Abstract
Modified natural zeolites (MNZ) are widely used in pollutant removal, but how to address these MNZ that have adsorbed pollutants must be considered. Selenium is an essential trace element for metabolism and is also a water pollutant. Selenium is adsorbed in the water by MNZ in this study first. Then the Brassica chinensis L. was planted in the soil which contains the MNZ loaded with selenium (MNZ-Se) to explore selenium uptake. MNZ-Se release tests in water and soil were also considered. The results showed the following: (1) The maximum adsorption capacity of MNZ for selenium is 46.90 mg/g. (2) Water release experiments of MNZ-Se showed that regardless of how the pH of the aqueous solution changes, the trend of the release of selenium from MNZ-Se in aqueous solution is not affected and first decreases before stabilizing. (3) Soil release experiments of MNZ-Se showed that the selenium content in the soil increased and reached the concentration in the standard of selenium-rich soil. Addition amount and soil pH value will affect the release ratio. The release ratio of MNZ-Se in the water was higher than that in the soil. (4) With an increase in the soil MNZ-Se content, the selenium content in the soil and B. c increases. Above all, MZN can be a good medium for water pollutant removal and soil improvement.
Introduction
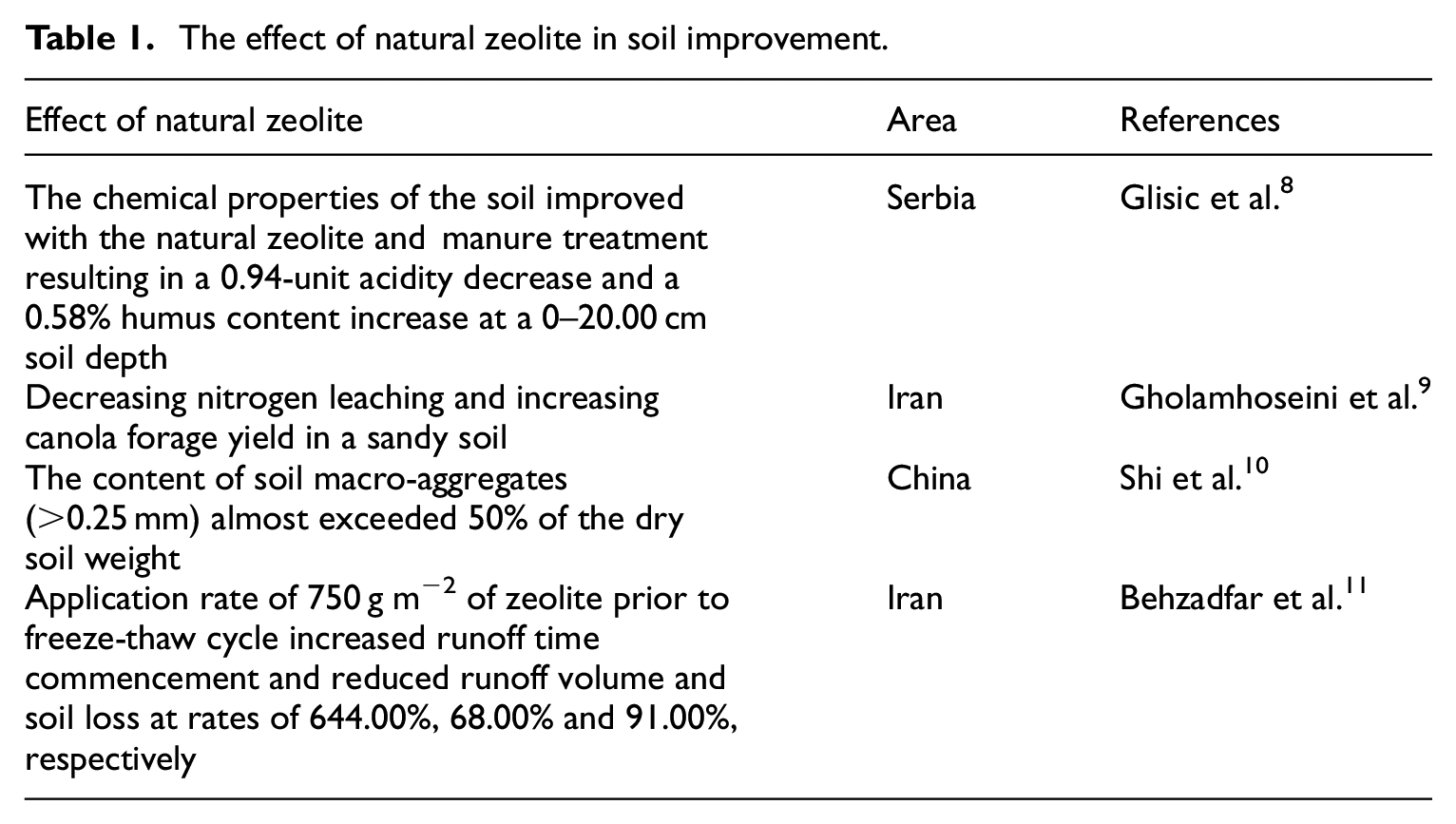
Natural zeolite (NZ) is an environmentally friendly material that usually used in the field of soil improvement, water contamination removal or material property enhancement. 1 The application of NZ is beneficial because of its good ion exchange ability, pore structure and cost-effectiveness.2–4 In terms of soil improvement, the application of NZ could improve the soil physical and chemical properties, 5 decrease soil erosion, 6 and increase crop yields, 7 as shown in the Table 1.
The effect of natural zeolite in soil improvement.
NZ was also used as a low-cost material to remove different water contaminants, including heavy metals, metalloids, salts, organic pollutants and other materials.12–15 Zeolites are formed by silicon and aluminum tetrahedra connected by oxygen atoms, the aluminum presence in the zeolite gives origin to an unbalanced framework having as a consequence the intrinsic cationic exchange capacity but low to negligible affinity for anions.16,17 Consequently, many approaches have been taken in the laboratory to modify NZ to increase adsorption ability of anions and adsorption capacity of cationic.18–20
However, after these modified NZ (MNZ) have completed the adsorption of pollutants, their next step of treatment has not been clearly clarified. The current research has aimed to improve the adsorption ability of NZ by various means or focused on regeneration of adsorbents.21,22 The way to address these MNZ that have adsorbed pollutants must be considered. It would be interesting if we could combine the use of MNZ for removing water pollutants and improving the soil quality. Some substances, such as ammonium, phosphate, selenium, not only cause water pollution but can also be used for improving plant quality crops in the soil.23,24 Shang et al. 25 studied the adsorption of phosphate by quaternary ammonium based sugarcane bagasse (QA-SB) and its release of laden phosphate in aqueous solutions and soil. This research indicated that the laden phosphate on the QA-SB could be desorbed efficiently and the recovery of phosphate onto the regenerated QA-SB still attained about 94.50% after four cycles. The slow-release of laden phosphate in aqueous solution could be continuously conducted for 12 days, with 43.60% of laden phosphate (11.60 mg/g) released. In contrast, phosphate (7.4 mg/g) released in soil reached equilibrium within 4 days, accounting for only 27.80% of laden phosphate released.
Selenium (Se) is an essential trace element that has multiple biological functions for metabolism. 26 A healthy adult needs 25–35 μg intake of selenium every day, which is recommended by the World Health Organization (WHO). 27 It is estimated that up to one in seven people worldwide, including approximately 39.00%–61.00% of Chinese residents, have insufficient dietary Se intake. 28 In fact, the selenium content of edible plant parts is often closely related to the soil selenium content where the crops are cultivated. 29 Selenium is also a common and intractable metalloid contaminant with high toxicity in water, and the WHO recommends a maximum permissible selenium level in drinking water of 10.00 μg/L. 30 Many materials for adsorption of selenium have been studied, such as metal nanoparticles, metal oxides and zeolite.31–35
Selenium-contaminated wastewater contains a large amount of selenium, but soils have very low selenium levels, with an average concentration of 200.00 μg/kg. 36 Overall, it is a feasible method to use MNZ to absorb selenium in wastewater and reuse the MNZ loaded with selenium from adsorption test to supplement selenium in soil and increase the selenium content in the vegetable. In this research, NZ modified with Fe2O3 (MNZ) was used to adsorb selenium in a water solution. Then the modified natural zeolite loaded with selenium (MNZ-Se) was placed in the water and soil to explore the release of selenium. Finally, MNZ-Se was placed in soil, and vegetable seeds were planted to observe and explore the uptake of selenium by the vegetables. The vegetable species was Brassica chinensis L. Vegetables planted in soil without zeolite were used as blank controls. We could make the following assumptions: (1) The selenium adsorbed on the MNZ can be released into the water and soil, and the release ratio of MNZ-Se in the water was higher than that in the soil; and (2) B.c that is grown in soil with MNZ-Se can effectively absorb selenium, and the amount of selenium content in the vegetables increases.
Materials and methods
Chemicals
All of the chemicals used in this work were of analytical grade or higher. Sodium selenite (Na2SeO3), sodium hydroxide (NaOH), nitric acid (HNO3), hydrochloric acid (HCl), ethanol (C2H6O), ferric chloride (FeCl3), citric acid (C6H8O7), and slaked lime (Ca(OH)2) were purchased from Beijing Chemical Works. NZ was obtained from a zeolite plant located in Hebei Province, China. The experimental soil was obtained from Zhangjiakou, Hebei Province, China. The fertilizers and vegetable seeds were purchased from Taobao.Com, and their qualities are compliant with Chinese standards GB/T 23348-2009 and GB 16715.2-2010.
Preparation of MNZ
The NZ was modified based on a China patent. 37 The modification steps are as follows: First, 5.00 g of NZ and 5.00 g of FeCl3 were weighed, respectively, and then poured these two ingredients into the conical flask (1000 ml) with 500.00 ml C2H6O. Second, 20.00 g of C6H8O7 (mass fraction ≥99.50%) were weighed and poured into the conical flask and mixed with a glass rod. Then, the conical flask filled with solution was placed in a water bath and stirred and heated for 24 h; the water temperature was 65°. Subsequently, the reaction solution was filtered by filter paper in order to separate the MNZ. Finally, the MNZ was placed in an oven for 4 h, of which 2 h were spent drying the moisture at 108° C and 2 h were spent calcining the material at 200°C, and collected after passing through a 140 mesh sieves.
Determination of the zeolite characteristics
The chemical compositions of NZ and MNZ were determined by X-ray fluorescence (XRF) (Philips PW2404, Philips Co., Holland). The percentages and oxide content of each element were determined based on the chemical analysis method of silicate rock in GB/ t14506.28-93. The mineralogical compositions of the zeolites were determined using a powder X-ray diffraction (XRD) instrument (Rikaku D/max-RB, Rikaku, Japan). Scattering patterns were collected from 5° to 70° with a scan time of 1 min per two steps. Photomicrographs of the two aforementioned materials before and after adsorption were obtained using a scanning electron microscope (SEM) (Gemini SEM 300, ZEISS, German). Before the experiment, The Specific surface area (SSA) and pore structure of the zeolites was determined using the nitrogen adsorption method. Nitrogen adsorption experiments were conducted at 77 K using QuadraSorb SI (Cantata Instruments Co., USA). The zeta potential of the NZ and MNZ under different pH were determined by Zetasizer Nano ZS90 (Malvern, England). The Fourier Transform infrared spectroscopy (FTIR) of NZ and MNZ before and after adsorption were recorded with a Perkin Elmer Frontier spectrometer (Perkin Elmer, USA). The X-ray photoelectron spectroscopy (XPS) of NZ-Se and MNZ-Se was determined by K-Alpha X-rays spectrometer (Thermo Scientific, USA).
Experiments on the adsorption of selenium by zeolites
Stock solutions of selenium (IV) (1000.00 mg/L) were prepared from Na2SeO3 (analytical grade) and diluted to the desired experimental concentrations by adding distilled water. The adsorbent dosage of 1.00 g/L of MNZ and 5.00 g/L NZ was added to 100.00 ml selenium (IV) solution with different selenium concentrations at 25°C. The experiments were conducted in 100 mL stoppered conical flasks on a temperature-controlled shaker (HZQ-F160, Aozuo, China) with continuous stirring at 240 rpm/min. The specific adsorption test setup is fully described in the supplementary materials. According to the preliminary tests, the optimum adsorption conditions of NZ and MNZ were pH = 2.0, contact time = 240 min for NZ, and pH = 3.0, contact time = 120 min for MNZ. Each solution’s pH was adjusted via the addition of the appropriate volume of 1 M HNO3 or NaOH, and the pH was measured using a pH meter (PHS-3C, Leici, China). After the experiment, the suspensions of the batch experiments were centrifuged and then filtered with a 0.45 μm cellulose acetate membrane.
The concentrations of selenium (IV) in equilibrium in the supernatants were analysed by inductively coupled plasma optical emission spectroscopy (ICP-OES, Optima 2000, PerkinElmer Co., USA). The amounts of selenium (IV) adsorption by the two zeolites (q(mg/g)) were calculated using equation (1)

where C0 and Ce are the initial and equilibrium selenium (IV) concentrations of the test solution (mg/L), respectively. V is the testing solution volume (L), and W is the mass of the adsorbent (g). All of the experiments were repeated twice.
The following pseudo-first-order (equation (2)) and pseudo-second-order (equation (3)) kinetic models were applied to simulate the experimental data 38 :

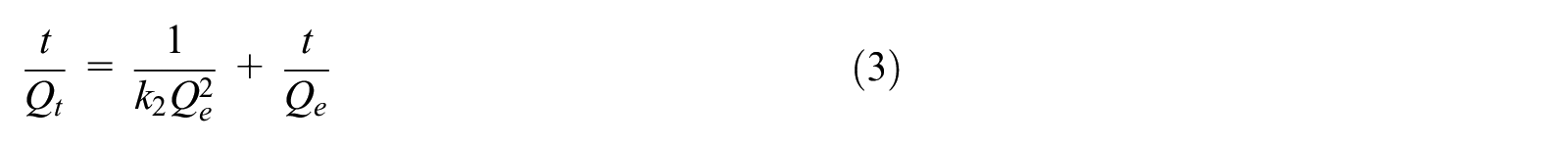
where Qe and Qt are the amounts of Se(IV) adsorbed on the synthesized zeolites (mg/g) at equilibrium and at time t, respectively. The values of k1 (min−1) and k2 (g·mg−1·min−1) are the sorption rate constants of the pseudo-first- and pseudo-second-order kinetic models, respectively.
The mechanism of interaction between adsorbent and adsorbate can be described by adsorption isotherms. Of the many models that describe this process, the commonly used Langmuir and Freundlich isotherms were applied in this study to fit the adsorption isotherm data.
The Langmuir isotherm assumes that monolayer adsorption occurs on an adsorbent surface, and there is no interaction between the adsorbate molecules. The Langmuir equation is as follows:

where KL is the Langmuir coefficient (L/mg). Qm is the maximum monolayer adsorption capacity, and Qe is the amount of adsorbed Se (IV) on a mass unit of the adsorbent at equilibrium (mg/g). Ce is the equilibrium Se (IV) concentration (mg/L).
The Freundlich isotherm is an empirical equation, as follows:

where KF and nF are the Freundlich parameter and adsorption intensity, respectively. The nF should be in the range of 0.1–1 for beneficial adsorption.
Release of selenium
According to the optimal reaction conditions, 1000 mg/L of selenium (IV) solution was prepared in a 100 ml conical flask with pH = 3 and then 0.50 g of MNZ was added. After a full reaction of 120 min, the MNZ-Se was filtered by a 0.45 μm cellulose acetate membrane and then dried in an oven at a temperature of 115° and a drying time of 3 h. The selenium content of the loaded MNZ was 35.46 mg/g. The zeolites used in all subsequent experiments were prepared in this manner.
For the release experiment, the MNZ-Se was prepared according to appropriate reaction conditions and placed in distilled water and soil. The reaction of the aqueous solution was performed in a 1000.00 ml conical flask with 500.00 ml distilled water, and the pH of the aqueous solution was adjusted via the addition of the appropriate volume of 1 M HNO3 or NaOH. The pH was measured using a pH metre (PHS-3C), and the pH of the aqueous solution was 3, 5, 7, and 9. Then, 0.20 g MNZ-Se was added into the aqueous solution. The selenium content in the solution was determined after MNZ-Se was placed into the solution for 2, 4, 8, 24, 48, 96, 120, and 240 h. These steps were performed to evaluate the release of selenium by the MNZ-Se in the solution and to determine the effects of the solution pH on the release. All of the experiments were repeated twice.
For the release of selenium by the MNZ-Se in the soil, we first considered the effect of the amount of zeolite, and 0.50and 1.00 g MNZ-Se were evenly mixed into 3.00 kg of soil. Second, the soil was placed in a flowerpot, and the soil water content was adjusted to 30% while the initial pH of the soil was 7.8. To consider the effect of the soil pH on the selenium release, 45.00 g Ca(OH)2 was added into each 3.00 kg soils to adjust the soil pH to 10.8. The soil without MNZ-Se was used as the control in the above experiment. The soil selenium contents were determined after MNZ-Se was placed into the soil for 5, 25, 35, 45, and 60 d by microwave digestion and the atomic fluorescence spectroscopy method. All of the experiments were repeated twice.
Experiment on the uptake of soil selenium by B.c
To study the uptake of selenium in soil by B.c, the following steps were performed. First, different types of fertilizers and MNZ-Se were mixed evenly with the soil, the weight of the MNZ-Se were 1.00 and 2.00 g. The fertilizers were phosphate, nitrogen and potash; the weight of the phosphate was 5.00 g, and the weight of the nitrogen and potash was 3.00 g for each. The effect of the MNZ-Se content on the release was also considered. Second, the mixed soil was placed in a flowerpot, and the B.c seeds were spread on the soil surface evenly. The soil water content was adjusted to 30.00%. The blank control was the same as that described in the step above except that MNZ-Se was not added into the soil. Finally, after the growth of B.c basically stopped, the B.c was collected, and the selenium content in the B.c and the soil were determined. The selenium content was determined twice.
Results and discussion
Characterization of zeolites and adsorption of selenium by zeolites
The chemical compositions of NZ and MNZ are listed in Table 2. The dominant elemental compositions of the zeolite sample showed that aside from the main Si and Al components, Fe is the next highest component, followed by K. The silica to alumina ratios (Si/Al) of NZ and MNZ were 2.25 and 2.29, respectively. The XRD patterns of NZ and MNZ are shown in Figure 1. The main components of NZ and MNZ were quartz and kaolinite. The formula of the zeolite was K90.1Al92Si100O384. By comparing the XRD patterns (Figure 1), SEM images (Figure 2(a) and (b)), and FTIR (Figure 7) of NZ and MNZ, the modification did not change the structure and functional groups of the zeolite. The MNZ was smoother than NZ, and its specific surface area and average pore size (Table 2) were higher than those of NZ.
Dominant chemical compositions, specific surface area, and average pore size of NZ and MNZ.

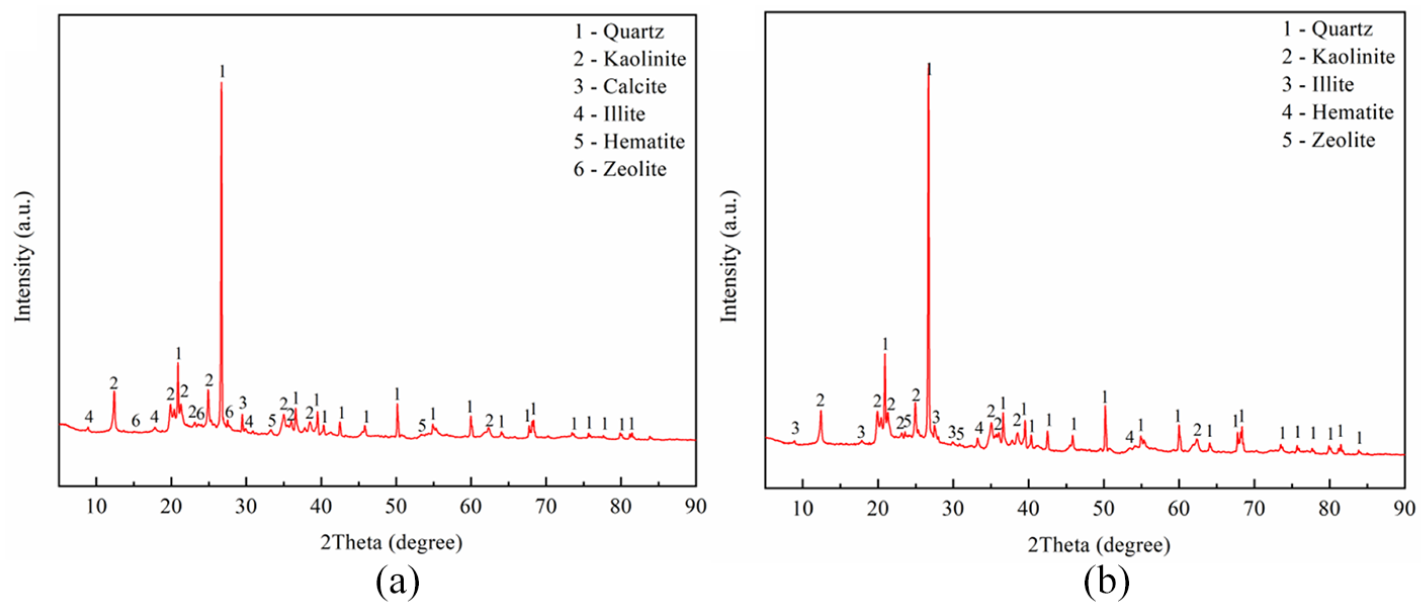
XRD patterns of: (a) NZ and (b) MNZ.

SEM of: (a) NZ, (b) MNZ, (c) NZ-Se, and (d) MNZ-Se.
The results of adsorption kinetics and adsorption isotherms are shown in Figures 3 and 4, Tables 3 and 4. In the sorption of Se (IV) by the NZ, the coefficients of determination for the pseudo-first-order kinetic model (R2 = 0.99) were higher than that for the pseudo-second-order kinetic model (R2 = 0.78). Hence, the pseudo-first-order kinetic model better explained the kinetic process of NZ, suggesting the main effect of physical adsorption. The first-order and second-order kinetics have good fitting results for the adsorption process of selenium on the modified zeolite (R2 = 0.95 and 0.96, respectively), indicating that the adsorption of selenium by the MNZ had both physical and chemical adsorption. The adsorption isotherm fitting results illustrating that the process of adsorption of selenium by NZ and MNZ had both single-layer adsorption and multilayer adsorption, mainly single-layer adsorption.

Adsorption kinetics of Se (IV) by: (a) NZ and (b) MNZ.

Isotherm nonlinear plots on selenium (IV) adsorption by: (a) NZ and (b) MNZ.
Rate constants and correlation coefficients for the studied kinetic models.

Rate constants and correlation coefficients for the studied isotherm models.

In aqueous media, Se (IV) species existed as selenious acid (H2SeO3), biselenite (HSeO3−), and selenite (SeO32−) based on the relationship between Se (IV) dissociation and pH.30,39 The HSeO3− is the dominant species when the pH between 3.5 and 9.0. However, the SeO32− species is dominant above pH 9.0. On the other hand, the H2SeO3 is dominant when the pH is below 3.5.40,41 The point of zero charge (pHPZC) of the NZ and MNZ was at a pH~3.1 and 3.3, respectively, as characterized by zeta potential measurements (Figure 5). At this pH, zeolites are electrically neutral. Below those pH values, the zeolites surface sites carried a positive charge, the acidic water donates more protons, and the adsorbent surface is positively charged to take up the anions. This could enhance the Se (IV) attraction to the surface and increase the adsorption of the Se (IV) at low pH levels. Also, the high sorption at a low pH region is related to protons sufficiency, and it causes the reduction of Se (IV) to elemental selenium that will take place on the adsorbent surface according to the equations (6) and (7).42,43



Zeta potential of NZ and MNZ.
XPS results (Figure 6) also prove this point. Rovira et al. 44 found that the selenium binds strongly to iron oxides in acidic conditions because the surface charge on the iron oxides hematite and goethite are positive. FTIR results showed that the functional groups did not change before and after adsorption (Figure 7), it indicating that this process is not the main mechanism of selenium adsorption. SEM results showed that the adsorption did not change the structure of zeolite (Figure 2).
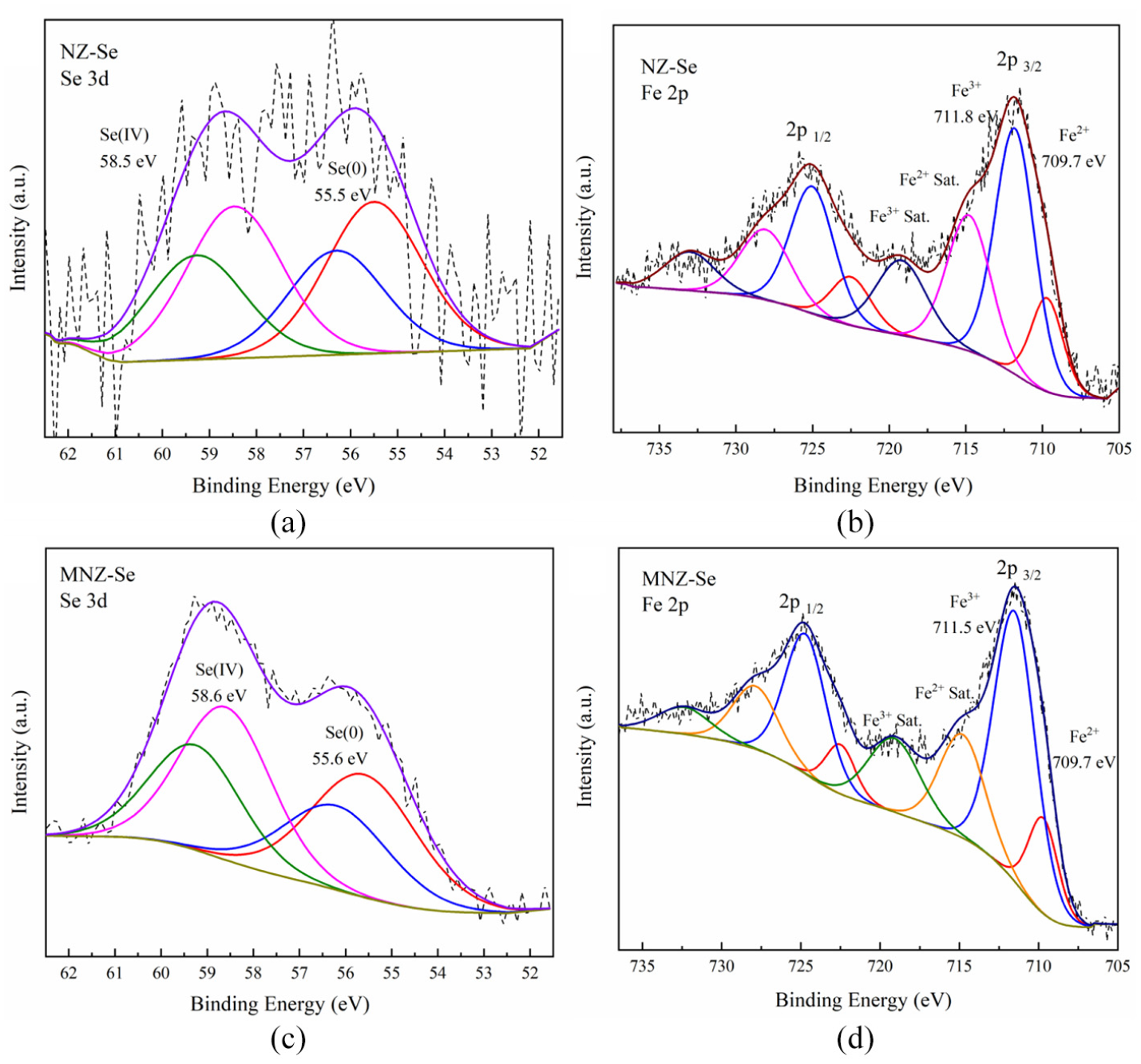
XPS of (a, b) NZ-Se and (c, d) MNZ-Se.
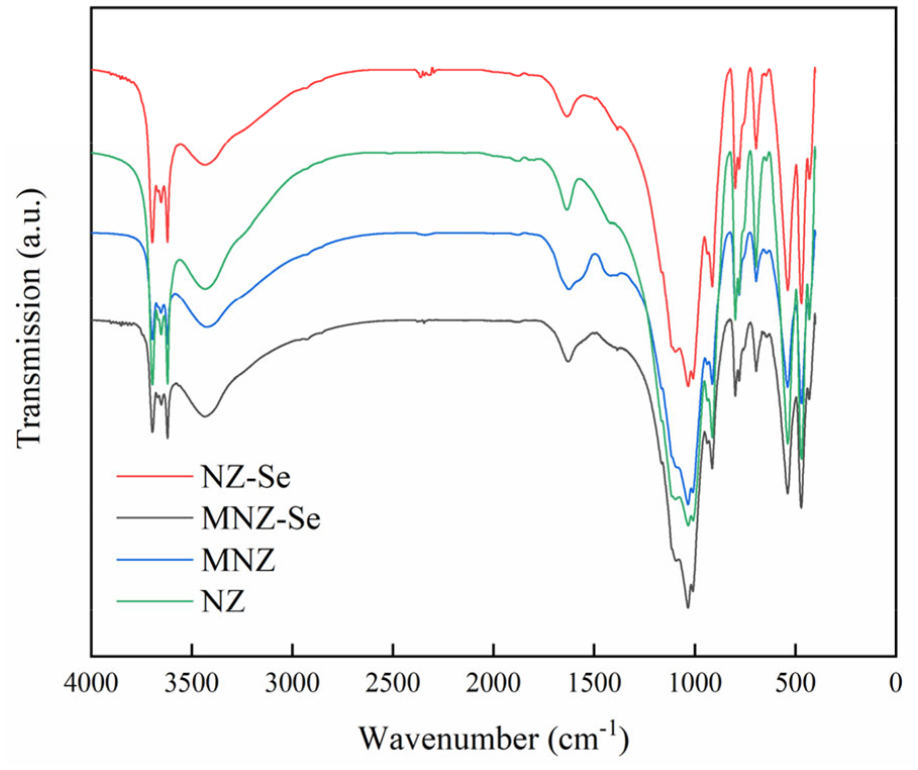
FTIR of NZ, MNZ, NZ-Se, and MNZ-Se.
The maximum selenium (IV) adsorption capacities of NZ and MNZ were 4.63 mg/g and 46.90 mg/g according to the theoretical calculation (Table 4). The results showed that the MNZ had a higher selenium adsorption effect than NZ, as expected, and the reason could be attributed to the increase in the Fe2O3 content and specific surface area (Table 2). The modification provides more iron ions to react with selenium, and the increase of specific surface area provides more contact sites for the adsorption of selenium. Thus, MNZ was chosen to use in further research concerning the release of selenium in the water and soil and the uptake of selenium by B.c.
A comparison of the maximum Se (IV) sorption capacities of adsorbents relating to zeolites is shown in Table 5. The MNZ in this study showed higher sorption capacity in comparison with various other adsorbents.
Comparison of maximum adsorption capacity of Se (IV) on different adsorbents.

Release of selenium in the water
The MNZ-Se was prepared according to the above method, and the average selenium content was 35.46 mg/g. As shown in Figure 8, regardless of how the pH of the aqueous solution changes, the trend of the release of selenium from MNZ-Se in aqueous solution is not influenced. With the increase of time, the content of selenium in the solution gradually decreases and finally tends to be stable. This indicated that release of selenium in water is a dynamic process and the release and adsorption exist simultaneously. When MNZ Se was put into water, a large amount of selenium was released, and the content of selenium in the solution increased significantly, while the amount of selenium adsorbed on zeolite decreases, the adsorption sites of zeolite increase, and thus, the liberated selenium will be adsorbed again. With development over time, this dynamic change process eventually tends to be stable. In the stable period of selenium release, the selenium content was 5.00–5.30 mg/L, and the selenium content on the MNZ-Se was 35.46 mg/g. Thus, 35.26%–37.38% of the selenium on the MNZ-Se was released into the aqueous solution regardless of the influence of pH.
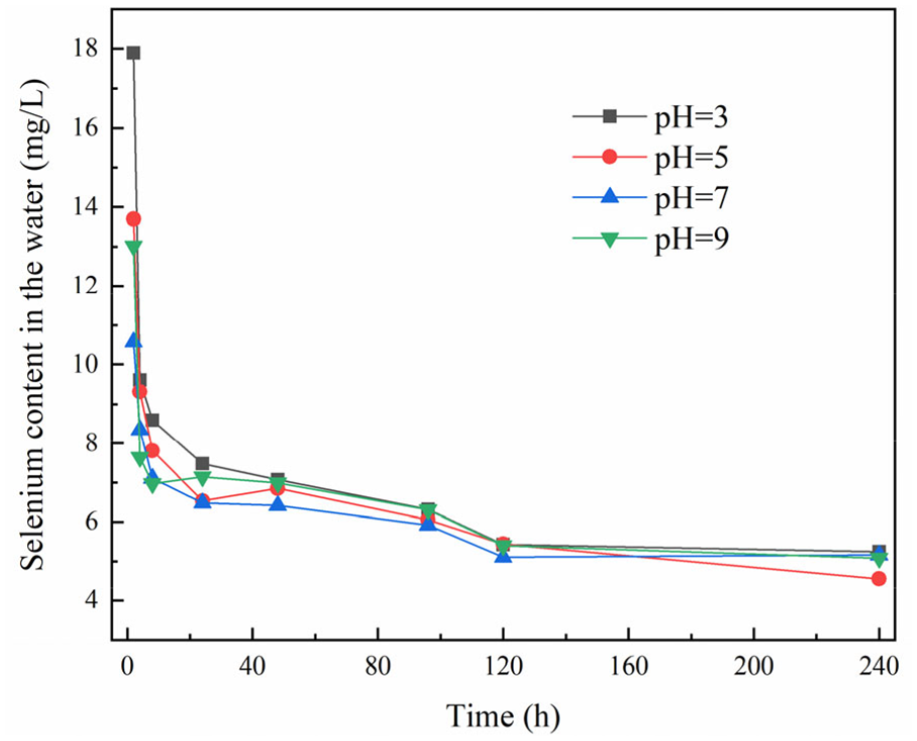
Release of selenium from MNZ-Se in aqueous solutions under different pH values (temperature = 25°C, adsorbent dosage = 0.2 g).
At the beginning of the experiment, the amount of selenium released from MNZ-Se in an aqueous solution is different due to the effect of the solution’s pH. The release of selenium in neutral water is due to the release of selenium adsorbed by the physical effect. In the acidic and alkaline solutions, except for the release of selenium adsorbed by the physical effect, the Se attached to the zeolite surface reacts with the solution and increases the concentration of selenium in the solution according to equations (8) and (9). As the acid and alkali in the solution are gradually consumed, the selenium was adsorbed by zeolite again. Therefore, the content of selenium released from the solution is basically the same with time, regardless of the initial pH of the aqueous solution.


Release of selenium in the soil
As was shown in Figure 9, the selenium content of the soil without zeolite was 0.10 mg/kg, which was lower than 0.40 mg/kg 46 and did not meet the standard of selenium-rich soil. However, after the MNZ-Se was added into the soil, the selenium content in the soil increased and reached the standard of selenium-rich soil. At the same time, when MNZ-Se released selenium to a stable level, the larger the amount of MNZ-Se added into the soil, the larger the amount of selenium released in the soil but the smaller the release ratio from MNZ-Se under the same soil pH conditions. For example, if the soil pH was under 7.8, and 0.50 g MNZ-Se was added into the soil, the content of selenium in the soil would be 0.82 mg/kg, and the release rate would be 13.84%. However, if 1.00 g MNZ-Se was added into the soil, the content of selenium in the soil would be 1.13 mg/kg, and the release ratio would be 9.52%. This relationship means that although the increase in the soil MNZ-Se content will increase the soil selenium content, it will restrain the release of selenium and affect the relative increase in the soil selenium. Therefore, the proper amount of MNZ-Se should be considered in the process of MNZ-Se addition.

Effect of the mass of MNZ-Se on the release of selenium in the soil under different pH values (temperature = 25°C).
Moreover, at the same soil pH, the variation in the selenium released by MNZ-Se over time was consistent. In the soil with a pH value of 7.8, the selenium content in the soil first decreased and then increased before remaining stable. In the soil with a pH value of 10.8, the soil selenium content first decreased and then basically remained stable. Under the same amount of zeolite added, the lower the soil pH, the greater the soil selenium content and the larger the release ratio of selenium from MNZ-Se. For example, when 0.50 g MNZ-Se was added into the soil, the content of selenium in the soil was 0.82 mg/kg, and the release rate was 13.84% when the soil pH was 7.8. However, the content of selenium in the soil was 0.67 mg/kg and the release rate was 11.39% when the soil pH was 10.8. This relationship means that the lower the pH of the soil, the greater the release of selenium from the MNZ-Se in the soil, which in turn increases the soil selenium content. This promotion can be related to the fact that zeolite is an alkaline material. 47 Due to the anionic form of Se, electrostatic forces are likely to dominate sorption-interactions in soil. Selenium can be adsorbed to layered double hydroxides or anionic clays through electrostatic interactions, which are highly pH sensitive. As pH decreases, Se adsorption increases due to fewer negatively charged clay surfaces and sesquioxide edges, resulting in stronger electrostatic interactions. Moreover, soil organic matter is known to influence the retention of Se in soils. Due to their strong adsorption capacity and large specific surface area, soil iron/aluminum oxides and minerals play important roles in the adsorption and fixation of soil Se.48,49 When the soil pH reached 7.8 and the zeolite content was 1.00 g, the soil had the maximum selenium content. When the pH value of the soil reached 7.8 and the zeolite content was 0.50 g, MNZ-Se had a maximum release ratio in the soil, and 13.84% of the selenium on the MNZ-Se was released in the soil; in this case, the release ratio was smaller than the release rate in the water.
Uptake of selenium by B.c in the soil
As was shown in Table 6, with the increase in the content of MNZ-Se in the soil, the selenium content in the soil and B.c increased. According to the standard of selenium-enriched agricultural products, the effective selenium content of B.c was 0.15–0.30 mg/kg. 50 When the B.c was planted in soil without MNZ-Se, the selenium content of B.c did not increase to the selenium-enriched standard, but the selenium content of B.c exceeded the selenium-enriched standard when it was planted in the soil with MNZ-Se. Jevtić et al. 45 used the iron(III)-modified natural zeolitic tuff as an adsorbent and carrier for selenium oxyanions and found that samples of zeolitic tuff that contain selenium could be a good soil supplement for the cultivation of Pleurotus ostreatus mushrooms. This finding proves that using zeolite to adsorb selenium and placing it in soil can promote the absorption of selenium by plants.
The selenium content in the soil and B.c under different content of MNZ-Se in the soil.

To obtain the proper amount of MNZ-Se, we used the data on the selenium content in B.c (Y) and the content of MNZ-Se (X) to make a linear fit. The equation Y = 2.09 × X + 0.03 (R2 = 0.98) was obtained. The results showed that 0.06–0.13 g/kg MNZ-Se which contained selenium content 35.46 mg/g should be added into the soil to obtain the selenium-enriched B.c. However, this type of fitting provides only a prediction, and the zeolite addition amount still must be tried and tested in practice.
Application prospects
The above research shows that MNZ can be a potential medium for water pollutant removal and soil improvement, which provides a new idea for the reuse of decontamination material and adsorption test production. However, in practical applications, there are many factors that must be carefully considered. First, multiple pollutants exist in actual wastewater. The application of MNZ to the adsorption of pollutants in wastewater must consider the impact of these pollutants other than the target pollutants. Second, the choice of target pollutants must be combined with actual life needs, such as ammonium and phosphate. Ammonium and phosphate are common coexisting pollutants in wastewater and can also be effectively adsorbed by zeolite; this approach is also a feasible method to use MNZ to adsorb and reuse them. Third, although MNZ is an environmentally friendly material, the application of MNZ to adsorb pollutants and apply it to the soil will affect the properties of the soil. The duration of this effect is not fixed. Therefore, in-depth research is required to avoid the secondary application of MNZ to soil pollution. Finally, the results also showed that the amount of MNZ after the adsorption of pollutants should be accurately calculated to obtain the appropriate soil improvement results.
Conclusions
Based on the above research results, we reached the following conclusions: (1) Water release experiments of modified natural zeolite loaded with selenium showed that regardless of how the pH value of the aqueous solution changes, the trend of the release of selenium from MNZ-Se in aqueous solution is not affected, and the trend first decreased and then remained stable. (2) For the soil release experiments, after the MNS-Se was added into the soil, the selenium content in the soil increased and reached the standard of selenium-rich soil. (3) With the increase of content of MNZ-Se in the soil, the selenium content in the soil and B.c increased. (4) MNZ can be a good medium for water pollutant removal and soil improvement, which provides a new idea for the reuse of contaminated materials and adsorption test production.
Supplemental Material
sj-docx-1-sci-10.1177_00368504211019845 – Supplemental material for Selenium in wastewater can be adsorbed by modified natural zeolite and reused in vegetable growth
Supplemental material, sj-docx-1-sci-10.1177_00368504211019845 for Selenium in wastewater can be adsorbed by modified natural zeolite and reused in vegetable growth by Xiao Zhang, Xinyuan Li, Zihao Jin, Sadam Hussain Tumrani and Xiaodong Ji in Science Progress
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major Science and Technology Program for Water Pollution Control and Treatment of China (No. 2017ZX07101002-002). We gratefully acknowledge the Beijing Municipal Education Commission for their financial support through Innovative Transdisciplinary Program “Ecological Restoration Engineering”.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
