Abstract
The indiscriminate application of synthetic pesticides on horticultural plants for the management of Bactrocera zonata has both economic and environmental implications due to the biomagnification of harmful residues in humans via the food chain. This necessitates the use of some eco-friendly control measures such as insect growth regulators (IGRs) as an alternative. A laboratory experiment was laid out to determine the potential chemosterilant effect of five IGRs including pyriproxyfen, novaluron, lufenuron, buprofezin, and flubendiamide at six different concentrations on B. zonata after treating adult diet. Using the oral bioassay, B. zonata were fed on IGRs impregnated diet (50–300 ppm/5 mL diet) which was replaced with the normal diet after 24 h of feeding. Ten pairs of B. zonata were placed in a separate plastic cage accommodating an ovipositor attractant guava for eggs collection and calculation. An analysis of the result revealed that fecundity and hatchability were higher at a low dose and vice versa. Lufenuron at 300 ppm/5 mL of diet significantly decreased the fecundity rate (31.1%) as compared to pyriproxyfen, novaluron, buprofezin and flubendiamide, which had 39.3%, 39.3%, 43.8%, and 47.5%, respectively. The lowest hatchability (19.9%) was noted in lufenuron treated diet followed by pyriproxyfen, novaluron, buprofezin, and flubendiamide, which had 22.1%, 25.0%, 30.9%, and 31.6%, respectively. Furthermore, in a population of crosses between the lufenuron treated male and female, a significant decrease in fecundity (45.5%) and hatchability (51.7%) was noted as compared to other IGRs. Overall, this study identified the chemosterilant potential of lufenuron on the population of B. zonata which can be integrated for its management strategy.
Introduction
Among tephritids, the peach fruit flies (Bactrocera zonata S.) (Tephritidae: Diptera) is one of the most devastating, dominant, and polyphagous pests of vegetables and fruits around the globe.1,2 It is widely distributed in the tropical and sub-tropical zones such as the Indian subcontinent, most parts of Africa, Arabian states, and also in the Americas.1,3–5 This obnoxious pest (B. zonata) occurring in different ecological regions of Pakistan often causes up to 100% losses in different fruits.6,7 European Plant Protection Organization categories B. zonata as an A1 key quarantine pest 8 by several countries 9 and being polyphagous, 10 tephritidae flies attack on several fruit and vegetable species such as mango (Mangifera indica), guava (Psidium guajava), peach (Prunus persica), fig (Ficus carica), apple (Malus domestica), apricot (Prunus armeniaca) and tomato (Solanum lycopersicum), pepper (Piper nigrum) and eggplants (Solanum melongena) as secondary hosts. 11 In addition, the environmental conditions (temperature, rainfall and relative humidity (RH)) are also known to influence the extent of infestation which may vary between 30% and 100%.12,13
Globally, numerous conventional control measures like behavioural, biological, chemical, genetic, mechanical and microbial are being used to eradicate fruit flies.14,15 Male annihilation techniques (MAT) against fruit flies with synthetic chemicals (methyl eugenol, med lure, cue-lure etc.) were used for monitoring, mass trapping, suppression and mating disruption. 16 In Pakistan, the management of fruit flies largely depends upon synthetic pesticides 17 due to their quick knockdown effect. 18 This might lead to the development of insecticide resistance and the critical issues of pest resurgence, environmental pollution, pesticide residues, detrimental effects on beneficial fauna, and health hazards. 19 Another drawback of the chemical sprays is that 3rd instar larva leaves the rotten fruits and pupates in soil, so both larvae in fruits and pupae in soils are well protected from the applied insecticides. 20 Furthermore, in the case of sterile insect technique, an incredible number of irradiated flies (billions per week) are needed which is not only regarded a costly eradication programme but also other issues such as personal safety, maintenance of machinery, and availability of the facility are associated with it. 21
IGRs have been proposed as insect control agents 22 as they harbour compounds that impede insect growth by interfering with chitin synthesis or mimicking the juvenile hormone (JH), 20-hydroxyecdsyone hormone and moulting hormone. 23 That is why this class of chemicals is known to kill insects more slowly than conventional insecticides. 24 IGRs interfere with the moulting process, metamorphosis processes, 25 normal growth and development, 26 resulting in abnormalities in the insect. Due to their activity through hormonal pathways, IGRs have no lethal effects on other vertebrates 27 and non-target organisms. 24 In chemosterilization, IGRs either upset the development of an insect's embryo or block the sexual maturity 28 which results in a decrease in the infestations as the young insects are unable to replicate. 29
Chemosterilant activity of IGRs (Methoxyfenozoide, Tebufenozide, Tepa, Hempa and Apholate) is well established against other insect-pests like Bactrocera oleae, 30 B. dorsalis, B. cucurbitae, B. latifrons, C. capitata, 31 A. ludens, A. obliqua, A. serpentina and A. striata.32,33 Typically, IGRs acting as chemosterilants affect the fecundity and hatching efficiency of the target insects. 34 Keeping in view the future needs of human beings in the context of organic farming, we evaluated the chemosterilant effect of IGRs against the population of B. zonata for the development of a successful management strategy for both sexes (male and female) of the fruit fly. Furthermore, using the bioassays under laboratory conditions, we assessed the toxicity of IGRs impregnated adult diet on the fecundity, hatchability, and malformation of B. zonata at various stages of development.
Materials and methods
Experimental layout
The experiment was laid out in an insect rearing laboratory at the Institute Plant Protection (IPP) in Muhammad Nawaz Sharif University of Agriculture (MNSUA), Multan under a factorial (in cages) completely randomized design (CRD) with five replications.
Insect collection, rearing and biological material
Mango fruits infested with the larvae of B. zonata were collected from different mango (Mangifera Indica L., Anacardiaceae) orchards in the vicinity of MNSUA, Multan. The infested fruits were taken into the rearing laboratory and stored in card boxes half-filled with sterilized sand and sieved out after 14 days by using a fine mesh until the larvae transformed into the pupal stage. The sand was oven-sterilized for 1 h at 160 °C. The collected pupae from the infested fruits were placed in a Petri dish inside wooden cages till hatchability. The emerging adults of B. zonata were reared on an artificial, unnatural diet in insect-rearing wooden cages (60×40×40 cm) made with (2×2 cm) wire meshing to maintain a constant temperature and ventilation. The room was provided with heat and fluorescent light systems. Insect rearing conditions were maintained at 65–75% of RH, 25 ± 2 °C temperature, and a photoperiod of 12 h of dark and 12 h of light. The emerged flies were fed on a mixture of sugar and yeast (3:1) with a small amount of water. In addition, to ensure moisture availability a plastic cup with a wet cotton wick was placed in the cage. A one-inch square green card soaked with guava juice and water was used to stimulate flies to lay eggs. Deposited eggs were collected using a camel hairbrush and the collection of eggs continued until the adults attained the age of 30 days after hatching from eggs.
Rearing of larvae
To prevent desiccation, collected eggs were kept in the semi-artificial diets inside plastic trays (15×10×3 cm) covered with a muslin cloth until egg hatching. Before larval maturation, 0.5 kg wet sterilized sand was kept beneath the plastic trays for pupation of the mature larvae.
Preparation of artificial diet
To prepare an artificial diet having a total weight of 500 mL (by volume), banana (250 g), sugar (50 g), honey (∼ 4.2 g), yeast (1 g), egg yolk (50 g), syrup of vitamin B complex (0.2–0.4 mL) and distilled water were mixed together in an electric blender to obtain a homogenous paste.
Species identification key
For species identification, the morphological features of captured flies were compared with the available standard taxonomic keys. 35 B. zonata is approximately the size of a house fly and is primarily red-brown in colour. The absence of a complete costal band (reduced to an isolated apical spot) in the wing. Examine the thorax in the dorsal view now. There should be a narrow lateral yellow vitta (stripe) down each side (in the posterior two-thirds only). A third (medial) vitta should not exist down the centre. Finally, examine the abdomen. The markings on the abdomen vary more than the markings on the face, wings and thorax. Except for tergite V, there is usually a pair of dark marks on tergite III and no medial dark line. 36
Evaluation of IGRs
Five synthetic insecticides (IGRs) including lufenuron, novaluron, buprofezin, pyriproxyfen and flubendiamide were evaluated against the population of B. zonata fly under laboratory conditions (Table 1).
List of chemicals used as chemosterilant against B. zonata bioassay.

Preparation of insecticide solution
Before preparation of the stock solution, the glass beakers were cleaned with 70% ethanol and distilled water and finally dried up with hot air. A stock solution was prepared for each mentioned chemical (Table 1). The volume of each IGR's formulated solution necessary to produce the requisite stock-solution was determined using the dilution equation: C1V1 = C2V2. Six serial dilutions of 50, 100, 150, 200, 250 and 300 ppm of each stock solution and one control were diluted with distilled water until the required original dilution was attained. 37 Each IGR was prepared in six concentrations of 50, 100, 150, 200, 250 and 300 ppm to study its chemosterilant effect on B. zonata in the insect bioassays.
Experiment 1
Insect bioassay
For each IGR, 35 experimental units were constructed and placed on the iron stand having 288×164×18 cm length, height and width, respectively, and the distance between the two sections was 24 cm. The experiment included five IGRs with six different concentrations (50, 100, 150, 200, 250 and 300 ppm) of IGRs and one control treatment, which was replicated five times in CRD. Newly emerged adults of the age of 10 days (10 pairs/cage) of B. zonata were placed in a separate plastic cage (6×4×4 cm) with a mesh net covering both opposite sides for ventilation. Prior to bioassay, all the flies were subjected to starvation stress for 24 h to enhance their feeding behaviour. The size of each plastic jar was 21 cm in height and 8 cm in radius (4224 cm3). For bioassays, the adult diet (30 mL) was admixed with various concentrations of IGRs. After 24 h of feeding on the treated diet, the IGR-admixed-diet was replaced with normal diet for the rest of the experiment. As mentioned above, an ovipositor attractant guava (egg-receptacle) inside each plastic cage served as one experimental unit for the collection of eggs. The eggs were collected from each cage every day until the last day of oviposition. To calculate the fecundity rate, total number of eggs per cage was divided by the total number of females per cage to calculate fecundity by the following formula:

Malformation parameters
In addition, various morphogenetic anomalies in adult fruit flies (small size body, crumbled wings, larval to pupal, and pupal to adult intermediates) were sorted and counted using a hand lens.
Experiment 2
Based on the findings of experiment 1, this experiment was designed to determine if increased dose contribution resulted in maximum sterility in the form of lower fecundity and hatchability of each sex in B. zonata. The methodology was identical to that described above, but the bioassays were performed at the highest concentration (300 ppm/5 mL of diet) of all IGRs. Based on the sex, the newly emerged adults (10 days old) were divided into males and females (50 flies per cage), of which one sex was fed a diet containing 300 ppm treated diet. After 24 h of diet exposure, the following groups, that is, 10 pairs per cage with five cages/treatment (replicates) were established treated males × treated females (TM × TF), treated males × untreated females (TM × UF) and untreated males × treated females (UM × TF). Fecundity and hatchability in percentage were assessed in the prior experiment, feeding time was finished after 24 h for each treatment and the rest of the trial was accompanied by a regular diet. The oviposition position was made up of a green linen cloth soaked in guava juice to prevent egg dehydration in each cage. Insects were provided with usual food and water until the exhaustion of the oviposition period. The eggs were collected until the natural mortality of all B. zonata adults.
Experiment 3
To determine which IGRs caused the sterility in B. zonata sex at a higher dose (300 ppm/5 mL of treated diet), on which application day, and at which cross. Oral toxicity of IGRs admixed diet on B. zonata adults (10 days old) after 1, 2, 3, 4, 5 and 6 days was evaluated separately in the plastic cages. The concentration of chemicals was mixed in acetone (5 mL) and added further to the artificially prepared habitual diet (described above) and then the solvent was allowed to evaporate. Feeding time of both males and females was exhausted after 24 h for 1 day treated diet, subsequently 48, 72, 96, 120, and 144 h from 2, 3, 4, 5, and 6 days subsequently of each treatment. After ingestion, the remainder of the trial was supplemented by a regular diet. The following groups, that is, 10 pairs per cage with five cages/treatment (replicates) established treated males × treated females (TM × TF), treated males × untreated females (TM × UF), untreated males × treated females (UM × TF) and untreated males × untreated females (UM × UF). Data pertaining to fecundity and hatchability in percentage was assessed as mentioned in the above experiment.
Statistical analysis
The experiment layout for all experiments was CRD with factorial arrangements. For the 1st experiment, factors included type of IGRs and doses of IGRs, for 2nd experiment, factors were conspicuous crosses and optimized doses of IGRs from experiment 1, and for 3rd experiment, the factors were crosses and number of feeding days. Data was subjected to two-way analysis of variance to determine the parameters of significance, and the mean values for different treatments were compared using the Bonferroni significant difference test, by using STATISTICA-10 statistical software. 38
Results
Effect of IGRs concentrations on fecundity reduction and hatchability
Analysis of data revealed the chemosterilizing effect of IGRs, as well as their concentrations (50, 100, 150, 200, 250 and 300 ppm/5 mL of diet) significant effects (P < 0.05) on the fecundity reduction (eggs/female) and hatchability (fertility) of B. zonata.
Effect of different IGRs concentrations on fecundity
The egg-laying potential of B. zonata was greatly reduced when fed upon varying concentrations of IGRs impregnated diet. Analysis of the data revealed that all the evaluated IGRs exhibited varying degrees of sterility (in fecundity reduction, hatchability and malformation) against the population of B. zonata (Table 2).
Analysis of variance parameters regarding fecundity reduction and hatchability of B. zonata treated with different concentrations of lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide (total df = 174; error df = 140).

The fecundity data (means ± standard error of means) of B. zonata adults fed upon 50 ppm/5mL of diet showed substantial variations across the treatments. Females feeding on a lufenuron treated diet deposited the lowest number of eggs (12.60 ± 0.65), followed by pyriproxyfen (14.40 ± 0.61), novaluron (15.40 ± 0.84), buprofezin (15.80 ± 1.34) and flubendiamide (18.80 ± 1.21) as compared to the control (21.50 ± 1.25). On a percentage basis, lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide exhibited the lowest fecundity of 58.6%, 67%, 71.6%, 73.5% and 87.4%, respectively, compared to the control (Figure 1(A)).

Effect of different concentrations at 50, 100, 150, 200, 250 and 300 ppm/ 5mL denoted by A, B, C, D, E and F on fecundity (percentage ± SE) of Bactrocera zonata, treated with lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide and control. Bars indicate standard errors; the same style letters don’t differ significantly at the probability level of 5%. Treatments are on the x-axis and different dependent parameters are on the y-axis.
Similarly, females fed on 100 ppm/5 mL of lufenuron treated diet also deposited the lowest number of eggs (11.80 ± 0.52), followed by pyriproxyfen (13.00 ± 0.69), novaluron (14.80 ± 0.66 eggs/female), buprofezin (14.20 ± 1.02) and flubendiamide (17.80 ± 4.05) in comparison to the control (20.80 ± 1.52 eggs/female). On a percentage basis, lufenuron, pyriproxyfen, buprofezin, novaluron and flubendiamide exhibited the lowest fecundity of 56.7%, 62.5%, 68.3%, 71.2% and 85.6%, respectively, as compared to the control (Figure 1(B)).
A significant variation was noticed in the fecundity of B. zonata females feeding on 150 ppm/5 mL of diet. The females feeding on the lufenuron treated diet deposited the least number of eggs (11.20 ± 0.82), followed by pyriproxyfen (11.40 ± 0.77), novaluron (12.40 ± 0.34), buprofezin (13.00 ± 0.75) and flubendiamide (16.60 ± 2.07) as compared to the control (23.60 ± 1.62). On a percentage basis, lufenuron, pyriproxyfen, novaluron, pyriproxyfen, buprofezin and flubendiamide exhibited the least fecundity of 47.5%, 48.3%, 52.5%, 55.1% and 70.3% as compared to the control (Figure 1(C)).
Fecundity of adult B. zonata fed on 200 ppm/5 mL of diet showed substantial variations between the treatments. The females feeding on lufenuron mixed diet laid the lowest number of eggs (10.00 ± 0.79) followed by pyriproxyfen (10.30 ± 0.46), novaluron (12.40 ± 0.40), buprofezin (12.00 ± 0.97) and flubendiamide (15.40 ± 3.07) as compared to the control (22.50 ± 1.82). On a percentage basis, lufenuron, pyriproxyfen, novaluron, pyriproxyfen, buprofezin and flubendiamide exhibited the lowest fecundity of 44.4%, 45.8%, 55.1%, 53.3% and 68.4%, respectively, as compared to the control (Figure 1(D)).
The productivity of adult females B. zonata fed with 250 ppm/5mL of diet. The fruit flies fed on lufenuron treated diet deposited the least number of eggs (8.80 ± 0.54) followed by pyriproxyfen (9.60 ± 0.46 eggs/female), novaluron (10.40 ± 0.73), buprofezin (11.00 ± 0.79) and flubendiamide (13.00 ± 4.15) as compared to the control (23.80 ± 1.89). On a percentage basis, lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide exhibited the lowest fecundity of 36.9%, 40.3%, 43.7%, 46.2% and 54.6% as compared to the control (Figure 1(E)).
Fecundity of B. zonata adults fed with 300 ppm/5 mL of diet, females, treated lufenuron diet deposited the fewest number of eggs (6.80 ± 0.92), followed by pyriproxyfen (8.60 ± 0.52), novaluron (8.60 ± 0.61), buprofezin (9.60 ± 0.67) and flubendiamide (10.40 ± 0.75) as compared to the control (21.90 ± 2.31). On a percentage basis, lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide exhibited the lowest fecundity of 31.1%, 39.3%, 39.3%, 43.8%, and 47.5%%, respectively, as compared to the control (Figure 1(F)).
Effect of different IGRs concentration on hatchability
Different IGRs used in this study significantly influenced the hatchability of adult B. zonata from pupae. Higher hatchability was seen at lower concentrations (50 ppm/5 mL), while the lowest hatchability was noted at higher concentrations (300 ppm/5mL). Analysis of data revealed that IGRs impregnated diet (50 ppm/5 mL of diet) of B. zonata, lufenuron exhibited the lowest hatchability of 57%. Whereas pyriproxyfen, buprofezin, novaluron and flubendiamide treated female B. zonata diet exhibited the hatchability of 64.5%, 64.5%, 68.8% and 88.2%, respectively, as compared to control (Figure 2(A)).

Percent egg hatching of Bactrocera zonata treated with lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide irrespective of various concentrations at 50, 100, 150, 200, 250 and 300 ppm/5mL denoted by A, B, C, D, E and F and control. Bars indicate standard errors and the same style letters don’t differ significantly at the probability level of 5%. Treatments are on the x-axis and different dependent parameters are on the y-axis.
Female B. zonata treated diet with the IGRs at 100 ppm/5 mL diet that lufenuron exhibited a minimum hatchability of 52.7%. However, the hatchability of female B. zonata treated with novaluron, buprofezin, pyriproxyfen and flubendiamide showed to be 64.5%, 65.6%, 70.5% and 83.9%, respectively, as compared to the control (Figure 2(B)). Data of the female adults of B. zonata fed on diet treated with IGRs at 150 ppm/5 mL of diet exhibited that the least number of adults (37.4%) emerged from the lufenuron treatment followed by pyriproxyfen (39%), novaluron (39.8%), buprofezin (45.4%) and flubendiamide (56.5%), respectively, as compared to control (Figure 2(C)).
For the adults of B. zonata fed on 200 ppm/5 mL of diet, the hatchability data differed considerably across the treatments. Lufenuron treated diet exhibited the least hatchability of 27.3% followed by pyriproxyfen (29.5%), novaluron (36%), buprofezin (38.1%) and flubendiamide (46%), respectively, as compared to the control (Figure 2(D)). B. zonata diet treated with the IGRs (250 ppm/5 mL diet) revealed the least hatchability (23.5%) of the lufenuron treatment. However, the percentage hatchability with pyriproxyfen, novaluron, buprofezin and flubendiamide was found to be 26.5%, 25.7%, 33.8% and 41.9%, respectively, as compared to the control (Figure 2(E)).
The percentage of hatchability of B. zonata fed on a diet treated with IGRs at 300 ppm/5 mL diet was also found to be the lowest (19.8%) in lufenuron treatment. However, with pyriproxyfen, novaluron, buprofezin and flubendiamide it was noted at 22.1%, 25%, 30.9% and 31.6%, respectively, as compared to the control (Figure 2(F)).
Crosses among B. zonata at 300 ppm/5 mL diet of different chemicals
Overall, an analysis of the data in the above experiments revealed that a higher fecundity was found at a lower concentration of 50 ppm/5 mL diet while a lower fecundity was noted at a higher concentration (300 ppm/5 mL diet). Next, conspecific crosses were created to determine the substantial influence of these crosses on the fecundity and hatchability at 300 ppm/5 mL of diet in conjunction with different crosses made among B. zonata males with females (Table 3).
Analysis of variance parameters regarding fecundity reduction and hatchability of B. zonata treated with different concentrations of lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide (total df = 74; error df = 60).

Effect of different IGRs on the fecundity reduction of B. zonata crosses
While crossing treated males with treated females (TM×TF), it was found that the female B. zonata fed with lufenuron oviposited the lowest percentage of eggs (36.6%). While for novaluron, buprofezin, pyriproxyfen, and flubendiamide the percentage of laid eggs was 58.7%, 57.3%, 57.2% respectively (Figure 3(A)). Meanwhile, crossings were conducted between treated males and untreated females (TM×UF) at 300 ppm/5 mL of the diet of various IGRs. The highest fecundity (81.5%) was noted in the flubendiamide treatment followed by pyriproxyfen (72.2%), novaluron (67.6%), buprofezin (59.1%) and the lowest fecundity (51.8%) was observed in the lufenuron treated diet (Figure 3(B)).

Percentage fecundity (eggs/female) of Bactrocera zonata treated with different Insect Growth Regulators (IGRs) of lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide at concentrations of 300 ppm/5mL of treated diet during different crosses of TM × TF (A), TM × UF (B), UM × TF (C) and UM × UF (D). The same style letters don’t differ significantly at the probability level of 5%. Treatments are on x-axis and different dependent parameters are on y-axis. TM (treated male), TF (treated female), UM (untreated male) and UF (untreated female).
However, crosses were developed between untreated males and treated females (UM×TF) at 300 ppm/5 mL of the treated diet of various IGRs. For these crosses, the highest fecundity rate (73%) was noted in the flubendiamide treatment followed by pyriproxyfen (66.6%), novaluron (63%), and buprofezin (57.6%) respectively and the lowest fecundity (45.5%) was observed in with lufenuron (Figure 3(C)). Overall, the results showed that lufenuron was an effective IGR that had a significant effect on B. zonata fecundity during crosses, with the highest fecundity affected when crosses were made between both treated males and females followed by treated females with untreated males, treated males with untreated females and untreated male with untreated females (Figure 3(D)).
Effect of different IGRs on hatchability of b. zonata crosses:
The hatchability of adults from pupae during crossings with different IGRs at an optimal dose (300 ppm/5 mL of diet) had a significant impact on the population of B. zonata. While crossing treated males with treated females (TM×TF), the highest hatchability (76.2%) was noted in flubendiamide followed by novaluron (65.6%), pyriproxyfen (61.1%), buprofezin (51.9%) while the lowest hatchability (36.5%) was observed in the lufenuron treatment (Figure 4(A)). Meanwhile, crossings were conducted between treated males and untreated females (TM×UF) at 300 ppm/5 mL of treated bait. For these crosses, the highest percentage hatchability (80.8%) was noted in the flubendiamide treatment followed by novaluron (74.8%), pyriproxyfen (74.2%), buprofezin (62.2%) and the lowest number (59.1%) was recorded in the lufenuron treated diet (Figure 4(B)).
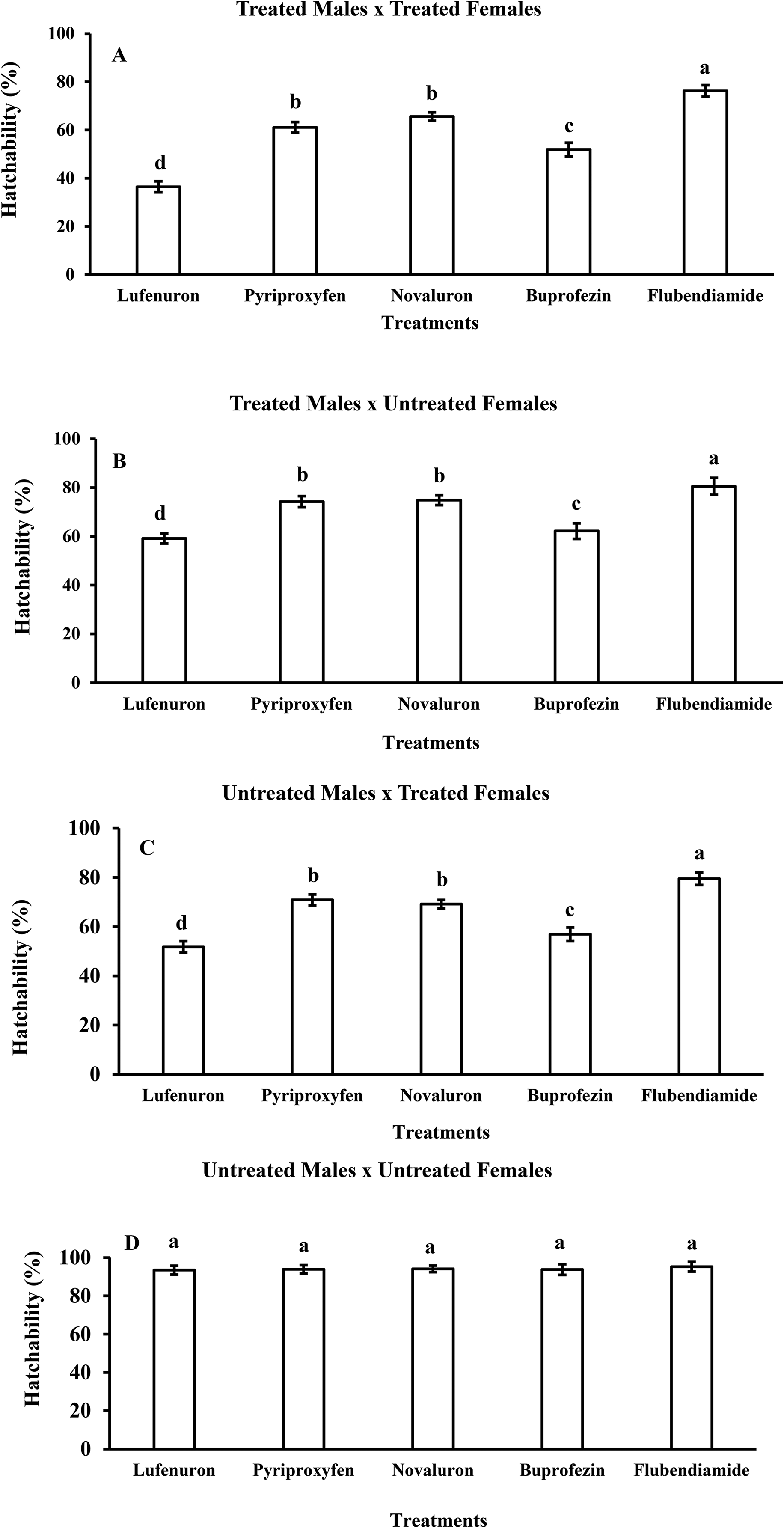
Percentage hatchability of Bactrocera zonata treated with different Insect Growth Regulators (IGRs) of lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide at concentrations of 300 ppm/5mL of treated diet during different crosses of TM × TF (A), TM × UF (B), UM × TF (C) and UM × UF (D). The same style letters don’t differ significantly at the probability level of 5%. Treatments are on the x-axis and different dependent parameters are on the y-axis. TM (treated male), TF (treated female), UM (untreated male) and UF (untreated female).
Similarly, crosses were developed between untreated males and treated females (UM×TF) at 300 ppm/5 mL baits of various IGRs. For these crosses, the highest hatchability (79.5%) was noted in the flubendiamide treatment followed by pyriproxyfen (70.9%), novaluron (69.2%), buprofezin (56.9%) and the lufenuron treatment exhibited the lowest hatchability of 51.7% (Figure 4(C)) as compared to the control (Figure 4(D)). Overall, an analysis of the results showed the potency of lufenuron as an effective IGR that considerably affected the hatchability of pupae. During the crosses, the lowest hatchability was affected when both treated males were crossed with females, followed by treated females crossed with untreated males, treated males crossed with untreated females and untreated males with untreated females.
Effect of different IGRs at 300 ppm/5 mL diet on fecundity reduction and hatchability during crosses of continuously feeding 1 to 6 days application
To investigate the effect of continuously feeding of a treated diet of different IGRs for 1 to 6 days application, crosses were made between treated males and treated females, treated males and untreated females, and untreated males and treated females to assess fecundity reduction and hatchability of B. zonata during crosses (Table 4).
Analysis of variance parameters showing fecundity reduction and hatchability of B. zonata treated with 1 to 6 days application in conspecific crosses (TM×TF, TM×UF, TF×UM and UM×UF) at a concentration of 300 ppm/5 mL diet of lufenuron, pyriproxyfen, novaluron, buprofezin, flubendiamide (total df = 119; error df = 96).

TM: treated male; TF: treated female; UM: untreated male; UF: untreated female.
Effect on fecundity reduction during crosses for 1–6 days application of treated diet
The fecundity of the B. zonata was likewise affected in various crosses when the number of applications treated diet in days (1–6 days) was fed continuously. The number of applications in days at the same concentration (300 ppm/5 mL) decreased the fecundity observed.
A. Effect on fecundity reduction during crosses for 1-day application
Assessment of crosses (TM×UF) revealed that the highest fecundity was observed in flubendiamide (24.41 ± 2.18) followed by buprofezin (23.16 ± 2.41), novaluron (21.82 ± 1.68), pyriproxyfen (20.04 ± 1.66), while the lowest (18.27 ± 1.43) was noted with lufenuron. For UM×TF crosses, flubendiamide exhibited the highest fecundity (23.75 ± 2.74) followed by buprofezin (21.95 ± 1.62), novaluron (19.97 ± 1.53) and pyriproxyfen (17.92 ± 1.42) while the lowest (15.83 ± 1.42) rate was found in the lufenuron treatment. In case of TM×TF crosses, the highest fecundity (23.29 ± 2.19) was noted in the flubendiamide treatment followed by buprofezin (21.30 ± 1.19), novaluron (18.35 ± 2.44) and pyriproxyfen (15.02 ± 1.22) while the lowest (12.04 ± 2.72) was observed in lufenuron treatment. However, for UM×UF crosses, it was found to be statistically similar across all the treatments including flubendiamide (25.90 ± 2.26), novaluron (25.20 ± 3.70), pyriproxyfen (25.10 ± 2.77), lufenuron (25.10 ± 2.30) and buprofezin (24.90 ± 2.04) (Figure 5(A)).

Conspecific crosses of B. zonata (means ± SE) on the fecundity reduction of different crosses TM×TF, TM×UF, UM×TF and UM×UF at a concentration of 300 ppm/5mL diet of lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide. Fecundity is often influenced in days when it comes to crosses, for one 1, 2, 3, 4, 5 and 6 applications of diet denoted A, B, C, D, E and F at a significant probability level of 5%.
B. Effect on fecundity reduction during crosses for 2-day application
While assessing the effect of continuous 2-day application of treated diet (300 ppm/5 mL) of different IGRs, it was found that the highest fecundity was observed in crosses TM×UF for flubendiamide (23.03 ± 1.65) which was followed by buprofezin (21.78 ± 1.88), novaluron (20.44 ± 1.15) and pyriproxyfen (18.66 ± 1.13) while the lowest fecundity (16.89 ± 2.90) was noted with lufenuron. For UM×TF crosses, the highest fecundity (22.11 ± 2.21) was observed in flubendiamide followed by buprofezin (20.31 ± 1.09), novaluron (18.33 ± 2.10) and pyriproxyfen (16.28 ± 1.89) while the lowest fecundity (14.19 ± 1.89) was noted with lufenuron (Figure 5(B)). In case of TM×TF crosses, the highest fecundity (22.01 ± 1.66) was found with flubendiamide followed by buprofezin (19.92 ± 1.66), novaluron (16.97 ± 2.11) and pyriproxyfen (13.64 ± 1.69) while the lowest fecundity (10.66 ± 2.19) was noted in lufenuron treatment in contrast to untreated males with untreated females.
C. Effect on fecundity reduction during crosses for 3-day application
While assessing the effect of continuous 3-day application of treated diet (300 ppm/5 mL) of different IGRs (Figure 5(C)), it was found that the highest fecundity was noted in crosses TM×UF for flubendiamide (22.43 ± 1.58) which was followed by buprofezin (21.18 ± 2.81), novaluron (19.84 ± 2.08) and pyriproxyfen (18.06 ± 1.06) while lufenuron exhibited the lowest fecundity of 16.29 ± 2.73. For UM×TF crosses, the highest fecundity (22.21 ± 2.14) was noted with flubendiamide followed by buprofezin (20.41 ± 2.02), novaluron (18.43 ± 1.93) and pyriproxyfen (16.38 ± 1.42) while the lowest fecundity (14.29 ± 1.82) was found in lufenuron treatment. In case of TM×TF crosses, the highest fecundity (21.65 ± 1.59) was observed in flubendiamide followed by buprofezin (19.56 ± 2.59), novaluron (16.61 ± 1.84) and pyriproxyfen (13.28 ± 1.62) while the lowest fecundity (10.30 ± 2.12) was noted with lufenuron as compared to control untreated males with untreated females.
D. Effect on fecundity reduction during crosses for 4-day application
While assessing the effect of continuous 4-day application of treated diet (300 ppm/5 mL) of different IGRs, the highest fecundity was observed in control (UM×UF) of all respective IGRs, that is, flubendiamide (25.33 ± 2.34), buprofezin (22.90 ± 1.34), novaluron (21.90 ± 2.17), pyriproxyfen (23.20 ± 1.34) and lufenuron (26.43 ± 1.34). Whereas, in line with TM×UF, flubendiamide (22.01 ± 2.22) had the highest fecundity followed by buprofezin (20.76 ± 2.45), novaluron (19.42 ± 1.72) and pyriproxyfen (17.64 ± 1.70) while lufenuron exhibited the lowest fecundity (15.87 ± 1.47).
For UM×TF crosses, the highest fecundity (21.81 ± 1.78) was noted with flubendiamide which was followed by buprofezin (20.01 ± 1.66), novaluron (18.03 ± 1.57) and pyriproxyfen (15.98 ± 1.46) while the lowest fecundity (13.89 ± 1.46) was found in lufenuron treatment. In case of TM×TF crosses, flubendiamide had the highest fecundity (20.88 ± 2.23) followed by buprofezin (18.79 ± 1.23), novaluron (15.84 ± 1.48), pyriproxyfen (12.51 ± 1.26) while the lowest fecundity (9.53 ± 2.76) was noted with lufenuron (Figure 5(D)).
E. Effect on fecundity reduction during crosses for 5-day application
While assessing the effect of continuous 5-day application of treated diet (300 ppm/5 mL) of different IGRs, the highest fecundity was observed in control (UM×UF) of all respective IGRs, that is, flubendiamide (22.90 ± 1.60), buprofezin (23.90 ± 2.90), novaluron (21.20 ± 1.96), and pyriproxyfen (22.10 ± 1.60) while lowest fecundity (21.50 ± 2.60) was found with lufenuron. For TM×UF crosses, flubendiamide had the highest fecundity (21.87 ± 1.48) followed by buprofezin (20.62 ± 2.71), novaluron (19.28 ± 2.98) and pyriproxyfen (17.50 ± 2.96) while the lowest fecundity (15.73 ± 2.73) was found with lufenuron. In the case of UM×TF crosses, flubendiamide had the greatest fecundity (20.06 ± 2.04) followed by buprofezin (18.26 ± 1.92), novaluron (16.28 ± 1.83) and pyriproxyfen (14.23 ± 2.72) while the lowest fecundity (12.14 ± 1.72) was noted in lufenuron treatment (Figure 5(E)). Similarly, for crosses TM×TF, the highest fecundity (19.93 ± 1.48) was noted with flubendiamide followed by buprofezin (17.84 ± 1.49), novaluron (14.89 ± 1.74) and pyriproxyfen (11.56 ± 1.52) while the lowest fecundity (8.58 ± 1.02) was observed in lufenuron treatment.
F. Effect on fecundity reduction during crosses for 6-day application
While assessing the effect of continuous 6-day application of treated diet (300 ppm/5 mL) of different IGRs, in TM×UF crosses, the highest fecundity (20.90 ± 1.58) was noted with flubendiamide followed by buprofezin (19.65 ± 1.81), novaluron (16.31 ± 2.08) and pyriproxyfen (13.53 ± 1.46) while the lowest fecundity (11.76 ± 1.83) was found with lufenuron. For UM×TF crosses, flubendiamide had the highest fecundity (19.91 ± 1.64) followed by buprofezin (18.11 ± 2.02), novaluron (14.13 ± 1.33) and pyriproxyfen (12.08 ±1.32) while the lowest fecundity (10.99 ± 1.82) was found in lufenuron treatment. In the case of TM×TF crosses, the highest fecundity (17.66 ± 1.79) was observed with in flubendiamide followed by buprofezin (17.57 ± 1.99), novaluron (11.62 ± 1.84) and pyriproxyfen (8.29 ± 2.62) while the lowest fecundity (5.31 ± 1.12) was noted with lufenuron. For the crosses (UM×UF) serving as control, the highest fecundity was noted with all respective IGRs, flubendiamide (23.19 ± 2.68), buprofezin (24.33 ± 2.71), novaluron (22.45 ± 2.69), pyriproxyfen (21.90 ± 2.70) and lufenuron (23.90 ± 1.70) (Figure 5(F)).
Overall, all the IGRs in the population of different crosses exhibited the lowest fecundity and with a continuous increase in the number of applications from 1–6 days of feeding, the fecundity of the B. zonata decreased. The lowest fecundity was noted in the population of B. zonata treated continuously with a sixth-day dose of lufenuron at 300 ppm/5 mL of diet in crosses treated males with treated females.
Effect on hatchability during crosses for 1–6 days application of treated diet
The hatchability of the B. zonata was also affected in various crosses with an increase in the number of applications of IGRs in days (1–6 days). The number of applications in days at the same concentration (300 ppm/5 mL) also lowered hatchability.
A. Effect on hatchability during crosses for 1-day application
For 1-day application of the treated diet (300 ppm/5 mL) of IGRs in crosses TM×UF, flubendiamide (22.18 ± 1.78) had the highest hatchability followed by buprofezin (20.41 ± 1.41), novaluron (18.68 ± 1.18) and pyriproxyfen (16.66 ± 2.26) while the lowest hatchability (13.43 ± 1.93) was found with lufenuron (Figure 6(A)). For UM×TF crosses, the highest hatchability (21.74 ± 1.99) was noted with flubendiamide followed by buprofezin (19.62 ± 2.72), novaluron (17.53 ± 1.83) and pyriproxyfen (14.42 ± 2.23) while the lowest hatchability (10.42 ± 2.11) was found in lufenuron treatment. In case of TM×TF crosses, the highest hatchability (21.19 ± 1.69) was recorded with flubendiamide followed by buprofezin (18.19 ± 2.19), novaluron (15.44 ± 1.49) and pyriproxyfen (11.22 ± 1.98) while lufenuron exhibited the least hatchability of 5.72 ± 1.82. In contrast, the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 22.60 ± 2.26, 20.34 ± 2.04, 23.70 ± 1.97, 22.77 ± 2.34 and 21.30 ± 1.83, respectively.

Conspecific crosses of B. zonata (means ± SE) hatchability of different crosses TM×TF, TM×UF, UM×TF and UM×UF at concentration of 300 ppm/5mL of lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide. Hatchability is often influenced in days when it comes to crosses, for 1, 2, 3, 4, 5 and 6 applications of diet denoted A, B, C, D, E and F at a significant probability level of 5%.
B. Effect on hatchability during crosses for 2-day application
While assessing the effect of continuous 2-day application of treated diet (300 ppm/5 mL) of various IGRs, it was found that in TM×UF crosses, the highest hatchability (21.65 ± 1.65) was observed in flubendiamide followed by buprofezin (19.88 ± 1.88), novaluron (18.15 ± 1.15) and pyriproxyfen (16.13 ± 1.13) while the lowest hatchability (12.90 ± 2.90) was noted with lufenuron (Figure 6(B)). For UM×TF crosses, flubendiamide had the highest (21.21 ± 2.21) hatchability followed by buprofezin (19.09 ± 1.09), novaluron (17.00 ± 2.10) and pyriproxyfen (17.00 ± 1.89) while the lowest hatchability (9.89 ± 1.79) was found with lufenuron. In the case of TM×TF crosses, flubendiamide exhibited the highest hatchability (20.66 ± 1.66) which was followed by buprofezin (17.66 ± 1.66), novaluron (14.91 ± 2.11) and pyriproxyfen (10.69 ± 1.69) while the lowest hatchability (5.19 ± 2.19) was noted in lufenuron treatment. In contrast for the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 21.77 ± 2.77, 23.60 ± 1.60, 21.34 ± 2.34, 23.70 ± 3.70 and 22.70 ± 2.70, respectively.
C. Effect on hatchability during crosses for 3-day application
While assessing the effect of continuous 3-day application of treated diet (300 ppm/5 mL) of various IGRs, it was found that in TM×UF crosses, the highest hatchability (22.58 ± 2.58) was noted with flubendiamide followed by buprofezin (20.81 ± 1.81), novaluron (19.08 ± 2.08) and pyriproxyfen (17.06 ± 1.96) while the lowest hatchability (13.83 ± 1.73) was observed in lufenuron treatment (Figure 6(C)). For UM×TF crosses, flubendiamide exhibited the highest hatchability of 22.14 ± 1.64 followed by buprofezin (20.02 ± 1.42), novaluron (17.93 ± 1.93) and pyriproxyfen (14.82 ± 1.82) while the lowest hatchability (10.82 ± 1.92) was found with lufenuron. In the case of TM×TF crosses, the highest hatchability (21.59 ± 2.09) was noted in flubendiamide followed by buprofezin (18.59 ± 1.59), novaluron (15.84 ± 2.44) and pyriproxyfen (11.62 ± 1.92) while the lowest hatchability (6.12 ± 1.72) was observed with lufenuron. In contrast for the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 21.34 ± 1.75, 22.47 ± 2.70, 23.70 ± 1.59, 21.70 ± 1.74 and 22.60 ± 2.97, respectively.
D. Effect on hatchability during crosses for 4-day application
While assessing the effect of continuous 4-day application of treated diet (300 ppm/5 mL) of various IGRs, it was found that in TM×UF crosses, the highest hatchability (22.22 ± 2.21) was noted with flubendiamide followed by buprofezin (20.45 ± 2.45), novaluron (18.72 ± 2.72) and pyriproxyfen (16.70 ± 2.70) while the lowest hatchability (13.47 ± 1.37) was found in lufenuron treatment (Figure 6(D)). For UM×TF crosses, flubendiamide exhibited the highest hatchability of 21.78 ± 1.78 followed by buprofezin (19.66 ± 2.66), novaluron (17.57 ± 1.57) and pyriproxyfen (14.46 ± 1.46) while the lowest hatchability (10.46 ± 1.96) was found with lufenuron. In case of TM×TF crosses, the highest hatchability (21.23 ± 1.83) was noted in flubendiamide followed by buprofezin (18.23 ± 2.23), novaluron (15.48 ± 1.48) and pyriproxyfen (11.26 ± 1.76) while the lowest hatchability (5.76 ± 2.06) was observed with lufenuron. In contrast for the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 23.34 ± 2.34, 21.34 ± 1.34, 23.17 ± 2.17, 21.34 ± 1.84 and 22.34 ± 1.74, respectively.
E. Effect on hatchability during crosses for 5-day application
While assessing the effect of continuous 5-day application of treated diet (300 ppm/5 mL) of various IGRs, it was found in TM×UF crosses, the highest hatchability (22.48 ± 1.98) was noted with flubendiamide followed by buprofezin (20.71 ± 2.01), novaluron (18.98 ± 2.08) and pyriproxyfen (16.96 ± 2.06) while the lowest hatchability (13.73 ± 2.03) was found in lufenuron treatment (Figure 6(E)). For UM×TF crosses, flubendiamide exhibited the highest hatchability (22.04 ± 2.04) followed by buprofezin (19.92 ± 1.09), novaluron (17.83 ± 1.08) and pyriproxyfen (14.72 ± 2.07) while the lowest hatchability (10.72 ± 1.02) was found with lufenuron. In the case of TM×TF crosses, the highest hatchability (21.49 ± 1.09) was noted in flubendiamide followed by buprofezin (18.49 ± 1.09), novaluron (15.74 ± 0.34) and pyriproxyfen (11.52 ± 1.02) while the lowest hatchability (6.02 ± 1.72) was observed with lufenuron. In contrast for the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 24.60 ± 2.1122.90 ± 2.02, 21.60 ± 1.76, 23.60 ± 1.76 and 20.60 ± 2.02, respectively.
F. Effect on hatchability during crosses for 6-day application
While assessing the effect of continuous 6-day application of treated diet (300 ppm/5 mL) of tested IGRs, it was found in the TM×UF crosses, the highest hatchability (21.58 ± 2.80) was noted in flubendiamide followed by buprofezin (20.81 ± 1.21), novaluron (20.08 ± 2.08) and pyriproxyfen (18.06 ± 1.46) while the lowest hatchability (14.83 ± 1.09) was found in lufenuron treatment (Figure 6(F)). For UM×TF crosses, flubendiamide exhibited the highest hatchability (19.14 ± 1.04) followed by buprofezin (20.02 ± 2.02), novaluron (18.93 ± 1.33) and pyriproxyfen (15.82 ± 1.32) while the lowest hatchability (11.82 ± 1.02) was found in lufenuron treatment. In the case of TM×TF crosses, the highest hatchability (18.59 ± 1.19) was noted with flubendiamide followed by buprofezin (17.59 ± 2.12), novaluron (16.84 ± 1.04) and pyriproxyfen (8.62 ± 1.08) while the lowest hatchability (3.12 ± 1.09) was observed with lufenuron. In contrast for the crosses (UM×UF) serving as control, flubendiamide, buprofezin, novaluron, pyriproxyfen and lufenuron exhibited the hatchability of 23.70 ± 2.03, 22.70 ± 2.11, 24.70 ± 2.09, 20.70 ± 2.07 and 21.70 ± 1.70, respectively.
Overall, the crossings, optimized dose and interaction of crosses with the number of applications in days significantly affected the fecundity and hatchability of B. zonata. With an increase in the number of dose applications, the fecundity and hatchability of the B. zonata decreased. The lowest fecundity and hatchability were observed in B. zonata treated with one- to six-time doses of lufenuron continuously at 300 ppm/5 mL of diet during the crosses of treated males and treated females.
Effect of IGRs on morphological characteristics
Ten days old adults were provided a diet for 24 h after treatment with various IGRs at 300 ppm/5 mL of diet. All the morphological parameters studied in this work are mentioned in Table 5, whereas no morphological disorder was observed in the control treatment. While assessing the effect of other concentrations of IGRs, a lower population of malformed individuals during the life span of both sexes was noted.
Effect of tested chemicals on morphological parameters of B. zonata.

• (+) sign shows the presence of characters while (-) sign showed the absence of characters. n=25.
Effect of IGRs on the life span of malformed B. zonata adult
Malformation is defined as an irregular, anomalous, abnormal, or faulty formation or structure of an insect's body, especially if it has been like this since birth.
Analysis of the data showed a significant reduction in the life span of malformed individuals. The male population of B. zonata feeding on lufenuron treated diets on the occurrence of morphological malformations exhibited the shortest life span of 2.4 days. However, for pyriproxyfen, novaluron, buprofezin and flubendiamide treated diets, it was found to be 3.4 days, 3.4 days, 5 days and 5.6 days, respectively. In case of female populations having the morphological aberrations showed a life span of 3 days, 3.8 days, 5.6 days and 6 days after feeding on lufenuron, pyriproxyfen, novaluron, buprofezin and flubendiamide treated diets, respectively (Table 6).
Impact of IGRs at 300 ppm/5mL on the life span of malformed adults (days).

*SEM of adult malformation of Bactrocera zonata.
**Means of values sharing similar letters don’t differ significantly at the probability level 5%.
A. Pupal weight
The effect of different IGRs (Mean ± SE) on the pupal weight was measured with the help of a digital vernier caliper.
All the tested IGRs significantly reduced the pupal weight. The maximum decrease (0.012 g) in weight was noted with lufenuron (0.012 g) while for pyriproxyfen, novaluron, buprofezin and flubendiamide it was found to be 0.014, 00.016, 0.018 and 0.018 g, respectively, as compared to control (Table 7).
Effect of IGRs on pupal weight, pupal length and pupal width of B. zonata treated at 300 ppm/5mL diet.

*SEM of adult malformation of Bactrocera zonata.
**Means of values sharing similar letters don’t differ significantly at the probability level of 5%.
B. Pupal length (size)
Pupal length (Mean ± SE) also depends upon the species and rearing techniques (food, temperature and humidity) under laboratory conditions. In comparison to the control diet, the diet treated with IGRs (Table 7) showed the greatest reduction in pupal duration (lufenuron > pyriproxyfen > novaluron > buprofezin > flubendiamide).
C. Pupal diameter
Pupal diameter (Mean ± SE) was also affected after the ingestion of baits. The effect resulted in B. zonata of barrel-shaped pupae. The lowest effect was observed in lufenuron (2.04 mm) and the highest in flubendiamide (3.13 mm) (Table 7).
Discussion
Management of fruit flies, that is, B. zonata is very challenging for researchers and pest managers. 39 The demand for horticultural crops especially fruits free from the infestation of fruit flies is on rise around the world. 40 Although numerous control measures are in practice 41 yet there is always a demand for eco-friendly and effective management.14,15 In this context, the use of chemosterilants has proven effective for the management of many insect pests including the species of fruit flies. In this regard, the IGRs have been used as chemosterilants against a variety of insects, especially tephritid pests (fruit flies), to reduce the fertility and fecundity of their adult stages. 42
The results of our bioassay revealed that all five chemicals inhibited sterility and reproduction in B. zonata to varying degrees due to their different modes of action. 43 All factors, namely fecundity reduction and hatchability showed a positive correlation with dose. 44 A high insecticide dose has long been advocated as a strategy for achieving high levels of control over agricultural insects pest while reducing resistance development. 45
Lufenuron was more toxic and had higher chemosterility impacts on B. zonata as compared to the rest of the treatments. Significantly lower fecundity and hatchability were observed in lufenuron treated B. zonata. Our current findings regarding sterility induced by lufenuron are in accord with the study of Navarro-Llopis, who reported higher sterility due to lufenuron in wild Ceratitis capitata populations. 46 Similar effects of lufenuron on the hatching efficiency of C. capitata eggs have also been reported by Nisar 14 and Casana-Giner. 47 Our results regarding fertility are in agreement with the findings of Chang et al., and Zhou et al., who have reported reduced fertility in onion flies due to lufenuron without affecting the survival of newly emerged adults.31,34 In addition to controlled studies, chemosterilant activity of lufenuron has also been reported by Navarro-Llopis in field studies. 48 It was evident that lufenuron has affected the ovaries of treated females which could lead to a decrease in egg laying, although they had a reduced time for egg laying and a reduced longevity of adults could contribute to the decrease in oviposits.
Similarly, the crosses of lufenuron treated males and females resulted in decreased fecundity, hatchability and mortality. These findings are in accordance with the results of Moya, who have reported that A. ludens produces sterile offspring by mating males with females both have been treated. 32 Decrease in hatchability and increased mortality have been reported by Rolim in cotton boll weevil. 49 It can be attributed to the fact that the insect cuticle is composed of intertwined chitin chains embedded in a protein matrix, where the absence of chitin may result in mechanical weakness and death of the embryo. 50 Similar to other chitin biosynthesis inhibitors, lufenuron acts by impairing cuticle deposition, as reported with boll weevils. 51
The IGRs treated insects had malformed, swollen heads and smaller body sizes, that is, pupal deformities, pupal length (2.25 mm), pupal weight (0.012 g) and pupal diameter (2.04 mm). These results were similar to Avila, 52 who found that lufenuron had a major impact on pupae due to the loss of a required quantity of water contents, 53 reduced the intensity of protein biosynthesis 54 and internal metabolic temperature increase resulting in reduced body fluid hindering proper sclerotization of the newly formed puparium leading to decreased pupal parameters that were studied during the experiment and agrees with the results obtained on M. domestica. 55 Lowest activity was observed in the case of flubendiamide where fecundity (81.56–99.44) and hatchability (80.00–99.60%) were higher. Weak chemosterilant activities of flubendiamide as compared to other IGRs have also been reported by Kong in armyworm. 56 This weak activity in flubendiamide is due to the bio-quality of the flubendiamide action by activating the Ca2+ release channel, which is sensitive to ryanodine and leads to insect muscle paralysis. The Ca2+ release (RyR) is increased by the influx into cells by the lumen and caused intracellular paralysis. This kind of activity is good for lethal effects but does not exhibit transgenerational activity.57,58
Conclusion
Concluding, lufenuron was most effective with respect to dose, time and mating time. It significantly reduced the fecundity and hatchability of the treated insects. Additionally, it affected the generation obtained from the cross of treated males/females with untreated ones. Farmers in developing countries mostly used synthetic insecticides as a cover spray to control tephritid fruit flies, which resulted in toxic residues in fruits that were not only harmful to human health but also polluted the atmosphere. Chemosterilants based on IGRs are an alternative solution to reducing B. zonata infestations without causing harm to humans or the ecosystem. Lufenuron baited diet was found to be toxic and to have chemosterilant effects on both male and female B. zonata flies in this study. The findings of the current biological studies also show that the use of IGRs prevented normal development of B. zonata at various developmental stages. During the development phases of B. zonata, all IGRs can reduce pupal length, weight and diameter, shorten adult life span and cause deformities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Muhammad Hasnain is a PhD Scholar who is mainly engaged for the management fruit fly against different fruit crops.
Shafqat Saeed is a Dean/Professor (Entomology) who is mainly engaged Integrated pest management of different major crops.
Unsar Naeem-Ullah is an Associate Professor (Entomology) who is mainly engaged as consultant for Dengue Mosquito/ Taxonomist for identification of Insect pest.
Sami Ullah is an Assistant professor (Horticultural) who area of specialization is pomology and postharvest science and technology of fresh horticultural fruit and vegetables.
