common features of fibrosis, necroptosis, and regenerative nodules, which affect the structure of hepatocytes to decrease their functional mass and modify the vascular architecture. 31 Globally, in 2022, cirrhosis was one of the top 10 leading causes of death. 7 Patients with cirrhosis are usually prescribed several medications for both curative and prophylactic purposes. 32 Thus, polypharmacy can lead to poor adherence which have been associated with PIMs. 33
Studies on patients with liver cirrhosis focusing on medication appropriateness are scarce in the literature. To the best of our knowledge, the present study appears to be the first to identify the prevalence of PIMs among patients with cirrhosis using the MAI as a reference standard.
We omitted the question related to medication cost due to the complex payment system in the country. This complexity may cause issues in identifying which drug is the least expensive since this will vary according to the way medications are purchased as wholesale using bidding tenders. Public hospitals in the country usually buy their medicines through tenders at special prices that differ from the common price for the public. These tenders can rapidly change over time, limiting the validity of cost analyses.
This study shows a high prevalence of PIMs, consistent with previous literature.34,35 Patients with cirrhosis attend to have higher disease burden, more medications, frequent prescription changes, and more hospitalizations, making them more susceptible to PIMs. 27 Therefore, these findings highlight the importance of medication prescription quality in reducing harm.
All participants in our study had at least one PIM, with the prevalence higher than in some previous studies.14,36 This could be due to the fact that Beers criteria are the commonly utilized criteria to evaluate medication appropriateness among individuals aged 65 years and older in healthcare settings. 37 Our study used MAI as a reference standard which mainly focuses on the patient's complete drug regimen with clinical conditions, whereas Beers criteria has inflexible criteria that focus solely on drug classes without addressing patients’ preferences. 14
In this study, we evaluated inappropriate medication prescribing using two approaches: the summated weighted MAI score per patient and the number of medications with at least one inappropriate MAI criterion. The MAI score provides a detailed criterion-based evaluation of medication appropriateness, while the number of PIMs counts medications with any inappropriate MAI criteria.
The median (IQR) summated MAI score per patient was 13.5 (8–21) indicating that treatment of a relatively small group of participants was considered inappropriate. This aligns with previous studies.38,39 The median (IQR) summated MAI score per medication was 0 (0–2), comparable to that reported by Lopez-Rodriguez et al. 40 and by Lund et al. 38
The most common issue with inappropriate prescribing was related to the treatment duration, which accounted for 18% of all inappropriate prescribing. This includes instances where medication was prescribed for either a longer or shorter period than necessary. This is particularly significant for cirrhosis patients, as liver function deterioration can lead to medication accumulation, increasing the risk of adverse effects and potentially worsening the progression of cirrhosis due to altered pharmacokinetics and pharmacodynamics. For example, prolonged exposure to some sedative drugs like benzodiazepine, NSAIDS, and some opioids metabolized by the liver can increase the risk of hepatic encephalopathy, gastrointestinal bleeding, and renal impairment.41–43 Additionally, polypharmacy with inappropriate medication duration increases the risk of drug interactions, cumulative toxicity and reduced treatment adherence, complicating the therapeutic plan. Therefore, it is essential to ensure an appropriate treatment period to avoid these harmful effects.
Regarding risk factors for inappropriate prescribing based on the summated MAI score of the patient, both renal function and number of medications were significant as found in previous studies.42–44 However, when considering the total number of PIMs per patient, only age and the number of medications were found to be associated with increased medication inappropriateness. As people get older, changes in pharmacokinetics and pharmacodynamics, decreased renal function, and impaired hepatic function that even small changes in the prescribing process can lead to significant clinical consequences, highlighting the need for caution and frequent medication reviews in the elderly. Additionally, consistent with other studies,40,44 patients prescribed ≥ 13 medications are more likely to experience medication errors. This underscores the challenges of polypharmacy, common in patients with multiple comorbidities. These patients are at increased risk of drug interactions, monitoring difficulties, adherence issues, and cumulative toxicity. These findings emphasize the need for a validated tool to improve rational drug prescribing in the elderly population.
A strong agreement was found between the two clinical pharmacists’ scores, with an overall kappa coefficient of 0.74, indicating significant reliability between the raters. This value is similar to or slightly higher than the values reported in other studies.11,22 The kappa coefficient value can vary based on differences in the ratters’ clinical experience, background, and familiarity with the MAI instrument. Therefore, the selection, training, and pretesting of the raters are crucial for obtaining reliable results.
This study highlights the importance of regular medication reviews and tailored treatment plans for elderly patients with cirrhosis, who often experience polypharmacy, hepatic impairment, and other comorbidities. Healthcare professionals should use validated tools like the MAI to assess medication appropriateness and focus on managing medication duration to prevent drug-related issues. It is essential to take into consideration the degree of renal impairment when prescribing medications to elderly people with cirrhosis. Prescribers should aim to reduce polypharmacy, when possible, address drug interactions, and ensure prescriptions are both safe and necessary. Future research should explore how cirrhosis-related changes in pharmacokinetics affect the body's handling of medications, especially considering the increased medication burden in these patients.
The strengths of our study include being the first to evaluate the prevalence of PIMs using the MAI criteria in elderly cirrhosis patients attending outpatient gastroenterology clinics. Additionally, we considered several patient-specific factors when assessing PIM prescribing, and we evaluated a relatively large number of medications. Data were collected from a teaching hospital, where physicians’ notes, prescriptions, and laboratory results were thoroughly reviewed.
Our study has several limitations. Firstl, the relatively small sample size may limit the generalizability of the findings, making it more challenging to draw definitive conclusions. However, our results are comparable to those from previous studies. Additionally, the single-site design may affect generalizability, as the findings may not fully represent patients from diverse geographical and clinical settings. The cross-sectional nature of the study and the absence of patient interviews could also influence the outcomes. Furthermore, excluding the medication cost is a crucial factor in medication selection. Incomplete data reporting including undocumented medications like over-the-counter medications or herbal supplements, may underestimate the magnitude of PIMs. Lastly, not interviewing the prescribers to understand their rationale for prescribing PIMs. Considering hepatic impairment in their decisions could provide further insights.
To address these limitations, future prospective, multicenter and interventional studies are needed to validate these observations. Overall, this study emphasizes the need for future research on optimizing medication and dosage selection for elderly patients with hepatic cirrhosis. These studies should focus on how cirrhosis affects pharmacokinetics and pharmacodynamics, influencing drug class selection and dose adjustment based on liver function. Additionally, evaluating the clinical implications of inappropriate treatment and drug interaction will offer valuable insights to improve treatment guidelines, particularly for patients on polypharmacy.
Conclusions
This study highlights the high prevalence of PIMs among patients with liver cirrhosis emphasizing the critical need for medication review and management. Our findings underline the need for improving prescribing practices in this population particularly regarding treatment duration, which can lead to more drug-related problems in patients with hepatic cirrhosis. The number of medications and renal function (GFR) were identified as significant contributors to medication inappropriateness. This reinforces the need for regular medication reviews and the use of validated tools such as the MAI to optimize patient therapy. In conclusion, the study suggests that the MAI tool can enhance the medication review process and facilitate personalized prescribing for elderly cirrhosis patients. Additionally, reducing the number of prescribed medications, when feasible, can minimize medication inappropriateness. Future research should build upon the findings of this study. First, larger multicenter studies across various geographical regions are needed to validate the results. An interventional study evaluating the impact of implementing the MAI tool would also be valuable in understanding the long-term effects of PIM prescribing in cirrhosis patients. Additionally, further studies should focus on gathering more data through patient and prescriber interviews to better understand the rationale behind prescribing decisions. Moreover, research involving other clinicians in reviewing medications and assessing their appropriateness using the MAI tool, while also comparing it with other tools, would provide valuable insights.
Footnotes
Acknowledgements
The authors would like to thank the IT department at KAUH for their technical support.
Ethical considerations
The study was approved by the IRB committee at King Abdullah University Hospital (Approval No. 43/141/2021) before any study-related activities started. All collected data was treated in a confidential manner with patient privacy being protected, and no patient private information disclosed.
Author contributions/CRediT
Qusai Y. Al-Share: idea conception, study design, data analysis and interpretation, drafting and submission of manuscript, approval of final version, and overall supervision; Aseel Y. Tahrawi: data analysis and interpretation, drafting of the manuscript, and approval of final version; Dyala M. Khasawneh: data analysis and interpretation, drafting and submission of the manuscript, and approval of final version; Aya W. Alshare: data analysis and interpretation, drafting of the manuscript, and approval of final version; Shrouq A. Alkhasawneh: data analysis and interpretation, drafting of the manuscript, and approval of final version.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research in Jordan University of Science and Technology, Jordan (Grant No. 43/141/2021).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data is available upon request from the corresponding author.
