Abstract
Objective
Cigarette smoking accelerates kidney disease progression. Emerging evidence suggests that dapagliflozin (DAPA) exerts protective effects against various kidney injuries. This study aimed to investigate the effects of DAPA on nicotine (NIC)-induced apoptosis in human kidney proximal tubular epithelial (HK-2) cells.
Methods
HK-2 cells were treated with NIC, DAPA or selective mitogen-activated protein kinase (MAPK) inhibitors (SP600125 and SB203580). Cell viability, apoptotic cell death and reactive oxygen species (ROS), namely intracellular ROS and MitoSOX, were assessed using a CCK-8 assay and flow cytometry. The concentrations of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) were determined by enzyme-linked immunosorbent assay. Protein expression levels related to mitochondrial function, endoplasmic reticulum (ER) stress and the MAPK signalling pathway were analysed by immunoblotting.
Results
Dapagliflozin and MAPK inhibitors significantly attenuated NIC-induced apoptosis and improved HK-2 cell viability, as evidenced by increased Bcl-2 expression and decreased Bax and Caspase-3 expression. Apoptosis attenuation was closely associated with the suppression of p-ASK1/p-JNK/p-p38 MAPKs and p-MEK3/p-MEK4 activation. Dapagliflozin and MAPK attenuations regulate the expression of oxidant and antioxidant proteins, reducing intracellular ROS and MitoSOX overproduction and thereby alleviating mitochondrial dysfunction and ER stress. Both agents also significantly reduced pro-inflammatory cytokine levels, including TNF-α and IL-1β.
Conclusions
These findings suggest that DAPA protects HK-2 cells from NIC-induced apoptosis by modulating the ASK1/p38/JNK MAPK signalling pathway, reducing oxidative stress and alleviating inflammation.
Introduction
Cigarette smoking is the leading cause of premature death and morbidity worldwide. Despite widespread public education and government policies banning smoking in public areas, the number of cigarette smokers remained at 1.1 billion in 2015. By 2030, cigarette smoking is projected to cause over 8 million deaths annually.1,2 In addition to increasing the risk of atherosclerosis, cancer and chronic obstructive pulmonary disease, cigarette smoking contributes to the progression of progressive chronic kidney disease (CKD) and worsens renal function in patients with diabetes 3 and hypertension. 4 Moreover, cigarette smoking by both kidney donors and recipients has been shown to reduce graft survival rates.5,6 Nicotine (NIC), a major toxic component of cigarette smoke, exacerbates acute kidney injury (AKI), accelerates CKD progression and worsens diabetic nephropathy (DN) through mechanisms involving oxidative stress, inflammation and apoptosis.7–9
Apoptosis signal-regulating kinase 1 (ASK1), also known as MAP3K5, is an upstream signalling kinase that activates c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) by phosphorylating MAPK kinase 3/6 (MEK3/6) and MEK4/7 in response to various stimuli. 10 Prolonged activation of the ASK1/P38/JNK signalling pathway is associated with inflammatory responses, cell death and fibrosis in multiple organs. 11 In the kidney, genetic deficiency or selective inhibition of ASK1 reduces p38 MAPK and JNK activation, thereby decreasing oxidative stress-induced apoptosis and fibrosis.12,13 Thus, inhibiting ASK1/P38/JNK signalling represents a potential therapeutic strategy for CKD management.
Sodium-glucose cotransporter 2 (SGLT2) inhibitors are a novel class of antidiabetic drugs that lower glucose and reduce HbA1c levels by 0.5–1% through direct SGLT2 blockade, inhibition of renal glucose reabsorption and promotion of urinary glucose excretion. 14 In addition, SGLT2 inhibitors are associated with a lower incidence of hypoglycaemia and reduced body weight compared to other antidiabetic agents. 15 The CREDENCE trial with canagliflozin and the EMPA-REG OUTCOME trial with empagliflozin demonstrated that SGLT2 inhibitors lower the risk of CKD progression and cardiovascular events in patients with type 2 diabetes and albuminuric CKD.16,17 Similar renoprotective effects have been observed in models of AKI, 18 chronic tacrolimus nephrotoxicity, 19 and unilateral ureteral obstruction (UUO). 20
Given these molecular mechanisms, we aimed to assess whether dapagliflozin (DAPA) inhibits NIC-induced apoptosis by modulating the ASK1/p38/JNK MAPK signalling pathway in the human kidney proximal tubular epithelial cell (HK-2) line.
Methods
Cell culture and treatment
HK-2 cells (CVCL_0302, ATCC) were cultured in Dulbecco's modified eagle's medium (DMEM; Viva Cell, China) supplemented with 10% foetal bovine serum (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA), 100 U/mL penicillin and 100 μg/mL streptomycin (Viva Cell, China). The cells were maintained in a 37°C incubator containing 5% CO2. Cells were treated under the following conditions for 24 h: (1) control; (2) Dapa (461432–26–8, 99.74%, TargetMol) (10 μM); (3) NIC (54–11–5, ≥ 99%, MERCK) (200 μM); (4) NIC + Dapa; (5) NIC + SP600125 (129–56–6, 99.73%, MCE) (20 μM); and (6) NIC + SB203580 (10 μM) for 24 h.
Cell viability assay
HK-2 cells (1.0 × 104 cells/well) were seeded in a 96-well plate, and cell viability was assessed using the Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan). After treatment as described above, 10 μL of CCK-8 solution was added to each well, followed by incubation at 37°C for 3 h. Absorbance was measured at 450 nm using a VersaMax Microplate Reader (Molecular Devices, LLC, Sunnyvale, CA, USA).
Measurement of reactive oxygen species production
Intracellular reactive oxygen species (ROS) production was detected using a ROS assay kit (Invitrogen). HK-2 cells (2.0 × 105 cells/well) were seeded in 6-well plates and subjected to the respective treatments. After trypsinisation and centrifugation, the cells were incubated with DCFH-DA (final concentration: 10 μmol/L) for 30 min at 37°C. Following three washes with PBS, fluorescence intensity was measured using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA).
Measurement of mitochondrial ROS
Mitochondrial ROS levels in HK-2 cells following different treatments were assessed using the MitoSOXTM Red mitochondrial superoxide indicator (M36008, Invitrogen). After staining with MitoSOXTM Red for 30 min at 37°C, samples were analysed by flow cytometry. Forward- and side-scatter data were collected, and values were obtained for the % gated R3 region. All samples were analysed in triplicate.
Apoptosis assay
Apoptosis was assessed using an Annexin V-FITC Apoptosis Detection Kit (MBL, Japan) and analysed using flow cytometry (BD FACSCalibur, USA). Cells were seeded and treated with DAPA, NIC or MAPK inhibitors for 24 h, then washed twice with PBS. Annexin V-FITC and propidium iodide staining was performed at room temperature under light-protected conditions. Each assay was conducted independently in triplicate.
Immunoblotting
Protein expression was analyzed by immunoblotting using an Odyssey Li-COR CLx infrared laser scanner following previously described methods. 20 Target proteins were quantified using ImageJ software with β-actin (AB_449644, 1:1000, Abcam) as the internal reference. Antibodies included: Abcam: Bax (AB_351500), Bcl-2 (AB_2064155), Caspase-3 (AB_2827742), TRX (AB_10863650), TXNIP (AB_2923036), MnSOD (AB_2191667), p-ASK1 (AB_867740), p-MEK3 (AB_1658840), p-MEK4 (AB_11156912), p-p38 (AB_2576214), p-JNK (AB_1523840) and XBP1 (AB_778939). WanLeiBio: CHOP (AB_3675430), PINK1 (AB_3678575) and Parkin (AB_3665473). All antibodies were used at 1:1000 dilution. LI-COR Biosciences IRDye 800CW goat anti-Rabbit (AB_621843, 1:20000,).
Enzyme-linked immunosorbent assay
The concentrations of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) secreted by the treated cells were measured using enzyme-linked immunosorbent assay (ELISA). Each experimental group included at least three replicates. The ELISA kit was purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd (Shanghai, China), and all procedures were carried out according to the manufacturer's instructions.
Measurement of oxygen consumption rate
Cells were seeded in 96-well plates and treated according to the experimental conditions for 60 min (three wells per group). Following treatment, the cells were incubated in Seahorse XF DMEM medium at 37°C without CO2 for an additional 60 min. Oligomycin, carbonyl cyanide 4- (trifluoromethoxy)phenylhydrazone and rotenone/antimycin A were sequentially added to the sensor cartridges. The oxygen consumption rate (OCR) and four mitochondrial respiratory parameters – basal respiration, ATP production, maximal respiration and spare respiratory capacity – were measured using a Seahorse XF96 extracellular flux analyser (Seahorse Bioscience, USA).
Statistical analysis
Data are presented as the mean ± SEM. Multiple group comparisons were conducted using one-way ANOVA followed by the Bonferroni post-hoc test using the SPSS software (version 21.0; IBM, Armonk, NY, USA). Statistical significance was set at p < 0.05.
Results
Dapagliflozin attenuates NIC-induced apoptotic cell death in HK-2 cells
We investigated the effects of DAPA on apoptotic cell death in NIC-stimulated HK-2 cells. Flow cytometry analysis demonstrated that DAPA reduced NIC-induced apoptotic cell death (Figure 1A) and improved cell viability (Figure 1B). These effects were accompanied by increased Bcl-2 expression and decreased Bax and Caspase-3 expression (Figure 1C).

DAPA treatment markedly decreased apoptotic cell death and improved cell viability in NIC-stimulated HK-2 cells, accompanied by regulation of apoptosis-controlling genes. Apoptotic cell death, cell viability and gene expression were assessed using flow cytometry (A), the CCK-8 assay (B) and immunoblotting (C). #p < 0.05 vs. CON; *p < 0.05 vs. NIC; mean ± SEM; n = 3 independent experiments. DAPA: dapagliflozin; NIC: nicotine.
Dapagliflozin suppresses NIC-activated ASK1/p-38/JNK MAPK and MEK3/MEK4 in HK-2 cells
Next, we examined the effects of DAPA and MAPK inhibitors on the expression of ASK1/p38/JNK MAPK and MEK3/MEK4 in NIC-stimulated HK-2 cells. Immunoblotting analysis revealed that NIC activated ASK1 and increased the expression of downstream MAPKs, including p-MEK3/p-MEK4 and p-JNK/p-p38. In contrast, treatment with DAPA or MAPK inhibitors suppressed their expression (Figure 2). These findings suggest that DAPA mitigates NIC-induced apoptosis by interfering with the ASK1/p38/JNK MAPK signalling pathway in HK-2 cells.

DAPA or selective MAPK inhibitors deactivated MAPK signalling pathway proteins in NIC-stimulated HK-2 cells. The expression of p-ASK1/p-p38/p-JNK and p-MEK3/p-MEK4 MAPKs was assessed by immunoblotting. #p < 0.05 vs. CON; *p < 0.05 vs. NIC; mean ± SEM; n = 3 independent experiments. DAPA: dapagliflozin; MAPK: mitogen-activated protein kinase; NIC: nicotine; p-ASK1: phosphorylated apoptosis signal-regulating kinase 1; p-JNK: phosphorylated c-Jun N-terminal kinase.
Dapagliflozin attenuates NIC-induced intracellular ROS and MitoSOX production in HK-2 cells
Nicotine-induced renal injury is closely associated with ROS-mediated oxidative stress, where an imbalance between oxidant and antioxidant enzymes plays an integral role. Dapagliflozin significantly reduced intracellular ROS levels (Figure 3A) and MitoSOX overproduction (Figure 3B) in NIC-stimulated HK-2 cells. Concomitantly, DAPA counteracted oxidative stress by upregulating MnSOD and TRX expression while downregulating TXNIP expression (Figure 4A). Similar renoprotective effects of DAPA have been observed in DN 21 and nondiabetic cardiorenal disease, 22 suggesting its potential as an oxidative stress scavenger.
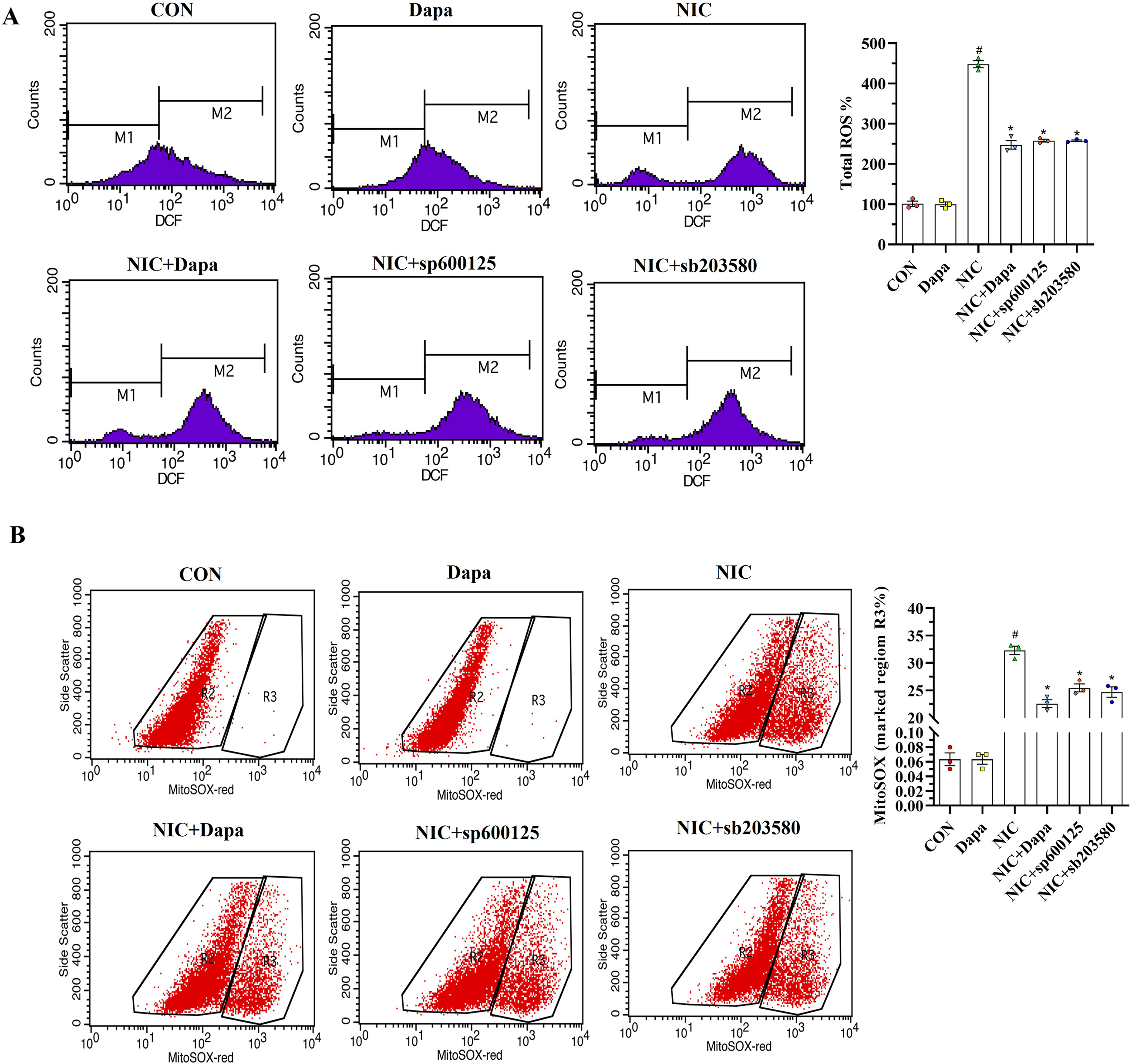
DAPA treatment significantly reduced ROS and MitoSOX overproduction in NIC-stimulated HK-2 cells. Intracellular ROS (A) and MitoSOX levels (B) were measured using flow cytometry. M1 represents the population with baseline fluorescence (low ROS levels), and M2 denotes the population with elevated fluorescence (high ROS levels). #p < 0.05 vs. CON; *p < 0.05 vs. NIC; mean ± SEM; n = 3 independent experiments. DAPA: dapagliflozin; NIC: nicotine; ROS: reactive oxygen species.

DAPA treatment restored the balance between oxidants and antioxidants and reduced TNF-α and IL-1β concentrations in NIC-stimulated HK-2 cells. Immunoblotting (A) was used to assess oxidant and antioxidant protein expression, while ELISA (B) measured TNF-α and IL-1β in cell supernatants. #p < 0.05 vs. CON; *p < 0.05 vs. NIC; mean ± SEM; n = 3 independent experiments. DAPA: dapagliflozin; ELISA: enzyme-linked immunosorbent assay; IL-1β: interleukin-1β; NIC: nicotine; TNF-α: tumor necrosis factor-α.
Dapagliflozin attenuates NIC-induced inflammation in HK-2 cells
Oxidative stress-mediated inflammation is strongly implicated in NIC-induced kidney injury.8,23 To determine whether DAPA inhibits NIC-induced inflammation, we measured inflammatory cytokine levels using ELISA. Dapagliflozin treatment reversed the NIC-induced increase in IL-1β and TNF-α levels in HK-2 cells (Figure 4B), indicating its anti-inflammatory effects.
Dapagliflozin protects against NIC-induced mitochondrial dysfunction and endoplasmic reticulum stress in HK-2 cells
Emerging evidence suggests that mitochondrial dysfunction and endoplasmic reticulum (ER) stress contribute to renal injury following NIC exposure.23,24 Immunoblotting analysis revealed that the dysregulation of mitochondrial quality control genes (Pink1 and Parkin-1)- and ER stress-regulating genes (CHOP and Xbp1) in NIC-treated HK-2 cells was mitigated by DAPA administration (Figure 5A and B). Furthermore, DAPA enhanced mitochondrial function by increasing the OCR, basal respiration, ATP production, maximal respiration and spare respiratory capacity in NIC-treated HK-2 cells (Figure 5C).
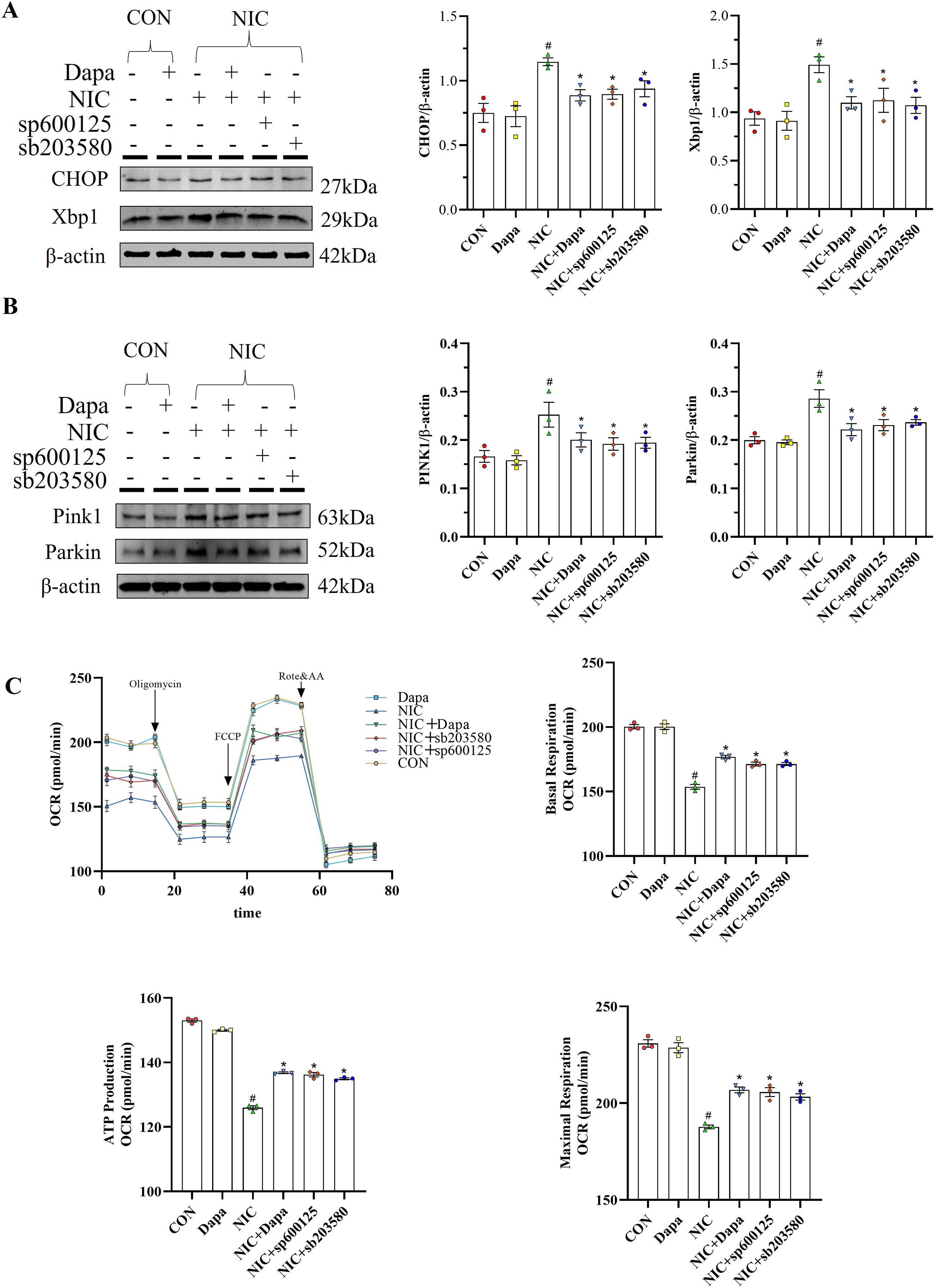
DAPA treatment suppressed CHOP, Xbp1, Pink1 and Parkin expression in NIC-stimulated HK-2 cells while enhancing mitochondrial respiration, as indicated by increases in the oxygen consumption rate (OCR), basal respiration, ATP production, maximal respiration and spare capacity. The expression of mitochondrial (A) and ER stress-related (B) genes was assessed by immunoblotting, and mitochondrial respiration (OCR) was assessed using an XF24 Extracellular Flux Analyzer(C). Dapa #p < 0.05 vs. CON; *p < 0.05 vs. NIC; mean ± SEM; n = 3 independent experiments. DAPA: dapagliflozin; ER: endoplasmic reticulum; NIC: nicotine.
Discussion
Cigarette smoking contributes to albuminuria and accelerates the progression of renal failure in individuals with 25 and without diabetes. 26 Moreover, smokers have a 30% higher risk of graft failure among renal transplant recipients, while smoking cessation significantly reduces this risk by 34%, while smoking cessation significantly reduces this risk by 34%. 27 In this study, we demonstrated that DAPA effectively mitigates NIC-induced HK-2 cell injury, potentially by inhibiting ASK1/p38/JNK MAPK-mediated apoptosis. Our findings provide supporting evidence for the clinical use of DAPA in managing CKD in smokers.
Nicotine induces apoptotic cell death in podocytes and HK-2 cells by generating ROS via activation of the NF-κB and MAPK signalling pathways.24,28 Previous studies have shown that DAPA reduces cardiomyocyte apoptosis following acute myocardial infarction in H9c2 cells 29 and protects against apoptosis in diabetic human retinal microvascular endothelial cells. 30 Consistent with these findings, our study, along with others, demonstrated that DAPA improves HK-2 cell viability and decreases apoptosis.20,31 Specifically, we found that treatment with DAPA or MAPK inhibitors significantly reduced NIC-induced HK-2 cell apoptosis, accompanied by modulation of apoptosis-related gene expression and suppression of key MAPK signalling proteins, including p-ASK1, p-MEK3, p-MEK4, p-JNK and p-p38. These results suggest that DAPA inhibits HK-2 cell apoptosis, likely by interfering with the ASK1/JNK/p38 signalling pathway, similar to its protective effects previously observed in the heart, 32 pancreas 33 and kidney. 31
The precise mechanism underlying the anti-apoptotic effect of DAPA in NIC-treated HK-2 cells remains unclear; however, oxidative stress is a likely contributor. Extensive evidence suggests that NIC induces renal injury via oxidative stress and apoptosis.23,34,35 This is supported by findings that the antioxidant curcumin alleviates NIC-induced renal damage by enhancing antioxidant defences. 36 Zaibi et al. 37 reported that DAPA exhibits antioxidant properties by reducing cytosolic and mitochondrial ROS overproduction in HK-2 cells under oxidative stress. Additionally, DAPA suppresses NOX-4 expression and decreases ROS production in a hypoglycaemia-dependent manner in mProx24 cells. 38 Consistent with these findings, we found that DAPA counteracted oxidative stress by decreasing intracellular ROS and MitoSOX production while balancing the expression of antioxidants (MnSOD and TRX) and oxidants (TXNIP). The anti-apoptotic effects of DAPA in this study may, at least in part, be attributed to its antioxidant properties. The anti-apoptotic and antioxidant effects of DAPA observed here may also involve cAMP signaling, which is known to regulate NF-κB activity, ROS production and inflammatory responses in renal cells.39–41 For example, NIC has been shown to elevate cAMP levels in cardiomyocytes, exacerbating oxidative stress, 40 while cAMP modulation can attenuate NF-κB-driven inflammation in HK-2 cells. 41 Although cAMP levels were not measured in this study, future investigations should explore whether DAPA alters cAMP signaling to suppress NIC-induced ASK1/p38/JNK activation, thereby mitigating apoptosis and oxidative stress. Such findings would further clarify the pleiotropic mechanisms underlying SGLT2 inhibitors’ renoprotective effects.
Regardless of ROS levels, oxidative stress-induced renal inflammation is a hallmark of NIC intoxication. Amer et al. demonstrated that chronic NIC treatment increases aortic IL-1β and TNF-α levels in a rat model of aortic calcification. 42 In the kidney, NIC activates NLRP3 inflammasomes, leading to the recruitment of apoptosis-associated speck-like protein containing a CARD (ASC), caspase-1 activation and increased IL-1β and IL-18 production.9,43 In this study, ELISA analysis showed that DAPA significantly reduced IL-1β and TNF-α concentrations in NIC-treated HK-2 cells. Our results align with previous studies on DN 44 and UUO, 20 supporting the anti-inflammatory effects of DAPA.
It is also evident that DAPA inhibits NIC-induced HK-2 cell apoptosis by preserving intracellular organelle integrity, as mitochondria and ER play critical roles in cellular pathophysiology. Previous studies have shown that NIC triggers mitochondrial dysfunction and persistent ER stress in chronic tacrolimus nephrotoxicity and HK-2 cells.23,24 Dapagliflozin has been reported to alleviate renal fibrosis by enhancing the mitochondrial-respiratory chain complex and suppressing ER stress in adenine-induced renal injury and diet-induced obesity models.45,46 In this study, DAPA administration decreased the expression of mitochondrial-related proteins (Parkin and Pink 1) and ER stress-related proteins (CHOP and Xbp1), coinciding with decreased oxidative stress and apoptosis. Thus, the anti-apoptotic effects of DAPA in this study were associated with the preservation of intracellular organelle function.
Dapagliflozin was originally approved for the treatment of type 2 diabetes mellitus at all stages. However, it has also been shown to lower the risk of major adverse kidney and cardiovascular events, as well as all-cause mortality, in patients with diabetic and nondiabetic CKD. 47 Our findings demonstrate that DAPA attenuates NIC-induced HK-2 cell apoptosis via the ASK1/JNK/p38 MAPK signalling pathway. The attenuation of oxidative stress and preservation of intracellular organelle function may be key mechanisms underlying its renoprotective effects. Therefore, early treatment with DAPA in patients with DN may provide additional benefits beyond glycemic control. This study has certain limitations. First, the experiments were conducted solely in the HK-2 cell line. While HK-2 cells are a well-established model for human renal proximal tubular injury and have been extensively utilized in studies investigating NIC toxicity and drug interventions, the use of a single cell line may limit the generalizability of our findings. We prioritized HK-2 cells due to their relevance to tubular pathophysiology and their widespread validation in apoptosis and oxidative stress research. Future studies will incorporate additional renal cell lines (e.g. HEK293, NRK-52E) and in vivo models to confirm these results and explore tissue-specific responses. Furthermore, we focused on the ASK1/p38/JNK pathway as a key mechanism, NIC is also known to modulate other signaling pathways, such as Akt and ERK, which may contribute to apoptosis and oxidative stress in HK-2 cells. Our study did not investigate whether DAPA exerts its protective effects through these additional pathways. Future studies should evaluate the potential role of Akt and ERK signaling in DAPA-mediated renoprotection to determine whether its effects are pathway-specific or involve broader network regulation. Second, this study was conducted in an in vitro HK-2 cell model, which may not fully replicate the complex in vivo kidney microenvironment. Second, while we identified MAPK inhibition as a key mechanism, other pathways, such as autophagy or metabolic adaptations, may also contribute to DAPA's protective effects. Additionally, no formal sample size calculation was performed, and the use of triplicates may limit the robustness of statistical conclusions. Further, in vivo studies are needed to confirm the clinical relevance of our findings.
Conclusion
Dapagliflozin attenuates NIC-induced apoptosis in HK-2 cells by targeting the ASK1/p38/JNK MAPK signaling pathway, reducing oxidative stress and preserving mitochondrial and ER integrity. These findings suggest that DAPA may have therapeutic potential in mitigating NIC-induced kidney injury, particularly in smokers at risk of CKD. Future studies should explore its clinical efficacy and underlying mechanisms in in vivo models and patient populations.
Footnotes
Abbreviations
Acknowledgements
This study was supported by the National Natural Science Foundation of China (No. 81560125; No. 81760293; No. 81760132); Science and Technology Research “13th Five-Year Plan” Projects of Education Department of Jilin Province (JJHK20180890KJ; JJHK20180915KJ).
Ethical considerations
This study was approved by the Medical Ethics Committee of the Affiliated Hospital of Yanbian University (Yanji, Jilin, China; Date of Approval: February 3, 2017). This study also complied with the principles of the Declaration of Helsinki and the Ethical Review of Biomedical Research Involving Human Beings.
Author contributions
Can Li, Byung Ha Chung and Chul Woo Yang designed the research; Songyu Li, Sheng Cui, Qi Yan Nan, Xue Jing Lin, Hai Lan Zheng performed in vitro study; Shang Guo Piao and Ji Zhe Jin analyzed the data and performed molecular works; Songyu Li wrote the manuscript; Lei Wang and Can Li revised the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 81560125; No. 81760293; No. 81760132); Science and Technology Research h13th Five-Year Plan” Projects of Education Department of Jilin Province (JJHK20180890KJ; JJHK20180915KJ).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
