Abstract
Tomato (Solanum lycopersicum L.) is cultivated and consumed worldwide, including in Indonesia. It is used in the food, cosmetic, and pharmaceutical industries, due to its high content of carotenoid (lycopene) compounds that have antioxidant and anticancer activities. In Indonesia, although several cultivars of tomato are cultivated, including Opal, Permata, Mutiara, and Rewako, studies on their genetic information are limited. Unpredicted climate change as well as heavy metal contamination, especially Pb pollution, has threatened Indonesian food security. Therefore, our study aimed to analyze the genetic diversity of the four local tomatoes using random amplified polymorphic DNA (RAPD) markers and to determine the growth responses of several local tomato genotypes under Pb stress. In this study, morphological responses to Pb, including plant height and root length were observed. The RAPD analysis showed that Rewako and Permata were distinct, whereas Opal and Mutiara were closely related, possessing 81.8% similarity. Pb stress influenced plant height and root length in the four tomato genotypes, with each genotype exhibiting different morphological responses than others. However, the closely related Mutiara and Opal genotypes demonstrated similar responses to Pb stress to Permata and Rewako. Our study demonstrates that RAPD are sensitive and efficient for elucidating the genomic profile of the tomato genotypes. In addition, our results suggest that genetic variation among tomato genotypes might influence the morphological responses against Pb stress.
Introduction
Tomato (Solanum lycopersicum L.), an important horticultural vegetable extensively cultivated worldwide, is used in the food, cosmetic, and health industries, owing to its high content of carotenoids (lycopene) that has antioxidant and anticancer activities. 1 In addition, tomatoes also contain high levels of vitamins and minerals. 2 Similar to other crops, several environmental factors determine the productivity of tomato plants. It includes nutrients, exposure to salinity, heavy metals, drought, temperature stress, and ultraviolet pollution. These factors can disrupt the plant's physiological, cellular, and molecular processes. 3 An environmental factor that lowers plant productivity is exposure to heavy metals. 4 Heavy metals are present in nature and are defined as metal elements possessing a high density exceeding that of water by at least five times. Agricultural soils and irrigation water are often highly contaminated by toxic heavy metals, such as Cu, Pb, Cd, Fe, Al, and Hg. 5
Heavy metals, such as Fe, Zn, Br, Cu, and Mn, are important micro-nutrients and play key roles in plant cellular activities. However, at certain concentrations, these can negatively impact plant growth. 6 Pb is one such toxic heavy metal generally present in contaminated soil and aquatic ecosystem; this is a result of various anthropogenic activities, such as smelting, producing batteries and paints, using pesticides and fertilizers excessively, and burning fossil fuels. 7 In the soil, Pb is available as free metal ions, complex inorganic constituents (such as HCO3−, CO32−, SO42−, and Cl−), or organic ligands (amino acids and fulvic and humic acids).8–10 Pb toxicity in plants causes growth inhibition, low seed viability, mitotic perturbation, chlorosis, decreased photosynthetic efficiency, perturbation in mineral absorption, and changes in cell membrane permeability and structure. These conditions could result in biochemical disorders 5 and ultimately lead to cell death.11,12 A previous study suggested that the production of reactive oxygen species (ROS) during stress is also involved in generating negative plant responses. 12 Elevated ROS levels generate oxidative stress, leading to oxidative damage of lipid membrane, DNA/RNA damage, protein oxidation, and reduced enzyme activities. 13
Previous studies have revealed that Pb accumulates in plants in the roots, stems, and leaves, depending on the species. 14 For instance, Pb accumulates in Amaranthus and Brassica mainly in the leaves. 15 Whereas, in Solanum lycopersicum and Zea mays it mainly accumulates in the roots, consequently inhibiting root growth and development.15–18 In Indonesia, many farmlands are polluted by Pb due to the use of chemical fertilizers. 19 Previous study showed that Pb concentration in soils is about 1.6–6.05 mg/kg of soil (mean, 3.68 ± 1.08 mg/kg) before fertilizer application and about 2.75–12.85 mg/kg (mean, 7.07 ± 2.3 mg/kg) after crop harvesting. 19 Due to the broad range of Pb impact on agriculture, studies on its effect on plant productivity are essential. In addition, different plant genotypes might exhibit different responses to heavy metal stress including Pb. 20 To date, studies on the responses of local tomato genotypes to Pb stress are limited. Therefore, we investigated the morphological responses of four local tomato plants to Pb stress. The tomato cultivars grown in Indonesia include Mutiara, Opal, Rewako F1, and Permata F1, and their characteristics are usually evaluated by their phenotype. However, studies on genetic polymorphism within the local tomato in Indonesia remains limited, especially studies that use molecular approaches. 21
Some molecular markers are routinely used to determine both intra and inter-species diversity. These genetic markers include Random Amplified Polymorphism DNA (RAPD), Restriction Fragment Length Polymorphism (RFLP), Amplified Fragment Length Polymorphism (AFLP), Simple Sequence Repeat (SSR), and Inter-Simple Sequence Repeat (ISSR).21,22 We evaluated the genetic diversity among Indonesian tomato cultivars using RAPD markers. Advantages of RAPD include rapidity and easy handling.23,24 Since genetic diversity determines plant responses, data obtained from the genetic variability study can be used to complement the analysis of tomato morphological responses to Pb stress.
Materials and methods
Cultivation of Indonesian tomato genotypes and pb stress treatment
Seeds of Mutiara, Opal, Rewako, and Permata were soaked in tap water for 30 min and in antifungal solution for 2 min. Subsequent steps are germinating the tomato seeds in growth media containing soil and compost media (1: 1) as described previously in Jadid et al. 4 The seeds were watered twice a day. 25 Pb treatment is prepared by firstly drying the media for 24 h. Prepared soils were then placed in polybag, sizes 15 × 15 cm. 26 Three-week-old seedlings were transferred to the Pb-treatment media (0, 75, 150, and 300 ppm) and treated for 20 d.
Morphological assessments
Plant height and root length measurements were performed at the end of Pb stress treatment (32 days after planting (DAP)). Plant height was carefully calculated from the base of the rootstock to the apex of the main stem using a steel ruler. 27 Root length was measured after carefully removing the plant from the planting medium, soaking the roots in a tub filled with water to dislodge the soil, and draining excess water. Root length was measured from the root tip to the root base.
Extraction and quantification of tomato genomic DNA (gDNA)
Tomato leaves of each genotype were subjected to total gDNA extraction as described previously. 28 The extraction was performed using “Plant Genomic DNA Mini Kit” (Geneaid®, Taiwan) according to the kit’s instructions. Measurement of gDNA quantity and quality was conducted using “Nano Drop” (Thermo Scientific™ nanodrop 2000).
DNA amplification using RAPD markers
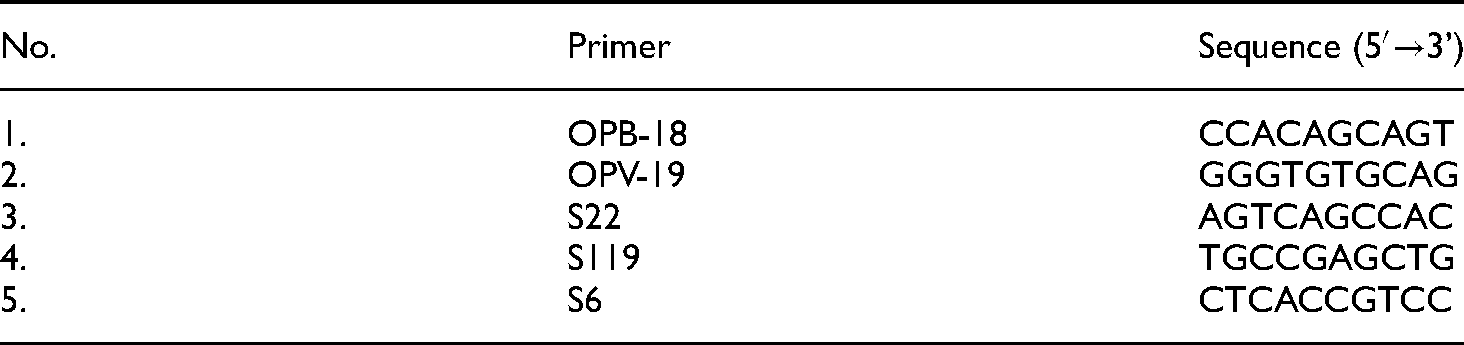
DNA amplification was performed using the previously extracted gDNA, which serves as template. We used five RAPD primers, namely, OPB-18, OPV-19, S22, S119, and S6 (Integrated DNA Technologies) (Table 1).29,30 PCR was performed with a final reaction volume of 25 µl using GoTaq® Green Master Mix (Promega, USA). The DNA amplification program consisted of an initial denaturation step at 95 °C for 2 min. 35 cycles of DNA amplification were performed. The latter consists of “denaturation” at 95 °C for 30 s, “annealing” at 37°C for 30 s, and “extension” at 72 °C for 1 min). The last step was “final extension” at 72°C for 5 min. The amplicons obtained from DNA amplification were migrated through electrophoresis at 100 V on a 2% of 0.5 µg/ml ethidium bromida (EtBr)-containing agarose gel. Amplicons were then visualized using UV-transilluminator.
List of the RAPD primers (10 bp) used in genetic diversity analysis of Indonesian tomato genotypes.
Data analysis
Morphological measurements were performed in triplicates and demonstrated as mean ± standard deviation (SD). The analysis of variance (ANOVA) analysis was performed using Minitab version 19. P values where less than 0.05 is considered significant. Data obtained from morphological responses of tomatoes under Pb stress were entered and analyzed using Microsoft Office Excel 2010. The PCR amplicons analyzed as RAPD bands were scored as 1 for presence and 0 for absence and evaluated as a binary data matrix. The polymorphic bands as well as the percentage of polymorphism were determined using the following equation by Kumla et al.
31
and Cavalli-Sforza et al.
32
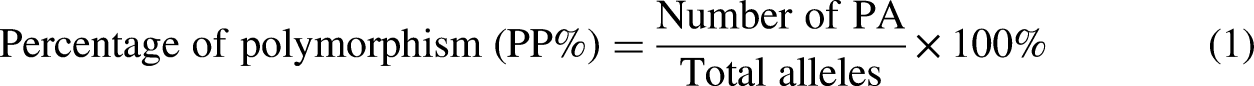

Results
RAPD profiling and genetic diversity analysis of the Indonesian tomato genotypes
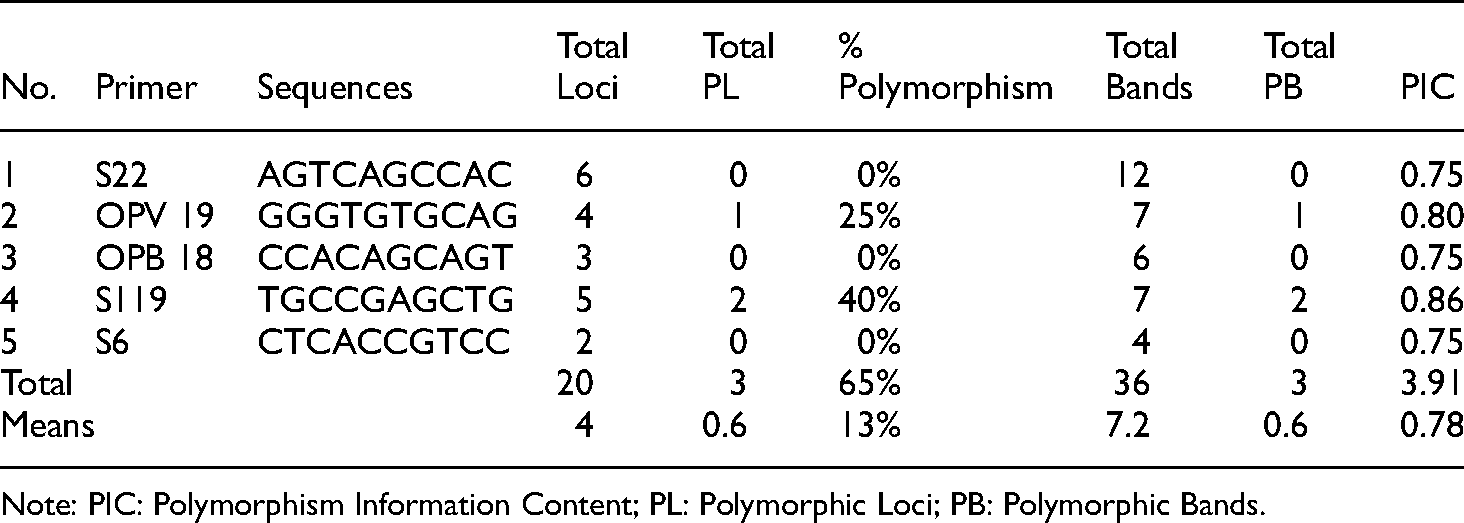
The genetic diversity of four Indonesian tomato cultivars have been assessed using five RAPD molecular markers (Table 1). The RAPD PCR produced distinct amplicons in the range between 300–1000 bp (Figure 1). A total of thirty-six DNA bands were generated in this amplification with an average of 7.2 bands per primer. In the present study, we also observed that three bands were polymorphic (Table 2).

PCR-amplified RAPD pattern of four tomato genotypes using five RAPD primers (S119; OPV19; S6; OPV18 and S22). L, ladder; O, Opal; P, Permata F1; R, Rewako; M, Mutiara.
Amplified fragment and polymorphic information obtained from five RAPD markers in four Indonesian tomato genotypes.
Note: PIC: Polymorphism Information Content; PL: Polymorphic Loci; PB: Polymorphic Bands.
The highest number of DNA bands were produced by S22, followed by OPV19 and S119 with 7 bands. The amplification of tomato gDNA using OPB18 generated 6 bands, while the lowest number of appearing DNA bands were found in S6 as 4 bands. We also observed a total of twenty loci and three polymorphic loci. In addition, our results showed that Permata F1 did not produce any DNA amplification in the five primers used. It might be due to the absence of Permata F1 sequence in the genome that might be homolog to the primer used. 35 In the recent study, the total percentage of polymorphism across the four tomato cultivars was 65%. It ranged between 0 to 40%.
In many genetic diversity studies, the discriminatory level of the primer used is analyzed using Polymorphism Information Content (PIC). Therefore, we assessed the PIC in this present study (Table 2). Our results showed that the PIC ranged between 0.75 and 0.86. The average value of the PIC in this study was 0.78 per primer. The highest PIC value (0.86) was obtained from S119, followed by OPV 19 with 0.80 of PIC value. Meanwhile, the lowest PIC value was obtained from S22, OPB 18 and S6 possessing PIC value as 0.75 (Table 2). The overall results demonstrated that all PIC values were more than 0.75.
The DNA band patterns from RAPD amplification were used to determine the similarity among plant genotypes and construct a phylogenetic tree. In this study, the relationship between tomato genotypes was evaluated using the Jaccard similarity coefficient index, which ranged from 0 to 1. The genotypes Mutiara and Opal were closely related, indicated by the high similarity (0.818) (Figure 2). Meanwhile, Rewako and Permata genotypes were presented in different clusters.

Unweighted pair group method with arithmetic mean (UPGMA) dendrogram illustrating the genetic relationship between four genotypes of Indonesian tomatoes based on RAPD fingerprinting.
Effect of pb contamination on morphological characters of the Indonesian tomato genotypes
Our results demonstrated that Pb treatment significantly alters the root length in all the four tomato genotypes tested (P < 0.05). Although a dramatic decrease in root length was observed in all the genotypes after treatment with 300 ppm of Pb compared to that in control, Pb-treated (300 ppm) Rewako showed a significant decrease (P-value < 0.05) in root length (3.25 cm) compared to non-treated Rewako (4.18 cm) and other genotypes (Figure 3). A minimal decrease in root length was exhibited by Pb-treated (300 ppm) Mutiara and Opal genotypes (9.48% and 17.4%, respectively) compared to their controls. This was consistent with our genetic diversity analysis, which indicated that Mutiara and Opal were closely related genotypes.

Effect of pb contamination on root length of the Indonesian tomato genotypes at 32 days after planting (DAP). The value represents means of plant height ± SD. Different letters indicate statistically significant differences (p < 0.05).
Our results also demonstrated that Pb contamination influenced the tomato plant height (p < 0.05). Figure 4 shows that all Pb-treated genotypes demonstrated an increase in plant height after 30 d, with Mutiara exhibiting the highest increase (24.4%) compared to the control plant. Pb-treated Mutiara possessed a plant height of 50.4 cm, while Mutiara control plant exhibited only 40.5 cm of height. In contrast to the Mutiara performance, Opal and Rewako performed a minimal increase of plant height after being treated with 300 ppm of Pb. Opal and Rewako only showed 11.18% and 17.7% of plant height increase when they were treated with 300 ppm of Pb compared to their control plant, respectively (Figure 4).

Effect of pb stress treatment on plant height of the Indonesian tomato genotypes at 32 days after planting (DAP). Value represents means of plant height ± SD. Different letters indicate statistically significant differences (p < 0.05).
Discussion
Tomato (S. lycopersicum L.), a Solanaceae plant, is the most cultivated and commonly consumed vegetable worldwide. 1 Their cultivation is spread from tropical to temperate regions. 36 High level of carotenoid-type compounds such as lycopene and β-carotene as natural scavenging of free radicals makes its market demand increases significantly. 37 Therefore, development of new tomato cultivars as well as a method of cultivation become a great challenge to ameliorate tomato quality and productivity. This present study investigated the genetic variation of four Indonesian tomato cultivars, namely Permata, Opal, Rewako and Mutiara. In addition, their morphological responses to Pb contamination have also been assessed.
Commercial cultivars often result from vegetative propagation. Thus, newly cultivars might exhibit high heterozygosity. 38 An efficient molecular tool to detect the genetic profile of the cultivars is necessarily needed. DNA-based markers are an efficient technique that might help to distinguish plant genotypes. RAPD is one of the DNA-based markers used to detect genetic variation among species. 39 It is a rapid and inexpensive technique that uses short primers (10–15 bases) for amplification. These primers will then randomly but directly bind to the genomic DNA. 24 In this present study, we used five RAPD primers producing three polymorphic bands, which distinguished the four tested tomato cultivars. The RAPD primers will attach to the genome sequences. Therefore, primer binding to the genome sequence determines the genetic diversity among samples. 35 Our results showed that none of the five primers could bind to the Permata F1 sequence. It could be noted that the primers might be less effective when used to discriminate between samples. This result is also observed in a previous study demonstrating that some primers might be specific to certain genotypes and might be not applied. 40 The percentage of polymorphism observed in this study ranged between 0 and 40%. Greater polymorphism indicated greater genetic diversity among the cultivars. 23 The use of RAPD markers to discriminate between tomato cultivars has been reported in previous studies. 29,41–43
Our results also demonstrated that the PIC values were greater than 0.75. PIC value is used to determine the informativeness of the RAPD markers. 44 A previous study concerning the genetic diversity of other tomato cultivars demonstrated a lower PIC value compared to this present study. Meng et al. 30 reported that the PIC value among Chinese tomato cultivars was between 0.31 and 0.50. According to Mateescu et al., 34 all our RAPD markers are considered highly informative markers to discriminate the four Indonesian tomato cultivars. The evaluation of genetic relationship, using Jaccard similarity coefficient index 45 in this current study revealed the existence of genetic variability among four Indonesian tomato cultivars. Mutiara and Opal were presented as closely. Whereas Rewako and Permata were distinct (Figure 2). Altogether, these current results might be beneficial for providing the molecular diversity among Indonesian tomatoes. In addition, this information could be further developed for agricultural management, genetic conservation and the development of new vegetable crops. 46
Lead (Pb) accumulation in soils has been reported to affect plant morphology. 16 Decreased root length and leaf chlorosis 47 are common symptoms exhibited in plants exposed to lead contamination in soils.48,49 Our assessment demonstrated that Pb contamination negatively affects the root growth in the four Indonesian tomato genotypes (Figure 3 and 5). Similar studies have shown that Pb contamination severely affects the root growth in Solanum melongena and Solanum lycopersicum cv. Falcon.50,51 Therefore, genetic factors might influence tomato responses to heavy metal stress. Studies have also reported that tomato performs a diverse array of responses under heavy metals 20 and other abiotic stresses. 52 Hence, evaluating the tolerance of tomato plants against Pb or other heavy metal contamination is a challenging task, involving a wide range of physiological, biochemical, and molecular approaches. 20

Root length of the Indonesian tomato (Solanum lycopersicum L.) genotypes after being treated with Pb stress at 32 days after planting.
Several studies have shown that Pb retention in the roots of dicots is greater than that in monocots.13,53 Therefore, its effect on root growth might also be different. Since the root is the first plant organ to come in contact with soil contaminants, Pb induces a reduction in root growth rate. Pb in the soil is bound to organic material including carboxyl groups of mucilage uronic acids. This binding might protect the root system from over-uptake of Pb. 13 However, once Pb is absorbed, it passes into the root apoplast and accumulates in the root epidermis, and trace amounts of Pb are also detected in the vascular tissues. 16 In general, root systems possesses a physical barrier (endodermis) to prevent the Pb movement into aerial parts of the plants. At lower concentration, Pb moves via apoplast However, at higher concentrations, Pb damages the plasmalemma. 54 Previous studies demonstrated that Pb is highly concentrated in the roots than in shoots. 55 In addition, reports have demonstrated the deposition of Pb in plant cell organelles.56,57
Inhibition of root growth as a consequence of the Pb contamination is mainly due to the cell division inhibition in the root tips. 58 Pb as well as other heavy metals reduce mitotic rates at the metaphase stage in the root meristematic zone. 13 According to Fahr et al., 16 plant roots under Pb stress respond by synthesizing callose. This formation might serve as a natural barrier that inhibits the uptake of Pb. 59
In this study, all Indonesian tomatoes demonstrated an increase in plant height after Pb treatment (Figure 3). In many reports, a high concentration of heavy metals may result in decreased plant growth performances. This may be due to the production of free radicals as a consequence of plant exposure to heavy metals. 60 It causes subsequential changes to plant physiology including plant cell division, photosynthesis, deformation of the cellular membrane and gene expression alterations. 61 In addition, Pb contamination also alters enzymatic antioxidant activities. 61 However, while some heavy metals are devoid of beneficial roles in plant growth and usually contribute to some adverse effects,60,62 other heavy metals, at specific concentrations, could enhance plant growth. Jayakumar et al. 63 reported an enhancement in tomato growth after Cobalt (Co) exposure. Similarly, growth in cluster bean (Cyamopsis tetragonoloba (L.) Taub) was enhanced after Zn treatment. 64 Nevertheless, plant responses to heavy metals might be genotype-dependent.
Studies on plant genetic diversity using RAPD markers are crucial in uncovering intra and inter-specific polymorphisms. 36 In addition, it can be used to explain the variation in plant responses to environmental stress, including heavy metal contamination. In this investigation, we successfully discriminated the four Indonesian tomato genotypes. Rewako and Permata genotypes were genetically distinct, while Opal and Mutiara showed a greater genetic similarity. In addition, the present study demonstrated that the genotypes displayed different morphological responses to the Pb treatment. It is important to note that this work is specifically limited to the genetic diversity of Indonesian tomato varieties and their several morphological responses, including plant height and root length. However, we realized that physiological, phytochemical, cytological and molecular responses also need to be further investigated. For instance, gluthatione and super oxide dismutase (SOD) might play important roles in Pb detoxifications. 65 Investigation of Pb content in different organs of tomato might also help to determine the toxicity of Pb in tomato plants. 66
It is also worth mentioning that some secondary metabolites produced by the plants are not only beneficial for human 67 but also important for protecting the plant against oxidative stress. 68 These metabolites include various compounds belonging to the family of phenolic, anthocyanin, ascorbic acid and terpenoids. In addition, relative gene expression analysis of metal-tolerance proteins (MTPs) is interesting and should be to be studied further. 69 Finally, the genetic diversity of tomato varieties in this study might be used for further exploration to decipher the mechanisms underlying Pb stress responses, choose Pb-resistant tomato cultivars, and develop new Indonesian tomato varieties tolerant to heavy metal stress.
Footnotes
Author contributions
Conceptualization, N.J.; data curation, C.E.S and A.L.J.; writing—review and editing, N.J and CES.; supervision, N.J, W.M, K.I.P and FM.; funding acquisition, N.J. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia, (grant number 927/PKS/ITS/2021).
