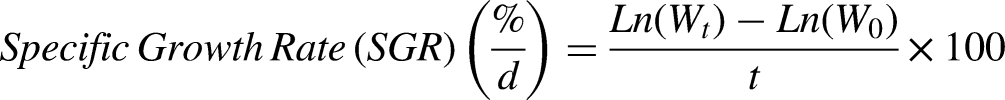Abstract
This study was conducted to investigate the effect of methomyl (MET) on water quality, growth and antioxidant system of genetically improved farmed tilapia (GIFT, Oreochromis niloticus) in the presence of peppermint as a floating bed. The concentration of NH3-N, NO2−-N, NO3−-N and TP in T3 (with 200 g wet peppermint) was significantly lower (P < 0.05) than that in T2 (100 g), T1 (50 g) and control, and the nutrient removal rates were 61.90%, 31.59%, 59.86% and 45.92% in 20 days, respectively. Juveniles GIFT (5.1 ± 0.2 g) were exposed to sub-lethal concentrations of 0.2, 2.0, 20 and 200 µg/L of MET for 45 days. After 6 weeks of a feeding trial, percentage weight gain (PWG), specific growth rate (SGR) and feed conversion ratio (FCR) were significantly decreased in 0.2, 2.0, 20 µg/L MET groups respectively and increased in the 200 µg/L MET group. Compared with the control, no significant changes in superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) were detected in the 0.2 µg/L group. The significant increase in activities of SOD, CAT and GPx was accompanied by a diminution in reduced glutathione (GSH) levels resulting with tilapia exposed to 2.0, 20, or 200 µg/L for 45 days. The highest rates observed in SOD, CAT, GPx were 157.63%, 164.05% and 167.46% of the control respectively, and the lowest inhibition rate in GSH was 66.42% of the control. Peppermint as a floating bed can alleviate the adverse effects of MET, such as growth retardation and oxidative stress.
Introduction
Phytoremediation, which uses plants to accumulate and remove contaminants from the environment, is considered promising because of its low cost, and safeness for the environment. 1 Constructed wetlands comprise emergent vegetation with harvested valuable products established upon a floating infrastructure, buoyant on surface waters. 2 Medicinal herbs also contain a complex chemical composition that present varied biological activities, making medicinal plants a suitable substitute for antibiotics and an effective alternative for multifactorial disease treatment. Plants have been reported to produce numerous bioactivity effects such as appetite stimulation, growth promotion, antistress and immunostimulation.3–8
The main routes of agricultural pesticide contamination are through drains from agricultural fields, spray drifts, disposal through waste and deliberate use in the aquatic environment. Methomyl (MET, C5H10N2O2S) is S-methyl-1-N-[(methylcarba-moyl)-oxy]-thioacetimidate, classified as the most toxic and hazardous pesticide by the WHO and EPA, which is functionally analogous to organophosphates for inhibiting enzyme activity of acetylcholinesterase in mammals and insects. The half-life of MET is more than 20 weeks, 9 and MET is considered to be highly toxic to aquatic animals. 10 The LC50 values of crucian carp were 0.9–3.4 mg/L, and the LC50 values of Daphina magna were from 0.022 to 0.026 mg/L. 11 Previous studies were conducted on Channa striatus by MET exposure, 12 genetically improved farmed tilapia (GIFT, Oreochromis niloticus), 13 etc. The results showed that significant changes in the serumal glutathione-S-transferase (GST), glutathione reductase (GR), glutathione peroxidase (GPx), and glutathione oxidized (GSSG) increased with the decreased reduced glutathione (GSH) following MET exposed to 2.0, 20, or 200 µg/L, and the activities of hepatic GST, GR, GPx, GSSG increased and GSH decreased.
Oxidative stress is an imbalance between the production of reactive oxygen species (ROS) and the cell's ability to reduce ROS, MET can cause an increase in ROS production in the cells of the exposed organisms. 13 Antioxidants can be categorized as a first-line defense. The antioxidants, superoxide dismutase (SOD), catalase (CAT) and GPx respectively dismutate superoxide radicals, breakdown hydrogen peroxides and hydroperoxides into harmless molecules (H2O2/alcohol and O2). GSH reduces H2O2 and lipid hydroperoxides with the GPx enzymes, which is an important part of the integrated antioxidant system and maintains other non-protein antioxidants in their reduced and biologically significant state.
The microbial degradation for MET and its related beneficial advantages has been well studied by other researchers.14–17 We constructed a determination method for MET in peppermint. 18 Peppermint showed significant antioxidant and antitumour activities, and strong antimicrobial and antiviral activities. 19 However, limited information on the capability of genus Mentha as a part of the biofilter is documented. 20 The present study aims to investigate nutrient removal rate and the antioxidative response using peppermint as a floating bed, and the results innovatively showed peppermint alleviated the adverse effects of MET exposure.
Materials and methods
Experimental design and sampling
MET was obtained from Shanghai Focus Biological Technology Co., Ltd, China), dissolved in pure water to form of stock solutions, and then the experimental concentration in aquariums was diluted immediately before use. This study was conducted in FFRC-CAFS in Wuxi, China. The study applied a static system with a floating bed. A total of 15 glass aquaria (40 cm × 40 cm × 60 cm, and 100 L) were used. Erstwhile to use, peppermint height of 10 cm was planted in several floating pots and placed in a tank. Peppermint was stored in a 1000 cm3 tank in 300 L water. There were no artificial nutrients added to the peppermint during the acclimatization and the experiment. Fingerlings and juveniles of GIFT were obtained from the fish farm in Yixing, China. After four weeks, the fish were transferred to the aquaria. The initial weight of fish was 5.1 ± 0.2 g for fingerlings, and 33.70 ± 2.65 g, 11.7 ± 0.4 cm for juveniles. Stocking density was 15 fish/100 L. Fish were fed twice daily with commercial feed (extruded pellets). The feeding rate was 5% of total body weight. This ration size was built on preliminary observations to drive the fish to feed on the roots, stems and leaves of peppermint, also to prevent water quality deterioration, as well as for cost-saving.
A total of 12 aquaria were set up, consisting of triplicate sets of four treatments T1, T2, T3, and control. The four different treatments tested in this phase were: control treatment with no vegetable (GIFT without peppermint); T1 (GIFT with 50 g wet peppermint), T2 (GIFT with 100 g wet peppermint) and T3 (GIFT with 200 g wet peppermint). 50, 100 and 200 g of peppermint as wet weight means life grass weight as treatment. An environmentally friendly material (bamboo) was used to construct the floating bed and net through which holes allowed the roots of the plants to elongate into water. The measurements of the floating beds were 15 cm wide and 30 cm long. All the aquariums were soaked and filled with fresh water at least 5 days before the experiment started to initiate the growth of bacteria in the water. This practice followed the standard procedure for the conditioning of the rearing system that helped the establishment of the process of nitrification.
Half of the water was changed in the semi-static system daily. Glass aquaria of 100 L were used with 250 g wet peppermint as a floating bed, and the addition of different sub-lethal concentrations of MET in four triplicates were P1 (0.2 µg/L), P2 (2.0 µg/L), P3 (20 µg/L), P4 (200 µg/L) and P5 for control (0 µg/L). The initial concentrations of 0.2, 2.0, 20, 200 µg/L groups were 0.23, 2.04, 21.40, 206.12 µg/L respectively, and the real MET concentrations in the aquaria were measured according to the method of Zheng et al. 18
Water quality
Water quality was measured daily for two weeks in aquariums. Parameters collected were dissolved oxygen (DO), pH, temperature, and conductivity. The ammonia-nitrogen (NH3-N), nitrite-nitrogen (NO2−-N), nitrate-nitrogen (NO3−-N), and total phosphorus (TP) were tested once per 5 days, filtering with a Whatman filter paper of a pore size of 0.45 μm before laboratory analyses.
Growth performance evaluation
The initial body weight (IBW) of every group was obtained at the beginning and end of the experiment. Observations on fish were made to assess the final body weight (FBW), percentage weight gain (PWG), specific growth rate (SGR) and feed conversion ratio (FCR). These measurements were calculated using formulae as follows
Antioxidant status evaluation
On the 15th, 30th and 45th days of the experiment, prior to sampling, fish were food deprived for 1 day. Three fish from each aquarium were randomly picked, dissected and immediately anesthetized with 100 mg/L MS-222. Thereafter, the fish were dissected to obtain liver samples and subsequently stored at −80°C for antioxidant enzyme activities. Samples were homogenized on ice with cold 0.86% physiological salt water (1:9, w/v), and centrifuged at 5000 r/min at 4°C for 5 min. The supernatant was collected to analyze SOD, CAT, GPx and the content of GSH using the commercial kits obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Spectrophotometric quantification was assessed using Power-Wave XS2 (BioTek instruments Inc, Vermont, USA). Briefly, WST-1 method was used to measure SOD concentrations through nitroblue tetrazolium inhibition at 450 nm. CAT activity was calculated by measuring hydrogen peroxide based on stable complex production with ammonium molybdate at 405 nm. GPx activity was determined by measuring the enzymatic reaction of glutathione at 412 nm. Finally, the total glutathione content GSH was measured using the method of reacting with 5,5-dithiobis-2-nitrobenzoic acid (DTNB) and the absorbance was recorded at 420 nm. 21
Data analysis
SPSS ver. 25 was used to conduct one-way factorial analysis of variance (ANOVA) and Tukey's multiple range tests. The results are presented as means ± SEM (standard error of the mean), P < 0.05 considered the difference is statistically significant, and the relevant graphs were drawn in Origin 9.4.
Results
Water quality
NH3-N in all groups increased from day 0 to day 20 due to the accumulation of uneaten feed and fish feces. NH3-N in T3 was significantly lower (Table 1, P < 0.05) than that in T2, T1 and control. NO2−-N is an intermediate product of the nitrification process. Therefore, NO2−-N concentration is generally lower than NH3-N and NO3−-N. A high concentration of nitrite leads to poisoning in fish, and plants do not use nitrite as a nutrient source. Thus, NO2−-N concentration should not exceed 5 mg/L. The concentration of NO2−-N tended to increase significantly (P < 0.05) during the experiment. In all treatments, increased NO3−-N occurred over time. Nitrate concentration ranged from 0.15 to 1.58 mg/L. NO3−-N is relatively harmless to most aquatic organisms, and does not cause any health hazard except at extremely high levels (>90 mg/L). For tilapia, NO3−-N toxicity may occur if the concentration exceeds 300–400 mg/L. The concentration of TP in T3 was lower significantly (P < 0.05) than in T2, T1 and control. The low concentration of TP in T3 might be attributable to usage by peppermint for their growth. Phosphorus accumulated in plant roots tissues is crucial for plant nutrients. Consequently, TP concentration of tilapia cultivation wastewater underwent much more reduction in T3 than in T2, T1 and control.
Ammonia, nitrite, nitrate and total phosphorus fluctuation during the experiment.

Note: Different lowercase letters indicate significant differences.
In our study, T3 has a higher rate of nutrient removal and has more efficiency than T1 and T2 (Table 2). The percentage of nutrient removal on T3, namely 65.90%, 31.59%, 59.86% and 65.92%, respectively for NH3-N, NO2−-N, NO3−-N and TP.
Percentage of nutrient removal.

Growth performance
Neither mortality nor visible disease symptoms were remarked during the experiment. The decreases in PWG and SGR were revealed in low concentrations of MET dose-related, being the lowest and most significant decrement (Table 3, P < 0.05) in those of MET concentration (200 µg/L). In contrast, FCR of fish fed with peppermint and exposed to 200 µg/L of MET showed a significant increment compared to the group of control (P < 0.05), while there is no remarkable significance in other concentrations.
Growth performance parameters of juvenile GIFT exposed to sub-lethal dose of methomyl and fed on peppermint.

Note: Different lowercase letters indicate significant differences.
Oxidative status
Compared with control, no significant (P > 0.05) changes in SOD, CAT, GPx were detected in 0.2 µg/L group (Table 4). SOD and CAT activities were significantly (P < 0.05) increased in 2 µg/L group after 45 days of MET exposure and no significant (P > 0.05) changes in GPx activities. As the dose was increased to 20 µg/L, significant (P < 0.05) elevation was noted for GPx at 30 days, while for SOD and CAT the increment (P < 0.05) started at 15th day of MET exposure. In 200 µg/L group, all enzymes activities significantly (P < 0.05) increased after 15 days of the experiment. GSH contents were reduced in all other treatments except 0.2 µg/L treatment. Significant (P < 0.05) decrease in 2 µg/L was observed after 30 days, while, GSH contents were significantly (P < 0.05) decreased in 20 and 200 µg/L groups after 15 days.
Hepatic SOD, CAT, GPx and GSH contents in GIFT exposed to a sublethal dose of methomyl with peppermint.

Note: Different lowercase letters indicate significant differences.
Discussion
GIFT is an omnivorous fish with high sensitivity to the oxidative stress of pollutants, it has been considered a common test species in toxicology studies. In this study, exposure of GIFT to MET in the presence of peppermint as a floating bed for 45 days significantly reduced FBW, PWG and SGR in 200 µg/L group. This reduction in fish growth may have been due to worsened water quality and decreased feed utilization. Comparison of fish growth retardation has been reported previously with other pesticides such as chlorpyrifos, 22 penoxsulam, 23 and GIFT may have to use energy to detoxify the MET by altering their protein and carbohydrate metabolism with less food energy available for growth consequently. In the beginning, ammonia-oxidizing bacteria (AOB) oxidize NH3-N to nitrite, later converted to nitrite through nitrite-oxidizing bacteria (NOB). NO2−-N and NO3—N, was associated with the growth of AOB than NOB, AOB population grow faster than NOB. 24 However, vetiver can remove 65.16% of NH3-N from tilapia wastewater with increasing plant density. 25
ROS detoxification can be caused directly by CAT, SOD, GPx, which are radical-scavenging enzymes, and indirectly through GSH level stabilization collaborate with GST stabilization. Many studies have recommended that the liver of fish is high in relation to antioxidant activity. The presence of pesticides such as MET in living systems has been shown to promote the synthesis of ROS. MET exposure provoked a noticeable concentration-dependent decrease in GSH in tilapia liver. Furthermore, SOD and CAT activities increased after MET exposure, similar to the previous report. 13 Tilapia increased ROS-scavenging enzymes (SOD, CAT) converting superoxide anions into H2O and O2. The increments in ROS-related enzymes are to reduce the ROS level in the target cells by compensatory mechanisms. Our previous studies showed water spinach reduced part of the water quality indexes in the MET (<200 µg/L) groups. 26 Remarkably, compared with a previous study, ROS-related enzymes of exposed tilapia fed on peppermint presented in the system as a floating bed were significantly normalized in low concentration (0.2 µg/L). In addition, results showed a decrease in the levels of all ROS-related enzymes in the concentration of 2.0 and 20 µg/L. It is possible that peppermint can enhance the antioxidant enzyme system. These favorable effects may have been due to direct scavenging by peppermint due to their high content of phenolic compounds (rosmarinic and cafeic acids), flavones (luteolin glycoside) and flavanone (eriodityol glycosides), which are possibly the main infusion antioxidants. 27
Therefore, a growing interest has emerged in using medicinal herbs to protect aquatic organisms against pesticide toxicity, 22 especially in peppermint.28,29 However, using peppermint as a floating bed gives the possibility to use it as a dietary supplement for regulating growth retardation and oxidative stress in GIFT. The peppermint was a suitable herb for integrated aquaculture-agriculture farming and aquaponics systems. 30 Studies have shown that peppermint as a medical herb, is able to improve antioxidant capacity. Antioxidant-active polysaccharides (WMP) isolated from peppermint haploid are potent natural antioxidants that significantly increase antioxidant enzyme activity, including SOD, GPx, and CAT, and reduce MDA levels in serum and liver of d-Gal-induced senescent mice. 31 Biofloc technology also enhances the bio-antioxidant status. One experiment found that biofloc technology not only enhances its digestive enzyme activity and antioxidant state, but also upgrades its immune response and enhances its ability to resist ammonia stress. 32
In addition, herbal active substances also have many effects, which have great research value and application prospects. Resveratrol (RES) can be used to suppress the inflammatory response,4,7 continuously improve biological intestinal health and improve immunity.4–7 To improve the biological antioxidant capacity, in addition to the use of medical herbs to enhance immune mechanisms, it can also achieve the improvement of its antioxidant capacity through the improvement of water quality. 33 The addition of taurine to feed in the presence of amdoxic acid can alleviate toxicity and can be used as a promising bioactive substance for the functional food and pharmaceutical industries. 34 Taurine supplementation is a beneficial additive that effectively reduces MET toxicity mixed with aquatic O.niloticus aquafeed. 35 The study revealed parsley (Petroselinum crispum) seed meal alleviated MET-adverse impacts on tilapia growth. 36 Centella asiatica ethanol extract changes communities by increasing α-diversity, depleting the colitis-associated genera Helicobacter, Jeotgalicoccus, and Staphylococcus, capable of reshaping the gut microbiota of suffering mice. It affects several metabolic signaling pathways, which may contribute to the refurbishment of the damaged intestinal mucosal barrier. 37 Our study showed growing spinach with water may have a positive effect on the prevention of GIFT's liver toxic damage. 26 Astragaloside IV from the medical herb Radix astragali, revealed a protective effect against acute cerebral ischemic/reperfusion injury through a PPARγ-dependent manner, which has been taken as a promising therapeutic agent. 38 The most important thing is to find active substances that can improve the immune ability or disease resistance of fish from medical herbs, like RES in our previous studies.4–7 Our recent studies showed RES alleviated lipid metabolism through the PPARα/γ signaling pathway. 8
Conclusions
Peppermint was capable of reducing the concentration of NH3-N (61.90%), NO2−-N (31.59%), NO3−-N (59.86%) and TP (45.92%) in 20 days. A significant increase occurred in activities of SOD, CAT and GPx accompanied by a diminution in GSH level with tilapia exposed to 2.0, 20, or 200 µg/L of MET for 45 days. Peppermint as a floating bed can alleviate the adverse effects of MET, such as growth retardation and oxidative stress, and its usage for medical herbs requires further studies.
Footnotes
Acknowledgments
We thank Ampeire Yona for providing grammar and spelling checks, and giving revision suggestions of the manuscript.
Author contributions
X.J.J., Y.Z., G.C.X., T.Y.L. conceived and designed the experiments; B.F., T.Y.L., Y.Z. analyzed the data; J.Z.C. contributed reagents/materials/analysis tools; Y.Z. contributed to tables preparation; X.J.J., Y.Z., T.Y.L. prepared and wrote the manuscript. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by China Agriculture Research System of MOF and MARA (No. CARS-46), Central Public-interest Scientific Institution Basal Research Fund, Freshwater Fisheries Research Center CAFS (NO. 2021JBFM19).
Ethical approval
Ethical Approval is not applicable for this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.