Abstract
Introduction
In December 2019, a cluster of pneumonia cases with an unclear cause was reported in Wuhan, China. Since then, the world has witnessed an increasing number of such cases. 1 Infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the putative causative agent of coronavirus disease 2019 (COVID-19), resulted in this pneumonia. Analysis of the SARS-CoV-2 genome has shown that it is phylogenetically close to SARS-CoV. 2 COVID-19 was declared a public health emergency of international concern by the World Health Organization on January 30, 2020, and recognized as a pandemic on March 11, 2020. By 2022, the global number of confirmed cases of COVID-19 had reached an unprecedented peak and continues to increase. COVID-19 primarily manifests as an acute respiratory illness, with interstitial and alveolar pneumonia; however, it can also affect multiple organs including the kidneys.3–5 As such, patients with COVID-19 having chronic kidney disease (CKD) are a particularly vulnerable group. Among these special patients, innate and adaptive immune function impairment leads to increased susceptibility to bacterial and viral infections. 6 Angiotensin-converting enzyme 2 (ACE2) serves as a receptor for SARS-CoV-1 and SARS-CoV-2 and is expressed in multiple organs, including renal epithelial cells and bladder cells. SARS-CoV-2 enters renal epithelial cells via ACE2.7,8 The virus can bind to renal epithelial cells, injure them, and subsequently disrupt fluid, acid-base, and electrolyte homeostasis, causing further damage to the kidneys. Moreover, most patients with CKD are immunocompromised due to uremia and require routine dialysis in hemodialysis centres two to three times per week. 9 Hemodialysis centres, often characterized by crowded spaces and highly mobile dialysis patients, have an exceptionally high risk for exposure during an outbreak. Therefore, such patients are more susceptible to SARS-CoV-2 than the general population. The incidence of COVID-19 is particularly high among the elderly and comorbid populations. Pre-existing cardiovascular disease, diabetes, hypertension, and CKD have been identified as risk factors for severe disease and mortality.10–12 Thus, the hemodialysis population may have an extremely high risk of experiencing severe COVID-19. Currently, there are few studies investigating patients with COVID-19 who are also undergoing MHD. Therefore, to aid in the prevention and management of this patient population, it is critical to explore clinical manifestations, prognosis, and risk factors for death in this population.
Protocol and registration
The protocol adhered to the recommendations established by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (i.e. “PRISMA”) statement, and has been registered in the International Prospective Register of Systematic Reviews database under registration number CRD42021223665.
Methods
Search strategy
A literature search of the PubMed, Embase, and Web of Science databases, for articles published between January 2020 and March 2022, was performed using the following search terms: “Novel coronavirus 2019” OR “2019 nCoV” OR “COVID-19” OR “SARS-CoV-2” AND “Hemodialysis”. The detailed search strategy is presented in Figure 1. Additionally, the reference lists of the retrieved articles were manually searched for other potentially eligible studies. There were no language restrictions in the search.

Flow diagram of study selection.
Selection criteria
Inclusion criteria
Eligibility for inclusion was based on the following criteria: study design (randomized trials, non-randomized controlled trials, observational studies [cross-sectional, cohort, and case-control], and case series); participants (patients with CKD who required MHD and had laboratory-confirmed COVID-19); and selected variables (at least one of the following outcomes was reported: epidemiological characteristics, clinical symptoms, radiological manifestations, treatment[s], and prognosis).
Exclusion criteria
Duplicate studies, commentaries, editorials, guidelines, case reports, case series, and observational studies, in which the number of reported cases was < 10, and investigations with incomplete or missing data were excluded.
Two authors (PL and SZ) independently performed the literature search based on the search strategy and screened the retrieved studies according to the inclusion and exclusion criteria. Any disagreement between the two authors (PL and SZ) was resolved through discussion with a third author (RW).
Data extraction
Two authors (PL and YG) independently performed data extraction from the selected studies, including: study identification (first author, publication date, study region, and country); study design (participants, age, sex, sample size, study data, and inclusion and exclusion criteria); and variables (epidemiological data, clinical symptoms, radiographic findings, treatment, and prognosis). Basic information collected and analysed in the study is summarized in Table 1.
Main characteristics of the included studies in our analysis.

AHRQ, Agency for Healthcare Research and Quality; NOS, Newcastle-Ottawa Scale.
* Agency for Healthcare Research and Quality (AHRQ) Article quality was assessed as follows: low quality = 0–3; moderate quality = 4–7; high quality = 8–11.
* Newcastle-Ottawa Scale (NOS). Article quality was assessed as follows: high quality = 6–9 by Chinese Journal of Evidence-based Cardiovascular Medicine, August 2012, Vol. 4, No. 4.
Assessment of risk of bias in the included studies and risk of publication bias
Observational studies commonly used for meta-analyses include cohort, case-control, and cross-sectional studies. Currently, however, there is no universal evaluation tool. Therefore, the Newcastle Ottawa-Scale (NOS) was used to evaluate the included cohort and case-control studies. The Agency for Healthcare Research and Quality (AHRQ) cross-sectional study evaluation criteria were used to evaluate the included cross-sectional studies. Two authors, independently and in a double-blind manner, assessed the risk of bias and the study quality of each included observational study or case series. Any disagreement between the two authors was resolved through discussion with a third author. Of all included studies, the AHRQ scale was used to evaluate 11 studies while the NOS was used for the other 11 studies. The quality scores of all studies are summarized in Table 1. Fourteen studies were of high quality and eight were of moderate quality; there were no studies with low-quality ratings. When more than 10 studies were included, funnel plots and Egger's test were used to assess publication bias. Results with P < 0.05 indicated significant publication bias.
Statistical analysis
Previous research criteria were consulted13–15 and a meta-analysis was performed when a particular variable was assessed by more than five studies with at least 100 participants. For dichotomous variables, the weighted mean prevalence and corresponding 95% confidence interval (CI), and for continuous variables, the weighted average means and 95% CI were calculated. The I2 statistic was used to assess study heterogeneity, which was classified as follows: I2 < 25%, low heterogeneity; 25%–50%, medium heterogeneity; and >50%, high heterogeneity. Due to the high heterogeneity in this study, a random-effects model was used. When the heterogeneity of a study was high, sensitivity analysis was performed, and studies that had a deviation in the results according to the analysis were excluded. Subgroup analyses were performed according to study region (Asian countries versus non-Asian countries) and clinical outcome (survivor versus non-survivor). Data from the included studies were analysed using Stata version 16.0 (StataCorp, LLC, College Station, TX, USA).
Results
The initial literature search retrieved 4495 potentially eligible studies, including from Embase (n = 2071), Web of Science (n = 970), and PubMed (n = 1454). First, 1388 duplicate references were excluded. After reading titles and abstracts, 118 studies were selected from the remaining references for full-text reading, of which 96 more were excluded. After excluding 4473 non-conforming studies, a total of 22 retrospective case analyses were included in the present meta-analysis. The selection process is illustrated in Figure 1. The 22 included studies16–37 were from 17 countries, with a total of 13,191 patients. Meta-analysis of epidemiological, clinical, and radiological data from COVID-19 patients undergoing MHD are summarized in Table 2.
Meta-analysis of epidemiological, clinical and radiological of COVID-19 in MHD. (Before and after sensitivity analysis).
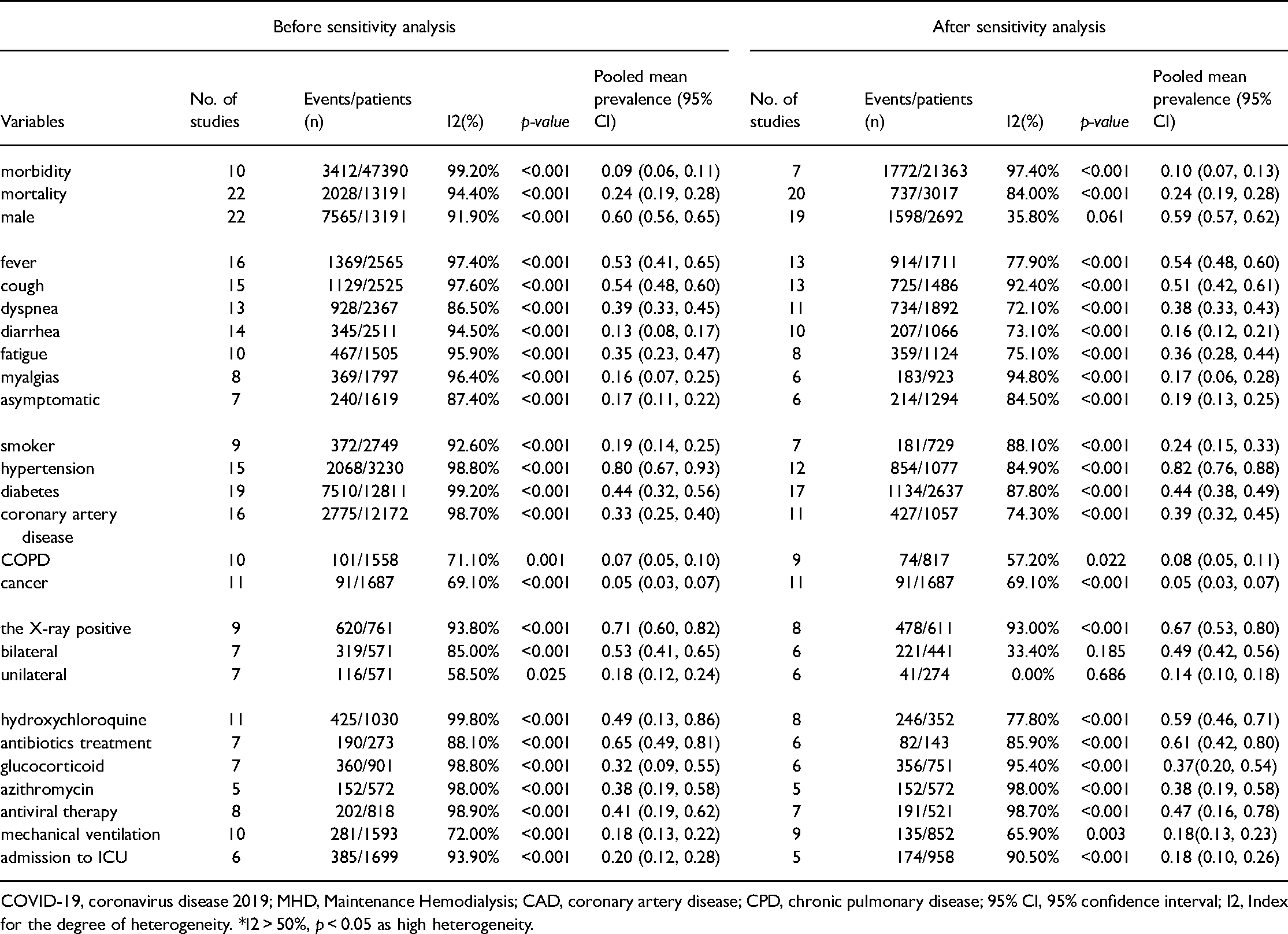
COVID-19, coronavirus disease 2019; MHD, Maintenance Hemodialysis; CAD, coronary artery disease; CPD, chronic pulmonary disease; 95% CI, 95% confidence interval; I2, Index for the degree of heterogeneity. *I2 > 50%,
Age and sex distribution
Most of the patients were middle age and older adults, with ages ranging from 51 to 83 years; the proportion of male patients was greater. Among the 13,191 patients undergoing MHD with laboratory-confirmed COVID-19, 7565 were male patients (60% [95% CI 56%–65%]).
Clinical features
In this population, the incidence of COVID-19 was as high as 9% (95% CI 6%–11%), as shown in the forest plot in Figure 2A. The majority of studies reported clinical characteristics of the patients. As shown in the forest plot in Figure 2C, the most prevalent symptoms included fever (53% [95% CI 41%–65%]) and cough (54% [95% CI 48%–60%]), while dyspnea (39% [95% CI 33%–45%]), fatigue (35% [95% CI 23%–47%]), diarrhea (13% [95% CI 8%–17%]), and myalgia (16% [95% CI 7%–25%]) were occasionally reported. There was also a relatively large percentage of asymptomatic patients (17% [95% CI 11%–22%]), as shown in the forest plot in Figure 2B.
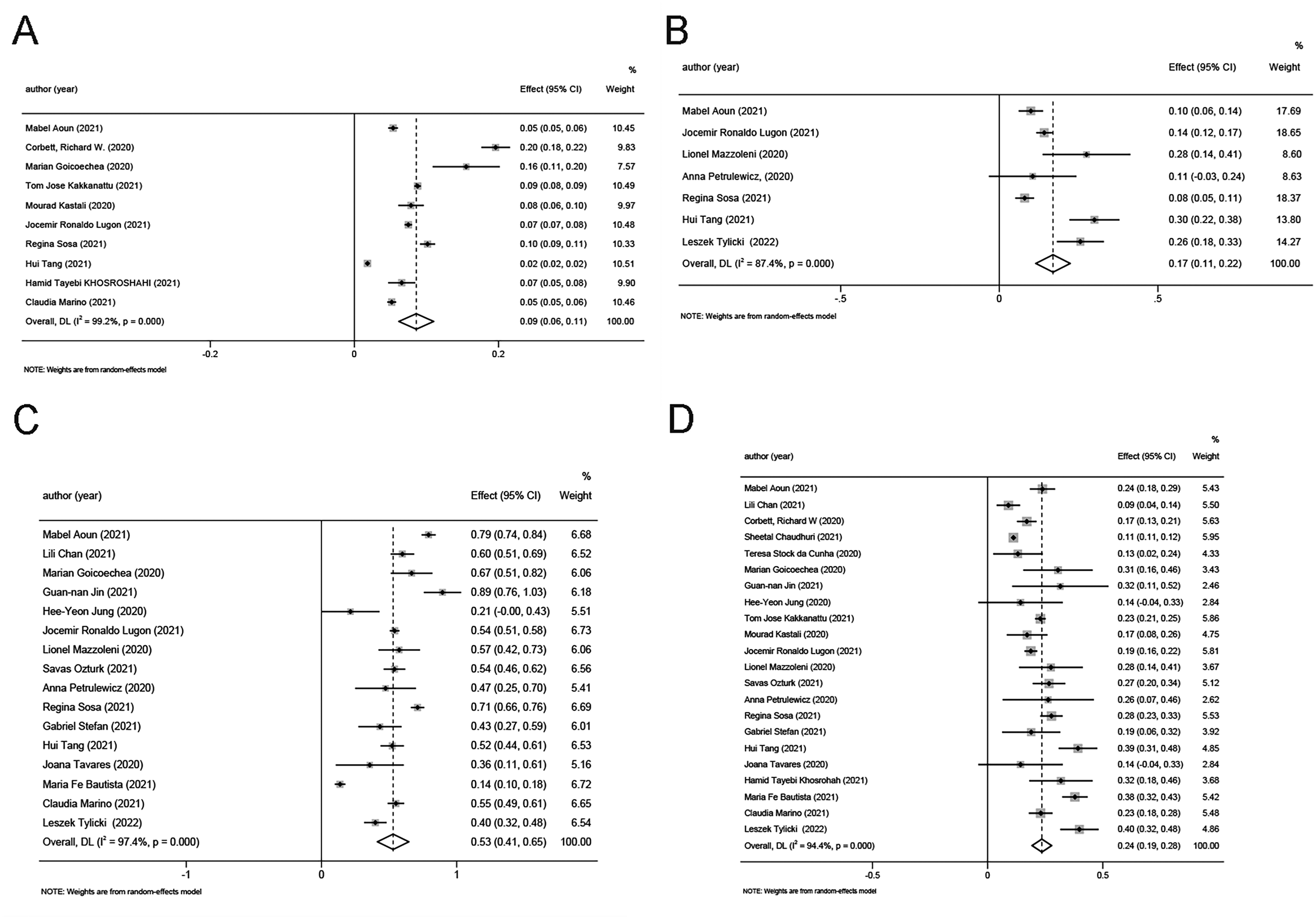
(A) The forest plot of morbidity, (B) The forest plot of asymptomatic, (C) The forest plot of fever, (D) The forest plot of mortality.
Radiographic findings
As shown in Table 2, nine studies20–21,23,27–29,31–32,35 reported radiographic findings. Positive computed tomography findings were common (71% [95% CI 60%–82%]), and bilateral lesions (53% [95% CI 41%-65%]) were more common than unilateral lesions (18% [95% CI 12%–24%]).
Complications and prognosis
Virtually all of the included studies reported that patients undergoing MHD often had a variety of underlying diseases. The most significant comorbidities included hypertension (80% [95% CI 67%–93%]) and diabetes (44% [95% CI 32%–56%]). The remainder included coronary artery disease (CAD) (33% [95% CI 25%–40%]), chronic obstructive pulmonary disease (COPD) (5% [95% CI 5%–10%]), and cancer (5% [95% CI 3%–7%]), and were relatively limited. Patients with COVID-19 undergoing MHD exhibited a high mortality rate (24% [95% CI 19%–28%]), as shown in the forest plot presented in Figure 2D.
Treatment
Thirteen studies16–17,21–23,25,27–33,35 reported data regarding treatment(s). Almost all patients underwent symptomatic treatment. Antibiotics were administered to 190 of 273 patients (65% [95% CI 49%–81%]), hydroxychloroquine to 452 of 1030 patients (49% [95% CI 13%–86%]), and glucocorticoid to 360 of 901 patients (32% [95% CI 9%–55%]). The use of other antivirals, such as interferon-α ribavirin, oseltamivir, lopinavir, ritonavir, and lopinavir were also reported, although relevant data were limited.
Subgroup analyses
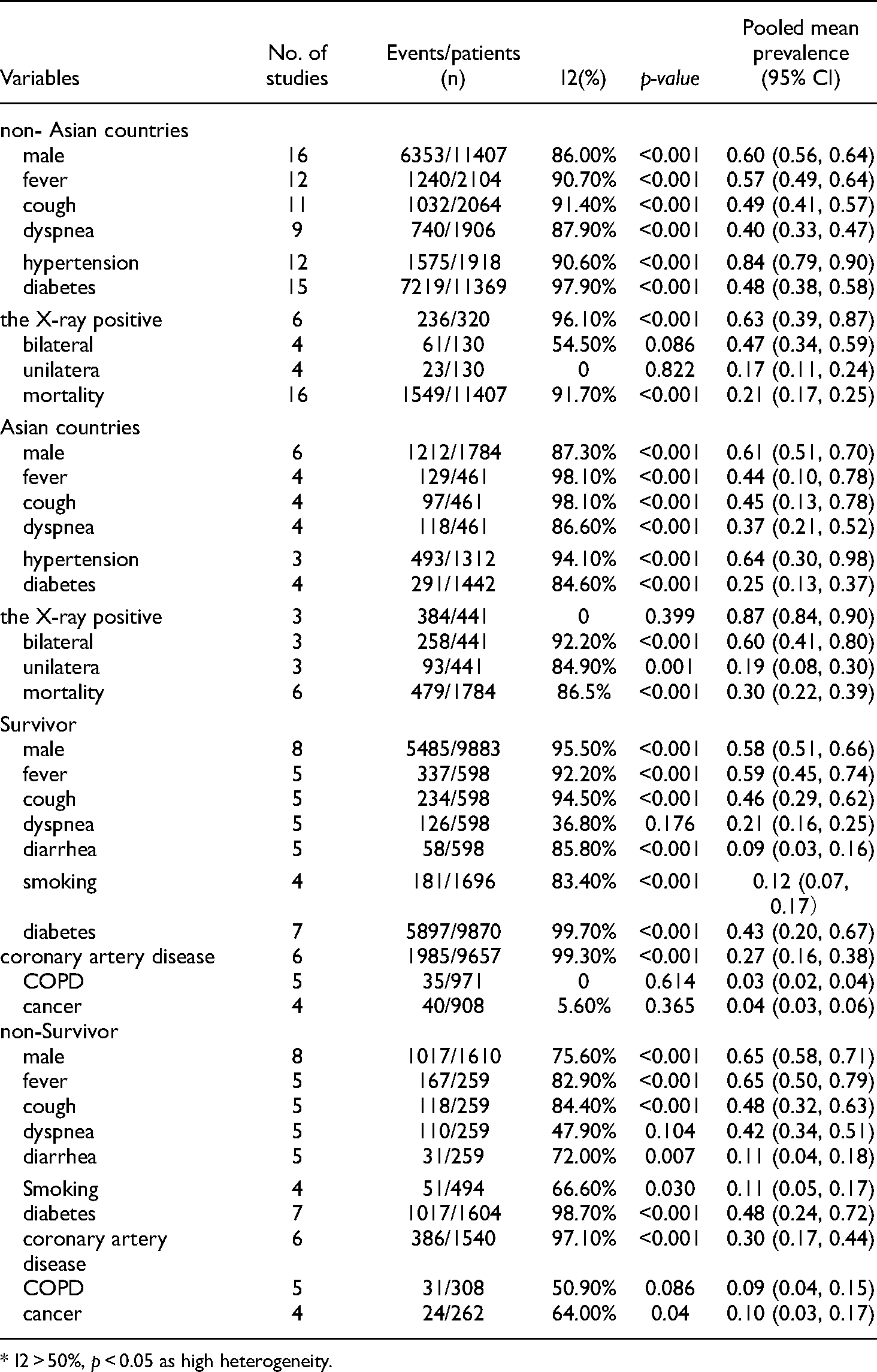
There was significant heterogeneity among the included studies. To explore the source of heterogeneity, subgroup analysis according to disease severity (e.g. mild, severe, and critical) was planned. However, due to varying definitions of disease severity among different countries, it was decided not to conduct a subgroup analysis. Instead, a subgroup analysis of the major outcome indicators of the study-by-study region (non-Asian versus Asian countries) was performed. In addition, a subgroup analysis based on clinical outcomes (survivor versus non-survivor) was performed. Detailed results of the subgroup analyses are summarized in Table 3. The subgroup analyses revealed a decreased proportion of patients with fever and a significant increase in the proportion of asymptomatic patients in Asia. Moreover, the proportion of patients with diabetes and hypertension in non-Asian countries was higher than in Asia. In the subgroup analysis, the proportion of male patients (65% [95% CI 58%–71%]), and patients with diabetes (48% [95% CI 24%–72%]), CAD (30% [95% CI 17%–44%]), and COPD (9% [95% CI 4%–15%]) was greater in the non-survivor group compared with the survivor group. The results are summarized in Table 3. Thus, male patients were at greater risk for death than female patients, and complications increased the risk for death. Forest plots are presented in Figure 3.

Subgroup analysis: (A) The forest plot of survivor male, (B) The forest plot of non-survivor male, (C) The forest plot of survivor diabetes, (D) The forest plot of non-survivor diabetes, (E) The forest plot of survivor coronary artery disease, (F) The forest plot of non-survivor coronary artery disease.
Subgroup analysis of different clinical symptoms. (Asian countries and non-Asian countries; Survivor and Non-survivor). .
* I2 > 50%,
Sensitivity analysis
Sensitivity analysis was performed on all variables. Possible outlying studies were removed based on the results of sensitivity analysis; the results of all variables before and after sensitivity analysis are summarized in Table 2. However, only a few variables excluded an outlying study, and the I2 was significantly reduced. The results of the sensitivity analysis are shown in Figure 3 (Figure 4).

Sensitivity analysis of before and after excluding the outlying studies (A-B male, C-D The X-ray bilateral positive, E-F The X-ray unilateral positive).
Publication bias
Funnel plots and Egger's test were performed when more than 10 studies were analysed. The results of Egger's test revealed that most studies (morbidity, p = 0.071; male sex, p = 0.218; fever, p = 0.618; cough, p = 0.154; dyspnea, p = 0.846; fatigue, p = 0.173; hypertension, p = 0.420; diabetes, p = 0.082; coronary artery disease p = 0.197; mechanical ventilation, p = 0.322; hydroxychloroquine, p = 0.691) did not have significant publication bias. However, mortality (p < 0.001), diarrhea (p = 0.025), COPD (p = 0.049), and cancer (p = 0.017) had significant publication bias; however, the funnel plots (mortality, mortality, diarrhea, COPD, cancer) were not symmetric, which meant that the indicators did contain publication bias. Funnel plots for several important variables are presented in Figure 5.
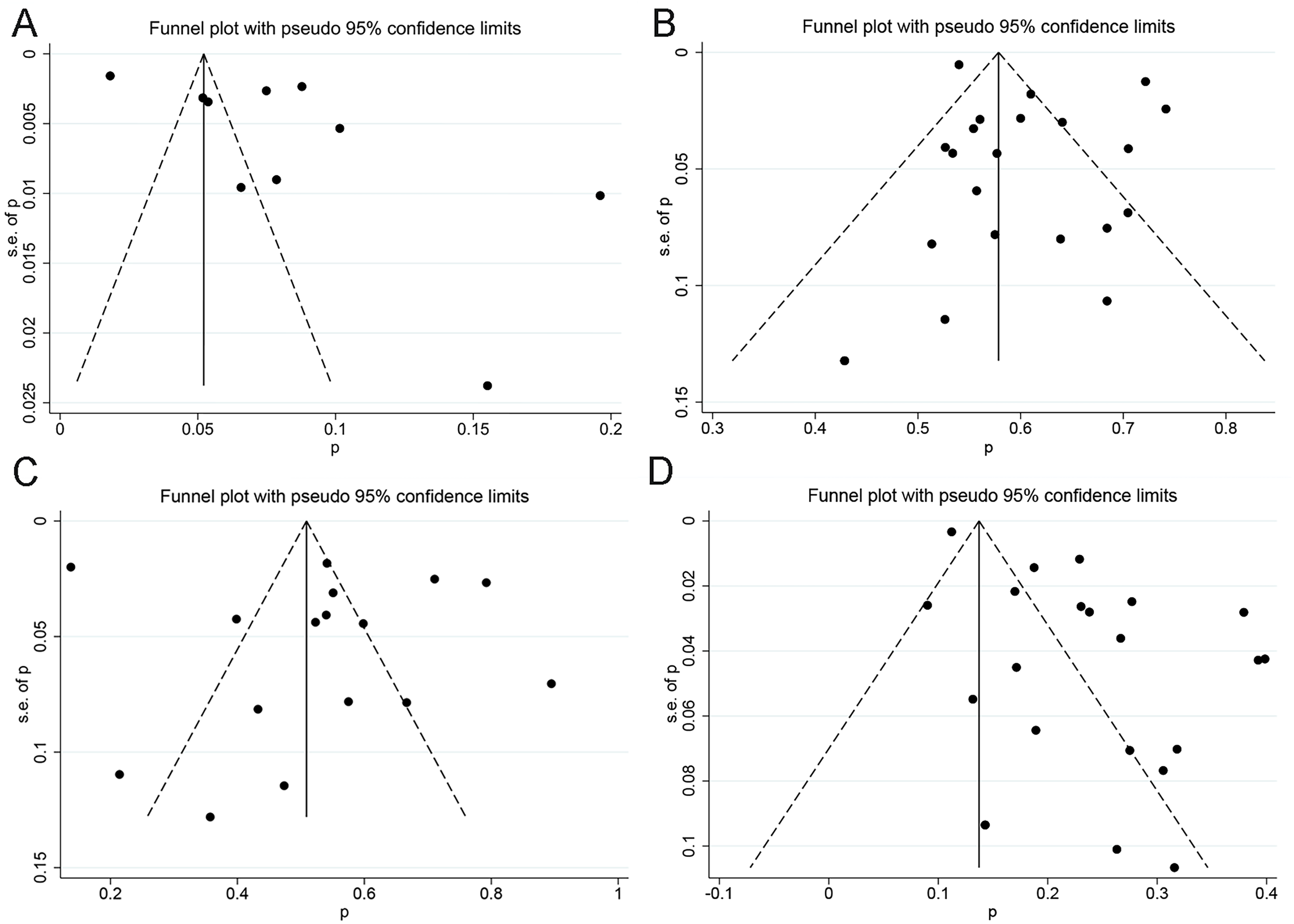
(A) Funnel plot of mortality. B) Funnel plot of male. C) Funnel plot of fever. D) Funnel plot of mortality.
Discussion
Despite tremendous global efforts to contain the transmission and spread of SARS-CoV-2, COVID-19 remains at a pandemic level. Patients undergoing MHD have a proinflammatory state with functional defects in their innate and adaptive immune cell populations and are reported to be at higher risk for upper respiratory tract infection and pneumonia,38,39 Infection is one of the leading causes of mortality among patients undergoing dialysis, second only to cardiovascular disease. 40 Moreover, hemodialysis centres tend to be in enclosed spaces that are more conducive to viral transmission and COVID-19 outbreaks. To control the spread of COVID-19, vaccines continue to be tested in clinical trials. 41 However, the effectiveness and safety of COVID-19 vaccines in patients undergoing MHD remain unknown. Therefore, clarifying the epidemiological and clinical characteristics of this special population and formulating effective preventive measures is the most effective intervention. Our results suggest that MHD patients with COVID-19 may initially present as asymptomatic or exhibit only mild symptoms; nevertheless, they have a higher risk for death compared with COVID-19 patients not undergoing MHD. Moreover, male sex and cardiovascular and respiratory diseases increase the mortality risk.
In this population, the incidence of COVID-19 was as high as 9%, which is significantly higher than that in the general population (0.5%–2%). Most dialysis patients tend to be elderly and have multiple complications. Therefore, they constitute a group at higher risk for infection with SARS-CoV-2. Furthermore, the proportion of MHD patients with COVID-19 who were asymptomatic in our study was 15%. This is much higher than that of COVID-19 patients in the general population. 42 Previous studies have reported that hemodialysis patients with COVID-19 experience milder symptoms, less lymphocytosis, and exhibit lower levels of inflammatory cytokines than other patients with COVID-19. 43 This is also consistent with the results of our study. Hence, patients may not exhibit overt clinical symptoms in the early stages of COVID-19. 44 Simultaneously, we performed subgroup analyses of patients from Asia and non-Asian countries, which revealed that the incidence of fever decreased in the Asia subgroup. Moreover, results of the subgroup analyses revealed that mortality rates among Asian patients were 30% higher among MHD patients compared to those in non-Asian countries. This suggests that COVID-19 patients in Asia were more likely to be overlooked if they were asymptomatic, thus, resulting in widespread epidemics. However, they have a poor prognosis and, as such, need early warning.
Male patients were more susceptible than female patients, which is consistent with previous reports on COVID-19. 15 This may be related to protection from the X chromosome and sex hormones, which play an essential role in innate and adaptive immunity. 45 Most MHD patients with COVID-19 are elderly and have other chronic diseases. Our results revealed that the most common comorbidities in this population were hypertension and diabetes. Previous studies have reported that COVID-19 patients with comorbidities tend to have a poorer prognosis. 3 In our study, the results of subgroup analysis of survivor and non-survivor groups also confirmed this. The subgroup analysis results revealed a greater number of non-Asian patients with diabetes and hypertension, which can be explained by different dietary and lifestyle habits in various parts of the world. Some studies have reported that the proportion of obese individuals in European and American countries is significantly higher than that in Asian countries. Obesity and cardiovascular disease are inseparable 46 and together may increase the likelihood of disease progression.
Almost all the patients underwent symptomatic treatment. Antibiotics were administered to 65% of the patients. Although there is no clear evidence that antibiotics can inhibit COVID-19, co-infection with pathogenic microorganisms are common among COVID-19 patients.47,48 Moreover, previous studies have shown that the human microbiome plays an important role in patients with COVID-19. 49 Hydroxychloroquine and glucocorticoids were administered to more than half of the patients. However, the effectiveness of these two drugs is also unclear.50,51 In particular, the use of glucocorticoids is controversial, due to their potential inhibition of viral clearance and prolongation of viremia. 52 Other antiviral agents used in the included studies were interferon-α, ribavirin, oseltamivir, lopinavir, ritonavir, and litonavir. However, we could not perform a meta-analysis because the available data were limited. Nevertheless, the clinical benefits of lopinavir and ritonavir treatment in the early stages of COVID-19 require further study. 53 Although several treatments have been proposed in several studies for this particular population, none are decidedly effective at present, and specific efficacy needs further research.
The results of this meta-analysis, including 22 studies, have implications for clinical practice and research. Patients undergoing MHD tend to experience relatively mild symptoms in the early stage(s) of infection. Moreover, dialysis centres are often in more enclosed spaces, and individuals who work at or attend dialysis centres could be an important source of transmission. Second, COVID-19 patients undergoing MHD have a worse prognosis, with a higher mortality rate, in fact, much higher than that of the population without MHD. It has been previously reported that kidney injuries increased the risk for death among patients with influenza A virus H1N1 infection or SARS.54,55 Furthermore, COVID-19 has important effects on many aspects of the body. 56 Renal events, including CKD and acute kidney injury, significantly increase the risk for death after infection. 57 Our results were similar in that patients with kidney disease also exhibited an increased risk for death resulting from COVID-19. However, the detailed mechanisms of renal involvement in COVID-19 remain unclear, and further clinical trials are needed in this regard.
Our study demonstrated higher mortality among MHD patients compared to the general population (3.6%). 58 In this special population, older age and multiple chronic diseases were factors for increased risk for infection. A study by Chun-Yu Chen 59 also explained this fact; however, their study included MHD patients with clinically diagnosed COVID-19, while our study only targeted patients with positive laboratory detection of SARS-CoV-2 nucleic acid, and we screened different literature that included the same cases. The clinical diagnosis criteria among different countries vary in that some used radiographic signs or clinical features as surrogate criteria for COVID-19. However, due to the immune-suppressed status in MHD patients, symptoms include radiographic signs of COVID-19 that may be less severe than those in the general population, which could be misleading for the diagnosis. As such, the SARS-CoV-2 nucleic acid test should be the gold standard criterion for COVID-19.
This meta-analysis had some limitations. First, the studies we included were retrospective in design and, as such, may have reduced our accuracy. There were also missing data for some clinical variables, and the number of infected patients enrolled in the included articles varied widely, which may have impacted the generalizability of our findings. Notably, there was significant heterogeneity between the groups, which may have been due to the studies being from different regions. The availability of resources has always varied between and among countries. Second, the funnel plots (mortality, mortality, diarrhea, COPD, cancer) were not symmetric, which meant that the indicators did contain publication bias. Due to the relatively small number of such studies, there may be omissions and small sample studies in the included studies, resulting in publication bias of some variables. Third, we only studied the clinical characteristics of hemodialysis patients, while the state of patients undergoing renal replacement therapy, such as peritoneal dialysis and kidney transplantation, remains unknown. Finally, in general, there are limited data regarding Patients with COVID-19 undergoing MHD. Some studies published in local medical journals may not have been identified and included in the review. Moreover, the included studies were from different countries, and due to national policies and ethnic differences, there may be significant differences among these studies.
Conclusion
COVID-19 was more likely to affect patients with MHD than the general population. Patients with COVID-19 undergoing MHD are often overlooked because they initially present with mild symptoms. In this special population, male sex and multiple chronic diseases were factors for increased risk for death. Moreover, COVID-19 may further damage the kidneys, thereby aggravating pre-existing kidney conditions in patients undergoing MHD, resulting in an increased mortality rate. Nevertheless, early detection and effective intervention may help reduce the mortality rate of patients with COVID-19 undergoing MHD. Therefore, it is necessary to implement preventive measures in advance for this specific population.
Footnotes
Abbreviations
Acknowledgements
The authors thank Prof Wenning Wu for providing guidance.
Authors’ contributions
PL, SZ, and YG analysed the data and wrote the first draft. RW, GF, and DZ designed the study, proofread, and revised the submission. EW directed the statistical analyses of the data. PS retrieved documents and extracted data. All authors contributed to the article and approved the submitted version.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This paper did not use experimental data from human subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the fund for Natural Science Foundation of China (grant number 81970051), Excellent Top Talent Cultivation Project of Anhui Higher Education Institutions (gxyqZD2017030), a fund from Reserve candidate for Anhui Province Academic and technical leader (2019), scientific research fund from Anhui medical university (2020xkj257), the Applied Medical Research Project of Hefei Health Commission (Hwk2021zd008), Construction Project of Provincial Teaching Demonstration Course of Anhui Province (1364), and Quality Engineering Project of Anhui medical university.
