Abstract
Introduction
The use of antiseizure medications (ASMs) is not limited to epilepsy or seizure control as most drugs in this class have multiple mechanisms of action that extend their use to the management of psychiatric disorders and pain syndromes. 1 Over the past few years, several new ASMs have come to market.2,3 As a result, maternal exposure to different ASMs at any time during pregnancy has become increasingly prevalent. 4 People with epilepsy exposed to ASMs during pregnancy may be at an increased risk of adverse pregnancy and neonatal birth outcomes including preterm birth, cesarean section (c-section) and fetal loss compared to people without epilepsy or people with epilepsy not exposed to ASMs. 5
Despite such increase in use and indications, evidence pertaining to the use of these ASMs is limited in pregnancy. Current literature describes outcomes such as major congenital malformations, however, other safety outcomes such as c-section and fetal loss are often not described. The objective of this review was to summarize the effects of maternal exposure to third generation ASMs, or any combination thereof during pregnancy regardless of dose, duration, or route of administration on the neonatal and obstetric outcomes of preterm birth, c-section, and fetal loss.
Methods
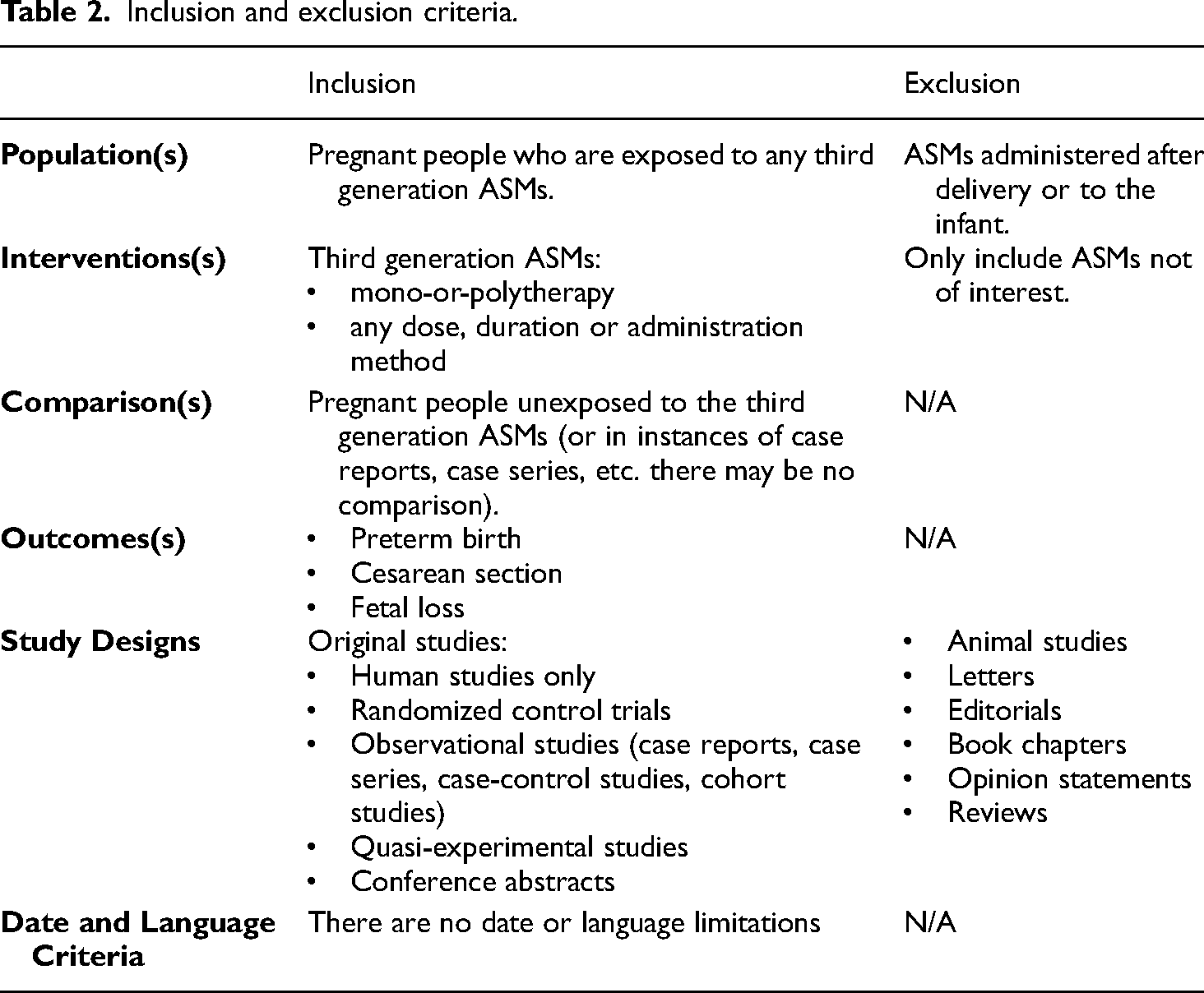
Inclusion and exclusion criteria
We followed the systematic review's reporting guidelines in Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020.6,7 The protocol has been registered and published (https://doi.org/10.17605/OSF.IO/T6HQ7). Studies were included if they reported pregnant people who were exposed to any third generation ASMs (Table 1), identified through the Anatomical Therapeutic Chemical (ATC) classification database of the WHO Collaborating Centre for Drug Statistics Methodology, and accounting for the date of approval and market availability.8–10 Table 2 details the eligibility criteria. The exposure of interest was mono-or-polytherapy in which one of the drugs must have been a third generation ASM for any dose, duration, or route of administration. The comparator group(s), when present, were pregnant people unexposed to third generation ASMs, including women on no ASMs. For inclusion, at least one outcome, either preterm birth, c-section, or fetal loss, must have been reported. Included study designs were randomized controlled trials, observational studies (case reports, case series, case-control studies, cohort studies), including both full reports and conference abstracts. Excluded study designs were animal studies, letters, editorials, book chapters, opinion statements, and reviews. Studies were excluded if the ASM was administered after delivery, to the infants themselves, or if none of our outcomes of interest were reported. There was no publication period restriction as the year of drug discovery may differ from the year the drug was approved for use. There was also no language limitation in our search. Table 2 details the eligibility criteria.
Third generation ASMs included in the review.

Inclusion and exclusion criteria.
Strategy search and information sources
The team developed a search strategy in collaboration with a librarian. Medline, Embase, International Pharmaceutical Abstracts, Cochrane Library and Scopus were searched in August 2022 (see Appendices 1 to 5 of Supplemental Material for the full search strategies). The search strategy was rerun for potential updates in September 2022: Medline (1946 to September 26, 2022), EMBASE (1974 to 2022 September 26), International Pharmaceutical Abstracts (1970 to September 2022), Cochrane Library (from inception to Issue 9 of 12 (last search on September 27)), and Scopus (inception to 2022). Citations were managed in Covidence, Mendeley, and the Systematic Review Accelerator was used for final deduplication.11,12
Study selection
Two authors (JG and OGO) screened titles and abstracts independently. Conflicts were resolved by consensus with a third reviewer (SE). Once a study was selected, the references of the study were reviewed to ensure eligible studies were captured. Additionally, citations included in related systematic reviews were searched for potential studies. Figure 1 includes the study selection PRISMA diagram.

Preferred reporting items for systematic reviews and meta-analyses flow diagram.
Data extraction
Data was extracted in a quality summary table by two independent reviewers (JG and OGO). The quality summary included the name of the study reference, study design, country of the study, data source, drug exposure period, name(s) of drug individual(s) were exposed to, number of individuals exposed, and % of events (preterm birth, c-section, fetal loss) in exposed individuals. After the extraction process, the reviewers (JG and OGO) resolved conflicts through consensus, in consultation with a third reviewer (SE). According to our protocol, the following variables were reported whenever available: definition of control, number of control participants, % of events in control individuals, effect size (RR (aRR or cRR)), Odds Ratio (aOR or cOR), prevalence, and CI/P-value. We planned to use the Newcastle Ottawa Scale (NOS) for risk of bias assessment. 13
Outcome definitions
Data for the following outcomes were included: preterm birth, c-section, and fetal loss. We defined preterm birth as a live birth where the gestational age of the infant is less than 37 completed weeks.14,15 We defined c-section as the birth of a fetus by performing a surgical incision through the maternal abdomen and uterus. 16 Fetal loss is a broad term that may include stillbirth, miscarriage, spontaneous abortion, or induced abortion, according to the reported studies, therefore we did not restrict our search to one definition. When additional details regarding mechanism of fetal loss were available, it was provided in the results (Table 3).
Studies examining the impact of maternal third generation ASM exposure during pregnancy on preterm birth, c-section and fetal loss events.

N/R, not reported
Results
Our search in the five databases resulted in 2978 articles. After excluding 869 duplicates, 2109 articles were screened. An additional 12 articles were further included through manual screening, with most being cannabis exposure studies. The updated search resulted in 29 publications to screen, and one additional case report was identified and extracted. Finally, 74 full text articles were screened, and 32 articles were eventually included (Figure 1).
Synthesis of results from included studies
Our search predominantly yielded results for lacosamide (n = 26), and the other included studies reported data on perampanel (n = 7), brivaracetam (n = 3), eslicarbazepine (n = 2), and fosphenytoin (n = 3). No studies were found regarding our population of interest and desired outcomes, for any of the other third generation ASMs, namely, cannabidiol, carabersat, carisbamate, cenobamate, ganaxolone, losigamone, remacemide hydrochloride, retigabine, rufinamide, safinamide, selectracetam, soretolide, stiripentol, or valrocemide. The included publications were grouped by the reported drug of exposure, cases of polytherapy were included in the results section of each drug. Table 3 presents an overview of the results obtained.
Lacosamide
The majority of publications included in this review were the 14 case reports for lacosamide.18–31 The other publications on lacosamide included nine case series,32–40 one retrospective cohort study, 41 and two pharmacovigilance safety reports.42,43
Ben-Menachem et al. reported in a multicenter open-label trial that two individuals became pregnant. Lacosamide monotherapy was discontinued at 7 weeks and 17 weeks after positive pregnancy tests. The method of delivery was not reported, but two live-birth infants were delivered with one infant being premature (50%, 1/2). 38 Dmitrenko et al. reported prenatal outcomes from the Russian Register of Pregnancy and Epilepsy for the latest ASMs, lacosamide and perampanel. Two individuals received lacosamide and four received perampanel, but the outcomes were reported collectively with all live-births and 66% (4/6) were delivered via c-section. 39 Gestational age was not reported in Dmitrenko et al. Herrero et al. in a retrospective cohort reported multiple prenatal outcomes for ASMs with one ASM being lacosamide. In the study, 55 people received lacosamide during pregnancy and six (10.9%) infants were preterm, 25 (45.5%) infants were delivered via c-section, but fetal loss was not reported. 41
There were two pharmacovigilance studies with data from the Berlin Embryotox pharmacovigilance center and UCB global safety database.42,43 Hoeltzenbein et al. reported the pregnancy outcomes of 52 pregnancies with lacosamide exposure; three (5.76%) were born prematurely and there were 12 (23%) cases of fetal loss (three spontaneous abortions, eight elective termination of pregnancy and one stillbirth). Golembesky et al. reported that out of 101 pregnant people exposed to lacosamide, there were 26 (25.7%) cases of fetal loss (11 spontaneous and 15 induced abortions).
From the 14 cases, 13 were exposed to other ASMs or treatment for acute medical conditions and one was lacosamide monotherapy. 20 In addition to lacosamide, two cases were exposed to fosphenytoin,18,19 one case was exposed to brivaracetam, 32 and perampanel. 29 The duration of exposure varied across the cases with some individuals taking lacosamide throughout the entire pregnancy while others started lacosamide therapy later in the gestation period. Overall, from the 14 cases, four infants were born prematurely, from the cases with reported methods of delivery nine were c-section and only one case resulted in fetal loss due to an elective abortion.
While Landmark et al. is a case series, only one person had taken lacosamide, and did not give birth prematurely, have cesarean section, or experience a fetal loss event. Similarly, Lee and Kim is a case series, but only one of the two individuals were on lacosamide and a live infant was delivered naturally at full-term.
In another case series, Li et al., included three of four individuals on direct brain–responsive neurostimulation and polytherapy with one drug being lacosamide. One of the pregnancies delivered prematurely at 34 weeks and 6 days. 33 In Alibas et al., the two individuals received lacosamide and they each delivered a live-premature infant via cesarean. Bóné et al., included two individuals on lacosamide; one person delivered an infant at full-term via cesarean and the other person delivered a premature infant. Lattanzi et al., included one polytherapy case and two monotherapy cases with lacosamide. The three cases (3/3) from Lattanzi et al. reported live, full-term births via spontaneous vaginal deliveries. Zutshi et al. included seven cases with three being lacosamide monotherapy and four polytherapy. Zutshi et al. reported two preterm deliveries (28.6%; 2/7) and one c-section delivery (14.3%; 1/7).
Perampanel
The seven publications reporting perampanel exposure included five case series,32,39,40,44,45 one case report, 29 and one pharmacovigilance safety report. 46 Sample sizes ranged from two to 71. Perampanel was the primary drug of exposure, however it was used in combination with other ASMs in some instances.
Maguire (2022) reported data for 18 pregnancies. They did not report the prevalence of preterm birth or method of delivery. Of the 18 cases exposed to perampanel, there was a total of 15 fetal loss events (77.8%; 15/18), nine of which were induced abortion events and five which were spontaneous. Four pregnancies resulted in live births, although one neonatal death was reported a few hours post-delivery, however this was deemed to be unrelated to perampanel as it was secondary to fluid aspiration. 45
Landmark et. al presented pregnancy outcome data for two individuals. One of the individuals was on a combination of perampanel, brivaracetam, and lacosamide. As mentioned above, the outcome of the pregnancy was a full-term birth via an unreported delivery method. 32
Alicino et al. identified four individuals who used perampanel during their pregnancies; 3 individuals used perampanel in combination with other ASMs while one individual used perampanel monotherapy for seizure management. The fetuses were exposed to an average of 250 days of perampanel throughout gestation, all resulting in live births, three of which were via c-section (75%; 3/4), with one natural vaginal delivery. 44
Lee and Kim (2022) present data concerning two cases of exposure to perampanel. One individual used a combination of perampanel, lacosamide and levetiracetam for the first four weeks of pregnancy; perampanel was discontinued at week five and lacosamide and levetiracetam were continued throughout the pregnancy. The other individual used perampanel and topiramate for seizure control; perampanel was discontinued at the fourth week of pregnancy while topiramate was continued. There were no events of fetal loss, preterm birth, or c-section as both cases resulted in full-term births via natural vaginal birth. 40
Dmitrenko et al. reported data on six participants who were exposed to a third generation ASM. Four participants were exposed to perampanel while the remaining two were exposed to lacosamide. 39 No distinction was made between the drugs participants were exposed to when reporting outcomes, as mentioned above.
In Kitamura et al. one individual was exposed to perampanel and lacosamide during pregnancy. Perampanel was only used for a total of 14 days between five- and seven-weeks’ gestation and the outcome of pregnancy was a full-term birth via c-section delivery (100%; 1/1). 29
The pharmacovigilance safety review performed by Vazquez et al. included data obtained from the Eisai global database for perampanel. A total of 71 pregnancies were reported; initially, 96 pregnancies in which maternal exposure to perampanel at varying times during pregnancy were identified, however, 18 were lost to follow-up and an additional seven were ongoing, so pregnancy outcome data could not be collected. Twenty-six participants used perampanel monotherapy, while the remaining participants used perampanel in combination with other ASMs. The primary outcomes reported that coincided with our outcomes of interest were preterm birth and fetal loss. C-section prevalence was not specifically reported and 43 live births via normal delivery were observed. One preterm birth was reported (1.4%; 1/71), however the method of delivery was not reported. Around 38% (27/71) of the pregnancies resulted in fetal loss events. 46 Eighteen of these events were induced abortions, eight were spontaneous miscarriages, and one still birth was reported. The stillbirth occurred at 30 weeks’ gestation with Tetralogy of Fallot, but was not considered to be associated with perampanel exposure.
Brivaracetam
Two of the included publications involving exposure to brivaracetam were case reports and the third publication was a case series.32,33,47 Sample sizes ranged from two to four participants. Paolini et al. examined brivaracetam monotherapy and 66% (2/3) of the cases reported c-section delivery with no preterm birth or fetal loss events. 47 Landmark et al. reported one of two patients had a c-section event (50%; 1/2) with maternal exposure to brivaracetam monotherapy during pregnancy; the other individual was treated with a combination of brivaracetam, lacosamide, and perampanel and reported a full-term delivery with no specification of delivery method. 32 One of the four participants in Li et al. was exposed to a combination of brivaracetam and eslicarbazepine, however, other participants used lacosamide. The only individual case exposed to brivaracetam and eslicarbazepine during pregnancy reported a full-term delivery via c-section. 33
Eslicarbazepine
Two publications involving eslicarbazepine included one pharmacovigilance safety report and one case series.33,48 Vieira et al., reported on the outcomes of an 11 years post-marketing surveillance for eslicarbazepine acetate. From 176 pregnancies, the outcomes were known for 117 pregnancies and 28 pregnancies (23.9%) ended in fetal loss. The gestational age and method of delivery were not reported. 48 As for Li et al., a case series, one out of four pregnancies (25%) in the study was exposed to eslicarbazepine along with clobazam, brivaracetam and direct brain–responsive neurostimulation. A full-term infant was delivered by c-section due to prolonged labor. 33
Fosphenytoin
All three studies involving fosphenytoin were case reports with sample sizes of one.18,19,49 The publications vary in the extent of fosphenytoin exposure. Two publications reported limited exposure at discrete times during pregnancy18,49; Kasai et al. reported exposure at 28- and 38-weeks’ gestation while Carrasco et al. reported exposure for a total of six days at 26 weeks’ gestation. Ylikotila et al. reported prolonged fosphenytoin exposure from 7 to 36 weeks’ gestation. All three publications reported the outcomes of interest, with all pregnancies resulting in live births. Kasai et al. reported a full-term delivery via c-section following exposure to fosphenytoin, along with other non-third generation ASMs for seizure control. Carasco et al. and Ylikotila et al. both report maternal exposure to fosphenytoin and lacosamide at varying times during pregnancy with outcomes of preterm deliveries (2 out of 2 exposed pregnancies) via c-section (2 out of 2 exposed pregnancies).
Discussion
Summary of evidence
In the current systematic review, we observed that the body of literature describing the relationship between third generation ASMs and obstetric and neonatal outcomes, specifically preterm birth, c-section, and fetal loss, is limited. Of note, there are limitations associated with the use of c-section as a marker of adverse outcomes as the reason for the c-section or if it is a repeat event may not be clearly documented. We identified 32 studies or case reports, however only one study included a control group, and was published only as an abstract. This is understandable considering the relative newness of this group of drugs. Furthermore, most studies exclude those who are pregnant, resulting in a dearth of pregnancy-specific safety data. Many of the drugs categorized as third generation ASMs were approved for use in the last twenty years, while some, most of which did not yield any results in our search, have not yet been approved for use. Although the body of evidence we found regarding third generation ASM exposure during pregnancy was limited, the results are valuable. However, conclusions cannot be definitively drawn as most of the publications are case reports. Even so, the results obtained provide a foundation for future studies for the relationship between maternal third generation ASM use during pregnancy and neonatal and obstetric outcomes to be better elucidated and identified.
Evidence on specific drugs
Lacosamide
From the third generation ASMs, lacosamide had multiple cases, case series, pharmacovigilance studies and a retrospective cohort study which reported preterm birth, c-section and/or fetal loss. All the case reports, except one case of an elective pregnancy termination, resulted in live births. Approximately 28.6% (4/14) births were premature, and 64.3% (9/14) were delivered by c-section. However, the safety of lacosamide or an association with preterm birth and c-section cannot be determined based on case reports alone due to heterogeneity in the cases and lack of a control group. The same limitation exists for the nine-case series,32–40 Combining the results from the case series is not feasible due to the varied circumstances in each series. For instance, the individuals in Alibas et al. experienced status epilepticus, in Bóné et al. received deep brain stimulation, Dmitrenko et al. did not separate lacosamide outcomes from perampanel outcomes and there were limited pregnancy details in Ben-Menachem et al. Additionally, these studies did not include comparison groups, which makes it difficult to conclude if the observed outcomes are linked to lacosamide, other ASMs, other medical treatments or seizures.
Pharmacovigilance studies included 153 people, highlighting the increasing use.42,43 However, without a comparator, determining if the 5.76% preterm births in Hoeltzenbein et al. or the fetal loss of 23% and 25.7% in Hoeltzenbein et al. and Golembesky et al. are related to lacosamide is currently not possible.
Finally, Herrero et al., was the only study out of the 32 included in our systematic review that included a reference group. Compared to people without a diagnosis of epilepsy, people on lacosamide had a non-significant risk difference (95% confidence intervals) of 4.6% (−3.6, 12.9) for preterm birth, and 9.7% (−3.5, 22.8) for c-section. It was not reported if the individuals on lacosamide were taking other ASMs and what other medical conditions other than seizure disorders were present in the population. Essentially, while the case reports, pharmacovigilance studies, and single cohort study show promise of safety during pregnancy for lacosamide, more studies with comparators are necessary.
Perampanel
The included publications provided results regarding a total of 101 pregnancies exposed to perampanel, some in combination with other ASMs of interest. Similarities and differences exist across each source. Similar outcomes were reported, although fetal loss and c-section events were most common. Differences arise in the study designs and perampanel exposure periods. Despite the larger number of cases, combining the outcomes of perampanel exposure during pregnancy from the included publications would not provide reliable conclusions regarding the relationship between perampanel exposure and neonatal outcomes.
The full-term c-section delivery reported by Kitamura et al. was elective in nature due to the individual's history of c-section births for prior pregnancies so no connection can be readily made between the c-section event and ASM therapy. The outcomes of the cases documented in Alicino et al. are relatively similar. All pregnancies resulted in live, full-term births and the participants were all on a form of combination antiseizure therapy which included perampanel although different doses were used. The three participants who reported c-section deliveries were on higher doses of perampanel than the participant who gave birth via a natural vaginal delivery. 44 A determining variable in this instance may be age as the participants who experienced c-section events were older. It is of note that only one participant's age (43 years old at delivery) increased their pregnancy outcome risk; resulted in a c-section delivery. Due to the differences in participants, a conclusion cannot be drawn regarding the influence of perampanel dose on pregnancy outcomes.
In keeping with the aggregate outcomes of the other included publications that investigated third generation ASM exposure during pregnancy, c-section was the most consistently reported event. In two publications a high prevalence of fetal loss was reported.45,46 Maguire reported the highest proportion of fetal loss events (77.8% of pregnancies ended in fetal loss). 45 Vazquez et al. had the largest sample size of all the included publications pertaining to perampanel exposure, and a high amount of fetal loss events were reported as well (38% of pregnancies resulted in fetal loss). 46 Fetal loss was not reported in any of the remaining publications.29,32,39,40,44 The adjunctive antiseizure therapies participants were exposed to across the different cases are not uniform, also the co-morbid conditions of each participant are not explicitly identified, so conclusions cannot be drawn regarding the influence of added therapy on obstetric and neonatal outcomes.
Lee et al. present data concerning two cases in which perampanel was stopped in the first trimester of pregnancy. These two cases resulted in full-term, natural vaginal births. Further studies investigating the effects of perampanel discontinuation at different weeks of gestation on neonatal and pregnancy outcomes would be needed in order to draw any conclusions regarding these results. The outcomes reported in Dmitrenko et al. cannot be accurately attributed to either lacosamide or perampanel as the results were not individually reported. 39 As a result, no conclusions can be drawn regarding the individual effects of each drug. Ultimately, the small sample sizes of the publications pertaining to perampanel exposure, the lack of uniformity in study design and exposure, along with the absence of a comparator, as well as the presence of multiple extraneous variables makes it challenging to elucidate the relationship between perampanel exposure during pregnancy and the outcomes of interest.
Brivaracetam
All three publications reported our outcomes of interest with c-section delivery being the most reported event across the publications. There was one incident of preterm birth and there were no fetal loss events reported with the use of brivaracetam. Regarding the three cases reported by Paolini et al., it is of note that the individual on the highest dose of brivaracetam also had the highest aggregate risk, considering the individual's age of 39 years.47,50 Despite this, the outcome of delivery was a natural vaginal birth, whereas the other participants reported c-section deliveries. This event is difficult to rationalize as additional information, such as the number and outcomes of prior pregnancies for participants were not reported.
Moreover, Landmark et al. contribute valuable cases of brivaracetam exposure. One of the participants reported brivaracetam monotherapy, while the second participant reported the use of brivaracetam, lacosamide and perampanel. Despite the similarities in reported outcomes of full-term birth, conclusions cannot be made.
Of the four cases presented by Li et al., only one of these individuals was exposed to brivaracetam, in combination with eslicarbazepine and another ASM during pregnancy which resulted in a full-term c-section delivery. 33 Since only one case of brivaracetam use was reported, no conclusions can be made. Six of the nine cases reported from the three included publications involved brivaracetam exposure and they all resulted in live births with varying number of c-section events. There are many variances present across these studies. The doses and duration of exposure to brivaracetam during pregnancy were different in all the cases reported, lacked a comparator, therefore conclusions cannot be made regarding the relationship between brivaracetam use and the risk of these outcome events.
Eslicarbazepine
Combining the eslicarbazepine studies did not yield a conclusion on the outcomes of preterm birth, c-section, and fetal loss. Vieira et al. only reported fetal loss and no other outcomes of interest. Li et al.'s individual case on eslicarbazepine was also on other ASMs and direct brain-responsive neurostimulation. Further study is required on eslicarbazepine to determine the relationship between its use and neonatal outcomes.
Fosphenytoin
C-section delivery was reported in all case reports of fosphenytoin exposure and fetal loss was not reported. All three individuals included in the three case reports were being treated for status epilepticus which is considered a medical emergency.18,19,49 The risks and adverse effects of prolonged seizure activity are increased in pregnancy as this may induce fetal distress.
Differences in individual characteristics and fosphenytoin exposure preclude the combination of the reported results in order to draw conclusions. Pre-existing medical conditions and advanced age are associated with increased risk in pregnancy. 50 All the pregnancies reported in these cases would be considered high risk as all the participants were experiencing seizures. 50 The association between the outcomes of maternal exposure to fosphenytoin during pregnancy and maternal age is one worthy of further investigation. Further studies including a larger sample size, comparators as well as methods to minimize confounding variables are necessary for any association to be elucidated.
Cannabidiol
Cannabis has a long history of use for many health-related conditions as well as recreational purposes. Cannabidiol specifically has been identified and approved for use as a third generation ASM. In many systematic reviews that exist summarizing cannabis use and associated outcomes, the method of use and dose unspecified, as opposed to cannabidiol, which is the component approved for medical use in seizure management. 51
The term “cannabidiol” was used in the search strategy to capture more studies for screening. With the increasing interest in cannabis for healthcare purposes, in pregnancy it is not limited to ASM use, but also for nausea and vomiting or hyperemesis gravidarum. 52 Details of cases using cannabidiol, including the administration method, dose and duration and the outcomes of the pregnancy are required to assess the safety of cannabidiol during pregnancy. While cannabidiol was the substance of interest, every paper found on cannabis/cannabidiol was eventually excluded, so there is no data on cannabidiol included in this review.
Strengths and limitations
The strength of this study is that five databases were searched in order to capture all available evidence. Reviewers were blinded in order to reduce risk of bias. It was planned to use a risk of bias tool, the NOS, in order to assess the studies’ quality. However, this review resulted in a qualitative summary as all of the publications were without comparison groups.
Of the publications included, most were case reports with very small sample sizes, which did not provide appropriate comparators. These factors limit our ability to draw meaningful conclusions from the reported data. Along with this, the included studies were heterogeneous in dose, length and timing of exposure. The differences in such variables can be rationalized as seizure events during pregnancy are a medical emergency and the primary focus of care is seizure control. Standardizing ASM exposure for individuals would be incredibly challenging considering the fact that seizure management requires individualized treatment plans which may involve varying doses and combinations of ASM to achieve and maintain a seizure-free state. Our review focused on specific neonatal outcomes, while excluding other commonly reported outcomes as birth defects risk. Our goal was to summarize the available evidence on a significant knowledge gap. We acknowledge that the teratogenicity of these medications is of high concern, which lead to a larger focus on birth defects and several safety outcomes become frequently missed. Consequently, to avoid redundancy, we focused our efforts on the evidence of other safety outcomes. Also, due to the study designs of the included publications, there were not enough studies with control groups to conduct a meta-analysis.
Conclusions
Overall, this systematic review highlights the scarcity of studies regarding the association between third generation ASM exposure during pregnancy and preterm birth, c-section, and fetal loss. The included studies lacked comparison groups, which further precludes quantifying the risks associated with third generation ASM exposure. Large and comparison safety studies are warranted.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241234781 - Supplemental material for Third generation antiseizure medications exposure during pregnancy and neonatal adverse birth outcomes: A systematic review
Supplemental material, sj-docx-1-sci-10.1177_00368504241234781 for Third generation antiseizure medications exposure during pregnancy and neonatal adverse birth outcomes: A systematic review by Joyce Goubran, Oreofe Grace Okunnu, Alekhya Lavu and Sherif Eltonsy in Science Progress
Footnotes
Registration and Protocol
The protocol has been registered in the Open Science Framework (OSF) database.
Registered name
Third generation antiseizure drug exposure during pregnancy and neonatal adverse birth outcomes: a systematic review
Acknowledgements
We thank Mê-Linh Lê, a University of Manitoba librarian, for the valuable and tireless guidance, advice, and assistance she provided regarding the development of our search strategy, and de-duplication.
Authors’ contributions
JG and OGO are co-first authors and contributed equally to the current review. JG, OGO, AL, and SE collaboratively contributed to the concept and designed the study. JG and OGO wrote the protocol while SE reviewed the protocol. JG and OGO published the protocol in the Open Science Framework database. JG and OGO developed the search strategies with AL. JG and OGO and ran the search strategies, screened, and extracted included results. JG and OGO drafted the final manuscript and AL and SE revised and approved the manuscript as submitted. JG and OGO are co-first authors and AL and SE are second and corresponding authors respectively.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded through a research grant from The Winnipeg Foundation.
Winnipeg Foundation,
Supplemental material
Supplemental material for this article is available online.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Informed consent for patient information to be published in this article was not obtained because * Not applicable. * manuscript titled “Third generation antiseizure medications exposure during pregnancy and neonatal adverse birth outcomes: a systematic review.”
Author biographies
Joyce Goubran holds a PharmD. Her area of research is drug safety and effectiveness.
Oreofe Grace Okunnu holds a PharmD. Her area of research is drug safety and effectiveness.
Alekhya Lavu holds a PharmD (PhD candidate). Her area of research is pharmacoepidemiology and population health.
Sherif Eltonsy is an Assistant Professor in Pharmacy. His area of research is pharmacoepidemiology, perinatal epidemiology and public health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
