Abstract
Numerous studies have demonstrated that anesthetics’ exposure to neonates imposes toxicity on the developing brain but the underlying mechanisms need to be further elucidated. Our present study aimed to explore the role of small conductance Ca2+-activated potassium channel type2 in memory and learning dysfunction caused by exposing neonates to sevoflurane. Postnatal day 7 Sprague-Dawley rats and hemagglutinin-tagged small conductance Ca2+-activated potassium channel type2 channel transfected COS-7 cells were exposed to sevoflurane and the trafficking of small conductance Ca2+-activated potassium channel type2 channels was analyzed; furthermore, memory and learning ability was analyzed by the Morris water maze test on postnatal day30–35 (juvenile period). Our results showed that sevoflurane exposure inhibited small conductance Ca2+-activated potassium channel type2 channel endocytosis in both hippocampi of postnatal day 7 rats and hemagglutinin-tagged small conductance Ca2+-activated potassium channel type2 channel transfected COS-7 cells and the memory and learning ability was impaired in the juvenile period after sevoflurane exposure to neonatal rats. Herein, our results demonstrated that exposing neonates to sevoflurane caused memory and learning impairment via dysregulating small conductance Ca2+-activated potassium channel type2 channels endocytosis.
Introduction
A large number of animal studies have demonstrated neurocognitive impairment effects on the developing brain and behavior in the short and long term after exposure to anesthetics.1,2 Our previous studies have shown neurotoxic effects of sevoflurane on the development of brains in neonatal rats; these neurotoxic effects may cause neurocognitive behavioral deficits later in life,3,4 and we have further shown that sevoflurane exerts its impairment effects by increasing the surface expression of small conductance Ca2+-activated potassium channel type2 (SK2) and increased synaptic recruitment of GluA2-lacking α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, which intensifies stronger inward rectification. 4
The SK channel family including SK1, SK2, and SK3 is expressed in several tissues and prominently throughout the adult central nervous system; however, SK2 channels have been the most studied. 5 The results show that SK2 channels regulate synaptic responses, plasticity, and hippocampus-dependent memory encoding. In addition, SK2 channels contribute to the expression and maintenance of long-term potentiation (LTP). 6 SK2 is expressed throughout cornu ammonis 1 (CA1) pyramidal neurons and SK2 overexpression diminishes synaptic plasticity and SK2-containing channels account for the physiological roles of SK channels in CA1 pyramidal neurons.7,8 Our previous study also verified that the increased expression of SK2 in the neurons of the CA1 area of the hippocampus caused an elevation in the threshold for LTP induction, facilitation of long-term depression (LTD) induction, and cognitive dysfunction. 4 However, the alteration of SK2 trafficking after sevoflurane exposure in the neurons should be further explored and verified in vivo and in vitro.
This study was designed to explore the effect of sevoflurane on the alteration of SK2 trafficking in both hippocampus of postnatal day 7 (PND7) rats and hemagglutinin (HA)-tagged SK2 channel transfected COS-7 cells and the role of SK2 on learning and memory function.
Methods
Animals
PND7 male Sprague-Dawley (SD) rats (n = 30) from the same natural litters, all of which were provided by the Institution of Animal Center of Guizhou University, were divided into various experimental groups randomly. All rats were caged in plastic cages with soft bedding under a standard 12 h light/dark cycle with free access to food and water.
Sevoflurane exposure
PND7 rats were separated from their mother and placed in a closed acrylic chamber and exposed to sevoflurane at 2% for 2 h in continuous 30% oxygen/air mixture, with the control group exposed to 30% oxygen/air mixture for 2 h. 9 The total gas flow was administered by calibrated agent-specific vaporizer; sevoflurane concentration was monitored in real-time feedback (PM8050; Dräger). The health of PND7 rats was assessed by the rhythm of breath (slow, but smooth), skin color (pink), and fast emergence after anesthesia. After gas exposure, pups were recovered and sent back to their mother. Rats were sacrificed for the following experiments at the age of 30–35 days.
Surface biotinylation of hippocampal slice preparation
At PND30–35, rats were sacrificed under anesthesia with pentobarbital sodium (50 mg/kg, i. p.) and the hippocampus was removed and 350 μm thick slices were cut transversely using a Vibratome (VT1200S, Leica Inc.) in ice-cold artificial cerebrospinal fluid (ASCF) buffer; then, the slices were incubated in ice-cold ACSF containing 100 mM NHS–SS–biotin (succinimidyl-2-(biotinamido)-ethyl-1,3-dithiopropionate) (Sigma) chamber for 45 min, and the chamber was placed onto a mixture of ice and cold water. 10 The slices were transferred to another well and washed twice with the ASCF buffer containing 1 mM Lysine (Sigma) to block reactive biotinylation.
Surface expression experiment
Non-biotinylated and biotinylated slices, obtained as described above, were homogenized in lysis buffer containing 0.5% Triton X-100, 1 mM ethylenediaminetetraacetic acid, 50 mM Tris, 150 mM NaCl and a protease inhibitor cocktail (Thermo Fisher Scientific). Homogenates were centrifuged at 13,000 r/min at 4 °C for 5 min and supernatants were harvested. To precipitate the biotinylated proteins from the homogenates, 40 μL Ultra-link immobilized streptavidin beads (Pierce) were added to the sample along with an extra 500 μL of cold ASCF, and the mixture was rotating overnight at 4 °C.
Protein extraction from non-biotinylated and biotinylated hippocampal slices was performed for a standard western blot. 11 Total protein concentrations were measured by a protein assay kit (Pierce Biotechnology, Inc. ). Equal amounts of total protein samples were subjected to 10% polyacrylamide gels and transferred to nitrocellulose membranes. The membranes were blocked with 5% milk solution at room temperature for 1 h and immunoblotted overnight at 4 °C with primary antibodies (rabbit anti-SK2 (1:1000, Millipore)) and rabbit anti-Cadherin (1:1000, Cell Signaling Technology) or rabbit anti-glyceraldehyde-3-phosphate dehydrogenase (anti-GAPDH) (1:2000, Abcam); then the secondary antibody donkey α-rabbit-horseradish peroxidase (HRP) (1:3000) (ECL NA 9340, Amersham Pharmacia Biotech) was incubated for 1 h at room temperature. The corresponding bands were developed using chemiluminescent reagents. Band density was measured by ImageJ.
Immunofluorescence assay
The PND30–35 rats were deeply anesthetized with sevoflurane and transcardially perfused with phosphate buffered saline (PBS) (pH 7.4) followed by 4% formaldehyde (pH 7.4). The hippocampus was collected and postfixed with the same fixative for 24 h at 4 °C. The hippocampus was cut into a thickness of 30 μm coronal sections on a freezing microtome. The sections were blocked in 3% bovine serum albumin (BSA) and 0.3% Triton X-100 (Sigma-Aldrich) for 1 h at room temperature followed by incubation with primary antibodies as required (mouse anti-SK2 (1:1000 Millipore,); rat anti-NeuN (1: 1000, Abcam)) in 1% BSA overnight at 4 °C. After the sections were washed, they were incubated for 1 h with secondary antibodies (Alexa Fluor 488 goat anti-mouse IgG, 1:500; Alexa Fluor 594 goat anti-rabbit IgG, 1:500; both from Cell Signaling Technology) and 4′, 6-diamidino-2-phenylindole (DAPI) solution for 10 min at 37 °C. Fluorescence was detected using a confocal laser microscope (LSM 800, Zeiss). The optical density of neuropeptide Y (NPY) was measured using ImageJ software (National Institutes of Health).
Morris water maze test
Spatial memory and learning were tested by the Morris water maze. 12 The Morris water maze consisted of a circular pool with 50 cm height and with a diameter of 96 cm filled water (22°C) to 40 cm deep and painted white. The cue flags were placed outside the maze, and its position remained unchanged during the whole test. The rats were given 90 s free swimming without the platform that is a pre-training session, which was performed 1 day before the training session. In the hidden platform test, the rats were required to find the location of a hidden platform submerged 1 cm below the surface of the water and underwent four trials per day for 5 consecutive days. The rats were released from different positions and allowed to swim for 90 s during each trial. The rats were allowed to stay on the platform for 15 s after reaching it. If a rat failed to find the platform within 90 s, it was guided to climb on the platform. On the sixth probe test, the platform was removed and the rats were allowed to search the maze for 45 s, whereas the time crossings in the target quadrant and the time crossing in the other quadrants were recorded. The swimming path of the rats was analyzed using EthoVision XT software (Noldus Information Technology).
SK2 channel constructs and generation of COS-7 expression SK channel cells
An extracellular HA-tagged human SK2 (GenBank™ accession number NM_021614.2) channel DNA was subcloned into the mammalian plasmid vector pcDNA3 (Invitrogen). COS-7 cells (Life Technologies) were transfected with HA-tagged SK2 channel constructs and the pRFP-C-RS vector (Origene) encoding for red fluorescence protein (RFP). Control cells were transfected with the RFP vector only. As much as 24 h after transfection, cells treated with the resistant RFP-positive cells were isolated by fluorescence-activated cell sorting (FACS). The expression of HA-tagged SK2 channel in single-cell clones was analyzed by western blotting.
Cell culture, exposure to sevoflurane, and experimental protocol
HA-tagged SK2 channel transfected COS-7 cells were cultured in Dulbecco's Modified Eagle Medium with 10% fetal bovine serum (Invitrogen) and 1% Pen/Strep (PAA) on glass-bottom Petri dishes (Wellco). HA-tagged SK2 channel transfected COS-7 cells were exposed to either sevoflurane/air mixture or air using an airtight glass chamber. The chamber atmosphere was kept saturated with water at 37°C continuously. In the control group, gas was prepared using a gas mixing unit by directing a 95% air/5% CO2 mixture at 3 L/min through calibrated vaporizers (Dräger) that were placed at the entrance of the chamber; in the exposure group, gas was prepared using a gas mixing unit by directing 95% air/5% CO2 mixture with 2% sevoflurane at 3 L/min through calibrated vaporizers, and sevoflurane concentration was monitored at the chamber exit port using a monitor (PM8050; Dräger). The cells were exposed to sevoflurane for 2 h; then cells in the dishes were taken out for antibody uptake assay and biotinylation internalization assay.
Antibody uptake assay
Antibody uptake assay with HA-tagged SK2 channel constructs transfected into COS-7 cells was performed after sevoflurane treatment. Cells were blocked with 1 × PBS containing 5% fetal calf serum (FCS) (Life Technologies) for 30 min at room temperature. Channels at the cell surface were labeled with goat anti-HA antibody (Abcam; 1:1000) at 4 °C for 60 min, then the cells were washed with PBS for three times to remove unattached antibodies. Subsequently, the labeled channels on the cell surface were allowed to internalize at 37 °C for 30 min; then, the cells were fixed with 4% paraformaldehyde for 10 min; the channels on the surface were labeled with Alexa Fluor 488-conjugated rabbit anti-goat secondary antibody (Abcam; 1:500). Following cell permeabilization with 0.1% Triton X-100 min at room temperature for 5 min, internalized channels were detected with Alexa Fluor 594-conjugated rabbit anti-goat secondary antibody (Abcam; 1:500). The coverslips were mounted on glass slides after three times of PBS washing. The labeled cells were photographed using a Leica fluorescence microscope (Leica). The results were analyzed with Image-Pro Plus 4.5 (Media Cybernetics).
Biotinylation internalization assay and western blotting
Biotinylation internalization assay with HA-tagged SK2 channel constructs transfected into COS-7 cells was performed after sevoflurane treatment at different time points. Biotinylation assay for internalization was performed as described previously. 11 COS-7 cells were biotinylated using the membrane impermeable sulpho-NHS-biotin (Pierce) at 4 °C for 20 min and the reaction terminated with 50 mM glycine. After washing, cells were incubated for 30 min at 37 °C to allow internalization. The remaining surface biotin was removed with glutathione and the cells were lysed for western blotting.
The expression status of proteins was determined by Western blotting. Detection of target proteins is performed using rat anti-HA antibodies (Abcam; 1:1000) and HRP-conjugated goat anti-rat secondary antibody (Abcam; 1:1000). The bands were detected using Odyssey Imaging Systems (Biotech).
Statistical analyses
Results were presented as the mean ± SEM. Results were analyzed by repeated measure (ANOVA) test and Student's t-test Differences with P values of <0.05 were considered significant.
Results
Neonatal sevoflurane exposure inhibited the endocytosis of SK2 channels in the hippocampus
The results of the Western blot assay showed that there was no difference in the total SK2 protein expression found in hippocampal lysates between sevoflurane and control rats (Figure 1(A)). However, surface SK2 protein expression increased significantly in the hippocampus of juvenile sevoflurane rats compared to that of juvenile control rats (Figure 1(B)). As a negative control, both total and cell surface Cadherin in the hippocampus did not differ between control and sevoflurane rats.

Neonatal sevoflurane exposure inhibited the endocytosis of SK2 channels in the hippocampus. (A) Western blot bands of biotinylated and total SK2s and N-Cadherin in the hippocampus from sevoflurane and control rats. (B) Statistical results show the ratios of biotinylated surface SK2 protein vs. total SK2 protein levels. Data are shown as means ± SEM. n = 6 in each group. Statistical significance was assessed by Student's t-test *P < 0.05. (C) Immunofluorescence images of SK2s distribution in the CA1 area of hippocampus from sevoflurane and control rats. (D) Statistical results show mean optical density (MOD) of SK2 immunoreactivity in the sevoflurane increased significantly compared to the control group. Statistical significance was assessed by Student's t-test *P < 0.05.
Next, the distribution of SK2s was examined in juvenile rat hippocampus using an immunofluorescence assay (Figure 1(C)). In the hippocampal CA1 region of juvenile rats, the mean optical density (MOD) of SK2 immunoreactivity in the sevoflurane increased significantly compared to the control group (Figure 1(D)).
These results indicated that neonatal exposure inhibited endocytosis of SK2s in juvenile rat hippocampus.
Sevoflurane exposure caused the impairment of memory and learning ability
The memory and learning ability of the juvenile rat was impaired after sevoflurane exposure at the period of PND7. As shown in Figure 2(A), sevoflurane rats spent more time than control rats to locate the hidden platform.

Effects of sevoflurane on spatial memory and learning in neonatal rats in Morris water maze tests. (A) Mean escape latency of reaching the submerged platform in the training period. (B) Crossing platform area times in target quadrant and other quadrants 24 h after the last training trial. n = 12–15 rats in each group. Data are shown as means ± SEM. Statistical significance in panel A was assessed by repeated measure ANOVA test Statistical significance in panels C and D were assessed by Student's t-test *P < 0.05; **P < 0.01; ***P < 0.001.
To assess spatial memory, probe trials were performed. The results revealed that sevoflurane rats spent an equal amount of time in the target quadrant and other quadrants, while control rats spent significantly increased time in the target quadrant than any other quadrant (Figure 2(B)).
These results revealed that sevoflurane exposure caused the impairment of memory and learning ability.
Sevoflurane treatment inhibited HA-tagged SK2 channel endocytosis in COS-7 cells
To distinguish cell surface and internalized HA-tagged SK2 channel protein, the channels that remained on the cell surface were labeled with Alexa 488-conjugated secondary antibody (green); the channels internalized were labeled with Alexa 594-conjugated secondary antibody (red). As shown in (Figure 3(A)), sevoflurane exposure resulted in a decrease in SK2 channel internalization. Quantifying the ratio of fluorescence intensity internalized channels demonstrated that the number of endocytosed HA-tagged SK2 channels decreased under sevoflurane exposure conditions (Figure 3(B)). These data provide direct evidence that sevoflurane could inhibit HA-tagged SK2 channel endocytosis in COS-7 cells.
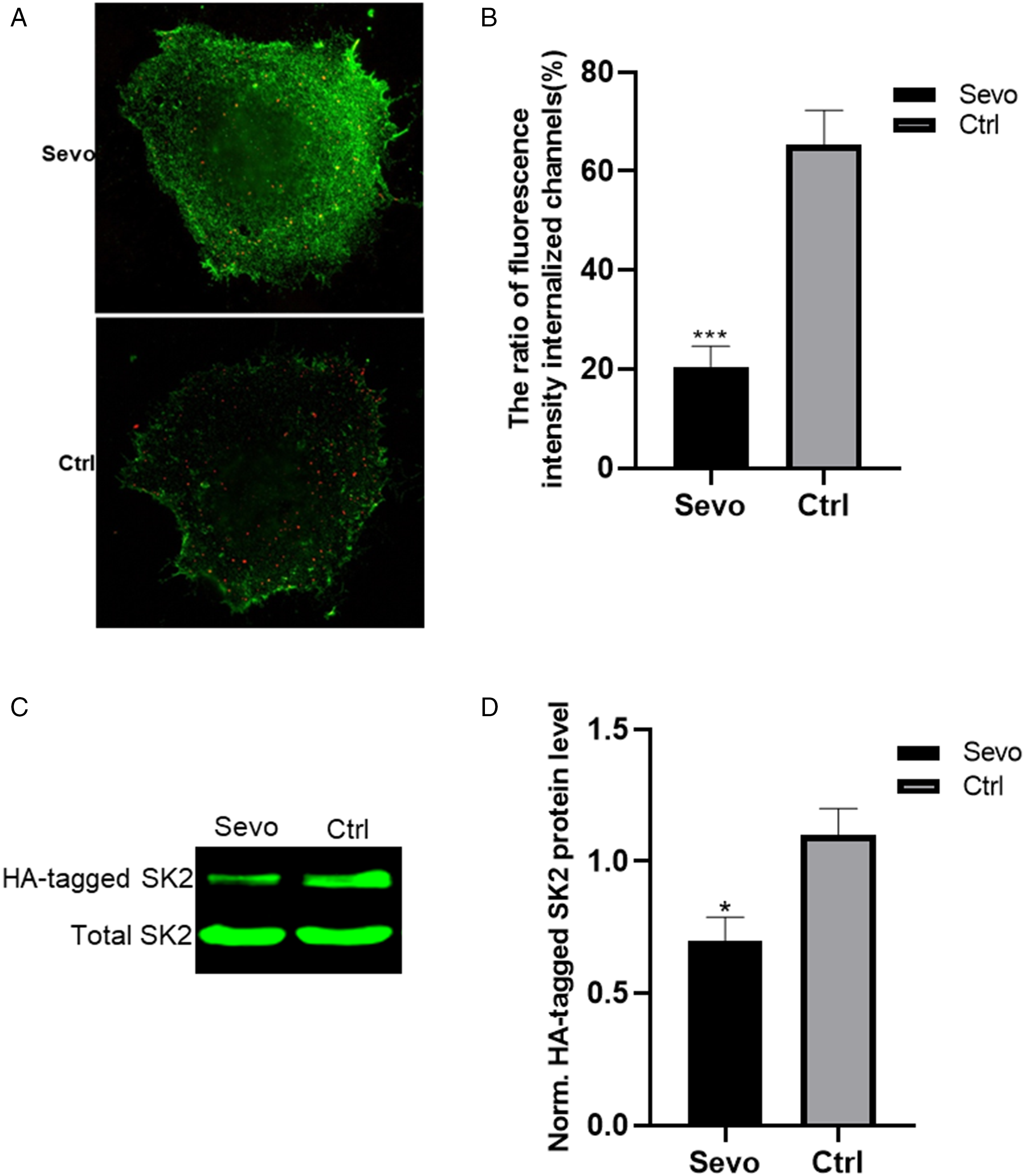
Sevoflurane treatment inhibited HA-tagged SK2 channel endocytosis in COS-7 cells. SK2 trafficking was measured by antibody uptake assay, biotinylation internalization assay and western blot after sevoflurane treatment. (A) Representative internalized SK2 channels (red) after sevoflurane treatment. (B) The ratio of fluorescence intensity internalized channels in sevoflurane vs. control group. (C) Western blot bands of internalization of HA-tagged SK2 channels in COS-7 cells. (D) Statistical results show the ratios of HA-tagged SK2 channels in the sevoflurane vs. control group. Statistical significance was assessed by Student's t-test *P < 0.05; ***P < 0.001.
To confirm the above conclusion, the internalized HA-tagged SK2 channel protein was measured using a biotinylation assay, and subsequent western blotting. The advantage of using this quantitative assay versus the fluorescent approach was that we could determine whether internalization was inhibited. As shown in Figure 3(C), compared to control groups, the internalization of HA-tagged SK2 channel was significantly reduced in the sevoflurane treatment group. These results are consistent with the data obtained by the antibody uptake assay.
Discussion
Our study indicated that a single exposure of sevoflurane (2% for 2 h) remarkably inhibited the endocytosis of SK2 channels in the CA1 area of the hippocampus; the newborn rat exposed to sevoflurane also showed memory and learning deficits in the juvenile period. These findings were consistent with our previous study. 4 It was demonstrated from in vitro experiments that sevoflurane treatment inhibited HA-tagged SK2 channel endocytosis in COS-7 cells. Taken together, the results suggest that endocytosis dysfunction caused by sevoflurane plays a vital role in sevoflurane-induced memory and learning dysfunction in neonatal rats.
In the hippocampus, SK channels modulate synaptic plasticity, neurotransmission, and learning.8,13,14 SK1, SK2, and SK3 are three subunits of SK channels that are expressed in the hippocampus.7,15 However, a lot of studies have focused on SK2 channels. The studies demonstrated that SK2 channels regulate hippocampus-dependent plasticity, synaptic response, and memory encoding. In addition, SK2 channels participate in metaplasticity that can change the threshold of LTP or LTD minutes to days later.4,16 Ca2+ influx activates SK2 channels that cause membrane hyperpolarization, which can be rescued by apamin, a selective blocker for SK2. 17 SK2 channels are functionally coupled with Ca2+-permeable N-Methyl-D-aspartic acid or N-Methyl-D-aspartate (NMDA) receptors in a series of events: NMDA receptors’ opening is followed by Ca2+ influx, activation of SK2s, membrane hyperpolarization, and feedback blockade of NMDA receptors. 18 The previous study indicates that SK2 is negatively associated with LTP induction: the transgenic mice with overexpressed SK2s show impairment of hippocampal-dependent learning and elevated threshold of LTP induction. 8 In contrast, blocking SK2s with apamin facilitates memory encoding and LTP induction. 19 Therefore, SK2s play important roles in the long-lasting effects of neonatal anesthetic exposure.
Our study first demonstrated that sevoflurane could regulate SK2 channel traffic by inhibiting endocytosis in vivo and in vitro; so, we could predict that more SK2 channels remained on the cell surface to conduct potassium ions down their electrochemical gradient; the consequence is that resting membrane potential is hyperpolarized and the excitability of the cell is inhibited. Our previous study also hypothesized that plasticity impairment caused by sevoflurane exposure was due to the high surface expression of SK2s. 4 The SK2 channel is functionally important in many cell types including neuronal and cardiac cells by involving in the after hyperpolarizing potential (AHP). 19 Enhanced intrinsic neuronal excitability of hippocampal pyramidal neurons via reductions in the postburst AHP has been hypothesized to be a biomarker of successful learning.20,21 The channel activity can be regulated in two ways, which is either by changing the open probability or by changing the number of channels in the cell membrane. The cell surface channel expression, in turn, is regulated by the forward trafficking of the channel or its endocytosis from the membrane. 22 Our study demonstrated that sevoflurane inhibited the endocytosis of SK2s, which increased the number of channels in the cell membrane.
There were two main limitations in our study. First, our study revealed that neonatal exposure to sevoflurane caused memory and learning impairment by inhibiting SK2 channels endocytosis. However, we lack transgenic mice with knockout SK2s to verify our result. Second, the exact mechanisms by which sevoflurane inhibited SK2 channels were not investigated in the current study. Further explorations of the molecular mechanisms by which sevoflurane inhibited SK2 channels endocytosis are needed.
In summary, our study predicted that neonatal exposure to sevoflurane caused memory and learning impairment by inhibiting SK2 channel endocytosis.
Footnotes
Availability of data and materials
All data and materials are available without restriction. Researchers can obtain data by contacting the corresponding authors.
Authors’ contribution
XY contributed to the conception, and design of the study. WK, YG, and YZ contributed to perform the experiments, data acquisition, and interpretation. XY drafted the manuscript. All authors contributed to the interpretation of the findings, and reviewed, edited, and approved the final manuscript.
Ethics approval
All animal care and experimental protocol were performed in compliance with the guidelines of animal ethics of Guizhou Provincial People's Hospital (approval no. 2020160).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China, grant number 82060239; GPPHNSFC-2020-9; Guizhou Provincial Natural Science Foundation of China, grant number Qiankehejichu-ZK [2021] Zhongdian006 and Guizhou Provincial Highlevel creative talents cultivation plan: Thousand plan, grant number GZSYQCC [2016]001.
