Abstract
Objective
Sepsis-induced coagulopathy (SIC) is a life-threatening complication that significantly impacts patient prognosis. This systematic review and meta-analysis aimed to evaluate clinical and biological indicators associated with the development of SIC to facilitate early risk identification.
Methods
We searched nine databases for studies reporting predictive factors for SIC. Data were pooled using random-effects or fixed-effects models as appropriate. Methodological quality was assessed using the Newcastle-Ottawa Scale.
Results
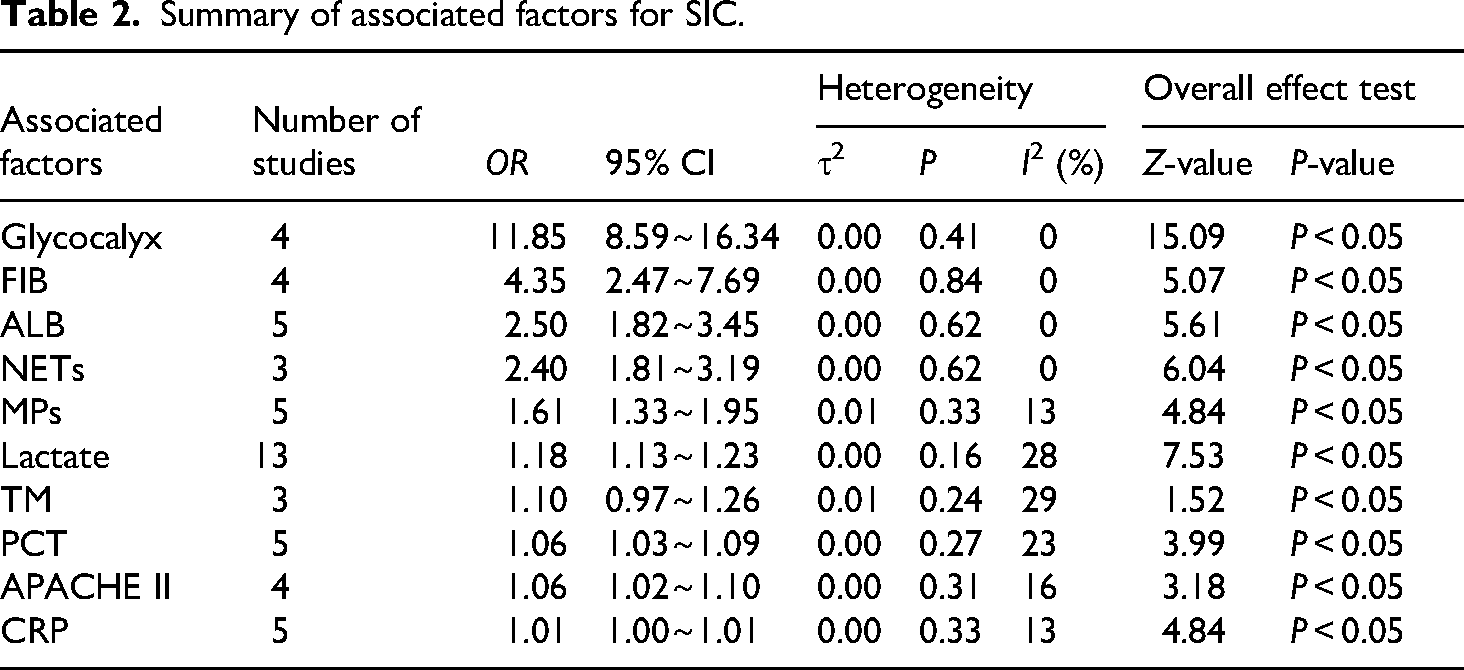
A total of 34 studies involving 37,459 patients were included. Meta-analysis revealed that several markers showed a significant association with SIC. Glycocalyx degradation emerged as a strong predictor (OR = 11.85, 95% CI: 8.59–16.34). Other associated markers included reduced Fibrinogen (OR = 4.35, 95% CI: 2.47–7.69), Albumin (OR = 2.5, 95% CI: 1.82–3.45), Neutrophil Extracellular Traps (OR = 2.4, 95% CI: 1.81–3.19), increased circulating Microparticles (OR = 1.61, 95% CI: 1.33–1.95), elevated Lactate(OR = 1.18, 95% CI: 1.13–1.23) and Thrombomodulin (OR = 1.10, 95% CI: 0.97–1.26), Procalcitonin, APACHE II score and C-reactive protein were also demonstrated predictive value for SIC occurrence.
Conclusion
Our findings identify multiple biological and clinical predictors associated with SIC. However, these results should be interpreted as statistical associations rather than direct causal links. These markers may assist clinicians in early recognition of high-risk patients, though further prospective trials are needed to confirm their clinical utility.
Registration
This study was registered on the PROSPERO (CRD420250653820).
Introduction
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. 1 A hallmark of its complex pathogenesis is the profound disruption of the coagulation system, primarily driven by vascular endothelial injury and excessive systemic inflammation.2,3 Sepsis remains a major global health challenge, affecting over 30 million people and causing 5 million deaths annually.4,5 Sepsis-induced coagulopathy (SIC) represents a critical stage of this dysfunction, where endothelial damage triggers a cascade of hemostatic imbalances that can progress to multiple organ failure. 6
Pathophysiologically, SIC evolves from beneficial immunothrombosis, which is a localized host defense mechanism, to dysregulated thromboinflammation. 7 Persistent systemic inflammation triggers extensive microvascular thrombosis, aggravating endothelial dysfunction, capillary leakage, and consumptive coagulopathy, which eventually culminates in disseminated intravascular coagulation (DIC) and multiple organ dysfunction syndrome (MODS). 8 Currently, SIC affects 24% to 68% of sepsis patients globally and is associated with a twofold increase in mortality.9–13
Given its multifactorial nature, identifying the exact predictors of SIC and understanding their interactions are essential for early detection and clinical management. 14 Despite extensive research, 15 significant gaps remain in characterizing these markers within the evolving landscape of sepsis care.
With the evolution of sepsis management, an updated and comprehensive analysis of factors associated with SIC is necessary. By integrating heterogeneous data, this study aims to provide robust evidence on the most critical predictors of SIC to guide clinical decision-making and optimize patient outcomes.
Methods
Search strategy and selection criteria
This systematic review and meta-analysis was conducted following the PRISMA 2020 guidelines, 16 and registered on the PROSPERO (CRD420250653820). A systematic search was conducted across international (PubMed, Embase, Web of Science, Cochrane Library, Ovid-Medline, Scopus) and Chinese databases (CNKI, Wanfang, CBM) for relevant studies published up to December 31, 2025. We employed a comprehensive search strategy combining controlled vocabulary (e.g. MeSH terms in PubMed and Emtree in Embase) and free-text keywords related to “sepsis,” “coagulopathy,” and “associated factors.” No language restrictions were applied to minimize publication bias. The complete search strategy is detailed in the text and Supplementary Table 1.
Inclusion criteria: (1) Peer-reviewed observational studies (prospective or retrospective cohort studies, case-control studies) published in English or Chinese. (2) Adult patients (
Exclusion criteria: (1) Publication types were reviews, conference abstracts, case reports, editorials, and animal experiments. (2) Studies with overlapping populations, inaccessible full texts, or insufficient data for effect size extraction. (3) Patients with significant co-morbidities (e.g. hematological malignancies) that could confound coagulation markers.
Literature screening process
Literature records were managed using EndNote X9, with duplicates systematically removed. Two reviewers (YW and FX) independently conducted a two-stage screening process: an initial title and abstract review followed by a comprehensive full-text evaluation based on the pre-defined eligibility criteria. Any discrepancies at either stage were resolved through consensus or, if necessary, consultation with a third reviewer (XP or ZY).
Data extraction and study quality assessment
Data extraction was performed independently by two researchers (YW and FX), capturing study characteristics, participant demographics, and adjusted effect sizes (OR with 95% CI). In this study, OR were extracted from the included literature as effect measures. Only multivariable-adjusted effect estimates were extracted when available. Importantly, multivariable-adjusted estimates were included regardless of their statistical significance, provided that the corresponding predictors were prespecified and reported in the original studies. Factors associated with SIC were excluded only when multivariable-adjusted effect estimates were unavailable or could not be reliably extracted.
Methodological quality was assessed by YW and LC using the Newcastle-Ottawa Scale (NOS). The NOS tool assesses studies through three major modules with a total of eight items, which specifically include: (1) selection of study population, (2) comparability between groups, (3) assessment of exposure/outcome. The NOS employs a semi-quantitative system with a maximum score of 9 stars, where scores of 7–9 indicate high-quality research. Two reviewers independently assessed study quality using the NOS. Inter-rater reliability was evaluated using Cohen's kappa statistic, which showed moderate agreement between reviewers (k = 0.8, P < 0.001), indicating acceptable consistency in quality assessment.
Data synthesis and analysis
Data synthesis was performed using Review Manager (version 5.3) and Stata (version 14.0). Adjusted OR and their 95% CI were extracted as the primary effect measures. Between-study heterogeneity was assessed using Cochran's Q statistic and the
Results
Study characteristics
A total of 34 studies met the inclusion criteria and were incorporated into the final analysis after title/abstract screening and full-text evaluation (Figure 1).

Flowchart for study selection process.
The characteristics of the eligible studies are summarized in Table 1. These comprised 14 prospective and 20 retrospective investigations, including 26 cohort studies and 8 case–control studies. Most included studies were conducted in Asian populations, predominantly from China and Japan, the majority originated from Asia (n = 26; primarily China and Japan), followed by North America (n = 3) and Europe (n = 5), while only a limited number originated from other regions.
Characteristics of the included studies.

Note: ED: emergency department; EICU: emergency intensive care units; RICU: respiratory intensive care units; SICU: surgery intensive care units.
①Microparticles; ②Neutrophil extracellular traps; ③Glycocalyx; ④Lactate; ⑤Thrombomodulin; ⑥Albumin; ⑦Procalcitonin; ⑧C-reactive protein; ⑨APACHE II; ⑩Fibrinogen.
Quality assessment
The methodological quality of the included studies was assessed using the NOS. Nine studies achieved the maximum score of 9, fourteen scored 8, eight scored 7, and three scored 6, reflecting an overall high quality and supporting the robustness and reliability of the pooled findings. Supplementary Table 1 provides a detailed breakdown of each study across domains of participant selection, comparability between groups, and outcome assessment, together with the overall score. In addition, two representative studies are presented in the Supplementary Materials to illustrate the step-by-step NOS scoring process at the item level.
Results of synthesis
These studies investigated a range of contributing factors, including Glycocalyx,24–27 Fibrinogen (FIB),39,41,44,45 Albumin (ALB),38,39,47,48,50 Neutrophil Extracellular Traps (NETs),19,23,51 Microparticles (MPs),18–22 Lactate,28–35,43,46,48–50 Thrombomodulin (TM),32,36,37 Procalcitonin (PCT),31,40,41,48,50 Acute Physiology and Chronic Health Evaluation II score (APACHE II)22,33,39,51and C-reactive Protein (CRP).23,29,40,42,51
Overall, ten factors were synthesized using random-effects models, with effect estimates expressed as OR comparing SIC versus non-SIC patients (Table 2). These factors were categorized into three groups based on their pathophysiological roles. Endothelial Injury Markers (Glycocalyx, TM, MPs),Clinical Severity and Metabolic Indicators(FIB, ALB, APACHE II, Lactate) and Inflammation and Immunothrombosis Markers(NETs, PCT, CRP).
Summary of associated factors for SIC.
Endothelial injury markers
Markers reflecting endothelial injury and Glycocalyx degradation showed the most robust associations with SIC development (Figure 2).

Forest plot for endothelial injury markers. (2A: Glycocalyx, 2B: TM, 2C: MPs).
Glycocalyx24–27: Demonstrated the strongest association with SIC (4 studies; pooled OR = 11.85, 95% CI: 8.59–16.34), with no detectable heterogeneity (
TM32,36,37: Was significantly associated with an increased likelihood of SIC (3 studies; OR = 1.10, 95% CI: 0.97–1.26), with mild heterogeneity
MPs18–22: Showed a consistent association with SIC (5 studies; OR = 1.61, 95% CI: 1.33–1.95), with mild heterogeneity (
Clinical severity and metabolic indicators
Factors reflecting systemic metabolic distress and overall disease severity exhibited substantial predictive value (Figure 3).

Forest plot for clinical severity and metabolic indicators. (3A: FIB, 3B: ALB, 3C: APACHE II, 3D: Lactate).
FIB39,41,44,45: Reduced levels were markedly associated with SIC and likely represent a consequence of consumptive coagulopathy rather than a primary upstream trigger. (4 studies; OR = 4.35, 95% CI: 2.47–7.69), with no detectable heterogeneity (
ALB38,39,47,48,50: Hypoalbuminemia was strongly associated with SIC (5 studies; OR = 2.50, 95% CI: 1.82–3.45), with no detectable heterogeneity (
APACHE II22,33,39,51: Higher scores were significantly associated with SIC occurrence (4 studies; OR = 1.06, 95% CI: 1.02–1.10), with mild heterogeneity (
Lactate28–35,43,46,48–50: Elevated Lactate levels were associated with SIC (13 studies; OR = 1.18, 95% CI: 1.13–1.23), with mild heterogeneity (
Inflammation and immunothrombosis markers
Markers related to the host's inflammatory response and immunothrombotic pathways showed consistent positive associations (Figure 4).

Forest plot for inflammation and immunothrombosis markers. (4A: NETs, 4B: PCT, 4C: CRP).
NETs19,23,51: Were significantly associated with SIC (3 studies; OR = 2.40, 95% CI: 1.81–3.19), with mild heterogeneity (
PCT31,40,41,48,50: Showed a positive association with SIC (5 studies; OR = 1.06, 95% CI: 1.03–1.09), with mild heterogeneity (
CRP23,29,40,42,51: Demonstrated a small but statistically significant association(5 studies; OR = 1.01, 95% CI: 1.00–1.01), with mild heterogeneity(
Conceptual framework
Based on the synthesized results, a conceptual diagram (Figure 5) was developed to illustrate the interplay between these multi-dimensional factors and the development of SIC.

Conceptual framework of the interplay between these multi-dimensional factors and the development of SIC.
Sensitivity analysis
Leave-one-out sensitivity analyses were performed for the ten factors associated with SIC. By sequentially excluding each study and recalculating the pooled effect sizes using a random-effects model, we observed that the pooled estimates remained consistent across all iterations. These results suggest that the overall findings were robust and not materially driven by any individual study.
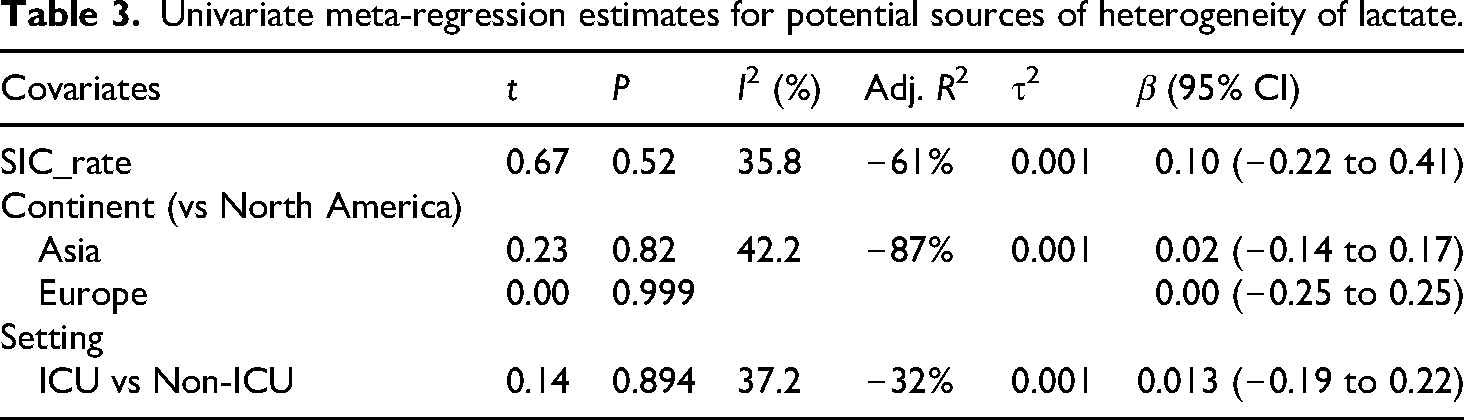
Meta-regression analysis for lactate
Following the methodological guidance from the Cochrane Handbook (version 6.4), meta-regression is generally recommended only when at least ten studies are available per covariate to ensure adequate statistical power. Among the ten factors analyzed, only Lactate (n = 13) met this criterion. For all other biomarkers, the number of contributing studies was
Univariate meta-regression estimates for potential sources of heterogeneity of lactate.
Publication bias
Publication bias was assessed with caution due to the limited number of studies available for most associated factors. Funnel plots were generated to visually explore potential publication bias; however, formal interpretation was restricted to factors supported by an adequate number of studies. Among the ten identified factors, only Lactate was reported in a sufficient number of studies (n = 13) to permit formal statistical assessment of small-study effects. For Lactate, Egger's regression test did not indicate significant publication bias (bias coefficient = 1.48, P = 0.105), and visual inspection of the funnel plot showed approximate symmetry, suggesting a low risk of publication bias for this factor (Figure 6).For the remaining factors, the number of contributing studies was limited (generally fewer than 10), which substantially reduces the reliability of both funnel plot symmetry assessment and formal tests such as Egger's regression. Therefore, no definitive conclusions regarding publication bias were drawn for these factors, and their pooled results should be interpreted cautiously and considered hypothesis-generating rather than confirmatory.

Funnel plot for lactate.
Discussion
This systematic review and meta-analysis identified a multi-dimensional set of clinical and biological predictors associated with SIC. By synthesizing data from both routine clinical parameters and novel biomarkers, our findings highlight three core pathological axes: endothelial injury, immune-inflammatory dysregulation, and systemic metabolic distress.
The endothelial injury axis: the initiating event of SIC
Endothelial dysfunction is increasingly recognized as the central mechanism in the pathogenesis of SIC. 14 In our analysis, markers of endothelial Glycocalyx degradation demonstrated the most potent predictive strength (OR = 11.85). The Glycocalyx is a delicate, carbohydrate-rich layer coating the vascular endothelium that regulates permeability and prevents the adhesion of platelets and leukocytes. 52 Under the systemic “insult” of sepsis, inflammatory mediators trigger the shedding of Glycocalyx components (such as Syndecan-1) into the circulation. This structural breakdown exposes the underlying endothelium to pro-thrombotic factors, initiating microvascular thrombosis.53,54 The finding in our study underscores that monitoring Glycocalyx integrity is paramount for the early identification of SIC.However, this pooled estimate was derived from only four studies, and extreme effect sizes in meta-analyses may arise from small-study effects or residual publication bias. Therefore, although the association appears strong, it should be interpreted cautiously and considered hypothesis-generating pending validation in larger, multicenter cohorts. TM (OR = 1.1) and MPs (OR = 1.61) serve as significant indicators within this axis. Under physiological conditions, TM acts as a key anticoagulant on the endothelial surface; however, widespread endothelial damage causes TM to be cleaved and released into the plasma. 55 This shedding not only signifies the loss of local anticoagulant capacity but also reflects a pathological “phenotype shift” of the endothelium from a thromboresistant to a procoagulant state. 56 Similarly, MPs act as pro-coagulant vesicles that transport tissue factor, amplifying the coagulation cascade intravascularly. 57 Integrating these markers allows for a sensitive biological assessment of the vascular endo-environment.
The immune-inflammatory cascade: driving thromboinflammation
Sepsis is characterized by a dysregulated host immune response, where inflammation and coagulation are inextricably linked. 2 Our findings demonstrate that NETs (OR = 2.4) are significantly associated with SIC development. As a cornerstone of “immunothrombosis,” NETs provide a physical scaffold for fibrin deposition and platelet entrapment, bridging the gap between innate immunity and pathological thrombosis. 58 Among traditional inflammatory markers, PCT (OR = 1.06) and CRP (OR = 1.01) showed statistically significant, albeit more modest, associations with SIC. These findings suggest that a persistent systemic inflammatory state acts as a continuous driver of hemostatic deterioration.59,60 Although their individual predictive power is lower than that of endothelial markers, their high clinical accessibility makes CRP and PCT valuable tools for monitoring infection progression and the secondary risk of coagulopathy at the bedside.
Clinical severity and metabolic indicators: reflections of systemic crisis
Clinical severity scores and metabolic markers are indispensable for refining the predictive landscape of SIC. 61 In our study, the APACHE II score (OR = 1.06) and Lactate levels (OR = 1.18) emerged as robust indicators of the cumulative physiological burden and tissue hypoxia. High APACHE II scores often reflect multiorgan dysfunction, where circulatory and respiratory failures synergistically exacerbate hemostatic imbalances.62,63 The stability of this association across different cohorts suggests that the overall severity of sepsis, as captured by this scoring system, is a universal prerequisite for the transition into a coagulopathic state. Complementing this, Lactate acts as a critical biomarker of metabolic distress and cellular hypoxia. In sepsis, elevated Lactate signifies a transition toward decompensated shock and mitochondrial dysfunction. 64 Our univariate meta-regression on the 13 studies involving Lactate further reinforced its reliability, suggesting its association with SIC remains stable across diverse healthcare environments and populations. In contrast, markers such as FIB (OR = 4.35) and ALB (OR = 2.50) provide complementary information regarding the consumptive and exudative phases of the disease,65,66Notably, decreased fibrinogen levels in sepsis most likely reflect ongoing coagulation activation and consumption, indicating an established coagulopathic state rather than serving as an early upstream predictor. Low albumin levels, in particular, are often a harbinger of capillary leak syndrome, which parallels the destruction of the endothelial Glycocalyx. 67
Methodological heterogeneity and clinical implications
A significant challenge in interpreting our findings is the lack of standardization across studies regarding detection methods and diagnostic thresholds. For key indicators such as Glycocalyx, NETs, and MPs, various assays were employed, which likely contributed to the statistical heterogeneity observed. Furthermore, the “optimal cut-off” for many biomarkers varied across cohorts, complicating the establishment of a universal threshold for clinical use. To date, no human trials have demonstrated that targeting these markers reduces SIC incidence or improves clinical outcomes. Consequently, the predictive values identified in this meta-analysis should be viewed with caution. Future research must prioritize the standardization of laboratory techniques and the validation of consensus-based thresholds before these markers can be formally integrated into SIC diagnostic protocols.
Based on these findings, we propose a two-tier risk stratification strategy for the early identification and management of SIC. First-tier screening relies on routine, readily available parameters, including APACHE II, Lactate, FIB, ALB, and CRP, for rapid bedside assessment. Patients flagged as high-risk through these routine tools should then undergo second-tier precision assessment using specialized biomarkers such as Glycocalyx and TM. This hierarchical approach allows for an “early-warning” phase, where endothelial damage can be detected before the manifestation of irreversible clinical abnormalities, such as severe platelet consumption or multi-organ failure. This strategy not only optimizes resource allocation but also provides a roadmap for future predictive model construction.
Limitations
This study provides valuable insights into the associated factors associated with SIC; however, several limitations should be acknowledged. First, Given the observational nature of the included studies, the identified factors should be interpreted as strong statistical associations rather than definitive causal links. Second, the number of eligible studies was relatively limited, which constrained our ability to fully elucidate the specific roles and predictive value of certain factors, thereby restricting their immediate clinical applicability. Third, the literature search was confined to studies published in English and Chinese, raising the possibility that relevant evidence reported in other languages may have been overlooked. Fourth, biological collinearity remains a challenge in sepsis research, and multivariable models in original studies may not have fully eliminated these inter-relationships. Last, most of the included studies were conducted in Asian populations, which may introduce regional bias and limit the generalizability of our findings to more diverse patient cohorts.
Conclusion
In conclusion, the development of SIC is driven by a triad of endothelial injury, inflammatory surges, and metabolic crises. Among the 10 predictors identified, endothelial glycocalyx markers and lactate demonstrate high predictive value and clinical robustness. Future research should focus on developing integrated, dynamic scoring systems that incorporate these multi-dimensional indicators, and prospective trials are needed to determine if early interventions targeting these pathways can improve clinical outcomes in sepsis.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261435291 - Supplemental material for Biomarkers and clinical factors associated with sepsis-induced coagulopathy: A systematic review and meta-analysis
Supplemental material, sj-docx-1-sci-10.1177_00368504261435291 for Biomarkers and clinical factors associated with sepsis-induced coagulopathy: A systematic review and meta-analysis by Wei You, Xiaoyu Fan, Ping Xu, Cheng Lei and Yanli Zeng in Science Progress
Footnotes
Acknowledgements
The authors would like to thank all investigators and participants of the original studies included in this systematic review and meta-analysis.
Author contributions
Wei You: conceptualization, methodology, software, visualization, and writing—original draft. Xiaoyu Fan: methodology, data curation, and visualization. Ping Xu and Cheng Lei: methodology, data curation, and visualization. Yanli Zeng: conceptualization, methodology, supervision, and writing—review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data analyzed in this study are derived from previously published articles. The extracted data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
