Abstract
Background
Intradialytic hypotension (IDH) is a common and serious complication in renal replacement therapy, especially in hospitalized patients. The absence of a standardized definition complicates data synthesis and the development of evidence-based guidelines. Current definitions vary, including different blood pressure thresholds, clinical symptoms, and the need for medical intervention during dialysis. IDH is linked to increased mortality and cardiovascular morbidity and may impede renal recovery in patients with acute kidney injury and chronic kidney disease.
Methods
A systematic review was conducted using MEDLINE via PubMed, Embase, and Web of Science to identify studies reporting IDH prevalence. A meta-analysis of proportions was performed to determine the global prevalence of IDH, with subgroup analyses to explore heterogeneity. The Joanna Briggs Institute's checklist was used to assess the risk of bias in prevalence studies. The PRISMA guidelines were followed to report the results of this study, PROSPERO registration number CRD42024500622.
Results
The meta-analysis found a global IDH prevalence of 31% (95% CI 0.18–0.44) across nine studies. Significant heterogeneity was observed (I²: 97.87%; p < 0.01), with prevalence rates ranging from 10.7% to 64% based on patient demographics and session characteristics. Sensitivity analysis suggested prevalence could range between 27% and 33% depending on study criteria.
Conclusions
IDH is a significant complication during hospital-based renal replacement therapy, with a global prevalence of 31%. These findings highlight the need for a standardized, evidence-based definition of IDH to improve diagnostic consistency and clinical outcomes through more accurate diagnosis, better treatment strategies, and tailored patient management.
Keywords
Introduction
Cardiovascular instability refers to the clinical state of abnormal vital signs, including blood pressure and heart rate, which can lead to inadequate tissue perfusion. 1 Intradialytic hypotension (IDH) is a common and distressing complication of renal replacement therapies; however, despite its clinical significance, there is no consensus medical definition for this condition. Over the years, numerous definitions have been implemented in clinical and research settings. The inconsistencies in definitions have difficulted data synthesis and the development of evidence-based guidelines for the prevention and treatment of IDH.2,3
The various definitions found in the literature incorporate three main components: a criterion for blood pressure, the occurrence of symptoms (nausea, dizziness, cramps, etc.), and the need for interventions by medical and nursing staff during the IDH session.2,3 The Kidney Disease Outcomes Quality Initiative (K/DOQI) guidelines propose a definition of IDH as a decrease of ≥20 mmHg in systolic blood pressure (SBP) or a decrease of ≥10 mmHg in mean arterial pressure (MAP), accompanied by symptoms of hypotension such as headache, fatigue, seizures, nausea, vomiting, and restlessness during dialysis. However, this definition is not applicable to critically ill patients who cannot report typical symptoms of hypotension or hypoperfusion and whose blood pressure is heavily influenced by concurrent illnesses (e.g. sepsis, cardiogenic shock) and treatments (e.g. mechanical ventilation, vasopressors). 4
It should be clear that IDH is a complication of all commonly used modalities of renal replacement therapy, including intermittent hemodialysis, sustained low-efficiency dialysis (SLED), and continuous renal replacement therapy (CRRT).5–8
The pathophysiology of IDH is diverse; it may be related to the underlying cause of acute kidney injury (AKI), decreased cardiac output from various origins (hypovolemia, hypocalcemia, diastolic dysfunction, etc.), or alterations in vasomotor tone related to membrane/circuit bio incompatibility, ultrafiltrate/dialysate, temperature, or ionic imbalance, among others.8–11
There is sufficient evidence suggesting that IDH negatively impacts the outcomes of patients with renal disease requiring renal replacement therapy. IDH is associated with higher mortality and cardiovascular morbidity and may limit renal recovery in patients with AKI.11–17
In this context, we conducted a systematic review to determine the prevalence of cardiovascular instability during hemodialysis in hospitalized patients to support the development of guidelines for the prevention or management of this complication during renal replacement therapy.
Methods
This study follows the PRISMA guidelines for reporting systematic reviews. 18 The systematic review was registered in PROSPERO with the following registration number (CRD42024500622).
Eligibility criteria
Primary studies related to renal replacement therapy as an exposure in a hospital or critical care setting adults (over 18 years old) were considered, regardless of publication status, language, and year of publication, including conference abstracts, case reports, original manuscripts, and those uploaded to preprint servers. Manuscripts that focused on ambulatory patients, as well as letters, comments, expert opinions, editorials, and other non-original studies, were excluded. Studies involving patients undergoing CRRT or SLED were also excluded.
Information sources
Search Strategies: Between January 2024 and February 20, 2024, a systematic search of information was conducted in the following databases: MEDLINE via PubMed, Embase, and Web of Science. Different combinations of terms, such as instability, hypotension, and dialysis, were used to identify eligible articles. The detailed search strategy is provided in Supplemental Appendix 1. Additionally, the search was supplemented by a manual exploration of the bibliographies of all included studies and a search of the grey literature through preprint repositories.
Study selection process
The selection process used the Rayyan–Intelligent Systematic Review software. 19 Two researchers (KA-V and MV-M) removed duplicate documents from all search results. Subsequently, an initial screening for relevance (whether the study addresses the research question) was independently performed by reading the title and abstract.
A second review was conducted on the selected manuscripts, in which the documents were assessed in full text and the selection criteria applied. A citation search was also conducted to obtain the final number of included documents. Two researchers (KA-V and MV-M) worked independently on including studies. A third investigator (CC-S) resolved disputes.
Data extraction and synthesis process
The data extraction form followed the Joanna Briggs Institute's Data extraction form for prevalence studies (JBI) 20 to record the following information: study details, study methodology, and results (authors, title, journal, year of publication, pages, date of data extraction completion, region and country, design, setting, number of participants and dialysis sessions, data collection period, inclusion and exclusion criteria, primary outcome, secondary outcomes, comorbidities, mean age, sex, severity scores, IDH definition, ethical approval, data analysis method, prevalence, confidence intervals (CI), ICU admission causes, hypotension by sex and age subgroups).
Data list
The form also included fields to capture relevant data for risk of bias (RoB) assessment. Prevalence figures and 95% CI were extracted or calculated from available data using the Wilson method. 21
RoB assessment of individual studies
The RoB in the selected studies was assessed through the Joanna Briggs Institute's critical appraisal checklist for studies reporting prevalence data.22–24 This tool evaluates nine domains 24 : (D1) adequacy of the sampling frame, (D2) recruitment method, (D3) sample size, (D4) study subjects and setting, (D5) coverage, (D6) diagnostic methods, (D7) reliability and standardization of measurements, (D8) statistical analysis, and (D9) response rate. Two evaluators (KA-V and MV-M) independently assessed the eligible studies and rated each question as “yes,” “no,” “unclear,” or “not applicable.” Discrepancies in the assessment were discussed until a consensus was reached. The final score for each study applied to the JBI questions was calculated based solely on the percentage of positive “yes” responses. The RoB for each study was categorized according to the final score as “high” (score of 49% or lower, leading to article exclusion), “moderate” (score ranging from 50% to 69%), or “low” (score higher than 70%).
Graphs considering each RoB domain across all studies were prepared using the ROBVIS R package v. 0.3.0.900. 25 Cohen's Kappa index was used to measure the agreement between the two evaluators; values between 0.61 and 0.8 were categorized as substantial inter-rater agreement, 0.81 and 0.88 as almost perfect agreement, and a Kappa index of 1 as perfect. 26
Synthesis methods
First, tables present a narrative synthesis of the general characteristics of all included studies. These characteristics include authors, title, journal, year of publication, region–country, design, setting, number of participants, number of renal replacement therapies, inclusion criteria, exclusion criteria, primary outcome, mean age, sex, IDH definition, and prevalence.
Given the relevance of the topic and the need to obtain an aggregate value for the prevalence of IDH, a quantitative synthesis is presented by a meta-analysis using a random-effects model, and a restricted maximum likelihood method was performed. Prior to the meta-analysis, a double arcsine transformation was conducted using the Freeman–Tukey formula to control the variance between studies. To improve comprehension, results were re-transformed to prevalence after the meta-analysis. Studies with a high RoB were excluded from the meta-analysis.
To assess heterogeneity, Cochran's Q test and I² were used. Cochran's Q test was considered statistically significant when it reached a value of <0.05. A univariable meta-regression analysis was conducted based on the covariates: RoB, sample size, and the number of sessions vs. the number of participants.
Finally, sensitivity analyses were conducted based on subgroups, correcting heterogeneity with an I² of 10%, a leave-one-out meta-analysis, and standard error corrections of the effect size (prevalence) using the Sidik–Jonkman and Knapp–Hartung truncated methods. All analyses were performed using Stata 18 statistical software. 27
Publication bias assessment
To objectively evaluate publication bias, Egger's test was measured, a funnel plot was graphed, and a trim-and-fill analysis was performed.
Certainty of evidence rating
This assessment was not performed because there is no specific instrument for evaluating the certainty of evidence in prevalence systematic reviews.
Results
Figure 1 shows the flow diagram for article selection. The electronic search identified 8446 articles: 2906 from MEDLINE/PubMed, 2532 from Embase, and 3008 from Web of Science. After screening, 1055 duplicates were removed. A total of 7391 articles were examined by title and abstract, resulting in 82 full-text articles being assessed for eligibility. Of these, 76 articles were excluded for the following reasons: 38 were studies including ambulatory patients, and 38 had a different design. Additionally, three articles were identified from the reference lists of relevant studies. Ultimately, nine articles were included in this systematic review.

PRISMA flow diagram for studies selection.
The nine included articles collected data from 2002 to 2021. Three articles (33.3%) were from Africa,28–30 three (33.3%) were from North America,31–33 one (11.1%) was from South America, 34 and two studies (22.2%) were from Europe.35,36 The countries where studies were conducted included Nigeria (n = 2; 22.2%), the United States (n = 2; 22.2%), France (n = 2; 22.2%), Canada (n = 1; 11.1%), Brazil (n = 1; 11.1%), and Egypt (n = 1; 11.1%) (see Figure 2).

World map with the origin countries of the included studies.
Characteristics of the studies and individuals
Six studies were found to have a low RoB,28,29,32–35 while three (n = 3; 33.3%) presented a moderate RoB.30,31,36 The selection process achieved a Cohen's kappa of 0.737, considered a substantial agreement (see Figures 3 and 4).
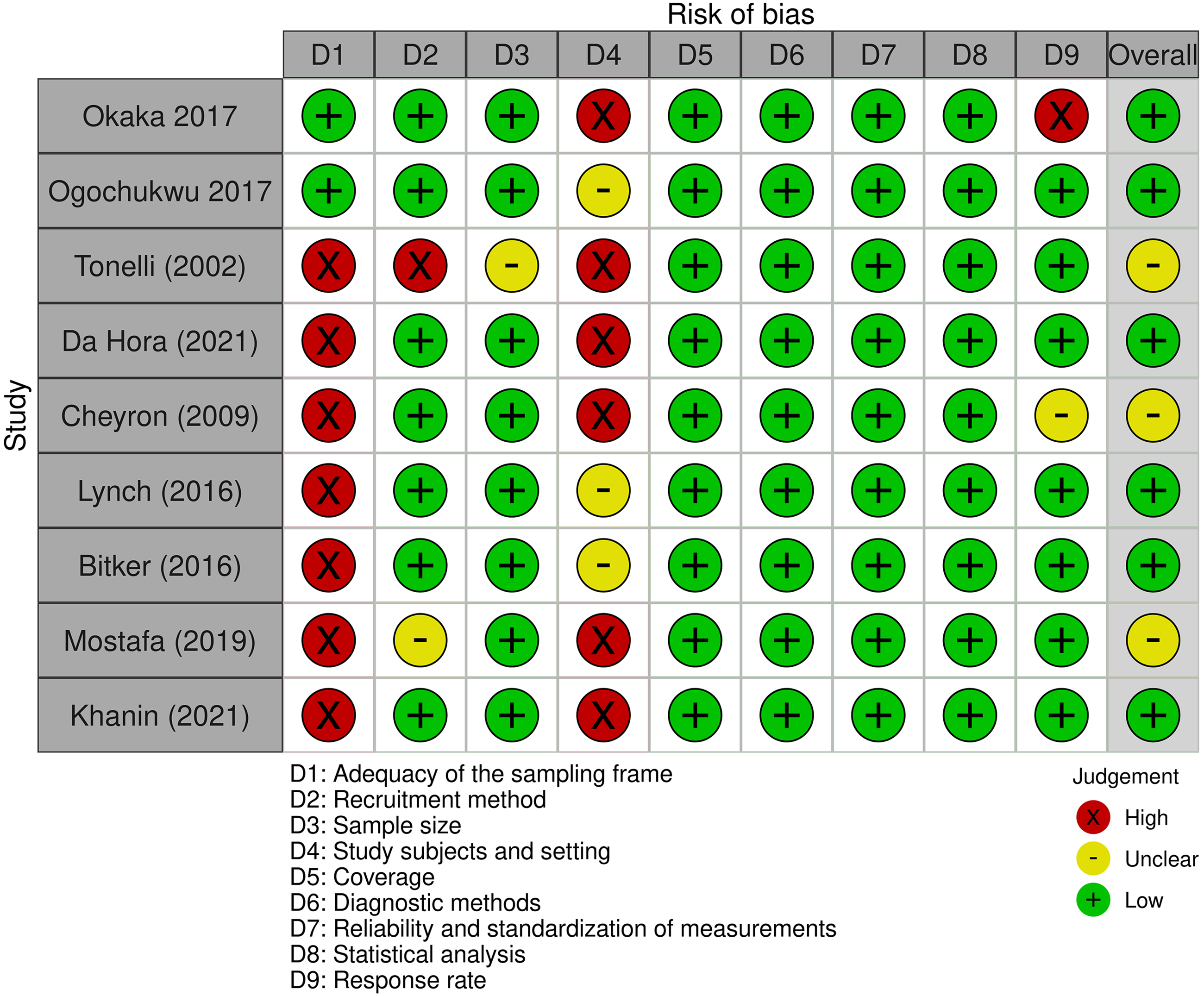
Risk of bias across studies through the Joanna Briggs Institute's critical appraisal checklist for studies reporting prevalence data.

Risk of bias across the domains of the Joanna Briggs Institute's critical appraisal checklist for studies reporting prevalence data.
Three types of study designs were used among the included studies: cross-sectional studies, prospective cohort studies, and retrospective studies. Most studies aimed to investigate the frequency of IDH and identify patient factors, treatment variables, or other variables associated with IDH.
Table 1 summarizes the patient characteristics of the nine articles. Five studies reported their findings by the number of patients included, which ranged from 20 to 404. On the other hand, four studies included the number of hemodialysis sessions.31,32,35,36 The mean age of patients ranged from 42.5 to 68 years. All studies specified the inclusion and exclusion criteria and reported the sex distribution. In seven studies, most patients were men28,29,32–34 and in two studies women.31,36
Characteristics of included studies.
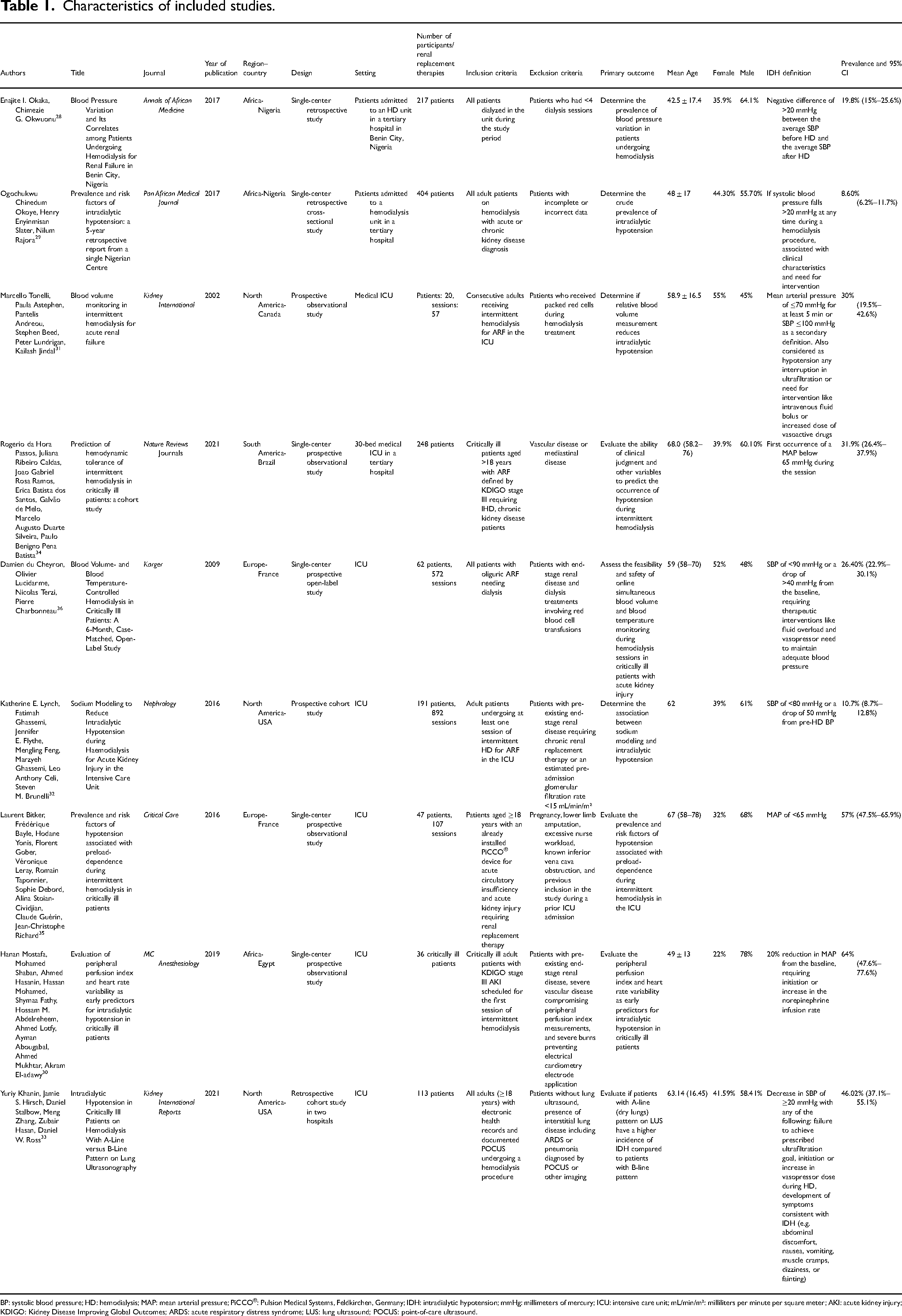
BP: systolic blood pressure; HD: hemodialysis; MAP: mean arterial pressure; PiCCO®: Pulsion Medical Systems, Feldkirchen, Germany; IDH: intradialytic hypotension; mmHg: millimeters of mercury; ICU: intensive care unit; mL/min/m²: milliliters per minute per square meter; AKI: acute kidney injury; KDIGO: Kidney Disease Improving Global Outcomes; ARDS: acute respiratory distress syndrome; LUS: lung ultrasound; POCUS: point-of-care ultrasound.
Seven studies were conducted in intensive care units (ICU),30–36 and two studies were conducted with patients admitted to tertiary hospital hemodialysis units.28,29 The seven ICU studies indicated the most common admission causes, including sepsis and cardiovascular causes. Six articles reported the percentage of individuals undergoing mechanical ventilation at the time of inclusion.30,31,33–36
Six articles reported prognostic and severity scores, such as Charlson, SAPS II, SOFA, and APACHE at the time of inclusion.30,31,33–36 Respectively, one study reported a statistically significant higher APACHE 30 and SOFA 35 score in patients with IDH; however, others found no differences regarding the IDH status.34,36
All articles described the cause of renal replacement therapy. In six articles, the patients had AKI,30–32,34–36 and in three, the patients had both AKI and chronic kidney disease.28,29,33 In five articles, patient comorbidities were detailed, with the most relevant being chronic glomerulonephritis, hypertension, diabetes mellitus, and heart failure.28,29,32,33,36
Definitions of IDH
Of the selected studies, one included a decrease in SBP of ≥20 mmHg 31 as a primary component of its definition; two other studies added clinical symptoms or the need for intervention to this element for diagnosing IDH.29,33 In two studies, hypotension was defined as the first occurrence of a MAP below 65 mmHg during the session.34,35
One defined IDH as a 20% reduction in MAP from the baseline, requiring the initiation or increase of the norepinephrine infusion rate. 30 Two studies identified IDH when SBP values were <80 and <90 mmHg or a drop of ≥50 and ≥40 mmHg, respectively, relative to pre-hemodialysis blood pressure.32,36
One study defined IDH when patients reached a MAP of <70 mmHg for at least 5 min or an SBP of <100 mmHg, or required any pause in ultrafiltration, or needed an intervention such as the administration of intravenous fluid bolus or an increase in vasoactive drug dosage. 31
Prevalence of IDH
From the studies that used the number of sessions, the prevalence of hemodialysis complicated with IDH varied from 10.7% to 57%, as shown in Table 2.
Prevalence by number of sessions.

Studies measuring the prevalence of IDH according to the number of patients showed values ranging from 8.6% in a study with 404 participants 28 to 64% in a study with 36 patients, 29 as shown in Table 3.
Prevalence by number of participants.
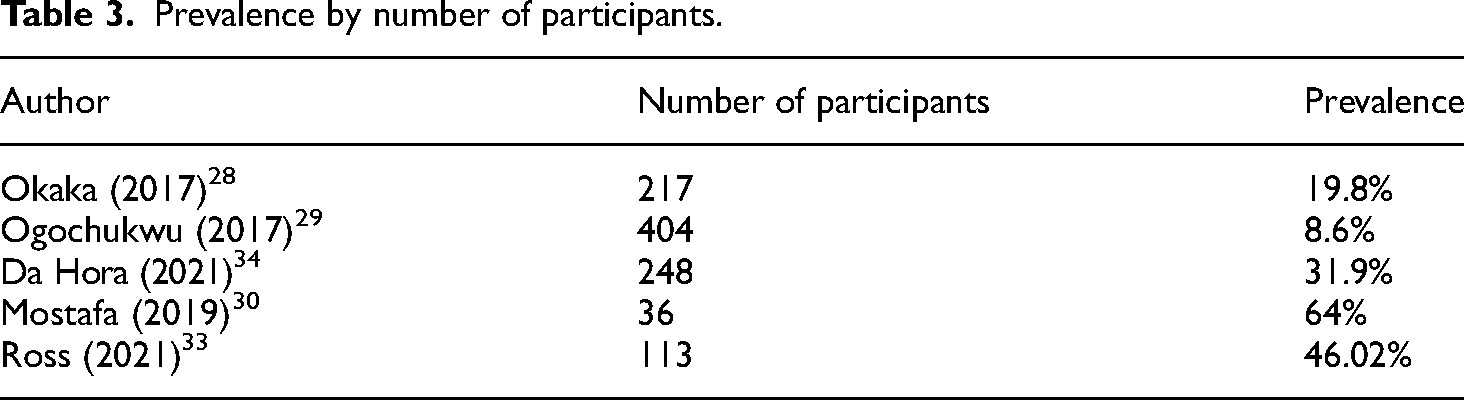
Table 4 shows the prevalence of IDH by sex. In all studies, the prevalence was higher for men.
Prevalence of IDH by sex.

Quantitative synthesis results
Heterogeneity
The heterogeneity among the nine included studies was assessed, and it was concluded that there is high heterogeneity across studies (I²: 97.87%; p < 0.01). It did not differ after subgroup analysis based on the RoB and the number of sessions or participants, where heterogeneity remained considerable. The Galbraith plot is shown in Supplemental Appendix 2.
A meta-regression analysis was performed with the variables sample size, RoB, and number of sessions versus number of participants.
Regarding the number of participants, an I² of 95.5% was identified, of which 46.6% corresponds to this variable. The meta-regression model was significant (p = 0.006). The corresponding bubble plot is shown in Supplemental Appendix 3. Other covariates, such as the RoB and the number of patients versus the number of sessions, showed no significance in the meta-regression model (p = 0.45 and p = 0.85, respectively).
Effect measure
The meta-analysis's overall IDH prevalence across the nine included studies was 31% (95% CI 0.18–0.44). The cumulative prevalences by subgroups of RoB were 27% (95% CI 0.13–0.44) for the low bias subgroup and 39% (95% CI 0.18–0.62) for the medium bias subgroup, as shown in Figure 5. Regarding the number of sessions versus the number of participants, Figure 6 shows the pooled prevalence, which ranged from 32% (95% CI 0.14–0.52) for the number of patient subgroup to 29% (95% CI 0.12–0.50) for the number of dialysis session subgroup. Another analysis was performed with studies involving only patients admitted to the ICU (Figure 7). Subgroups were created based on the measures taken to prevent IDH. The overall prevalence was 36% (95% CI 0.23–0.51). The subgroup with the highest prevalence was the one who received any invasive hemodynamic monitoring (64%; 95% CI 0.47–0.79), and the one with the lowest prevalence was the group that received any dialysis machine biofeedback (21%; 95% CI 0.10–0.35).
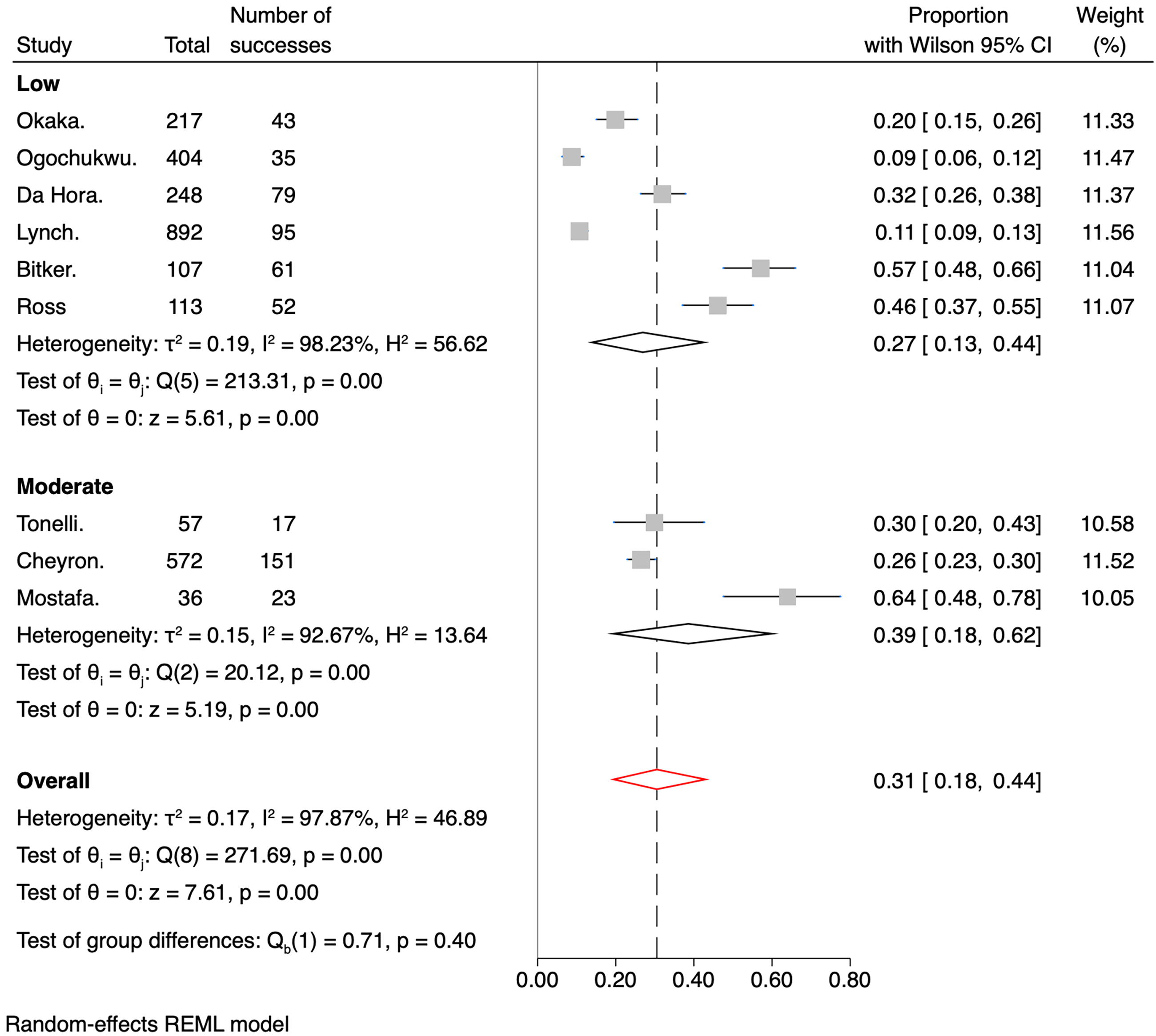
Forest plot for the prevalence of IDH divided by risk of bias.

Forest plot for the prevalence of IDH divided by the number of participants or sessions.

Forest plot for the prevalence of IDH in critical patients divided by the measures adopted to prevent IDH.
In the sensitivity analysis, assuming an I2 of 10%, the prevalence of IDH decreased to 20%. Analyzing the effect size (prevalence) by removing one study at a time, the prevalence ranged from 27% to 33% (Table 5).
Leave-one-out meta-analysis.

Through sensitivity analysis using the Sidik–Jonkman and the Knapp–Hartung truncated method for adjusting the standard error of the effect size, the prevalence of IDH was 30.5%.
Publication bias
The effect of small studies was assessed using Egger's test, which reached a significance level of p = 0.0015, subjectively confirmed by the asymmetric distribution observed in the Funnel plot (Figure 8). Finally, a Trim-and-fill analysis was conducted, and no new articles were imputed.
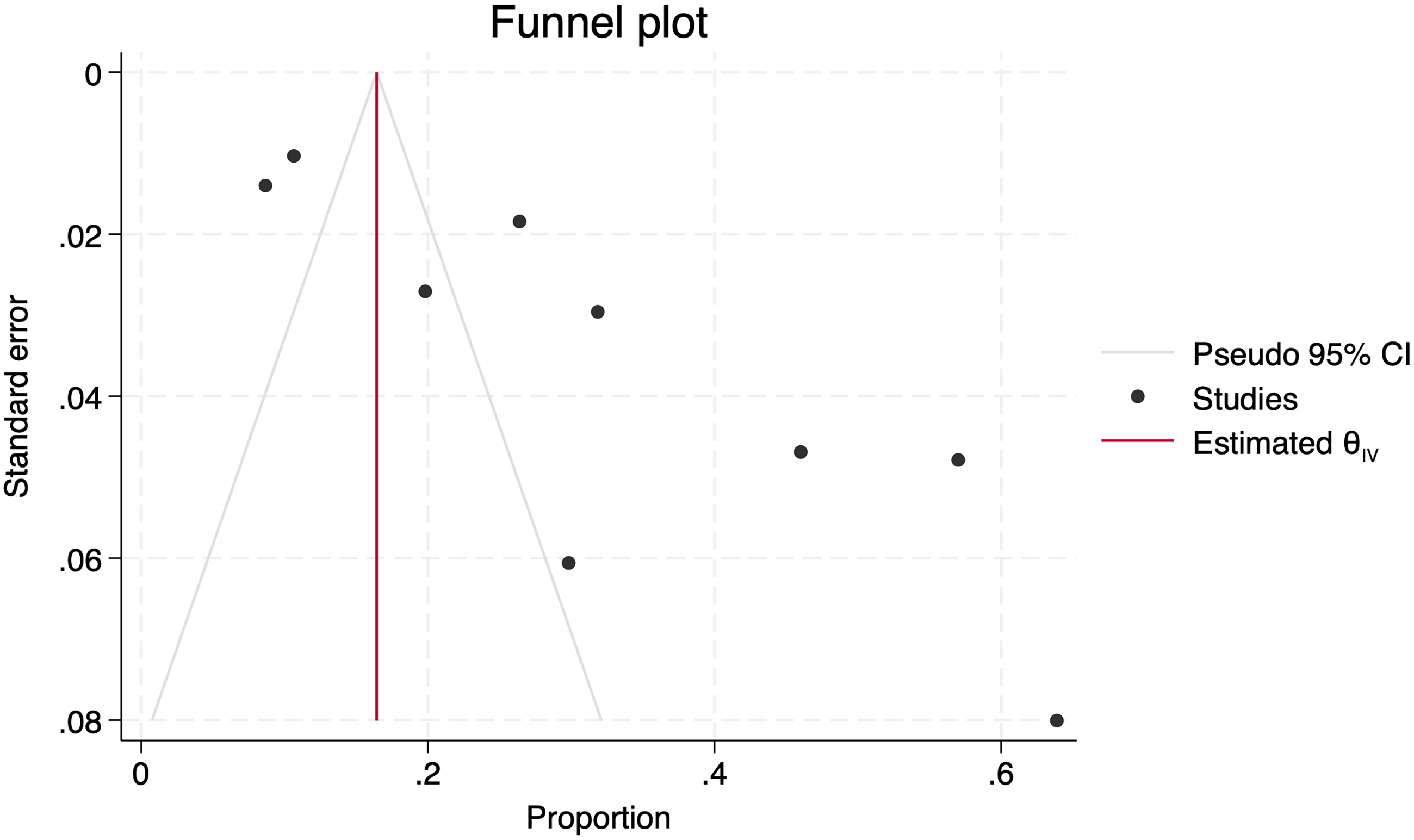
Funnel plot with asymmetric distribution of studies.
Discussion
In our systematic review, the overall incidence of IDH in hospitalized patients was 31%, with minimal variability when considering the study RoB and prevalence measured according to the different approaches for renal replacement therapy. However, the prevalence is higher than that reported in a large 2019 study that included non-hospitalized patients 36 and similar to the reported in other publications.14,37–41 Our prevalence significantly decreased to 20% when we performed the sensitivity analysis, assuming an I2 of 10%.
The wide range of prevalence rates also suggests the existence of multifactorial factors contributing to the occurrence of hypotension during dialysis sessions and the variety of definitions.2,3 Currently, there is no general consensus on the best evidence-based indicators of IDH; however, our report found definitions that include a reduction in SBP of ≥20 mmHg with or without additional symptoms or as a drop in MAP.
Cardiovascular instability during dialysis is linked to severe complications such as intestinal ischemia, myocardial infarction, and access thrombosis. 42 This condition may also hasten the decline of residual renal function and adversely affect renal recovery in patients with severe AKI.43,44 Given the high prevalence observed in our study, the critical challenge for nephrologists, intensivists, and nurses is to implement early interventions to prevent these complications.
We identified the demographic and clinical characteristics of patients, as well as causes and pre-existing conditions associated with IDH. Chronic glomerulonephritis, primary hypertension, diabetes mellitus, and heart failure were the most prevalent. From our results, we could also infer that sepsis was the most common cause of IDH in ICU-admitted patients. In addition, men were most affected by IDH, which differs from results previously published where the female sex is considered a risk factor for IDH.45–50
Some of the included studies also suggest measures to prevent IDH.29–36 However, we believe an individualized assessment based on the severity and the risk of IDH should be performed before and during renal replacement therapy in a hospitalization or ICU setting.
The data presented in this systematic review should be interpreted in terms of its limitations and strengths. The available evidence has limitations, such as heterogeneity between studies and the lack of standardization in the diagnostic criteria for IDH. We recommend an initial assessment of the patient before the onset of the renal replacement therapy and consecutive hemodynamic evaluations to determine the origin of the shock and to individualize the treatment based on current guidelines. 51
Among the strengths of our systematic review are that the search strategy was exhaustive and conducted according to a previously developed protocol and that studies with a broad demographic distribution were included, which ensures a global approximation of IDH prevalence. Finally, all patients included were hospitalized or admitted to the ICU, providing a clearer picture of IDH in these settings.
Conclusions
In our systematic review and meta-analysis, the prevalence of hospital-based renal replacement therapies with IDH in hospitalized patients was 31%.
Although it is challenging to establish a uniform definition of IDH that encompasses decreases in blood pressure, symptoms, and interventions taken in response to hemodynamic instability, standardizing a single definition would facilitate understanding and homogenization of diagnoses and improve decision-making by healthcare personnel, making it more appropriate and timelier.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241308982 - Supplemental material for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis
Supplemental material, sj-docx-1-sci-10.1177_00368504241308982 for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis by Karla Arcentales-Vera, María Fernanda Vera-Mendoza, Cristian Cevallos-Salas, María Fernanda García-Aguilera and Luis Fuenmayor-González in Science Progress
Supplemental Material
sj-jpg-2-sci-10.1177_00368504241308982 - Supplemental material for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis
Supplemental material, sj-jpg-2-sci-10.1177_00368504241308982 for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis by Karla Arcentales-Vera, María Fernanda Vera-Mendoza, Cristian Cevallos-Salas, María Fernanda García-Aguilera and Luis Fuenmayor-González in Science Progress
Supplemental Material
sj-jpg-3-sci-10.1177_00368504241308982 - Supplemental material for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis
Supplemental material, sj-jpg-3-sci-10.1177_00368504241308982 for Prevalence of cardiovascular instability during hemodialysis therapy in hospitalized patients: A systematic review and meta-analysis by Karla Arcentales-Vera, María Fernanda Vera-Mendoza, Cristian Cevallos-Salas, María Fernanda García-Aguilera and Luis Fuenmayor-González in Science Progress
Footnotes
Acknowledgements
We extend our sincere gratitude to Dr. Fabricio Picoita for his guidance throughout the course of this research. We also wish to acknowledge the support of the Universidad Central del Ecuador and the Consejo de Postgrado Rodrigo Yépez for providing the necessary resources and facilities that made this study possible.
Author contributions
A-V. K. contributed to data curation, formal analysis, resources, investigation, writing the original draft, and writing, reviewing, and editing the final draft. V-M. M. contributed to data curation, formal analysis, resources, investigation, writing the original draft, and writing, reviewing, and editing the final draft. C-S. C. contributed to project administration, investigation, validation, and writing, reviewing, and editing of the final draft. G-A. M. contributed to the formal analysis, methodology, validation, visualization, and reviewing and editing of the final draft. F-G. L. contributed to the conceptualization, formal analysis, methodology, project administration, validation, visualization, and writing, reviewing and editing of the final draft.
Data availability
Data will be made available upon reasonable request following communication with the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
