Abstract
Background
Systemic sclerosis-associated interstitial lung disease (SSc-ILD) is a prominent form of connective tissue disease-associated interstitial lung disease with high prevalence and poor prognosis. The therapeutic use of corticosteroids is often limited, and the efficacy of conventional treatment may be suboptimal in certain cases. This systematic review and meta-analysis assessed the clinical effects of rituximab in SSc-ILD compared to conventional treatments and investigated potential clinical benefits in patients with disease progression despite conventional therapy.
Methods
A comprehensive literature search was conducted in Ovid MEDLINE®, Cochrane Library, EMBASE, and KoreaMed databases to identify studies on rituximab in adult SSc-ILD patients. Comparative studies with complete trial results were included. The primary outcome was the effect of rituximab on disease progression, measured by the change in forced vital capacity (FVC) compared to conventional treatments.
Results
From 940 articles, six studies (two randomized, four non-randomized) were included for analysis. Standardized mean differences (SMD) were used in analyses due to the varied durations of the included studies. The meta-analysis demonstrated that rituximab reduced the FVC decline in patients with SSc-ILD compared to conventional treatment (SMD 0.65; 95% confidence interval, 0.08–1.22, P = .03), though no significant differences were found in lung diffusing capacity or modified Rodnan skin score. Rituximab showed a significantly lower incidence of leukopenia compared to cyclophosphamide (P = .02) and showed a non-significant trend toward fewer instances of pneumonia and urinary tract infections.
Conclusions
The meta-analysis revealed that rituximab may be clinically beneficial in SSc-ILD that progresses despite conventional treatment. Further research is necessary to validate the clinical benefits of rituximab in SSc-ILD.
Introduction
Systemic sclerosis (SSc) is a complex autoimmune disease involving multiple organ systems and is characterized by immune dysregulation, obliterative microvasculopathy, and fibrosis, with a diverse clinical course. 1 Lung involvement in SSc is common, with abnormal findings on high-resolution computed tomography (HRCT) scans in 75–90% of patients.2–4 Interstitial lung disease (ILD) represents the most prevalent and clinically significant form of lung involvement in SSc, with prevalence ranging from 26.1% to as high as 88.1% according to various studies.5–8 SSc-associated ILD (SSc-ILD) tends to occur early in the disease course; when present a high proportion of patients present with a progressive phenotype, that continues to worsen despite treatment.3,8,9 Additionally, SSc-ILD is a major contributor to mortality in patients with SSc and can be traced to 20–35% of SSc-related deaths, emphasizing the need for effective treatment and management.10,11 The recently revised 2023 American College of Rheumatologist (ACR)/American College of Chest Physicians (CHEST) guidelines included rituximab as a potential first-line therapy option for SSc-ILD in addition to previously indicated mycophenolate, cyclophosphamide, and tocilizumab.12,13 Nonetheless, various studies have shown that therapeutic options remained limited for SSc-ILD that continues to progress despite established conventional treatments, such as mycophenolate and cyclophosphamide. 14 Despite its emerging potential as a therapeutic agent, the clinical efficacy of rituximab in the treatment of SSc-ILD remains ambiguous, and the supporting evidence is inadequate. Consequently, this systematic review evaluates the clinical efficacy of rituximab in moderate to severe SSc-associated interstitial lung disease (SSc-ILD) compared to established treatments such as mycophenolate and cyclophosphamide, while also investigating potential clinical benefit in patients with disease progression despite conventional therapy.
Methods
This research adhered to the guidelines outlined in the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) statement. Since only publicly available data was used, institutional review board approval was not required. The protocol for the systematic review was registered with the International Prospective Register of Systematic Reviews, PROSPERO (registration number CRD42024486556).
Literature search strategy
The databases searched on January 1, 2024, were Cochrane Library, EMBASE, KoreaMed, and Ovid MEDLINE (R). All relevant literature published from January 1, 1970, up to the research date of January 1, 2024, was considered, with no restrictions on the publication period or language for the initial search. The comprehensive literature search focused on the use of rituximab in patients with SSc-ILD. The detailed search strategy, including keywords and combinations, is outlined in Table S1 in the supplemental material.
Inclusion criteria
The inclusion criteria were based on the Population, Intervention, Comparison, Outcomes, and Study (PICOS) design: (1) the population of interest (P) was patients with moderate to severe SSc-ILD and patients who progress despite treatment with conventional treatment including mycophenolate, cyclophosphamide (2) the intervention (I) was administration of rituximab, (3) the comparison (C) was with conventional treatments such as cyclophosphamide or mycophenolate mofetil, (4) the outcome of interest (O) was the effect of rituximab on the decline in forced vital capacity (FVC), and (5) the study design (S) encompassed both randomized controlled trials and non-randomized comparative studies. Additionally, only articles that provided complete results and were available in full text were included.
Diagnosis of SSc was defined according to the 2013 ACR and European League Against Rheumatism (EULAR) classification, 15 while SSc-ILD was defined based on the radiological criteria in the clinical guidelines. 14 Moderate to severe SSc-ILD was defined by an extent of fibrosis on HRCT of 10% or greater, accompanied by an FVC % predicted of less than 80%.16,17
Exclusion criteria
The exclusion criteria were as follows: (1) studies involving non-human subjects, including animal and laboratory experiments, (2) studies not focused on SSc-ILD, (3) studies lacking comparative interventions between rituximab and conventional treatments, (4) studies not published in English or Korean, (5) duplicate publications (i.e. articles with the same content published in different journals or in different formats), and (6) studies where the original text was unavailable.
Primary outcome
The primary outcome of this study was to assess the clinical impact of rituximab on the progression of SSc-ILD compared to conventional treatments. The effect of rituximab was measured by the change in FVC percent predicted over the periods evaluated. Rapid deterioration in lung function indicates disease progression and is linked to higher mortality rates in various interstitial lung diseases. Additionally, changes in FVC are an established measure for assessing the degree of lung function decline, which helps evaluate the progression of ILD and the effectiveness of treatments.
Secondary outcome
Secondary outcomes were (1) comparison of changes in the diffusing capacity of the lung for carbon monoxide (DLco) over the durations of each study between rituximab and conventional treatments, (2) assessment of changes in modified Rodnan skin score (mRSS) over the duration of each study, and (3) evaluation of the incidence of overall adverse events, along with severe adverse events.
Data extraction and analysis
Two reviewers independently conducted data extraction from all sources. De-duplicated studies were imported to Covidence online software (https://www.covidence.org, accessed on January 1, 2024). Two authors screened the titles and abstracts of the de-duplicated studies for selection. Any discrepancies were resolved through discussion. Subsequently, two reviewers independently reviewed the full text of the selected articles. Participant data, inclusion/exclusion criteria, intervention specifics, and outcome measures were extracted. Any discrepancies in data extraction were resolved through discussion and/or consultation with a third reviewer.
Assessment of quality and the level of evidence
The quality of selected studies was evaluated based on criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions, across seven domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. Each domain was categorized as presenting a low, high, or unclear risk.
Statistical analysis
The results from the finalized studies were combined using Review Manager 5.4. The heterogeneity of studies was evaluated using the Cochrane Q statistic, followed by an evaluation with the I2 statistics. Based on the results, either a random effect or fixed effects model was applied. For continuous variables, the effect size was initially assessed using the mean difference. However, given the variability in evaluation periods for FVC, DLco, and mRSS across the included studies, the standardized mean difference was utilized to enable a more comprehensive analysis. Additionally, the risk ratio was employed to compare the effect size of adverse events, categorized as dichotomous variables, all reported with a 95% confidence interval (CI). Statistical significance was set at P < .05. Assessment for publication bias was conducted through a funnel plot along with Egger's regression test and Begg and Mazumdar rank correlation test for plot symmetry assessment.
Results
Literature review and selection
A total of 940 articles was initially found through database searches, with 83 duplicates removed. Following the screening of titles and abstracts, 676 records were discarded due to irrelevant publication types of study. Hence, 181 articles remained for the study (Figure 1). Among these, six full-text articles met the predetermined inclusion and exclusion criteria and were eligible for further analysis.

Flow diagram of the preferred reporting items for systematic reviews and meta-analyses (PRISMA).
Characteristics of the included studies
The meta-analysis included six eligible studies, of which two were randomized controlled trials (RCTs) and four were non-randomized studies. A total of 275 patients were included across the six studies, with 132 in the rituximab group and 143 in the conventional treatment group. Since the studies reported both median and mean values, a weighted average calculation method was used to determine the average age, disease duration, FVC, and mRSS score of the included patients. The calculated averages were: an age of 47.3 years, a disease duration of 10.9 years, an FVC % predicted value of 70%, and an mRSS score of 15.1.
Among two RCTs, the 2010 study by Daoussis et al. examined the outcomes of eight patients administered two doses of rituximab at 24-week intervals, in comparison to six patients who continued with standard conventional treatment over a 1-year period. The primary focus of this analysis was on changes in FVC % predicted, DLco % predicted, and mRSS. The rituximab group exhibited statistically significant improvements at 1 year in all measured outcomes: FVC % predicted (P = .0018), DLco % predicted (P = .017), and mMRC (P < .001), demonstrating significant differences compared to the control group. In a separate RCT conducted by Sircar in 2018, a randomized parallel-group design was employed to compare 30 patients receiving rituximab with 30 patients receiving cyclophosphamide. By the 6-month assessment, the rituximab group demonstrated significant improvement in both FVC % predicted (P = .003) and mRSS (P = .001) in comparison to the cyclophosphamide group. Furthermore, the 6-minute walking distance in the rituximab group also showed significant improvement over the 6-month duration, with noteworthy differences observed when contrasted with the cyclophosphamide group.
Among the four non-randomized studies, the 2015 investigation by Jordan et al. conducted a post hoc nested case-control analysis utilizing data from the European Scleroderma Trial and Research (EUSTAR) cohort. This study comprised 25 patients with diffuse SSc-ILD who were treated with rituximab, alongside matched control patients. The primary outcomes assessed were changes in FVC % predicted and mRSS at the 6-month interval. The rituximab group demonstrated statistically significant improvements in both FVC % predicted (0.4 ± 4.4% vs. −7.7 ± 3.6%, P = .02) and mRSS (−24.0 ± 5.2% vs. −7.7 ± 4.3%, P = .03) when compared to the control group. The 2017 study conducted by Daoussis et al. was a 4-year, multicenter, open-label comparative study involving 33 patients in the rituximab treatment group, who were compared to a conventional treatment cohort receiving azathioprine, mycophenolate, or cyclophosphamide. The rituximab group exhibited a significant improvement in FVC at the 2-year follow-up (P = .041 compared to baseline), indicating a substantial advantage over the conventional treatment group (P = .013). Furthermore, mRSS improvements favoring rituximab were consistently observed across all time points assessed. The 2018 study conducted by Ebata et al. provided a direct comparison between rituximab and cyclophosphamide in a cohort of 39 patients diagnosed with anti-topoisomerase I-positive SSc-ILD. The cohort comprised nine patients in the rituximab treatment group and thirty patients receiving cyclophosphamide. The study assessed changes in FVC, DLco % predicted, and mRSS at the 24-month follow-up. The rituximab group exhibited significant improvements in % predicted FVC (3.9 ± 2.1% vs. 2.4 ± 1.1%, P < .05) and DLco (8.5% vs. no change) compared to the cyclophosphamide group at 3 months. Furthermore, the results for mRSS also indicated a favorable outcome for patients treated with rituximab (P < .05). The 2021 study conducted by Yilmaz et al. encompassed patients with SSc-ILD who were treated with either rituximab or cyclophosphamide for a minimum duration of 24 months, comprising 27 patients in the rituximab group and 34 patients in the cyclophosphamide group. The study compared changes in symptoms, FVC, and DLco from baseline to the 24-month endpoint. The rituximab group exhibited significant improvements in symptoms such as Raynaud's phenomenon, digital ulceration, diarrhea, and dysphagia, with the exception of dyspnea, when compared to those in the cyclophosphamide group. Regarding FVC, the rituximab group did not demonstrate an advantage in the improvement of FVC % predicted compared to the cyclophosphamide group at the 6th month (1.7 ± 5.6 vs. 3.9 vs. 9.1, P = .74). Conversely, the change in DLco in the rituximab group was superior to that in the cyclophosphamide group without statistical significance. The summarized results of the included studies are presented in Table 1.
Summary of included studies and outcomes.

RTX: rituximab; PD: prednisolone; CYC: cyclophosphamide; MMF: mycophenolate mofetil; FVC: functional vital capacity; DLco: diffusing capacity of the lungs for carbon monoxide; mRSS: modified Rankin skin score; HRCT: high-resolution computed tomography; 6MWT: six-minute walk test; EUSTAR: European Scleroderma Trials and Research Group; AZA: azathioprine; MTX: methotrexate; KL-6: Krebs von den Lungen-6; SP-D: surfactant protein-D; IgG: immunoglobulin G; CRP: C-reactive protein.
The effect of rituximab on disease progression in SSc-ILD as evaluated by changes in FVC
All six studies evaluated changes in FVC. Ebata et al. 22 measured changes in FVC % predicted at 12 weeks, while Jordan et al., 20 Sircar et al., 19 and Yilmaz et al. 23 assessed changes at 24 weeks. Daoussis et al. conducted two studies measuring changes at 52 weeks.18,21 Due to these differences in evaluation periods across studies, SMD was used instead of mean difference MD to assess the clinical impact of rituximab on FVC decline. The meta-analysis indicated significant heterogeneity among included studies (I² = 74%, P = .0005). Thus, a random effects model was applied for the analysis. The meta-analysis demonstrated that rituximab significantly reduced FVC decline compared to conventional treatment (SMD 0.64, 95% confidence interval (CI) 0.03–1.25, P = .04). When examining each evaluation interval separately, the study by Ebata et al., which assessed changes in FVC % predicted at 12 weeks, showed a significant reduction in FVC decline with rituximab (SMD 1.07, 95% CI, 0.28–1.85, P = .008). However, the three studies that assessed at 24 weeks and the two studies that assessed at 52 weeks, while showing a tendency to favor rituximab, did not reach statistical significance (P = .18, P = .35, respectively). Detailed results of the meta-analysis and the forest plot are summarized in Figure 2.

Forest plot of the impact of rituximab on FVC decline reduction in SSc-ILD patients after 12, 24, and 52 weeks of treatment.
The effect of rituximab on disease progression in SSc-ILD as evaluated by changes in DLco
Three studies assessed changes in DLco % predicted following rituximab treatment: Jordan et al. and Yilmaz et al. evaluated DLco changes at 24 weeks, while Daoussis et al. (2010) assessed DLco changes at 52 weeks. The pooled meta-analysis revealed moderate heterogeneity among the studies (I² = 34%, P = .55), which led to the application of a random effects model. In contrast to the positive impact of rituximab on FVC, the meta-analysis indicated no significant difference in changes of DLco between rituximab and conventional treatment (SMD 0.17, 95% CI, −0.4 to 0.73, P = .57). Furthermore, separate analyses at 24 weeks and 52 weeks also failed to demonstrate any significant improvement on DLco with rituximab compared to conventional treatment (P = .96, P = .42, respectively) (Figure 3).
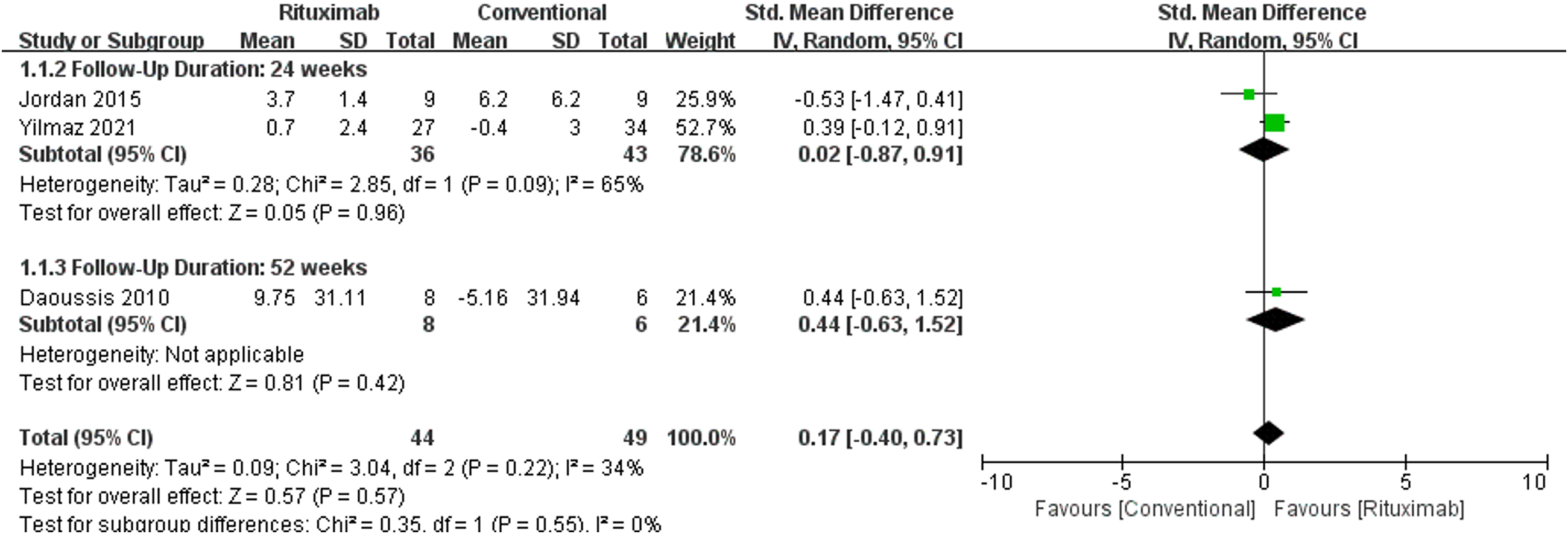
Forest plot of the impact of rituximab on changes of DLco in SSc-ILD patients after 24 and 52 weeks of treatment.
The effect of rituximab on disease progression in SSc-ILD as measured by changes in mRSS
The evaluation of changes in mRSS score encompassed findings from four studies: two conducted by Jordan et al. and Sircar et al. at 24 weeks, and two by Daoussis et al. published in 2010 and 2017 at 52 weeks. A random effects model was used in the analysis due to significant heterogeneity among the studies included (I² = 84%, P = .0004). According to the meta-analysis results, rituximab did not demonstrate a significant reduction in mRSS scores compared to conventional treatment (SMD −0.75, 95% CI −1.58 to 0.07, P = .07). Analysis of the data from 24 weeks and 52 weeks separately revealed a tendency for rituximab to achieve a greater reduction in mRSS scores compared to conventional treatment; however, these differences did not reach statistical significance (P = .18 and P = .23, respectively) (Figure 4).
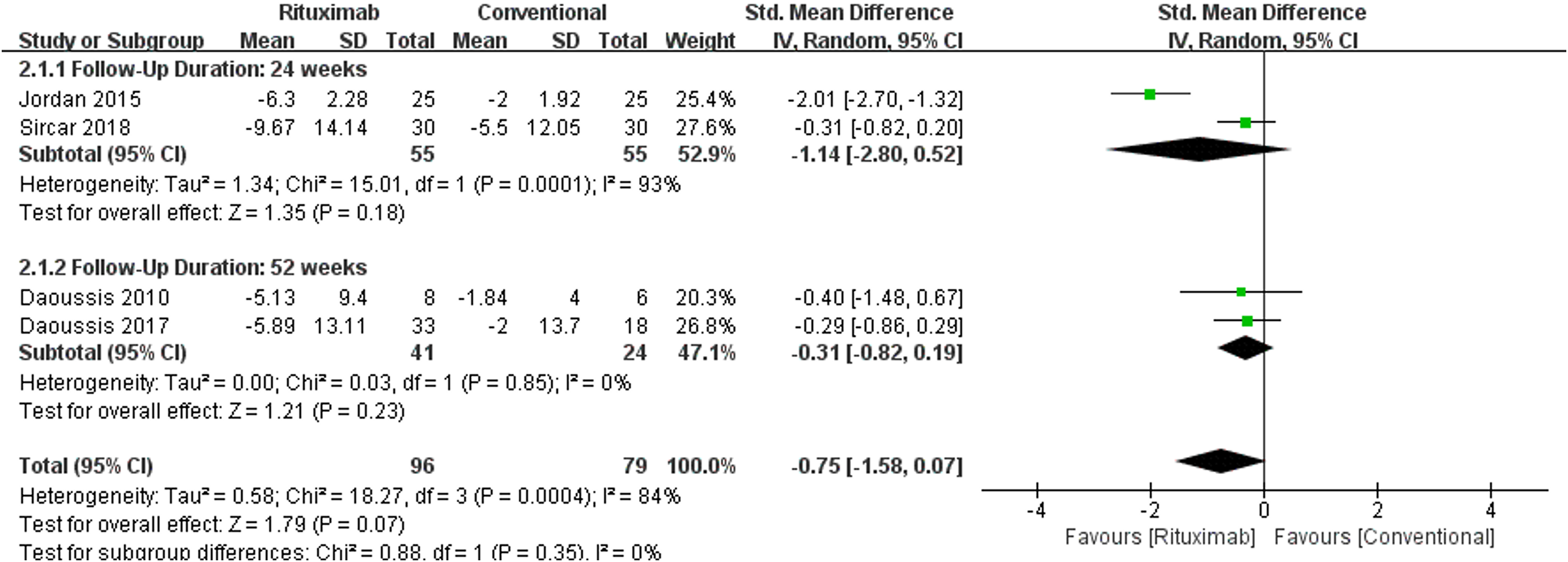
Forest plot analyzing the effect of rituximab on changes in mRSS score among SSc-ILD patients following 24 and 52 weeks of treatment.
Adverse events
The analysis of adverse events was limited to the two studies that directly compared rituximab with cyclophosphamide: Ebata et al. evaluated at 12 weeks, and Sircar et al. evaluated at 24 weeks. The adverse events in both studies were leukopenia, pneumonia, and urinary tract infection (UTI). Since all three analyses of adverse events showed no significant heterogeneity (I² = 0% each), a fixed effects model was employed. Additionally, some results included a zero incidence, so the analyses were conducted using the Peto odds ratio (POR). The meta-analysis revealed that rituximab was associated with a significantly lower incidence of leukopenia compared to cyclophosphamide (POR 0.17, 95% CI 0.04 to 0.76, P = .02). In contrast, in cases of pneumonia and UTI, while rituximab showed a trend towards lower occurrence compared to cyclophosphamide, statistical significance was not achieved (P = .11, P = .44, respectively). Forest plots for adverse events are presented in Figure 5.

Forest plots for adverse events of (A) leukopenia, (B) pneumonia, and (C) urinary tract infection.
Risk of bias assessment
In the 2010 study by Daoussis et al., participants were assigned based on their birthdate: those with even birthdates were allocated to the rituximab group, while those with odd birthdates were assigned to the control group. This method of allocation was deemed to pose a higher risk of bias due to a potential imbalance in the randomization sequence and concealment of allocation. Moreover, as it was an open-label study, the risk of performance bias associated with blinding of participants and personnel was considered high (Figure 6A).

Risk of bias assessment: A. Risk of bias summary and graph for randomized controlled studies, B. Risk of bias summary and graph for non-randomized studies. Red for high risk of bias, yellow for unclear bias, and green for low risk of bias.
Among non-randomized studies, a study by Daoussis et al. in 2017 did not provide uniform immunomodulatory therapy to the conventional treatment group. Consequently, it was deemed to have a higher risk of bias in terms of target group comparison and selection processes. A 2015 study by Jordan et al. compared the rituximab group with matched control patients from the EUSTAR cohort database, which raised concerns about a higher risk of bias in the target group comparison and selection processes. Additionally, the study noted that missing FVC values and the limited number of evaluated patients could potentially bias the results due to incomplete outcome data (Figure 6B).
Assessing publication bias
To assess publication bias in the studies included in the meta-analysis, we utilized a funnel plot, Begg and Mazumdar Rank Correlation, and Egger's regression. The funnel plot appeared asymmetric, and Egger's regression test confirmed this asymmetry (P = .004). However, the Begg and Mazumdar rank correlation test produced conflicting results (P = .1361), and no missing studies were identified using the trim and fill methods. Additionally, an examination of Cook's distances of the included studies revealed no overly influential studies. Therefore, despite these considerations, the study was continued while maintaining the results of the meta-analysis (Figure 7).

Funnel plot for assessing publication bias among studies included.
Discussion
This systemic review and meta-analysis aimed to evaluate the clinical efficacy of rituximab compared to standard treatment in patients with moderate to severe SSc-ILD and to explore its potential as a therapeutic option for SSc-ILD patients who exhibit disease progression despite conventional therapy. Our meta-analysis, which synthesized data from six studies (including two randomized controlled trials and four non-randomized studies) involving 275 patients, indicated that rituximab might be more effective in mitigating FVC decline than conventional treatments such as mycophenolate and cyclophosphamide in patients with SSc-ILD. Our findings underscore both the potential and limitations of rituximab as a treatment option for SSc-ILD, necessitating further research to solidify its clinical role.
Investigations seeking effective treatments for SSc-ILD have validated the clinical efficacy of several therapeutic agents, leading to their recommendation as first-line therapies. The Scleroderma Lung Study I and II trials support the use of cyclophosphamide and mycophenolate as first-line therapies due to their efficacy in reducing FVC decline.24–26 Similarly, the focused trial highlights the effectiveness of tocilizumab in preserving lung function in early SSc-ILD patients.27,28 However, there is a lack of evidence and discussion regarding the treatment of progressive SSc-ILD that does not respond to these initial therapies, indicating the need for further research. Although the clinical evidence for rituximab predominantly stems from few observational studies and small-scale RCTs, its unique mechanism of action—directly inhibiting B cells that are pivotal in the pathogenesis of SSc and its diverse manifestations—offers a pathophysiological rationale for its potential effectiveness in SSc-ILD treatment.29–32 Consequently, we hypothesize that rituximab may exhibit superior clinical efficacy compared to conventional treatments such as mycophenolate and cyclophosphamide, positioning it as a promising therapeutic option for progressive SSc-ILD unresponsive to first-line therapies.
Our primary analysis focused on the impact of rituximab on the decline in FVC % predicted, and demonstrated it to produce a significant reduction in FVC decline compared to conventional treatments (SMD 0.65, 95% CI 0.08–1.22, P = .03). Our finding is supported by previous studies with similar results. A systematic review with meta-analysis by Goswami et al., incorporating two RCTs and multiple observational studies, reported comparable findings. 29 Goswami and colleagues employed MD to assess the 6-month change in FVC, utilizing data from the studies of Jordan et al. 20 and Sircar et al., 19 both of which contributed to our meta-analysis. Their analysis revealed that the rituximab group exhibited a statistically significant improvement in FVC relative to the matched control group (MD 1.03, 95% CI 0.11–1.94, P < .001). Another systematic review and meta-analysis by Borrirukwisitsak et al. included eight RCTs and cohort studies to evaluate the clinical efficacy of rituximab in SSc-ILD by comparing rituximab with non-rituximab treatment. 33 They also used MD for comparison and did not divide the analysis by time period. While the overall analysis indicated that the rituximab group did not significantly reduce FVC decline compared to the non-rituximab group (MD 6.02, 95% CI −0.31 to 12.35, P = .06), a subgroup analysis of three RCTs revealed a significant improvement in FVC (MD 6.59, 95% CI 3.51 to 9.68, P < .0001). These differences are likely attributable to variations in the units employed for comparison and the methodologies used to address the timing of FVC measurements. Nonetheless, the overall trend suggests that the rituximab group exhibits a lesser decline in FVC than the non-rituximab group. These findings indicate that rituximab may serve as a potential treatment for progressive SSc-ILD unresponsive to mycophenolate and cyclophosphamide therapies, though further research is warranted.
In contrast to the findings for FVC, the meta-analysis revealed no significant difference in the reduction of DLco between rituximab and conventional treatments (SMD 0.17, 95% CI −0.4 to 0.73, P = .57). This result diverges from the study conducted by Borrirukwisitsak et al., 33 which incorporated two RCTs and four non-randomized studies to assess the differential impact on DLco between rituximab and non-rituximab groups. Their meta-analysis indicated that the rituximab group significantly reduced the decline in DLco compared to the non-rituximab group (MD 7.75, 95% CI 3.73–11.78, P = .0002). This discrepancy may be attributed to differences in the units of analysis (MD vs. SMD) and the methodologies employed for timing DLco measurements, factors that likely had a substantial influence on the outcomes.
In our study, rituximab did not achieve a significant reduction in mRSS compared to conventional treatment (SMD −0.75, 95% CI −1.58 to 0.07, P = .07), mirroring the results observed for DLco. Analyses at 24 and 52 weeks also failed to reach statistical significance, although a trend toward greater mRSS reduction with rituximab was noted. Conversely, research by Borrirukwisitsak et al. 33 demonstrated that the rituximab group showed significantly greater mRSS reductions compared to the non-rituximab group across classifications by study design (MD −3.31, 95% CI −4.95 to −1.68, P < .0001), at a 24-month mRSS change (MD −2.27, 95% CI −3.5 to −1.04, P = .0002), and in the overall analysis (MD −2.99, 95% CI −4.37 to −1.62, P < .0001). These discrepancies may be attributed to variations in the types of studies included, the measurement units of mRSS, and the analytical methods employed. However, the consistent trend of greater mRSS reduction with rituximab supports our analysis.
Regarding adverse events, rituximab demonstrated a significantly lower incidence of leukopenia compared to cyclophosphamide. Although the occurrences of pneumonia and UTI were generally lower in the rituximab group compared to the cyclophosphamide group, these differences did not reach statistical significance. These findings align with results reported in other studies comparing the adverse event profiles of rituximab and cyclophosphamide.34,35 In the study by Stone et al., which evaluated the clinical efficacy and safety of rituximab versus cyclophosphamide in patients with anti-neutrophil cytoplasmic antibody-associated vasculitis, the incidence of leukopenia was lower in the rituximab group than in the cyclophosphamide group (3 of 99 vs. 10 of 98). 34 The study did not differentiate between types of infections, yet there was no observed difference in the overall infection rates between the two groups (7 of 99 vs. 7 of 98). Nevertheless, given the limited number of patients and studies included, further research is warranted to evaluate the safety profile of rituximab.
Our review and meta-analysis have several limitations. First, the small number of included studies makes it difficult to generalize the results. However, inclusion of 275 patients, which is not non-significant, and the inclusion of two well-orchestrated RCTs, the results should not be underestimated given the scarcity of studies comparing rituximab directly with mycophenolate and cyclophosphamide. Second, the different timing of FVC, DLco, and mRSS measurements in the included studies might have introduced bias when evaluated through SMD. Last, by selecting a maximum follow-up period of 52 weeks, the study is limited in comparing the long-term clinical effects of rituximab versus conventional treatments. Future efforts should aim to include more studies and diverse analytical methods to provide a more insightful and generalized understanding of the clinical efficacy of rituximab.
Conclusions
The meta-analysis demonstrated that rituximab has a significant clinical effect in reducing FVC decline compared to conventional treatments such as mycophenolate and cyclophosphamide. This suggests potential for rituximab as a treatment for progressive SSc-ILD unresponsive to conventional therapies. Further research is needed to confirm the clinical advantages and potential of rituximab over conventional treatments in SSc-ILD.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251333912 - Supplemental material for Rituximab in systemic sclerosis-associated interstitial lung disease: A systematic review and meta-analysis
Supplemental material, sj-docx-1-sci-10.1177_00368504251333912 for Rituximab in systemic sclerosis-associated interstitial lung disease: A systematic review and meta-analysis by Ji Hoon Jang, Minyoung Her, Ju Hyun Oh, Joo Hun Park, So Young Jung, Junghae Ko and Jae Ha Lee in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504251333912 - Supplemental material for Rituximab in systemic sclerosis-associated interstitial lung disease: A systematic review and meta-analysis
Supplemental material, sj-docx-2-sci-10.1177_00368504251333912 for Rituximab in systemic sclerosis-associated interstitial lung disease: A systematic review and meta-analysis by Ji Hoon Jang, Minyoung Her, Ju Hyun Oh, Joo Hun Park, So Young Jung, Junghae Ko and Jae Ha Lee in Science Progress
Footnotes
Acknowledgments
The authors declare that this study was conducted without any financial or nonfinancial support.
Ethical considerations and consent for publications
Not applicable.
Author contributions/CRediT
Conceptualization: JH.J., M.H., JH.L.; methodology: JH.J., M.H., JH.L.; validation: JH.O., JH.P., SY.J.; formal analysis: JH.J., SY.J., J.K.; investigation: JH.J., M.H., JH.L.; data curation: JH.J., JH.O., JH.P.; writing-original draft: JH.J., M.H., JH.L.; writing-review and editing: all authors; visualization: JH.J., JH.O., J.K.; supervision and project administration: JH.L.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
All data are provided within the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
