Abstract
This review article discusses neonatal lupus syndrome (NLS), an immune-mediated disease caused by maternal antibodies. Maternal antibodies in the fetal circulation are mostly but not always protective. NLS is a disease caused by pathogenic maternal autoantibodies in the fetal circulation. The passive immunization of the fetus by NLS-causing maternal antibodies may occur in the absence of a previously known maternal systemic autoimmune rheumatic disease (SARD). Screening for NLS-related antibodies in patients with related SARD or those in whom there is a risk of NLS including first-degree relatives should occur before pregnancy. This screening is best performed as part of a collaborative relationship between obstetrics and rheumatology. Pregnancy preparations in those with SARD include transitioning to pregnancy-safe medications. The symptoms of NLS range from minor skin rashes to fetal demise from heart block. Fetal screening allows for maternal therapeutic interventions that may be beneficial, as well as the use of fetal pacemakers in the more severe cases that include cardiac NLS.
Keywords
Introduction
Neonatal lupus syndrome (NLS) is a condition wherein maternal autoantibodies lead to clinical symptoms in the fetus and neonate that resemble those of systemic lupus erythematosus (SLE). The passage of maternal antibodies into the fetal circulation is generally but not always protective for the child, and this protection may last up to the first six months of life. 1 NLS is a rare condition that occurs following the transfer of maternal Sjogren (SSA and SSB) and U1-ribonucleoprotein (U1-RNP) antibodies across the placenta into the fetus. 2 In the fetus, these antibodies can cause mild to life-threatening clinical symptoms. Mild clinical symptoms and laboratory abnormalities can be seen in over 25% of such births. Severe symptoms and findings are rare and may be life-threatening. Symptoms such as congenital heart block (CHB) can occur in up to 2% of neonates born to mothers with positive antibodies. The subsequent risk of CHB following a prior pregnancy with NLS-related CHB increases significantly and has been estimated at around 10%. 2 These autoantibodies may be present in the absence of a diagnosed maternal systemic autoimmune rheumatic disease (SARD) and also occur in higher frequencies in the first-degree relatives (FDR) of those with SARD, making the at-risk group larger than we may otherwise assume. Though NLS is a disease of the fetus and newborn, the antibodies are derived from the mother. NLS antibody screening is therefore directed towards the mother only. Treatment, if required is directed at the fetus, but generally administered to the mother (transplacental transfer) and on rare occasions directly to the newborn.
The NLS-related antibodies are most commonly associated with primary Sjogren syndrome (SS) but can be seen in other SARD with secondary SS, including SLE, systemic sclerosis, and less commonly in rheumatoid arthritis (RA), and related disease. The frequency of SS autoantibodies in SLE is approximately 50% and in RA is only between 10% and 30% of patients.2,3
To optimize pregnancy outcomes, it is important for patients with SARD who desire a healthy pregnancy to have a well-structured multi-disciplinary medical team involved as early as possible. To ensure a safe and successful outcome, preconception counselling is extremely important and should involve a rheumatologist whenever possible. 4 Screening for NLS-related antibodies should be performed before conception if possible. In addition, the SARD should be in remission and the rheumatic therapies should be safe to use during pregnancy. Screening and monitoring for NLS and its complications allow for treatment that could prevent long-term complications of NLS.
The goal of this article is to increase awareness of this condition, its pathophysiology, screening requirements, and treatment to improve the outcomes for our patients. Patients and providers must remember that NLS may occur in the absence of maternal SLE, but can affect the fetuses of mothers with a variety of SARD hence the importance of screening. We also should remember that even clinically asymptomatic patients and those with a diagnosis of preclinical autoimmunity may have children with NLS. 5
Epidemiology of NLS
For several decades now, NLS has been recognized as a disease related to maternal SARD, that can even occur before the development of symptoms in the mother. 6 NLS is a disease of transplacental passage of maternal autoantibodies and not fetal-produced antibodies. NLS is not related to any one specific maternal SARD, and maternal SARD symptoms may be present and/or a SARD may be diagnosed before, during, or following pregnancy. It is estimated that 64.8% of cases of NLS are seen in mothers with a pre-existing diagnosis of a SARD. Approximately 30% of the time a diagnosis of SARD is not made until after pregnancy. Due to the physiological changes in pregnancy and the similarity to some SARD, a diagnosis of a SARD is made only 5.5% of the time during pregnancy. 7 SLE is the maternal SARD most commonly associated with NLS (24.4%), followed by Sjögren's syndrome (13.4%), RA (7%), and other diseases like undifferentiated connective tissue disease, and mixed connective tissue disease accounting for fewer than 10% of cases.2,7
The most frequently associated antibodies are anti-SSA/Ro, and anti-SSB/La, making up 63.2% of the cases of NLS. Anti-SSB/La alone makes up only 3.4%, and less frequently anti-U1RNP antibodies make up 1.6% of cases.7,8 In rare cases, maternal antibodies have been reported as absent in NLS. In such cases, there remains questions about how extensive maternal antibodies need to be. Moreover, the antibody type may have little or no clinical impact on clinical outcomes even in the more severe cases of NLS such as those presenting with heart block. 9 Although the overall risk of NLS in pregnancies associated with SARD is low in the first pregnancy and pregnancy following previous healthy births, this risk significantly increases in pregnancies following previous cases of NLS and such pregnancies should be monitored closely. 10
Pathogenesis of NLS
Though the pathogenesis of NLS is not fully understood, we do know that there are both maternal and fetal risk factors. The risk of cardiac NLS is increased with the maternal presence of human leukocyte antigen (HLA)-A1, HLA-B8, HLA-DR3, 11 as well as the presence of HLA-B*15. The maternal and fetal presence of HLA-C*02, HLA-DQ5, and HLA-DR10 antigens also increase the risk of cardiac NLS. Less severe NLS including skin-limited disease is associated with the fetal presence of HLA DQB1*02, and DRB1*03 as well as polymorphism in the promoter region of the gene encoding TNF-α. 4 In addition, the amount of maternal SSA antibodies and the degree of placental transfer of the SSA antibodies are directly proportional to the risk of developing NLS. 12 The presence of HLA-DRB1*04 and HLA-Cw*05 allele variants in the fetus in the presence of maternal SSA antibody increases the risk of heart block. The presence of fetal DRB1*13 and Cw*06 alleles is protective against heart block. 13
It is postulated that cardiac NLS results from cardiomyocyte destruction via molecular mimicry and/or antigen–antibody interaction that causes cell death and fibrosis. The current understanding of the molecular mimicry pathway is that the NLS antibodies cross-react with L-Type calcium channels causing dysregulation of calcium homeostasis. The NLS-related antibodies also interact with intracellular cardiomyocyte antigens that are translocated to the surface as part of growth, development, and remodeling and this antigen–antibody interaction may result in cell death. Both of these pathways result in heart block. 14
Clinical features of NLS
NLS only causes symptoms in the fetus and/or in the neonate. The mother may experience clinical symptoms related to the underlying SARD, or side effects related to the treatment of NLS or the underlying SARD.
Symptoms: fetal/neonatal
The symptoms of NLS can be varied and multiple, with only a small number of babies developing severe disease. The symptoms range from mild skin rashes, cytopenia, elevated liver enzymes, to heart block, and fetal demise. Several of these symptoms are transient, lasting for a few months until the loss of the NLS-related maternal antibodies. 15 Heart block including third-degree heart block is one of the most severe complications and tends to be permanent. Third-degree heart block occurs in about 2% of anti-Ro/SSA and/or anti-La/SSB antibody-associated pregnancies without a prior history of NLS. 2 Following cutaneous or cardiac NLS, the risk of third-degree heart block in a subsequent pregnancy increases significantly and ranges between 13% and 18%. 16 There is significant fetal mortality associated with NLS, ranging from 15% to 20%. The mortality is related to complications including fetal hydrops, cardiomyopathy, endocardial fibroelastosis, low heart rate, and prematurity. 17 The associated maternal morbidity including preterm labor and spontaneous abortions is also significant. 18 In a prospective study of mothers with anti-SSA/La antibodies with or without anti-SSB/Ro antibodies, the most frequent symptoms of NLS noted were hematologic in 27% of cases, liver enzyme elevation in 26%, and cutaneous findings in 16% with CHB noted in only 1.6% of pregnancies. 7
Symptoms: women with SARD
In mothers with SARD, the level of disease activity or disease duration is not related to the risk of NLS. 16 In NLS, the most likely associated maternal SARD is SLE up to 24.4% of the time. NLS can be seen in other SARD including primary SS (13.4%) and undifferentiated connective tissue disease in 7% of cases. In 48.9% of NLS cases, no specific maternal SARD is diagnosed. 7 In patients with RA wishing to conceive, the frequency of NLS-related antibodies is low. It has been estimated as approximately 4%. 19
Symptoms: women without SARD
To develop NLS there must be NLS-related antibodies in the mother, and these antibodies must cross the placenta. A significant percentage of women who have children with NLS do not have a SARD. 10 Half of the mothers without a SARD who have babies with NLS, progress to a defined or undifferentiated SARD. 20 There is an increased risk of autoantibodies and the development of SARD in FDRs of those diagnosed with SARD. 21 Therefore, it is reasonable to screen for NLS-related autoantibodies in FDRs of those with SARD. Mothers with positive SSA/Ro and SSB/La antibodies were almost twice as likely to develop a SARD as patients with only SSA/Ro antibodies.22,23 Women without a SARD but who have antibodies to both SSA/Ro (especially the anti-Ro52 antibody) and SSB/La have an increased risk of giving birth to children with NLS. 22
In general, the overall risk of NLS is low with only a small percentage of exposed babies developing NLS. Sicca symptoms, sun-related skin rashes, and joint pain affecting the fingers that may be worse in the morning and associated with stiffness in the mother should lead to further evaluation for the development of SARD. 24 In the absence of a diagnosis of SARD, efforts should be made to become familiar with the symptoms of SLE, SS, MCTD, and RA as these are the most common maternal SARD associated with NLS.
Management of NLS
Managing NLS should ideally begin with pre-screening the mother and optimizing the management of pre-existing SARD. Screening for NLS-related autoantibodies is also a necessary part of pre-conception counselling. Treatment options include administering medication to the mother or directly treating the child who develops NLS.
Pre-conception counseling
Timing is especially important in patients with SARD such as SLE particularly those with active nephritis wishing to become pregnant. A significant number of these patients especially those with moderate or severe disease manifestations such as LN may be on medications that are not compatible with a successful pregnancy outcome (Figure 1). The best-known medications used in SARD that have deleterious effects on the developing fetus include methotrexate, leflunomide, mycophenolate, and cyclophosphamide. 25 Patients on any of these medications should be transitioned to pregnancy-safe medications (Table 1). In all cases, care should be individualized, and the patient's wishes must be incorporated especially as several medications used to care for rheumatic patients have little to no pregnancy or post-partum data. In such situations, the risks and benefits of the medications recommended need to be balanced with the management of the SARD and the pregnancy while considering what is best for the individual patient. The patients should also be in clinical and serological remission on these pregnancy-safe medications such as azathioprine and hydroxychloroquine (HCQ) before conceiving. 26
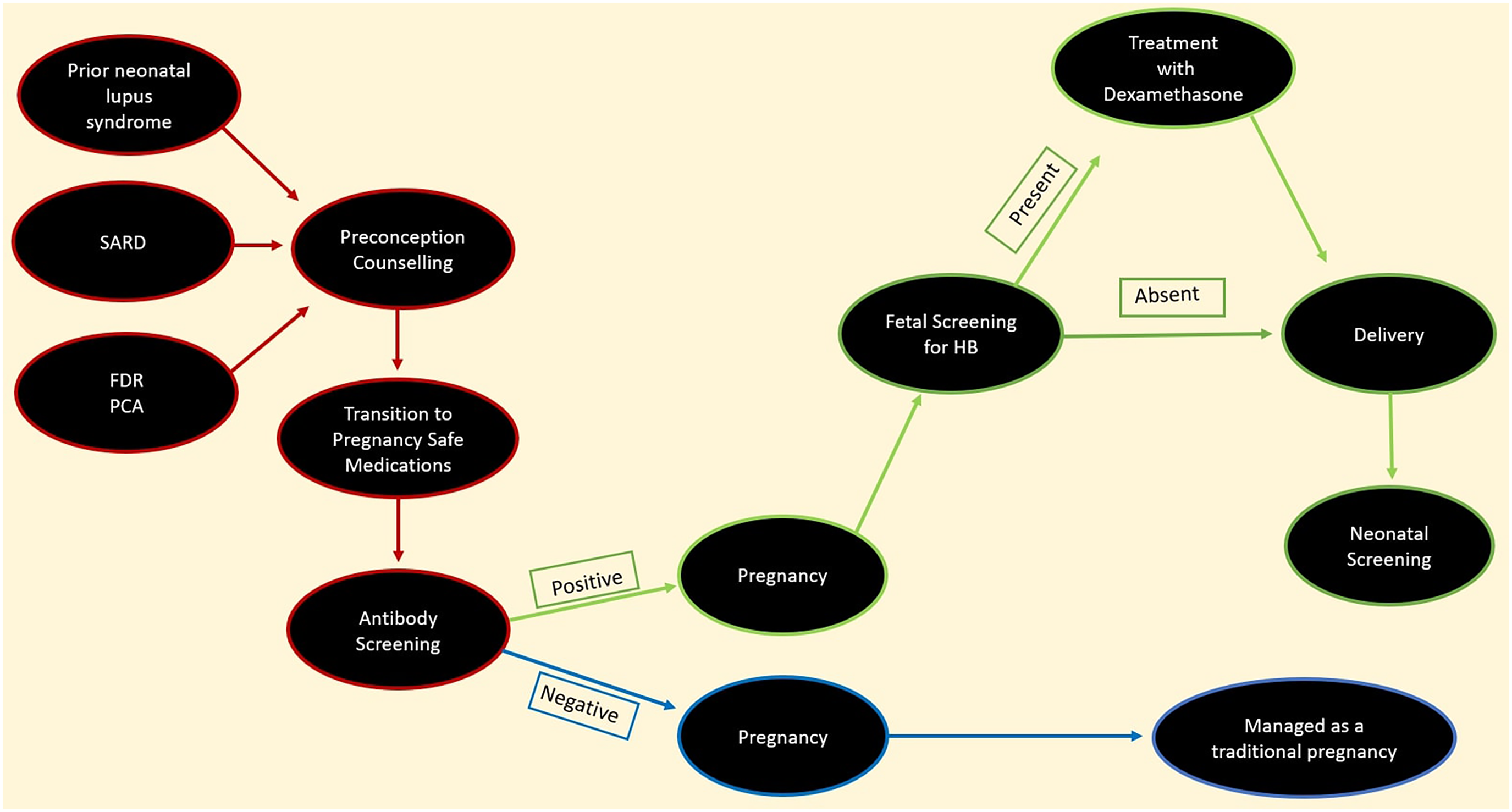
Maternal screening for neonatal lupus syndrome in patients with systemic rheumatic autoimmune disease.
Rheumatic medications and pregnancy.

Ideally, the screening for NLS-related autoantibodies in patients with pre-existing SARD should take place before conception. In our practice, we developed a practical application of the current guidelines keeping the central focus on patients. 26 The discussions around pregnancy typically occur early in the disease course. The outcomes of the conversation may influence the choice of contraception and therapeutic decisions. Pregnancy-safe medications are preferred for women of childbearing age who express an interest in conception. Patients with SARD on medications not considered to be safe in pregnancy should be transitioned to pregnancy-safe medications at the earliest opportunity once an interest in contraception has been expressed. There are always concerns related to potential disease flares with the changes in therapies as well as the general upheaval around the discussions of childbirth. Preconception counseling, medication changes, and NLS antibody screening help to improve pregnancy outcomes. We have found that addressing conception early and making the relevant changes help to relieve the stressors around this anxiety-provoking time for our patients. Close monitoring is required as these pregnancies may still be associated with flares of SARD or the development of NLS. Screening for autoantibodies that are related to NLS should occur early as this may have an impact on the decision-making process of our patients. In our practice, we revisit the conversation around conception and childbirth to allow patients who opt not to have children to revisit their decisions and options especially as their disease control improves. Should a patient become pregnant on medications where there is not enough data to indicate safety, our practice strives to enroll them in pregnancy registries.
Neither a diagnosis nor symptoms of SARD is necessary for there to be a risk of NSL. FDRs may not have come to medical attention before conception. In our practice, pre-pregnancy counselling involves educating our patients about the risk to FDR. We screen all patients with SARD and those in which there is a suspicion of SARD for NLS-related maternal antibodies. If NLS-related maternal antibodies are noted by non-rheumatologists, co-management with in a multidisciplinary care team involving a rheumatologist should be the goal. In pregnancies without NLS-related autoantibodies, management is similar to other standard pregnancies (Figure 1). In our practice, we screen patients at risk of possessing NLS related autoantibodies including FDRs, pre-clinical autoimmunity and asymptomatic SARD patients and follow anyone in whom NLS-related antibodies are present.
Screening of high-risk pregnancy
There are currently no universally accepted screening tools for NLS-related at-risk pregnancies. Nonetheless, prevention of more clinically significant manifestations of NLS via screening is the mainstay of management. Heart block is the most serious manifestation and complication of NLS, and it remains the main focus of preventive therapy. The goal of early detection and transplacental treatment of cardiac NLS is to prevent progression and possibly even cause regression of heart block. Appropriate fetal screening and monitoring for heart block is critical for early detection and allows for fetal-directed therapy using maternal dexamethasone if heart block is noted. Serial fetal echocardiography (FEC) is the most commonly used screening tool for the detection of NLS-related heart block. In women who have tested positive for NLS-related autoantibodies, screening with FEC is recommended between the 16th and 26th week of pregnancy. 10 The frequency of FEC depends on risk and may be weekly with a history of NLS in a prior pregnancy. 26 FEC can detect the full range of heart block including first-degree heart block to complete heart block. Although FEC cannot predict the progression to complete heart block, it remains a sensitive tool for early detection of heart block. This allows for the initiation of treatment that may improve outcomes. 17 Screening may also be necessary in the neonate for NLS-related postpartum heart block or postpartum progression of CHB in the neonatal period which is a rare and distinct subtype. 27
The care of the pregnancies at risk of NLS should be in conjunction with a rheumatologist and the frequency of follow-up with the various members of the health team should be individualized. 7 Within our practice, we screen for both NLS-related autoantibodies and other autoantibodies that may have an impact on pregnancy including antiphospholipid antibodies. We find that a clinically driven approach is more practical and cost-effective (Figure 1).
Prevention of NLS
There are currently no approved therapies for the prevention of NLS. HCQ has shown some benefits in the prevention of NLS in those at risk including those with high levels of autoantibodies 28 and has additional benefits in pregnant SARD patients. HCQ is a medication frequently prescribed by rheumatologists and has a good safety profile in pregnancy. 28 The maternal use of HCQ is customary in rheumatology practices and is commonly prescribed to prevent more serious manifestations of NLS including heart block. It is either prescribed alone or in combination therapy to pregnant patients with SARD at risk of NLS. 29 Although there are no official guidelines from the American College of Rheumatology (ACR) or the American College of Obstetricians and Gynecologists (ACOG), HCQ remains the most commonly prescribed prophylactic agent for NLS. 24 Data from registries in France, the United Kingdom, and the United States of America supports the use of HCQ in at-risk pregnancies as a method of preventing NLS cardiac disease in women with and without children with NLS. 28
Treatment of NLS
The symptoms of NLS range from a transient skin rash to hematological, hepatic, and cardiac abnormalities. 2 Other than NLS-related cardiac disease, supportive therapy is generally all that is required. This supportive therapy may include steroids and lead to a longer hospital stay. Currently, there is no uniformly agreed upon evidence-based treatment algorithm once heart block has been detected. 30 There is also no evidence-based directed management proven to reduce the progression to complete heart block once first-degree heart block has been detected. Following the diagnosis of NLS, the maternal use of HCQ seems to have a significant role in reducing the rate of recurrence of NLS in subsequent pregnancies. 28 Prednisone and other steroids frequently used in medical practice to inhibit inflammation are inactivated by the placental enzyme 11beta-hydroxysteroid dehydrogenase type 2 (11beta-HSD2). This results in the loss of efficacy of these steroids within the fetus, and therefore they are ineffective in the treatment of NLS and related heart block. 31 Fluorinated glucocorticoids are not inactivated by placental 11beta-HSD2 and remain therapeutic after crossing the placenta into the fetal system. The current data suggests that treating the fetus via transplacental transfer of maternal fluorinated glucocorticoids is effective and beneficial to the fetus in cases of NLS. 32 Although there are no prospective clinical trials, fluorinated steroids such as dexamethasone are commonly used to prevent CHB 27 and recent evidence including a systematic review and meta-analysis has shown it has benefits in the prevention of progression to complete heart block. Once NLS-related heart block is diagnosed the mainstay of treatment for most rheumatologists is dexamethasone 4–8 mg daily which is the most common of the fluorinated glucocorticoids used. 27 However, there is little or no evidence that they have any benefits for the management or regression of complete heart block once it is established. 30
As NLS is caused by the fetal transfer of maternal antibodies, the removal of the maternal antibodies with intravenous immunoglobulin (IVIG) does have biological plausibility. Due to the poor level of evidence showing benefits, 33 rheumatologists tend to offer IVIG only slightly more frequently than offering no therapy at all. 27 In some specialist centers, various protocols are used including triple therapy with dexamethasone, salbutamol, and IVIG. 34 In patients at high risk and with early signs of NLS-related cardiac disease, early intervention with IVIG and HCQ has been shown in some studies to prevent the development of heart block. 4 The effectiveness of IVIG may depend on dosing and timing of administration. Although well tolerated several older studies were terminated due to poor clinical response. 35 In cases of clinically significant NLS, a multidisciplinary approach is essential. Given the rarity of this condition, a specialist center should be involved in the management of these patients, as despite a combination of therapies, a number of children will need pacemakers. 34 A practical guideline for the screening and management of NLS that included the use of IVIG was published in 2021. 4 In addition, the European Alliance of Associations for Rheumatology also published guidelines on the care of patients with Sjogren's syndrome that included the management of women of reproductive age and the use of fluorinated steroids, IVIG and plasma exchanges in the management of heart block. 36
Limitations
At present there is a dearth of clinical trial evidence in the treatment of NLS, and this may be related to the difficulty in conducting clinical trials in pregnant individuals and neonates. Though there is currently increased interest in IVIG in NLS, there is very little evidence showing benefit. Though we attempted to include articles from around the world, regional publication biases limited our ability to do this.
Conclusion
NLS is a disease of the fetus caused by maternal autoantibodies that are transferred via the placenta from mother to child. Preconception evaluation of patients at risk for NLS is one of the most important steps in optimizing pregnancy outcomes. At risk individuals should be screened for NLS antibodies, and include those with SARD, FDR of those with SARD, those with preclinical autoimmunity, and asymptomatic SARD patients. Early input from a rheumatologist is important to ensure that patients with SARD are in clinical remission prior to conception. Rheumatologists also confirm that the patients are on pregnancy and lactation safe medications prior to conception, lowering the risk of teratogenesis from agents used in the treatment of many SARD.
Collaborative management facilitates decisions on screening, and treatment strategies for NLS including the use of HCQ, Dexamethasone, and IVIG, especially for those at risk of more serious complications including cardiac NLS.
Footnotes
Acknowledgement
The authors acknowledge the support of Dr Carl S. Henderson (literature review).
Author contributions
Adegbenga Bankole: performed the literature review, main writer and editor. Jane Nwaonu: performed the literature review and editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
