Abstract
This research investigates the viability of using Syzygium aromaticum (clove) as a natural dye for wool yarn through the application of microwave treatment and optimization using central composite design (CCD). As concerns grow over the environmental impact of synthetic dyes and their detrimental disposal in water bodies, the search for eco-friendly alternatives becomes imperative to revolutionize the textile industry. Microwave-assisted extraction of the colorant from clove powder is explored as an efficient and sustainable method, minimizing solvent usage and energy consumption compared to conventional techniques. To enhance colorfastness properties while eliminating the need for toxic mordants, green alternatives such as Al, Fe, and tannic acid, combined with plant phenolics from red sumac, pomegranate rind, and weld, are employed. According to the analysis of CCD, the higher color strength value 18.1653 was achieved using pH = 3, time = 50 min, temperature = 70 °C, and salt concentration = 1.5 g/100 mL. The optimized dyeing conditions also showed a maximum level of colorfastness properties of 5 for light, 5 for wash, 5 for dry rubbing, and 4 for wet rubbing. The findings from Fourier-transform infrared spectroscopy and scanning electron microscopy analyses provide valuable insights into the chemical and morphological changes induced by microwave treatment and dyeing with clove extract. The results affirm the presence of eugenol as a potential active molecule responsible for the captivating color of clove flower buds, validating its suitability as a natural dye source for wool. This study highlights the promising potential of microwave-assisted extraction and plant-based biomolecules as innovative and environmentally friendly approaches in natural dyeing, paving the way for a more sustainable future in the textile industry. Embracing these eco-friendly practices allows the textile sector to reduce its ecological footprint and contribute to a cleaner and greener environment. Further research and implementation of these techniques can foster a more harmonious coexistence with nature, ensuring a healthier ecosystem for all.
Introduction
Water pollution is a pressing environmental concern caused primarily by the discharge of substantial amounts of wastewater from industries such as textiles, paper, and leather. Among the various pollutants released, synthetic dyes have garnered significant attention due to their detrimental impact on the environment, even at low concentrations.1,2 The presence of colored wastewater containing highly toxic compounds poses serious environmental and health hazards. 3 In particular, the disposal of synthetic dyes into water bodies has led to increased biochemical oxygen demand and hindered photosynthesis, resulting in adverse effects on plant growth and overall ecosystem health. Moreover, the contamination of water resources, soil, and human health is a multifaceted consequence of synthetic dyes and their intermediate effluents. 4 The effluent produced by the textile industry contains a significant concentration of both organic and inorganic contaminants, including chlorinated compounds, heavy metals, sulfur, nitrates, naphthol, soaps, chromium compounds, formaldehyde, benzidine, sequestering agents, as well as dye and pigment finishes, among various other substances. 5 Even after undergoing specific treatment processes, wastewater often retains several toxic elements that pose a severe threat to both human and animal life, thus exacerbating environmental pollution. 6 Synthetic dyes are known to be recalcitrant, bio-accumulative, and exhibit toxic, mutagenic, and carcinogenic properties. 7 Consequently, the demand for eco-friendly products has surged, as environmental protection agencies have taken notice of the harmful aspects associated with synthetic dyes.
To address these environmental challenges, researchers and industries are turning toward natural dyes derived from plant sources as an eco-friendly alternative for textile dyeing. Natural dyes offer numerous advantages over their synthetic counterparts, such as biodegradability, non-toxicity, and renewable sourcing. 8 The revival of eco-friendly plant extracts as colorants has gained momentum in recent years due to the numerous advantages offered by natural dyes. 9 These colorants, derived from plant sources, present a promising alternative to synthetic dyes, imbuing textiles with unique, soft, and soothing shades. Beyond their esthetic appeal, certain natural dyes also possess additional benefits, such as anti-allergic properties, making fabric contact with the skin safer and healthier. 10 The versatility of bio-based colorants extends beyond textile dyeing, as they find applications in food coloring, leather tanning, and dyeing of natural fibers. 11 This broadens their potential usage, promoting sustainable practices in various industries beyond textiles. The adoption of natural colorants in these diverse fields aligns with the growing demand for environmentally friendly and non-toxic alternatives, fostering a safer and ecologically conscious approach to manufacturing processes. 12 As a result, we have also extracted natural colors from various plant sources, including tulsi leaves, 13 Rheum emodi plants, 14 gardenia yellow, 15 Triadica sebifera, 16 Amba-haldi, 17 black rice, 18 Coral Jasmine flower, 19 Butea monosperma plants, 20 Alkanna tinctoria roots, 21 and neem leaves extractions. 22
However, despite these benefits, natural dyes are not without limitations that significantly contribute to the fading popularity of this traditional dyeing art. Dyed textiles often exhibit color changes when exposed to sunlight and heat, and the act of washing and rubbing can likewise compromise the fabric's inherent properties. 23 These limitations may be attributed to the lack of technical advances in the extraction and dyeing techniques, which still need to be explored. To overcome these challenges, biochemical anchors are used to enhance the color strength and sustainability. 24 Bio-based mordants are also of medical importance as they possess antibacterial and antimicrobial properties, which can be beneficial for dyeing natural fabrics and in the medical textile industry. 25 In pursuit of enhancing technological advancements in extraction and dyeing methodologies, certain conventional techniques are being employed. These methods aim to amplify colorant extraction and bolster the fabric's capacity for absorption. 26 However, these methods not only require excessive solvent, energy, and economic resources but may also degrade the potency of the isolates (colorants). Modern methods, serving as alternatives, are gaining popularity due to their efficiency in using less solvent volume, requiring lower energy, and being more economical. 27 One such method is the microwave (MW)-assisted extraction tool, which has shown optimal results in a relatively short time. MW-assisted, especially when applied to plant materials, offers great potential due to its unique mode of action. 28 The MW rays transfer energy via solvent molecules, opening the derma of plant cells, allowing the molecules to come in contact with the solvent through powder solvent coordination, resulting in excellent colorant yield onto various matrices such as wool, silk, nylon, etc. 29
Among the diverse range of natural dyes available, Syzygium aromaticum (clove) holds promise as a potential dyeing agent for textiles due to its abundant availability and rich dye content (Figure 1(a)). Clove (S. aromaticum), belonging to the Myrtaceae family, has gained recognition for its health-promoting attributes attributed to its potential biomolecular constituents. The flower bud of clove contains flavonoids, polyphenols, alkaloids, and various bioactive ingredients, with tannins being the potential biomolecules responsible for imparting color to fabrics. 30 The primary biomolecules involved in the coloring process in the case of clove are tannins found in the eugenol (Figure 1(b)). During the dyeing process, these tannins interact with the fabric's structure to create complexes with the fibers that give the cloth its color. 31 Clove flower bud powder has been found to exhibit anti-inflammatory, antimicrobial, antifungal, and anticancer effects. 32 Utilizing clove as an eco-friendly bio-based dye could pave the way for dyeing medical textiles, such as medical suits, gloves, and masks. 33 To the best of our knowledge, a comprehensive exploration of clove powder extract as a source of bio-colorant has not been carried out yet. In light of the functional benefits of MW treatment for extraction, bio-mordanting, and shading, this research explored the coloring efficiency of clove flower bud powder as a source of a novel natural dye for wool yarn coloration. Previously, wool fabric was dyed using the natural dyes extracted from weld with a combination of synthetic dyes. 34 The utilization of yellow and black myrobalan extract was used as a bio-mordant for the dyeing of wool yarns with natural dyes from madder and reseda luteola. 35 The application of Adhatoda vasica natural dye in conjunction with bio-mordants as a substitute for metal mordants in the dyeing process of woolen yarn. 36 The dyeing of wool using natural dyes required high amounts of dyes and energy, which showed lower fastness properties.

Syzygium aromaticum (clove): (a) physical appearance and (b) chemical structure of eugenol.
In this study, we aim to revolutionize wool dyeing by exploring the potential of S. aromaticum natural dye through an innovative approach involving MW treatment and the application of central composite design (CCD) optimization. This research endeavors to establish an environmentally sustainable method for wool dyeing that minimizes the harmful impacts associated with synthetic dyes, contributing to the conservation of water bodies and overall ecosystem health. By systematically optimizing the dyeing conditions through CCD, we seek to enhance the color yield and fastness of the dyed wool fabric, while also reducing water consumption and effluent generation.
Experimental details
Materials
Clove (S. aromaticum) was procured as a natural dye source from Faisalabad, Pakistan. Bio-mordants, including red sumac, pomegranate, and weld, were chosen and processed to mesh up to 20 grades. The woolen yarn used in the study was provided by the University of Sestan and Baluchestan, Zehdan, Iran.
Extraction and irradiation procedure
To successfully extract and purify the natural colorant from clove powder, a dual approach was undertaken employing both aqueous (neutral) and acidic mediums. The extraction protocol involved the heating of 4 g of meticulously ground plant powder with 100 mL of water, brought to a boil for a duration of 60 min. Following this, the resultant extract was meticulously filtered using conventional methods, ensuring the acquisition of a pure, impurity-free extract. Subsequently, both the wool yarn and the extracts underwent exposure to MW radiation via a high-power MW irradiator. The treated and untreated extracts were subsequently utilized for dyeing both treated and untreated wool yarn at a temperature of 80 °C for 60 min, while preserving an optimal extract-to-fabric ratio of 1:25. The bio-mordants red sumac, pomegranate, and weld were collected, and dried under direct sunlight. Then, it was ground to make fine powder and mixed with water at a 1:10 ratio. The solution was then heated at 90 °C for 60 min and filtered using filter paper. Finally, the extracted bio-mordant was collected and stored for the dyeing process.
Dyeing variables optimization
In the pursuit of optimizing dyeing variables, the CCD method was harnessed, incorporating a comprehensive set of 32 design points. A Minitab 22 software was used to analyze the data in CCD. These points were meticulously allocated to four crucial dyeing parameters: time, temperature, pH of extracts, and salt concentration. The overarching goal was to attain the highest possible yield of colorant on the fabric surface, achieved through the meticulous optimization of dyeing conditions utilizing a second-order response surface methodology as a robust statistical model. Temperature influence was meticulously explored through dyeing experiments executed at distinct levels spanning 50 °C, 60 °C, 70 °C, 80 °C, and 90 °C. A parallel experimental series focused on optimizing the dyeing process based on various time intervals: 30, 40, 50, 60, and 70 min. Likewise, with a keen eye on the inherent characteristics of the wool yarn, dyeing endeavors encompassed a spectrum of pH values (3, 4, 5, 6, and 7) to extract the maximal potential. Furthermore, striving for the utmost exhaustiveness, dyeing operations were meticulously carried out with varied concentrations of table salt (0.5–2.5 g/100 mL). The intricate framework of these experiments, along with their pertinent parameters, have been systematically formulated and outlined in detail, as discernible in Table 1.
Utilizing a central composite design (CCD) to optimize dyeing variables with irradiated clove extract.

Shade improvement process
Novel shades were intricately fashioned onto wool yarns through a meticulous dyeing regimen, employing clove extract under specific conditions, both preceding and succeeding the application of eco-friendly chemical and green mordanting methodologies. The dyeing process transpired at a controlled temperature of 70 °C for 50 min, orchestrated with a carefully balanced mordant-to-wool yarn ratio of 25:1. The mordant-to-wool yarn ratio of 25:1 was used to prepare the mordanting solution in terms of the weight of wool yarn. The suite of mordants encompassed a range of concentrations, spanning from 0.5% to 2.5% for Al, Fe, and tannic acid. In a concerted effort to amplify efficacy and realize heightened colorfastness and serene shades, the integration of green anchors teeming with bioactive constituents was executed under identical conditions. To facilitate this, extracts were meticulously fashioned by amalgamating uniform particle powders in the range of 0.5% to 2.5% with 100 mL of water, which underwent a boiling process before being subjected to meticulous filtration. Each filtrate, meticulously adjusted in proportion to the wool yarn weight, was judiciously implemented both prior to and post the dyeing process under the meticulously selected conditions. For pre-mordanting, the wool yarn was treated using different concentrations of metal and bio-mordants at 70 °C for 30 min. Then the fabric was dyed in the presence of MW radiation at different concentrations. For post-mordanting, a cold wash was performed in dyed fabric and then it was treated with different concentrations of mordanting at 70 °C for 30 min. A comprehensive depiction of the radiation and dyeing conditions applied to the wool yarn is elaborated in Table 2 for clarity and reference.
Selected radiation and dyeing conditions for wool yarn using clove extract.

NRE: non-irradiated extract, NRWY: non-irradiated wool yarn, RE: radiated extract; RWY: radiated wool yarn; MAD: microwave-assisted dyeing.
Fabric analysis
The Fourier-transform infrared spectroscopy (FTIR) spectra were meticulously captured utilizing an attenuated total reflectance-FTIR instrument (Perkin Elmer, Waltham, MA, USA), with the spectral range strategically set from 400 to 4000 cm−1. Furthermore, the surface morphology of the wool yarn, both antecedent and consequent to irradiation, underwent meticulous examination through a scanning electron microscope (SEM De-scan = 5 kV). During this analysis, the fibers were scrutinized at an image power magnification of 1000×. To dissect the characteristics of the dyed fibers, a spectra flash SF600 (Data Color, USA) instrument was aptly employed, employing a D65 10° observer configuration. This comprehensive analysis enabled the determination of color strength (K/S) and subtle tonal variations. 37 The wavelength was selected based on the highest value of color strength obtained during the measurements. Rigorous evaluation of colorfastness attributes was conducted for the dyed fibers, encompassing crucial aspects such as washing (ISO 105 CO3), light exposure (ISO 105 BO2), and rubbing (ISO 105 X-12), meticulously adhering to the stringent ISO standards. The allocation of ratings was methodically accomplished by meticulous comparison of shade alterations against the standardized grayscale. 38
Results and discussion
MW radiation is a widely used commercial isolation tool in the world of natural products, known for its unique mode of action that uniformly heats the material by transferring energy via radiation. 39 The effectiveness of MW radiation depends on the treatment time for both the fabric and the extract. This energy transfer kinetics not only reduces the time required to extract the colorant into the solvent but also minimizes solvent consumption by rupturing the cell wall to maximize interaction with the biomolecules (dye). 40 Given its cost-effectiveness for extracting colorants from plant-based materials, MW radiation has been considered an economical and frequently used technique in various fields.
In this study, MW radiation was employed to extract the colorant (eugenol) from clove powder for dyeing wool yarn. Prior to irradiation, the use of neutral extract resulted in a better yield on the fabric, while after treatment for up to 8 min, the use of aqueous extract produced dark shades (K/S = 11.6567) (Figure 2(a)). On the other hand, changing the medium from aqueous to acidic before radiation yielded better results with the acidic extract, but after treatment for up to 6 min, excellent yield (K/S = 15.2221) was obtained on the wool yarn (Figure 2(b)). Based on the results, it is recommended that for dyeing wool yarn, both acidic extract and yarn should be stimulated with MW rays for up to 6 min using clove bud powder. This optimized method could lead to improved colorant yield and overall dyeing efficiency, making it a promising approach for dyeing wool yarn with clove natural dye.
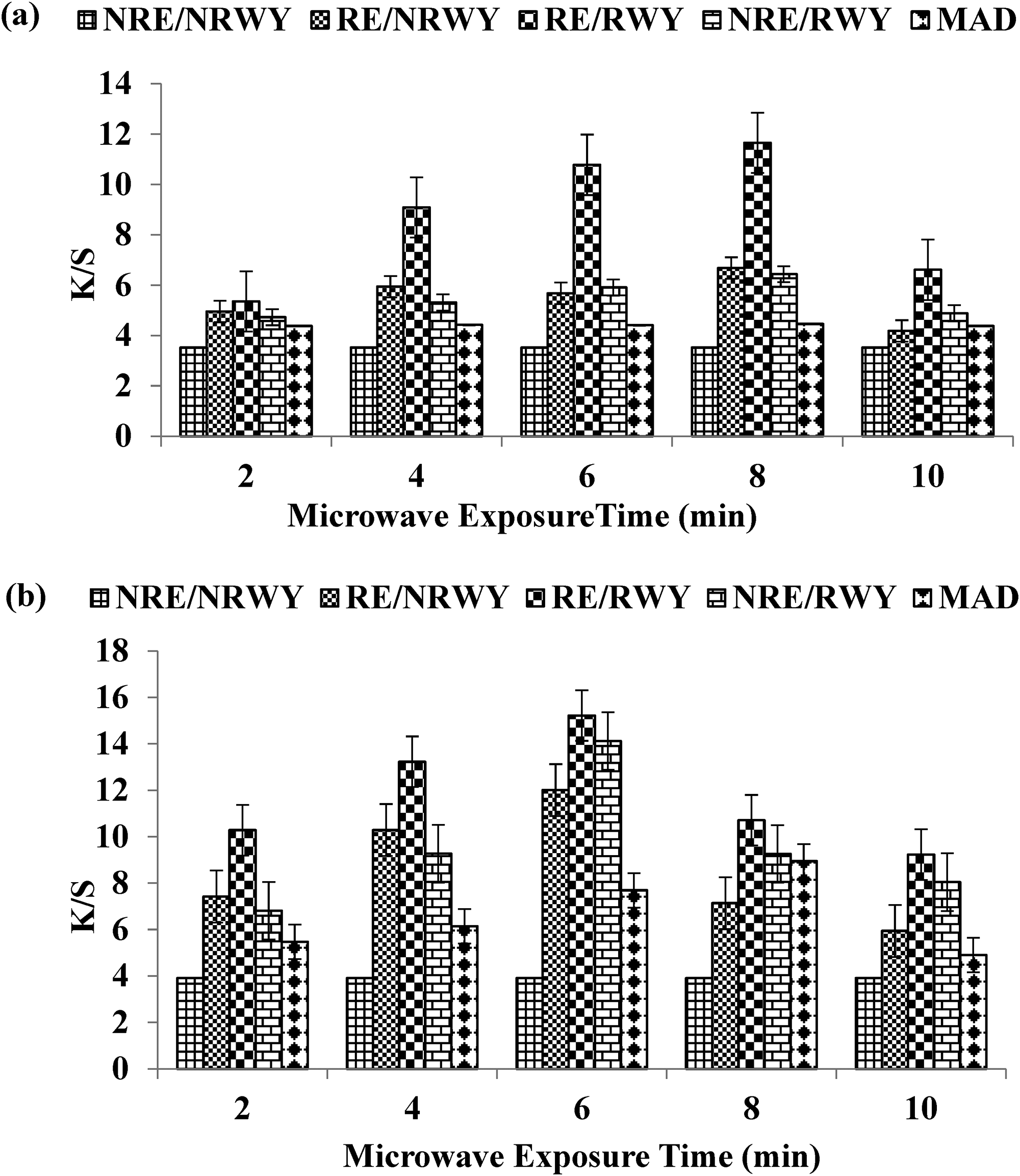
Effect of microwave treatment on wool yarn for dyeing using (a) aqueous extract and (b) acidic clove extract.
MW treatment has made it possible to reduce dyeing variables, leading to energy, time, and cost savings. When a CCD model was employed, it showed that MW treatment significantly reduced the dyeing time to 50 min (Table 3). This is because after radiation, the clusters of molecules are broken, and smaller size colorant particles take less time to evenly sorb onto the surface of the tuned woolen yarn. 41 However, for longer contact times (>50 min), more small molecules are sorbed, but they mostly remain on the modified surface. Upon washing, the unfixed colorant molecules are stripped, resulting in less yield when assessed under the CIE Lab system. Similarly, the amount of salt (1.5 g/100 mL) occasionally employed during dyeing has been shown to provide maximum color strength by producing leveled exhaustion. Using more than the selected amount of salt (>1.5 g/100 mL) might lead to over-exhaustion, causing poor fixation of color from the dye bath onto the wool yarn. Temperature also affects the shade strength because heating for less time does not accelerate the kinetics of the dye bath, whereas excessive heating can favor desorption, resulting in poor yield in both cases. 42 The thermal decomposition temperature of wool yarn is around 200 °C to 230 °C, which has no effect on dyeing temperature. 43
Central composite design (CCD) analysis for the optimization of dyeing parameters using irradiated clove extract.

Based on the results, it is recommended that after MW radiation treatment for up to 6 min, dyeing of MW-treated wool yarn for 50 min at 70 °C should be done with MW-treated extract having 1.5 g/100 mL of table salt as an exhausting additive. The nature of the dye bath is crucial because it plays a significant role in achieving maximum sorption of dye from the extract. Wool yarn is proteinaceous in nature and requires an acidic dye bath to achieve excellent dyeing results. The results show that clove, containing tannin, is also acidic in nature and enhances fabric when used at a pH of 3 for woolen yarn. As a result, it is evident that after MW radiation treatment for up to 6 min, dyeing of woolen yarn should be carried out at 70 °C for 50 min with clove extract of pH 3 and 1.5 g/100 mL of salt. Statistically, it can be observed that the employed model fits well, and the role of salt and extract pH is highly significant. The two-way interaction between extract pH and salt also plays a significant role in dyeing wool yarn (Table 4).
Analysis of variance of CCD using response surface methodology for the optimization of dyeing parameters using irradiated clove extract.
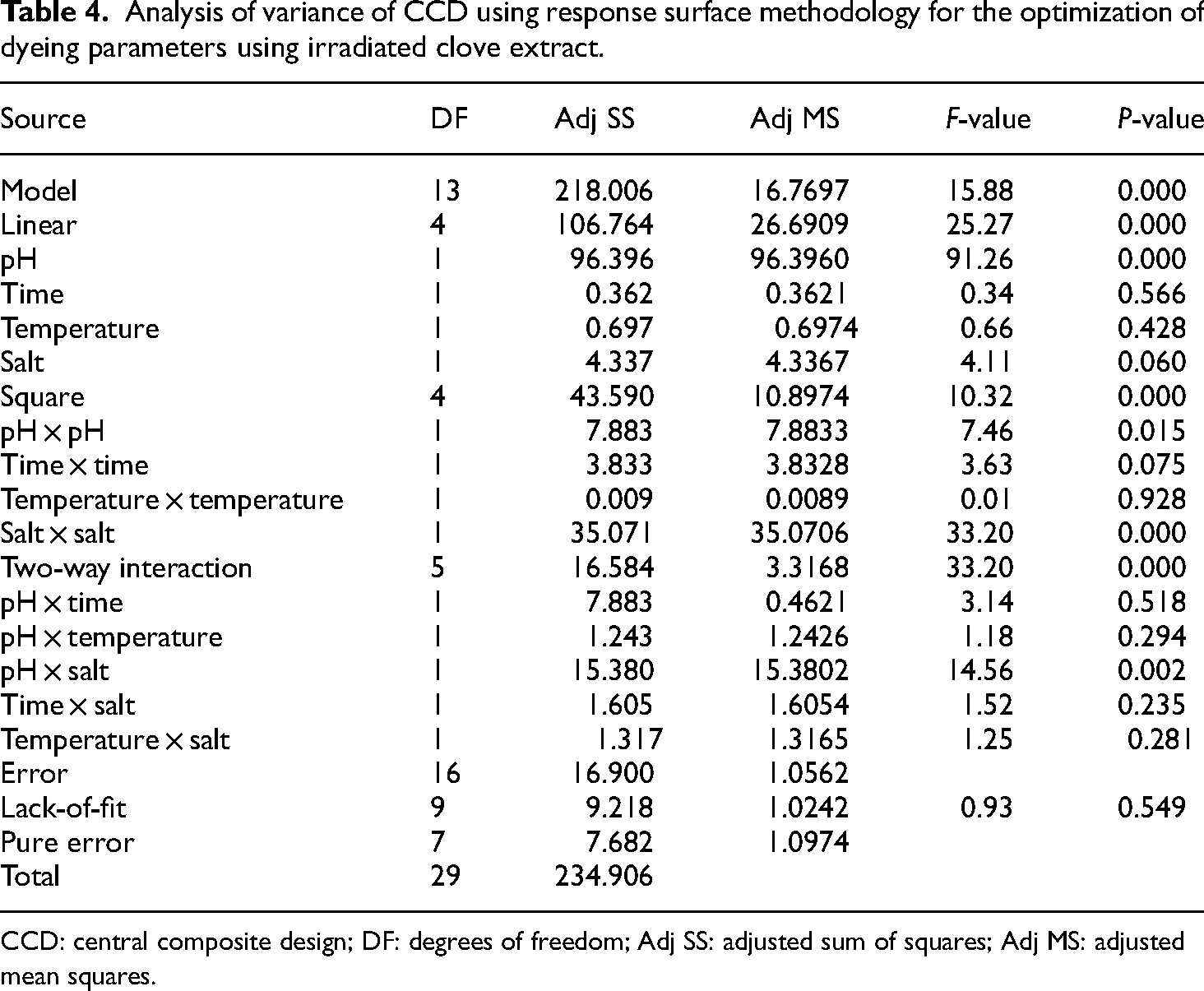
CCD: central composite design; DF: degrees of freedom; Adj SS: adjusted sum of squares; Adj MS: adjusted mean squares.
The spectral peaks displayed for un-irradiated wool yarn at 3278 cm−1 show the presence of the NH2 group, and at 2924.27 cm−1, they show the asymmetric vibrations of the CH3 functional groups (Figure 3(a)). The spectral bands at 1633 cm−1 indicate the presence of C=O, and at 1516 cm−1, they show the vibrations of C–N and N–H (amide II). 44 After radiation, these intermediates have not significantly altered their positions (Figure 3(b)). Regarding the un-irradiated clove extract, the spectral peaks indicate the presence of –OH at 3279.51 cm−1, vinyl group (–CH) stretching at 2919.45 cm−1, aromatic benzene stretching at 1614.79 cm−1, and CH3 at 1450–1300 cm−1. Additionally, the presence of eugenol has been confirmed by observing the spectral band at 1200.72 cm−1 (Figure 3(c)). The existence of eugenol in clove has been previously reported by the researcher, 45 supporting its potential as an active molecule responsible for the color of clove flower buds.

Fourier-transform infrared spectroscopy (FTIR) analysis of (a) un-irradiated wool yarn, (b) irradiated wool yarn, (c) un-irradiated clove extract, (d) radiated clove extract, (e) un-irradiated dyed wool yarn, and (f) irradiated dyed wool yarn.
The irradiation process has not caused significant changes in the positions of these functional groups in both the wool yarn and clove extract (Figure 3(d)). This consistency suggests that the essential chemical components responsible for the dyeing process, particularly the colorant eugenol, have remained intact and stable during the radiation treatment (Figure 3(e) and (f)). 46 The presence of eugenol as a potent active molecule further supports the suitability of clove extract for dyeing wool yarn with eco-friendly and sustainable natural colorants.
The SEM analysis presented in Figure 4 provides a comprehensive view of the impact of MW radiation and dyeing with clove extract on the surface morphology of wool yarn. The SEM images provide valuable insights into the structural alterations of the fibers under various conditions. Figure 4(a) showcases the un-irradiated wool yarn, serving as a reference point for the fiber's baseline structure in the absence of MW radiation. This image allows us to understand the initial state of the wool yarn. On the other hand, Figure 4(b) illustrates the SEM analysis of the irradiated wool yarn, allowing us to observe any changes in the fiber's surface caused by the MW treatment. The MW radiation's impact on the fiber surface is evident in this comparison. 47 Figure 4(c) displays the un-irradiated dyed wool yarn, giving insights into the fiber's surface morphology after dyeing without exposure to radiation. It can assess the alterations due to dyeing alone. Lastly, Figure 4(d) presents the irradiated dyed wool yarn, revealing the combined effects of MW radiation and dyeing on the wool fiber's surface structure. Notably, it allowed us to evaluate the presence and distribution of dye particles on the wool yarn's surface. Analyzing these four figures enables us to assess the influence of these processes and it was clearly shown that the dye particles were present on the surface of wool yarn, signifying successful dye absorption and adhesion. 48 The SEM examination highlights the visual validation of the structural modifications and the interplay between the wool yarn and the dye derived from cloves, hence elucidating the efficacy of the dyeing procedure. The present study provides a comprehensive analysis that showcases the feasibility and efficacy of employing MW methodologies in the process of natural dyeing, as well as their capacity to augment the interactions between dyes and fibers.

Scanning electron microscopy (SEM) analysis of (a) un-irradiated wool yarn, (b) irradiated wool yarn, (c) un-irradiated dyed wool yarn, and (d) irradiated dyed wool yarn.
In natural dyeing, it is essential to treat fabrics with either salt or plant extracts to achieve colorfast shades. Traditionally, salts of Al3+, Cu2+, Co2+, Cr2+, Ni2+, Fe2+, and tannic acid have been used for this purpose. However, it has been recognized by global communities that Co, Ni, Cr, and Cu salts are toxic and hazardous, causing harmful effects on aquatic life, water quality, and soil fertility. 49 Therefore, there is a growing trend to replace these harmful materials with eco-friendly and sustainable alternatives, such as plant bioactive. Recently, there has been a focus on developing alternate and new shades on fabrics using plant phenolics or other functional moieties.
In this study, green mordants such as Al, Fe, and tannic acid were used as chemical mordants, along with plant phenolics from red sumac, pomegranate rind, and weld, both before and after dyeing with clove bud powder extract (Table 5). The results showed that the metal salts formed dye complexes onto the wool yarn through coordinate covalent bonding (Figure 5(b)). 50 Al salt, lacking d-orbitals, brightened the shade, while iron in the salt, having d-orbitals, contributed to the formation of complex molecules, resulting in darker shades. Tannic acid, with its –OH group, reacted through hydrogen bonding with the –OH group of eugenols from clove, leading to the development of new shades. 24

Schematic presentation of mechanism between eugenols from clove and wool yarn in the presence of (a) bio-mordant and (b) metal mordants.
Shade quality parameters of selected wool yarn dyed with clove extract before and after chemical and bio-mordanting.

From the results displayed in Figure 6, it was observed that 1.5% of iron salt, 2.5% of Al, and 2% of tannic acid produced favorable outcomes. After dyeing, 2% of Fe, Al, and tannic acid resulted in excellent yields. Comparatively, before dyeing, 1.5% Fe salt and after dyeing, 2% Fe salt should be used for optimal results. This highlights the importance of selecting the appropriate mordants and their concentrations to achieve the desired shades while ensuring the use of eco-friendly and sustainable materials in natural dyeing processes.

(a) Pre-chemical mordant and (b) post-chemical mordant of wool yarn with clove powder extract at optimized conditions.
Nowadays, plant-based biomolecules are being increasingly utilized to make the dyeing process eco-friendly and more innovative. These selected plants contain biocomponents that exhibit Ayurvedic and biological properties. When these biomolecules interact with the fabric and colorant, they form additional H bonds, which contribute to the creation of new colorfast shades. 51 Moreover, the conjugation present in these biomolecules enhances the development of darker shades with stable hues, making them an attractive alternative to toxic mordants (Figure 5(a)).
The data presented in the form of graphs reveals that using extracts from 2.5% of red sumac and pomegranate rind, along with 1% of weld, before dyeing has resulted in the production of darker shades (Figure 7(a)). Among them, the extract of red sumac provided the highest color strength with dark tones. Similarly, after dyeing, 2.5% of pomegranate and 0.5% of red sumac extracts produced good shades, while the weld extract (2%) exhibited the darkest shades with the highest strength (Figure 7(b)).

(a) Pre-bio-mordanting and (b) post-bio-mordanting for wool yarn with clove powder extract at optimized conditions.
As a result, the preferred combination for developing colorfast shades is using 1.5% iron as a pre-chemical mordant, 2.5% red sumac as a pre-bio-mordant, 2% iron as a post-chemical mordant, and 2% weld as a post-bio-mordant. By incorporating these plant-based biomolecules as mordants in the dyeing process, it is possible to achieve sustainable and eco-friendly dyeing methods, replacing the need for toxic mordants and contributing to greener practices in the textile industry.
When engaging in the dyeing of fabric with plant pigments, the paramount emphasis revolves around securing robust shade fastness. A meticulous evaluation of colorfastness attributes becomes imperative in this context. To comprehensively gauge these attributes, fabric samples that have undergone dyeing with plant pigments are subjected to a thorough examination, both prior and subsequent to mordanting, facilitated by electrolytes and plant extracts (as exemplified in Table 6). This evaluation involves using a grayscale to determine the level of colorfastness achieved. The colorfastness of the dyed fabric is achieved by forming a durable and stable complex on the surface of the fabric. This is accomplished through surface modification, which includes using salts and enhancing hydrogen bonding with plant extracts. The attainment of deeper shades is also contingent upon the particular surface modification technique applied and the specific combination of colorants and mordants utilized. 52
Fastness grading of wool yarn dyed with clove powder extract before and after chemical and bio-mordanting.

LF: light fastness; WF: wash fastness; DRF: dry rubbing fastness; WRF: wet rubbing fastness.
The findings of the study revealed that through the judicious application of suitable mordant quantities, the fastness characteristics of dyed wool experienced significant augmentation, ultimately leading to an elevated level of colorfastness. This approach offers a sustainable and eco-friendly method for dyeing wool yarns, replacing the need for synthetic dyes and toxic chemicals. By utilizing plant pigments and appropriate mordants, textile industries can adopt greener practices while ensuring the longevity and vibrancy of the dyed fabrics.
The research findings present promising outcomes in the utilization of naturally derived dyes from clove buds, combined with MW treatment and bio-mordants, for dyeing wool yarn. However, it is essential to acknowledge certain constraints that hinder its suitability for widespread industrial adoption. The investigation was conducted under specific experimental conditions, encompassing prescribed solvents, durations, and temperature settings. The potential applicability and scalability of the proposed technique may be constrained by the possibility that these conditions may not accurately reflect real-world industrial or commercial scenarios. While the study prioritizes environmental sustainability and pollution reduction, it is imperative to assess the economic feasibility and practicality of implementing the proposed approach on a broader scale. The feasibility and implementation of the approach in industrial contexts can be influenced by various factors, including equipment costs, energy consumption, and process optimization.
Conclusion
This research highlights the potential of S. aromaticum (clove) as a natural dye for wool yarn, utilizing MW treatment and optimizing the process with CCD. The primary goal was to address environmental concerns related to synthetic dyes and their impact on water bodies. Our findings show that MW-assisted clove extraction is a more efficient and eco-friendly alternative to traditional methods, reducing solvent and energy use. The use of green mordants and plant phenolics enhances color fastness and reduces the need for toxic mordants. Analytical techniques confirm the presence of eugenol as the active dyeing molecule. This study emphasizes the significance of eco-friendly methods in natural dyeing, offering the textile industry a path to reduce its environmental impact and move toward sustainability. Further research and implementation of these techniques can lead to a cleaner and greener textile sector, coexisting harmoniously with nature.
Footnotes
Acknowledgements
The authors are highly grateful to the Department of Chemistry, and to the Department of Applied Chemistry, Government College University Faisalabad, Pakistan for providing the necessary funds for the smooth running of the experiments for this M.Phil. Studies. The authors are also thankful to the Textile Engineering Department of Marmara University, Turkey, for the characterization of fabrics and extracts.
Author's contribution
Muhammad Yameen: Conceptualization, data curation, and writing—original draft; Fariha Asghar: Investigation, methodology, and writing—original draft; Shahid Adeel: Supervision, funding acquisition, project administration, and writing—review and editing; Muhammad Zulqurnain Haider: Investigation, methodology; Meral Özomay: Resources, formal analysis, and software; Muhammad Aftab: Software, validation, and visualization; Rony Mia: Data curation, formal analysis, software, and writing—review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The necessary funds for the smooth running of the experiments for this M.Phil. Studies were provided by the Departments of Chemistry and Applied Chemistry, Government College University Faisalabad, Pakistan.
Author biographies
Muhammad Yameen is an Associate Professor of Biochemistry at Government College University, Faisalabad, Pakistan.
Fariha Asghar is an MPhil student of the Department of Biochemistry at Government College University, Faisalabad, Pakistan.
Shahid Adeel is an Associate Professor of Applied Chemistry at Government College University, Faisalabad, Pakistan.
Muhammad Zulqurnain Haider is an Assistant Professor of Botany at Government College University, Faisalabad, Pakistan.
Meral Özomay is an Associate Professor of the Department of Textile Engineering at Marmara University, Istanbul, Turkey.
Muhammad Aftab is a PhD scholar of the Department of Statistics at Government College University, Faisalabad, Pakistan.
Rony Mia is a lecturer in the Textile Engineering department at the National Institute of Textile Engineering and Research, a constituent institute of the University of Dhaka, Dhaka, Bangladesh.
