Abstract
In the industrial sector, vegetable residual materials have received attention in the production of bio-colorant for textile dyeing. The current research endeavor is centered on investigating the possibility of using sugar beet leaves as a natural source of dye for the purpose of dyeing cotton fabrics. Different extraction methods were utilized to isolate the bio-colorant present in sugar beet residual material, and the most favorable colorant yield was obtained using a 5% methanolic KOH solution. For optimal dyeing results, the cotton fabric performed dyeing for a duration of 45 min at a temperature of 60 °C, using a salt solution concentration of 6 g/100 mL and 50 mL of the extracted dye solution. Characterization of dye using Fourier transform infrared spectroscopy analysis confirmed the presence of quercetin in the leaf extract. For the creation of a range of color variations, mordants that were chemical in nature, such as tannic acid, iron sulfate, potassium dichromate, and copper sulfate, as well as mordants that were bio-based, such as onion peel, pomegranate peel, henna, golden shower bark, and turmeric, were employed in harmony. In comparison, the utilization of bio-mordants resulted in darker shades that exhibited enhanced color intensity and superior color fastness properties with the value of 4–5 for wash, 4 for wet rubbing, 4–5 for dry rubbing, and 4–5 for light. The findings of this study hold significant value in terms of ecofriendly waste management and contribute to advancements in the industrial sector by utilizing waste residual materials as a natural source of colorants.
Introduction
Recently, the textile industry has become a major source of environmental pollution on a global scale. Textile effluent released thousands of tons of toxic substances, including synthetic dyes, which disrupt the ecological balance as well as pose severe threats to public health. 1 To overcome this disquieting situation, natural dyes represent an attractive ecofriendly alternative option. 2 Most of the research has been conducted on different plant parts, algae, animals, lichens, and microbes. 3 Since the beginning of civilization, people have used natural resources; nevertheless, the usage of natural colors substantially decreased with the discovery of synthetic dyes in 1856. Since then, only artisans, hobbyists, and owners of small businesses in the cottage-style industry sector have used natural dyeing techniques.4,5 With the goal of advancing natural dyeing from small-scale, so-called “handicraft” or handmade items to industrial size, governments of several nations have recently begun to push textile manufacturers to employ ecologically friendly materials. Because of this, it is anticipated that the global market for natural dyes would develop and reach $5 billion in sales by 2024, with an average yearly growth rate of 11% from 2018 to 2025. 6
As a result, in the textile dyeing and printing industries, natural dyes have attained much greater popularity. 7 To fulfill the increasing demand for bio-colorants, exploration of pigment crops has become a research hotspot. 8 Different industries are appreciating the need to utilize the natural dyes in various domains of life, including food, pharmaceuticals, cosmetics, and dye-sensitized solar cells, as a result of their remarkable antimicrobial, antifungal, antioxidant, and deodorant properties. 9 For example, Ahmed et al. 10 modified dyed silk fabric using silver and palladium metal precursors for improvement of the functional properties where red peanuts’ extract was used as a natural dye. Emam et al. 11 functionalized the cotton fabric using natural dyes from red peanuts with the immobilization of silver and palladium particles. Therefore, we have also extracted natural color from mahogany wood waste, 12 Coral Jasmine flower, 13 gardenia yellow, 14 tulsi leaves, 15 wheat husk agro-wastage, 16 Butea monosperma plants, 17 Alkanna tinctoria roots, 18 Rheum emodi plants, 19 black rice, 20 and neem leaf extractions. 21 Because their effluents and residues become a part of the mixture and enrich the soil when they are combined with agricultural land, these bio-colorants do not do any damage to the environment or cause problems with wastewater sewage. 22 In addition to the studies conducted, it is important to acknowledge that natural dyes have certain disadvantages. These include their non-reproducibility, higher cost (especially when aiming for vibrant and consistent colors), lower color yield, and relatively poor color fastness when derived from plant materials. 23 As a solution to the issue, we discovered a vegetable plant known as sugar beet (Beta vulgaris L.), which has a long history of usage in the production of sugar crop, while searching for potential natural extracts.
Sugar beet (B. vulgaris L.), the 10th most important vegetable and important sugar crop, belongs to the Chenopodiaceae family. 24 Sugar beet contains a high level of sucrose and is commonly used for the production of sugar. 25 At the start of the 20th century, it was cultivated on a larger scale in the USA for sugar production, and now it fulfills 20% of the world’s sugar demand. 26 Refat et al. 27 reported that Egypt is a big producer of sugar beet in terms of higher sugar production in the world. Furthermore, it is well known for its value-added products such as sugar, fodder, human nutrition, biofuel, pharmaceuticals, food coloring, and plastics. 28 Literature studies revealed that annually, 14 million tons of sugar beet pulp is produced from sugar industries in European countries. It is a rich source of 75–80% polysaccharides, 22–24% cellulose, 30% hemicelluloses, and 25% pectin, making it an economically important fodder. 29 Leaves containing a variety of flavonoids, including aglycone, quercetin, quercetin 3-O-glucoside, galactose, and gallic acid, have been traditionally used for their potential medicinal properties in treating various diseases. 30 Phytochemical studies of sugar beet leaves have revealed the presence of flavonoids, primarily quercetin, which is responsible for their coloration.31,32 The different parts of sugar beet leaves including the vein, lamina, midrib, petiole, and leaf base produce red color in the extracted dye solution. 33 Furthermore, as a consequence, a number of researchers have utilized sugar beets as a natural dye source for the purpose of coloring textile products. For example, Benli et al. 34 investigated the dyeing of wool fabrics with a bio-based red beetroot dye derived from B. vulgaris L. Rotich et al. 35 undertook an extensive investigation, involving the analysis of a natural dye extract derived from B. vulgaris peels and pomaces, surface examination of optimally dyed and mordanted cellulosic fabric through Fourier transform infrared spectroscopy (FTIR) spectroscopy, and assessment of the color fastness properties of the dyed textiles. Popescu et al. 36 focused on achieving intense red coloring of wool through the utilization of a sustainable extract derived from beetroot peels, all accomplished without the use of metallic mordants. However, no one has used different media to find out the optimized condition of dye extraction and application of cotton fabric coloration with statistical analysis. Although cotton fabric was dyed using the different parts of B. vulgaris plants such as beetroot,34,37 whatever the process and chemicals they used, it was different from our current study. As a result, the current research focused on the natural dye extraction from sugar beet leaves using different extraction media, which will be applied for the coloration of cotton fabric.
To date, little attention has been given to utilizing agricultural residual material for textile dyeing. For example, Mohtashim et al. 38 employed banana bio-resource waste for dyeing cotton fabric. The coffee extraction have also been used for the dyeing of cotton fabric. 39 Therefore, the current study makes an attempt to isolate the colorant potential of sugar beet leaves. In addition, to provide an industrial application of residual material for the textile industry by converting non-valuable material into a value-added product, especially in developing countries like Pakistan, the utilization of cost-effective natural dye creates employment opportunities for farmers as well as uplifts the industrial progress.
Materials and methods
Materials
The leaves of the sugar beet were collected from the vegetable fields that are located at the Ayub Agricultural Research Institute in Faisalabad, Pakistan. The freshly obtained leaves were cleaned and dried properly before being pulverized into a powder. This powder was then utilized for the dyeing procedure on cotton fabric, as shown in Figure 1. The cotton fabric with a specification of yarn count 30/1 Ne, weight of 160 g/m2 (GSM), and single jersey knit fabric, used in the study, was purchased from the local market in Faisalabad, Pakistan. Bio-mordants such as henna, onion peel, pomegranate peel, turmeric, and golden shower bark were acquired from the herbal market in Faisalabad, Pakistan. All chemicals used in the study, including potassium hydroxide (CAS number: 1310-58-3), sodium hydroxide (CAS number: 1310-73-2), ethanol (CAS number: 64-17-5), methanol (CAS number: 67-56-1), hydrochloric acid (HCl, CAS number: 7647-01-0), iron sulfate (CAS number: 7782-63-0), tannic acid (TA; CAS number: 1401-55-4), potassium dichromate (CAS number: 7778-50-9), copper sulfate (CAS number: 7758-98-7), and sodium chloride (CAS number: 7647-14-5), were obtained from the Paragon Dyes and Chemicals Ltd, Faisalabad, Pakistan. All the chemicals were in commercial scale and used without any purifications.

Conversion of sugar beet leaves into extracted dye solution.
Methodology
Extraction of natural dye
Different extraction media were used to extract natural dye from sugar beet leaves. The media were aqueous (tape water), alkaline (NaOH, KOH (1–10%), organic (methanol, ethanol (20%, 40%, 60%, 80%, and 100%), methanolic KOH (1–5%), and acidic (acidified methanol, acidified ethanol, acid (HCl) (1–5%)). To extract the dye, a small quantity (4.0 g) of the powdered sugar beet leaves was boiled in the previously mentioned extraction medium for a duration of 40 min (Figure 1). Subsequently, the dye filtrates obtained from the extraction process were utilized for dyeing cotton fabric (Figure 2). The Economic Botany Lab, which is part of the Department of Botany at the Government College University in Faisalabad, Pakistan, was the location where the process of dyeing cotton was carried out.
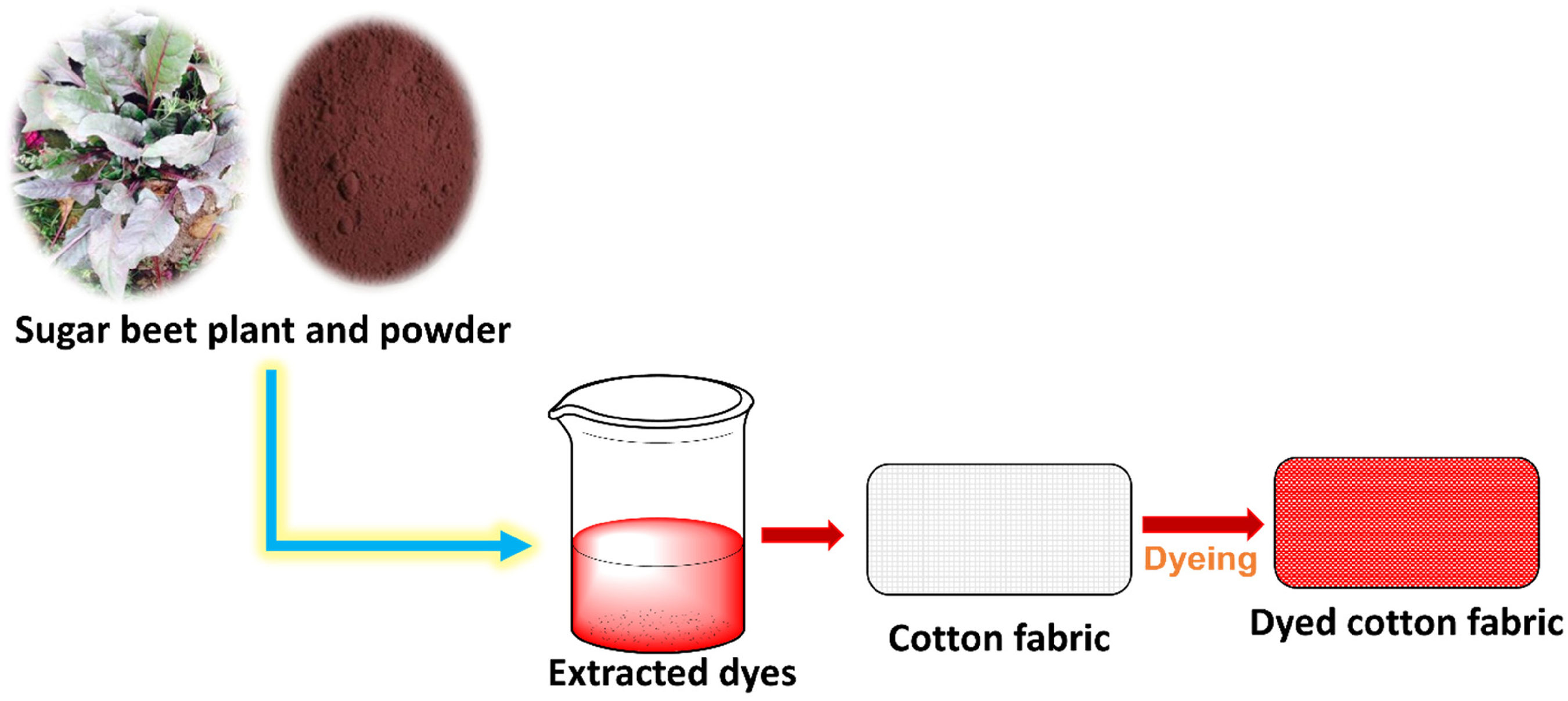
Demonstration of the dyeing process on cotton fabric using a natural dye obtained from sugar beet leaves.
Optimization of dyeing parameters
Varying dyeing conditions including time duration (15, 25, 35, 45, 55, 65, 75, and 85 min), heating level (30, 40, 50, 60, 70, 80, and 90 °C), salt concentration (1, 2, 4, 6, 8, 10 g/100 mL), and material-to-liquor ratio (30, 40, 50, 60, 70, 80, and 90 mL) were optimized. 40
Mordanting
To produce variety of shades of dyed samples, bio- and chemical mordanting were performed under optimized dyeing conditions. Different concentrations ranging from 1 to 10% of extracts obtained from henna (Lawsonia inermis L.), onion peel (Allium cepa L.), pomegranate peel (Punica granatum L.), turmeric (Curcuma longa L.), and golden shower bark (Cassia fistula L.) were used as bio-mordants. Additionally, chemical mordants such as iron sulfate (FeSO4), TA (C76H52O46), potassium dichromate (K2Cr2O7), and copper sulfate (CuSO4) were employed.
Characterization and assessment
The color intensity of the dyed and mordanted specimens was quantified using the Spectra flash SF 600 apparatus produced by Datacolor, USA. All the samples were prepared three times, and the results were measured three times for technical replicates and independent of the experiments. This assessment was conducted at the Department of Applied Chemistry, Government College University in Faisalabad, Pakistan. The colorfastness characteristics of the mordanted and dyed specimens were evaluated employing the following ISO methodologies: ISO 105-C03 assessed washing fastness, ISO 105 X-12 evaluated rubbing fastness, and ISO 105 BO2 determined light fastness. 41 Functional groups of sugar beet leaf dye were characterized by FTIR (Bruker, model Alpha).
Statistical analysis
A one-way analysis of variance (ANOVA) was performed with a completely randomized design (CRD) to identify the statistically optimal level for attaining increased colorant yield. The analysis was carried out using the Costat software window version, following the methodology outlined in a previous report. 42
Results and discussion
Effect of extraction media
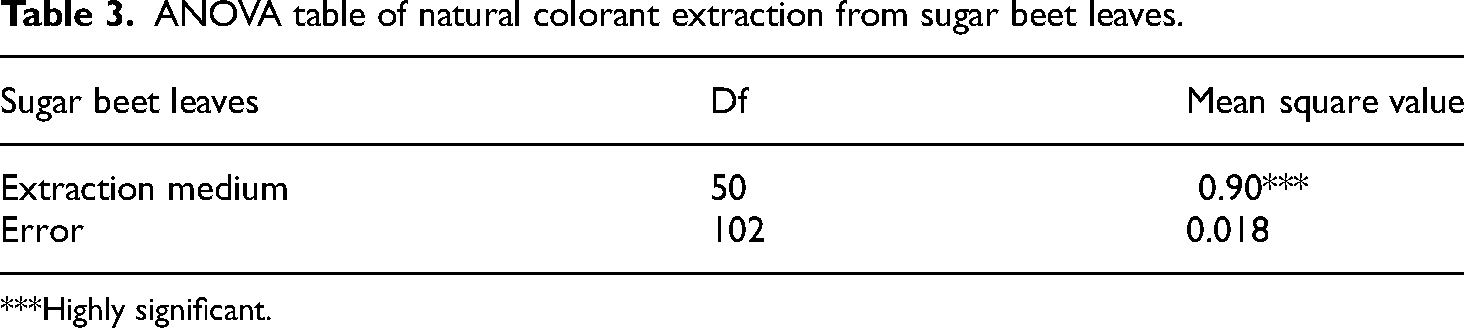
Extraction efficiency of natural dyes greatly depends on the suitable solvent. For achieving maximum colorant from sugar beet leaves, four extraction media (aqueous, alkaline, organic, and acidic) were chosen. Results in Tables 1 to 3 indicated that statistically 5% methanolic KOH gave highly significant colorant output. It was revealed that by increasing polarity of solvent, colorant extraction was increased. Previous literature studies showed that methanol was found to be an excellent solvent in isolating maximum dye molecules from sugar beet leaves. 37 Comparatively, combination of methanol and KOH approaches the maximum colorant extraction by rupturing the plant cell wall efficiently. 43
The effect of aqueous and alkaline solution for the colorant extraction media of sugar beet leaves.

The effect of organic and acidic solutions for the colorant extraction media of sugar beet leaves.

ANOVA table of natural colorant extraction from sugar beet leaves.
***Highly significant.
Dyeing conditions optimization
Effect of time duration on cotton dyeing
Color strength values mainly depend on dyeing time. 44 Dyeing time (15–85 min) was optimized to attain higher K/S value. Figure 3(a) illustrates that color strength was increased by a rapid rise in dyeing time duration, and dyeing attained equilibrium at 45 min and gave maximum color strength to the dyed sample. On the other side, color strength value declined beyond the optimal dyeing time. Moreover, short dyeing time failed the dye sorption on the fabric, while prolonged duration caused desorption of the dye molecules and results in minimum yield of K/S value to dyed fabric. 45

Effect of different dyeing parameters (a) dyeing time (min), (b) dyeing temperature (°C), (c) salt concentration, (d) material-to-liquor ratio on the color strength value of dyed fabrics using dye extract of sugar beet leaves. All the color strength data points are demonstrated as means of three replicates along with standard error (error bars represent the significant difference between the levels).
Effect of heating levels on cotton dyeing
For cotton dyeing, various heating levels (30–90 °C) were conducted to get optimum dyeing condition. Results in Figure 3(b) revealed that low heating level failed to penetrate maximum colorant molecules onto fabric upon dyeing. Therefore, initially at 30 °C, 5% methanolic KOH–dyed cotton fabric gave lower color strength value, while, in gradually rising temperature, sorption of colorant onto cotton fabric was increased up to 60 °C by increase in pore size of cotton fabric and ultimately gave higher color strength (K/S) value to dyed cotton sample. Hence, 60 °C is considered to be optimum heating level to obtain excellent result in the form of higher K/S value, while more than 60 °C heating level (excess heating) caused degradation of both colorant molecules and cotton fabric and ultimately produced minimum color strength value to dyed sample. 46
Effect of exhausting agent on cotton dyeing
The dyeing process can be considerably influenced by the concentration of salt, which affects the uptake of color. The incorporation of salt, also known as a salting-out or exhausting agent, is a prevalent technique in the process of dyeing textiles. The primary effects of this process are enhanced dye solubility, improved fiber affinity, prevention of dye aggregation, and increased interactions between dye and fiber. 47 In order to achieve a higher color strength value for the dyed samples, various salt levels ranging from 1 to 10 g were tested and optimized. The results, as depicted in Figure 3(c), demonstrated that the intensity of color in the cotton sample exhibited a significant augmentation as the salt concentration increased to 6.0 g/100 mL. It was optimum level to establish strong dye fabric interactions, which in result gave higher K/S value onto the dyed sample, while unstability was created by excess salt level and gave minimum K/S value to dyed sample. 23 This highlighted the optimal salt concentration for achieving higher color strength and dye–fabric interactions, while excessive salt levels induced instability and resulted in a diminished K/S value for the dyed sample. 48
Effect of dye extract ratio on cotton dyeing
Varying levels of dye-to-liquor ratio (1:30–1:90) were optimized. According to the results shown in Figure 3(d), the color intensity of the dyed fabric elevated as the dye-to-liquor ratio increased, reaching its peak at a ratio of 50 mL. It was economic and optimum level to achieve excellent color strength value by maximum dye sorption onto the cotton fabric, while color strength was declined beyond the optimum level due to aggregation formation of dye molecules into the dyeing bath. 49
Shade development
The utilization of mordants is a widely employed technique in natural dyeing to achieve a diverse range of color shades. 4 Salts of metals such as Fe, Al, Cu, Co, and TA act as chemical mordant, and extracts of varying plant barks, peel (mango bark, pomegranate peel, acacia bark, and henna) act as bio-mordant.22,50 Chemical mordanting results showed that among all chemical mordants, TA and FeSO4 gave best results (Figure 4(a) and (b)). The statistical data presented in Table 4 revealed that using 1% TA as a pre-mordant and 6% iron sulfate (FeSO4) as a post-mordant resulted in highly significant outcomes in terms of achieving a darker color shade on the mordanted cotton samples. Comparatively, 1% TA and 6% FeSO4 are cost-effective and ecofriendly levels in attaining maximum K/S value. The decreased reduction potential of iron plays a role in the creation of robust metal dye complexes, resulting in the production of darker shades and elevated K/S (color strength) values on the mordanted fabric. 51 Certainly, the elevated reduction potential of TA plays a pivotal role in the establishment of metal dye complexes. TA exhibits a pronounced affinity for metal ions and is capable of forming robust interactions with both the –OH groups of the dye molecule and the –OH groups present in the cotton fabric. This interaction enhances the bonding between the dye and the fabric, resulting in improved color retention and dye fastness properties. 52 Consequently, a diverse array of color shades is generated as a result of the robust hydrogen bonding between the metal salt and the –OH groups of the dye, along with those of the cotton fabric. The schematic mechanism is shown in Figure 5. Meanwhile, excess of metal salts form aggregation formation in the dyeing bath and in result fail to diffuse onto cotton fabric as well as cause unevenness on the dyed sample. 53
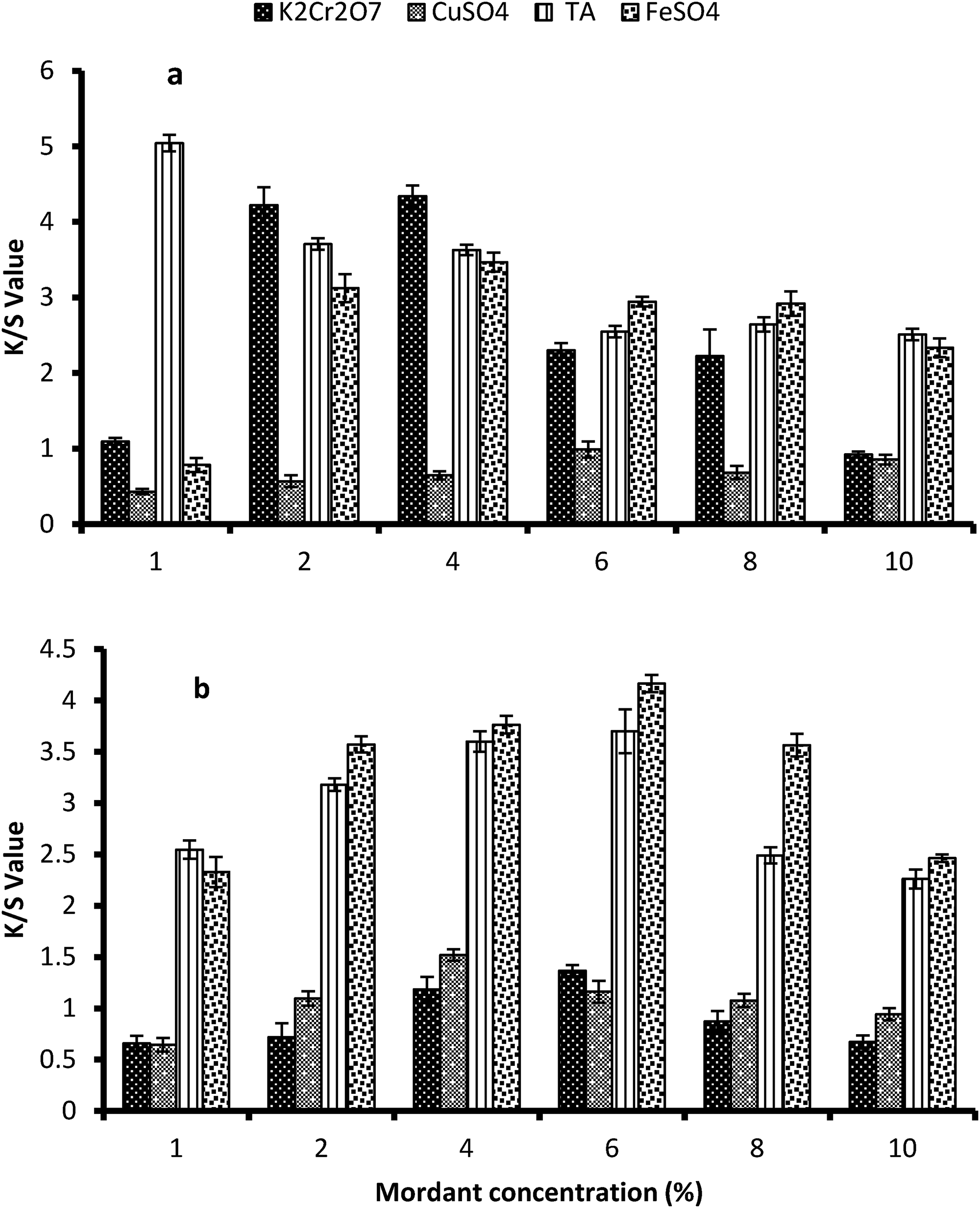
Effect of (a) chemical pre-mordant and (b) chemical post-mordant on the color strength value of dyed cotton fabrics using sugar beet leaf dye extract. All the color strength data points are demonstrated as means of three replicates along with standard error (error bars represent the significant difference between the levels).

Proposed mechanism of chemical mordants, dye, and fabric interaction.
ANOVA results for the mordanting processes involving chemical and bio-mordants on cotton fabrics with the use of sugar beet leaf dye extract.

***Highly significant.
Bio-mordanting is an environment-friendly approach to yield darker color shade onto the cotton. The results in Figure 6(a) and (b) show that bio-mordants produced varying shade with higher color strength values. The results presented in Table 4 indicate that bio-mordanting of cotton fabric yielded statistically significant outcomes. Particularly, the application of an 8% pomegranate peel extract (abundant in tannins) as a bio–pre-mordant, coupled with a 6% henna (Lawsonia) bio–post-mordant, led to the creation of a deeper hue on the cotton sample. The colorant derived from onion peel forms a strong or covalent bond with the primary colorant, quercetin, as well as with the cotton fabric during the dyeing process. Consequently, this interaction results in the production of a darker shade, specifically a brown color. These levels of bio-mordants proved to be highly significant in achieving the desired color intensity. Meanwhile, color shade was declined beyond the optimum levels. The proposed mechanism illustrated in Figure 7 suggests that bio-mordants, due to the presence of additional functional moieties, create robust bonds with the –OH groups present in both the colorant molecules and the cotton fabric. These firm bonding interactions contribute to enhanced color absorption and retention on the fabric surface, resulting in a deeper and more durable coloration. 54 In comparison, the utilization of bio-mordants demonstrated a more sustainable approach in attaining a darker shade, as indicated in Table 5.

Effect of (a) bio–pre-mordant and (b) bio–post-mordant on the color strength value of dyed cotton fabrics using sugar beet leaf dye extract. All the color strength data points are demonstrated as means of three replicates along with standard error (error bars represent the significant difference between the levels).

Proposed mechanism of bio-mordant, dye, and fabric interaction.
Color characteristics and shade of mordanted cotton fabrics dyed using sugar beet leaf dye extract.

Color fastness properties
The outcomes outlined in Table 6, related to chemically pre- and post-mordanted fabrics, and Table 7, related to bio–pre- and post-mordanted samples, exhibit favorable ratings in terms of light fastness, washing fastness, dry rubbing fastness, and wet rubbing fastness. In chemical mordanting, 1% tannic acid as pre-mordant, 6% iron sulfate post-mordanted sample, and 8% pomegranate peel bio–pre-mordanted cotton fabric as well as 6% henna bio–post-mordanted cotton sample gave excellent rating of fastness. This can be ascribed to the robust complex formation between the mordants and the colorant, along with the mordants and the fabric. These sturdy interactions diminish the leaching of the colorant from the fabric surface, resulting in enhanced color retention and durability. 2
Effect of chemical mordants on the color fastness properties of pre- and post-mordanted cotton fabrics dyed using dye extract of sugar beet leaves.

LF: light fastness; WF: washing fastness; DRF: dry rubbing fastness; WRF: wet rubbing fastness.
Effect of bio-mordants on the color fastness properties of pre- and post-mordanted cotton fabrics dyed using dye extract of sugar beet leaves.
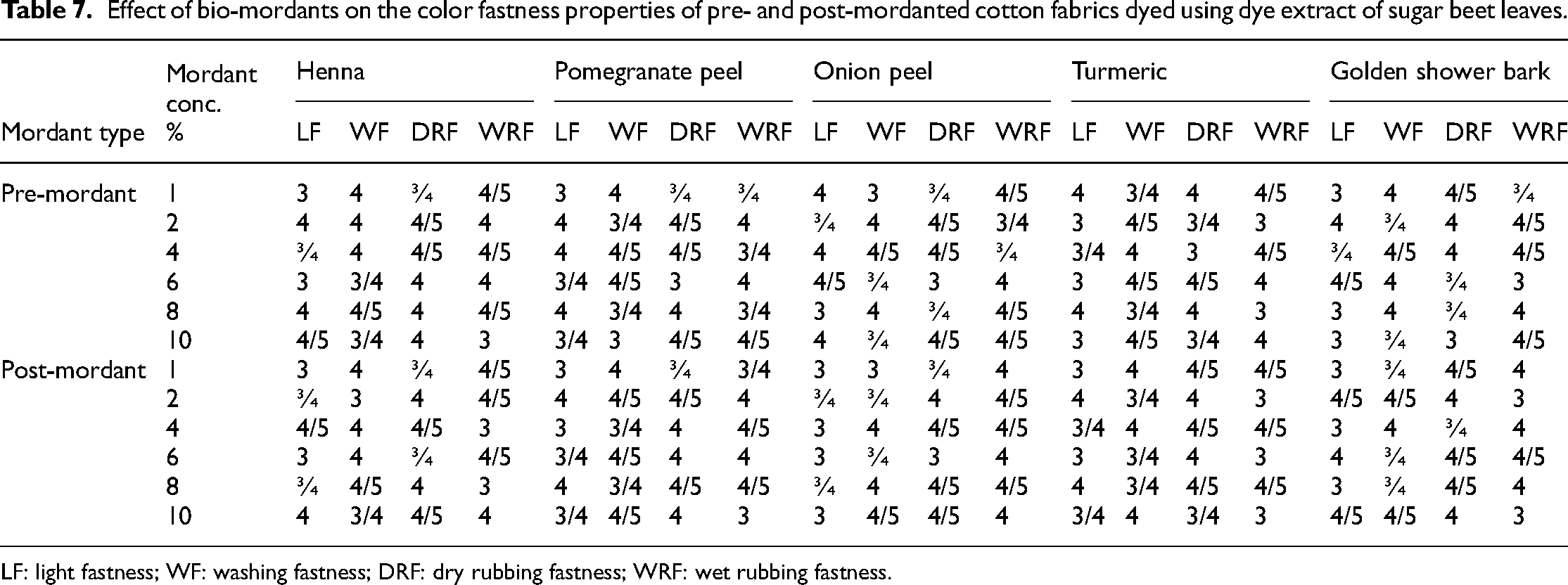
LF: light fastness; WF: washing fastness; DRF: dry rubbing fastness; WRF: wet rubbing fastness.
Characterizations
In the present research, various extraction media were utilized to isolate the actual colorant from sugar beet leaves. Consequently, 5% methanolic KOH was identified as an excellent medium, yielding the maximum colorant. Subsequently, for further optimization and mordanting processes, we exclusively employed the 5% methanolic KOH medium. Our emphasis in this regard was specifically directed toward the application of the 5% methanolic KOH medium for FTIR analysis. Other extraction media were disregarded due to their minimal solubility of the colorant, precluding their further utilization.
According to Popescu et al., 55 B. vulgaris L. (sugar beet) extracted solution absorbance peak was observed at 540, 270, and 424 nm on the ultraviolet–visible (UV–Vis) spectra in different pH conditions. The different peaks appeared for the different components present in the sugar beet solutions such as betanidines, cyclo-dopa-5-O-glycosides, and betalamic acid at a wavelength of 540, 270, and 424 nm, respectively. 36 The absorbance peak also confirmed the red–violet color exhibits peak absorption in the region of 530–540 nm, while yellow–orange color absorbs light at around 480 nm. The high performance liquid chromatography–mass spectrometry (HPLC–MS) results also indicated that the primary chemical of sugar beet leaves was betanin/isobetanin. 55 In the current research, the FTIR analysis of the dye extract revealed distinct characteristic peaks, as shown in Figure 8. The peak observed at 3276 cm−1 indicates the presence of the –OH group, which could correspond to compounds such as quercetin or gallic acid. The peak at 1611 cm−1 suggests the presence of the C=O group, which is typically associated with the phenolic group. 56 The peak at 1351 cm−1 suggests the presence of methyl and methylene groups, possibly indicating lipids or fatty acids, with certain flavonoids like quercetin contributing to this region. Additionally, the peak observed at 1047.91 cm−1 signifies the presence of an ester linkage, which could be attributed to quercetin or other flavonoids, known for their role as bio-colorants. The peak observed at 830 cm−1 corresponds to C–H out-of-plane bending vibrations, indicating the possible presence of aromatic compounds such as phenolic compounds or lignin, recognized contributors to coloration. Peaks registered at 607 and 500 cm−1 align with vibrations characteristic of aromatic compounds, potentially supporting the existence of anthocyanins or other pigments. Lastly, the peak at 452 cm−1 signifies skeletal vibrations of polysaccharides or lignin, suggesting the presence of cellulosic or lignocellulosic components that contribute to the overall color profile of sugar beet leaves. These interpretations offer valuable insights into the potential coloring compounds existing in sugar beet leaves. 57 The FTIR results were also consistent with the previous studies of UV–Vis and HPLC–MS spectra. 55

FTIR analysis of dye extract of sugar beet leaves.
The FTIR spectra of the dyed, undyed, and mordanted dyed cotton fabric are shown in Figure 9. The cellulose structure was present in the cotton fabric; therefore, the peaks appeared at 3283 cm−1 for O–H stretching in undyed cotton fabric, while in the dyed cotton fabric, whether bio-mordanted or chemically mordanted, the peaks of O–H stretching was shown at around 3334 cm−1, which means the peak was shifted after dyeing. For the other peaks that appeared on the control cotton fabric were 2898 cm− 1 for C–H stretching, 1426 cm− 1 for C–H bending, and 1105 cm-1 and 1028 cm−1 for C–O bending. On the other hand, when the fabric was dyed using the sugar beet solution, some of the peaks disappeared and new peaks point were shown (Figure 9(b)). Therefore, the FTIR results confirmed the successful dyeing of cotton fabric using sugar beet solutions. The results were also consistent with the cotton fabric dyed using gallnut by previous researchers. 58
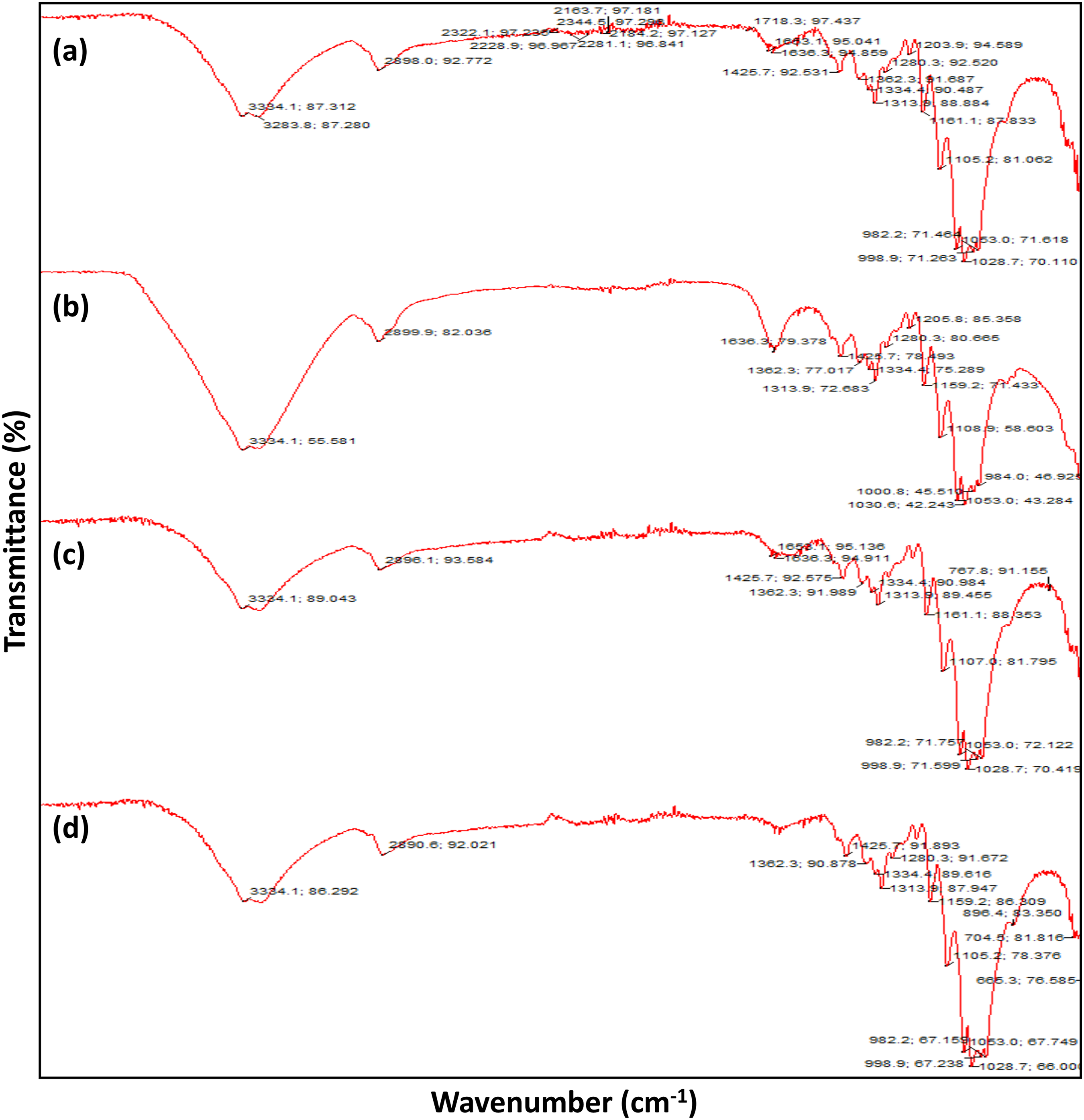
FTIR spectra of the (a) control cotton fabric, (b) sugar beet-dyed cotton fabric, (c) bio–post-mordanted dyed fabric (turmeric), and (d) chemical post-mordanted dyed fabric (CuSO4).
Discussion
The research findings highlight the substantial potential of sugar beet leaves as a renewable and organic resource for coloring textiles. The investigation of several extraction techniques, where a 5% methanolic KOH solution resulted in the most significant colorant extraction, highlights the necessity of improving the dye extraction procedure. The best dyeing conditions have been determined as follows: a dyeing period of 45 min at a temperature of 60 °C, using a salt solution concentration of 6.0 g/100 mL and 50 mL of dye extract. These circumstances are essential for obtaining effective and brilliant coloration on cotton fabric. The identification of quercetin via FTIR analysis provides more clarity on the primary pigment present in sugar beet leaves. The application of chemical and bio-mordants, with a special emphasis on bio-mordants, has demonstrated their efficacy in intensifying color and boosting the durability of color, offering essential knowledge for environmentally friendly and sustainable dyeing methods. The obtained color fastness values of 4–5 for washing, 4 for wet rubbing, 4–5 for dry rubbing, and 4–5 for light exposure demonstrate the possible practical use of colored fabric in real-life situations.
Nonetheless, it is crucial to acknowledge the limitations of this study. While the research centered on a specific set of bio-mordants and yielded significant results, the potential for exploring additional bio-mordants and their combinations remains untapped. Furthermore, the study primarily delved into examining the color quality and durability of the dyed fabric, neglecting a comprehensive exploration of other potential factors such as toxicity, scalability, and economic feasibility. To gain a more thorough understanding of the practical implications and constraints of utilizing sugar beet leaves as a natural colorant for textile dyeing, future research should concentrate on investigating these aspects. Other limitations of this research were to characterize the dyed cotton fabric using scanning electron microscopy (SEM) images, X-ray diffractions (XRDs), and mechanical properties to know more about the interaction chemistry between the sugar beet extracted dyes and cotton fabric. Therefore, considering the diverse composition of residual materials, conducting further research using dyed fabric characterization for dye–fabric interactions on the feasibility of incorporating other plant waste materials into the dyeing process would contribute to a more holistic and sustainable approach to textile coloring.
Conclusion
The investigation into sugar beet leaf residual material has revealed its potential as a promising natural colorant. Varying extraction media (aqueous, alkaline, organic, and acidic) have been employed for the extraction of the natural colorant of sugar beet leaves, and the maximum colorant was extracted in 5% methanolic KOH. The determined optimal conditions for effective dyeing encompass a process lasting 45 min at a temperature of 60°C, utilizing a salt solution concentration of 6.0 g/100 mL along with 50 mL of dye extract. FTIR analysis corroborated the presence of a specific coloring component, namely, quercetin. In comparison, the application of an 8% pomegranate peel as a bio–pre-mordant and 6% henna as a bio–post-mordant resulted in the creation of darker shades, accompanied by enhanced color strength and improved color fastness properties. Likewise, employing 1% TA as a pre-mordant and 6% iron sulfate (FeSO4) as a post-mordant also led to the development of a deeper shade. The fabric's quality, in terms of dye adherence and durability, experienced a significant improvement under these optimized conditions. Moving forward, further exploration in this field could delve into additional bio-mordants and their combinations, aiming to broaden the spectrum of sustainable and vivid color options in textile dyeing.
Footnotes
Acknowledgments
We are thankful to the vegetable section of Ayub Agricultural Research Institute, Faisalabad, Pakistan, for providing sugar beet residual material. The current paper is an outcome of the PhD studies of Ms. Fatima Batool.
Author contributions
Fatima Batool: conceptualization, data curation, and writing—original draft. Naeem Iqbal: investigation, methodology, and writing—original draft. Shahid Adeel: supervision, funding acquisition, project administration, and writing—review and editing. Muhammad Azeem: investigation and methodology. Muhammad Hussaan: resources, formal analysis, validation, and visualization. Rony Mia: data curation, formal analysis, software, and writing—review and editing.
Data availability
The work is an outcome of the PhD studies of Ms. Fatima Batool.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
