Abstract
Phytonanotechnology plays a crucial part in the production of good quality and high-yield food. It can also alter the plant's production systems, hence permitting the efficient, controlled and stable release of agrochemicals such as fertilizers and pesticides. An advanced understanding of nanomaterials interaction with plant responses like localization and uptake, etc. could transfigure the production of crops with high disease resistance and efficient nutrients utilization. In agriculture, the use of nanomaterials has gained acceptance due to their wide-range applications. However, their toxicity and bioavailability are the major hurdles for their massive employment. Undoubtedly, nanoparticles positively influence seeds germination, growth and development, stress management and post-harvest handling of vegetables and fruits. These nanoparticles may also cause toxicity in plants through oxidative stress by generation of excessive reactive oxygen species thus affecting the cellular biomolecules and targeting different channels. Nanoparticles have shown to exert various effects on plants that are mainly affected by various attributes such as physicochemical features of nanomaterials, coating materials for nanoparticles, type of plant, growth stages and growth medium for plants. This article discusses the interaction, accretion and toxicity of nanomaterials in plants. The factors inducing nanotoxicity and the mechanisms followed by nanomaterials causing toxicity are also instructed. At the end, detoxification mechanism of plant is also presented.
Introduction
Global production and distribution of food are facing major challenges due to the environmental contamination, increasing population, higher demands of water/energy and change in climate. 1 To meet the increasing challenges of expanding globe's population, the scientific community is striving their best to find alternative sources to enhance food crops’ yield. Of various strategies, nanotechnology has been shown to be one of the promising technologies in the field of agriculture. Current studies have revealed the auspicious competence of nanotechnology for improvement of the agricultural division by enhancing the productivity of agronomical contributions, also presenting solutions to the agricultural challenges for food security and productivity. 2
The use or exploitation of nanotechnology for the improvement of plants, control of diseases and increment of yield, called as phytonanotechnology. Phytonanotechnology has the potential to modify the plant's production systems, thus permitting efficient, controlled and stable agrochemicals release such as fertilizers, several pesticides and specific type of the biomolecules. 3 Understanding the interaction of nanoparticles at advanced level with the plant responses like localization and uptake can transfigure the production of crops with high disease resistance, efficient nutrients utilization and crop yields. 4 Nanomaterials are characterized by unique physicochemical features such as surface charge, surface area, agglomeration rate, surface coating and particle morphology. 5 These distinctive properties commonly result from the surface structure, stability, chemical properties and shape of NPs. 6 Phytonanotechnology could play its significant role in the production of “Smart Crops.” Target-specific, self-regulated, programmed, time-controlled, multifunctional capabilities are provided with the help of nanoscale particles. 7
In the environment, nanomaterials can originate from various natural processes like microbial activities, incomplete combustion, chemical precipitation, anthropogenic activities, geological effects, intentional release such as nanofertilizers or nanopesticides or unintentional release such as corrosion and garbage disposal. 8 However, the extensive use of NPs has raised concern regarding the detrimental effects of NPs on the environment, plants and human health. 9 In general, the residual NPs mainly contaminate the food chain by entering into the plant system. 10 In addition, NPs can be integrated into plant and microbial systems, thus leading to absorption of NPs in the food chain, that subsequently threatens the quality and safety of human health. 11
There are different types of nanomaterials that can be mainly categorized into two classes based on their chemical composition: inorganic and organic nanomaterials. In relation to plants, the use of single and multiwalled carbon nanotubes (CNTs) from organic nanomaterials class is common and their applications include early plant growth, pesticides, seed germination and biosensors analysis, 12 whereas inorganic metallic nanomaterials such as silver, gold, zinc oxide, iron oxide, titanium dioxide and copper oxide effects have been widely investigated in numerous plant species. Generally, these NPs exhibit different effects on the plants that may be neutral, positive or toxic depending on their types, size, dose, duration of application, exposure method, targeted plant species and environmental conditions.13,14
Because of their catalytic attributes, presence of active surfaces and their reduced size, NPs or nanomaterials can easily enter cell through cell membranes and induce toxicity through their impact on various intracellular biomolecules. The NPs-associated phytotoxicity might occur via their interaction with plant cells that may affect the nutrient absorption, besides, these accumulated NPs in plants can negatively regulate the cellular functions by interacting with various cellular structures. Different The phytotoxicity of NPs could reduce the number of seedlings, reduce root and stem growth, delay the flowering, restriction in seed's emergence that will ultimately decrease overall yield.15–18 Furthermore, NPs can cause breakage of cell wall, gene expression inhibition, condensation of chromatin and mitotic arrest by disturbing the metaphase.18–20
In this review, the uptake of these nanomaterials and their toxicity on environment and plants will be discussed. Moreover, factors affecting phytotoxicity of NPs and detoxification mechanism induced in plants have also been presented.
Uptake and translocation of nanomaterials in plants
In the field of agriculture, plants are more likely to be exposed to different nanomaterials via the applications of agricultural nanotechnologies such as pesticides, growth regulators and pesticides and by atmospheric deposition particularly in industrial and urban areas. 21 Incorporation of nanomaterials in edible and reproductive plant parts is of utmost importance as various NPs are dangerous to the living organisms. 22
Nanomaterials make their way to the agricultural field by various approaches, however, the most direct way to reach the soil is through the application of nano-formulated agricultural chemicals. NPs uptake in plants mainly occurs through the soil or due to foliar application.22,23 Transportation of NPs and their uptake by plants fluctuate with cultivars, plant species and growth stages. 24 The penetration of NPs into root epidermis occurs via convoluted events starting from entry of NPs into plant's xylem, stele and then to leaves (Figure 1). 25 The transport of NPs through undamaged cell membrane takes place via pores of cell membrane that are specific to size. 26 NPs assimilate themselves passively out of endodermis prior to approaching to the stele of plant. 27 NPs uptake usually occur due to active transport mechanism and processes like signaling, recycling and plasma membrane regulation.25,28 Normally, NPs can internalize the leaves via stomata. as cuticles restrain the entry of NPs to size limit of <5 nm. The NPs having size > 10 nm enter via stomata, and transportation is carried out via apo-symplastic routes in plant's vascular system. 3 The internalized NPs are transported through phloem tubes alongside sugar flow. Due to vascular transportation via phloem the NPs travel two-way and ultimately accumulated in varying organs such as fruiting bodies, stems, roots and young leaves because these plant parts can act out as potential sinks for cell sap.29–31
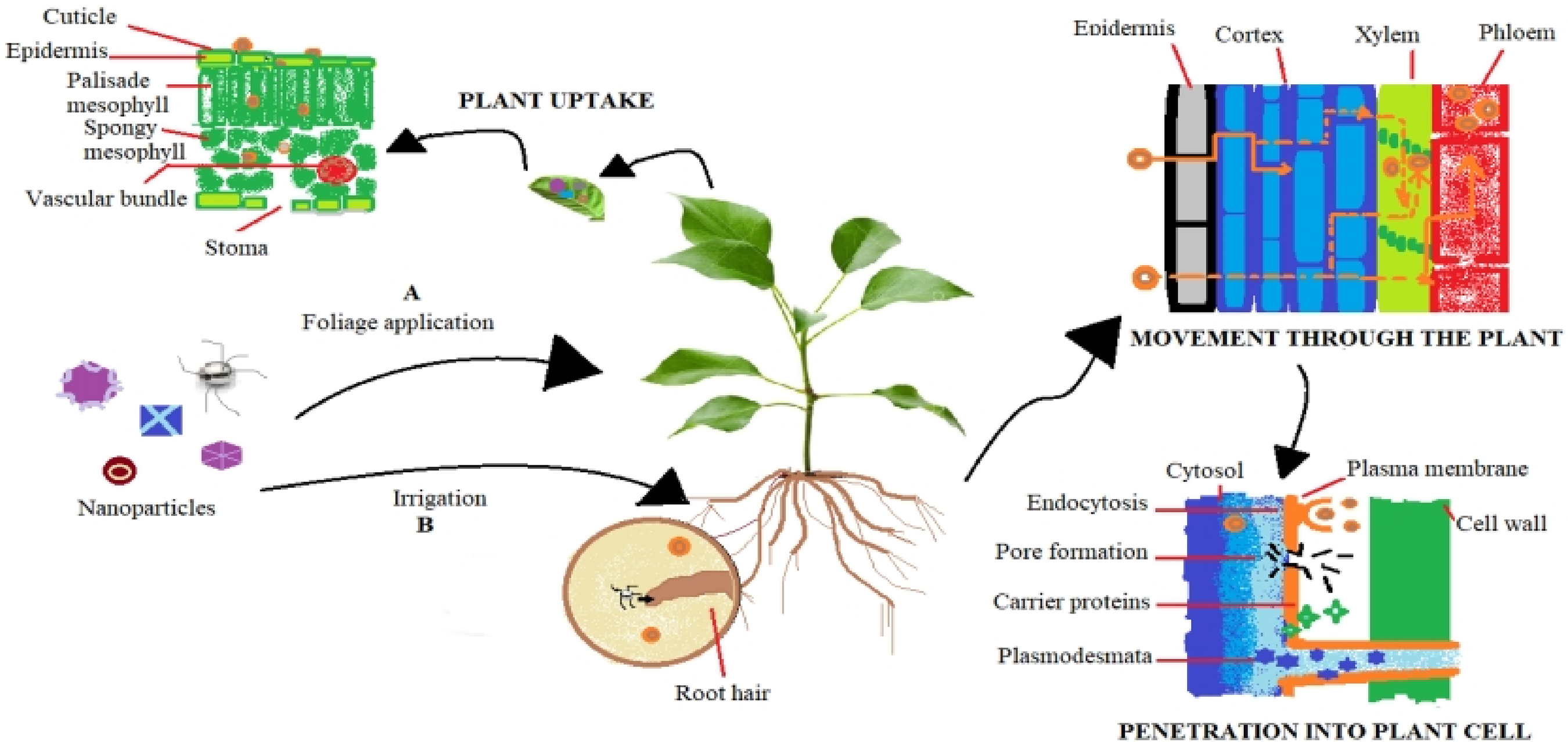
Mechanism of translocation and uptake in plants (A) Uptake of nanoparticles via foliar application. Penetration of nanoparticles into leaf's cuticle and further entry into palisade, spongy mesophyll and ultimately in vascular bundles (B) Uptake of nanoparticles through roots, they get penetrated in root hairs then into channels (xylem and phloem) by apoplastic and symplastic pathways. 25
Phytotoxic effects of nanoparticles
Due to the technological advancements, the substantial increase in quality and yield of crops has been observed in the agriculture sector, however, serious concerns have also been raised owing to the toxicity of nanomaterials toward growth, root and shoot extension, seeds germination and biomass of plants.32–34 Once NPs are translocated inside plants, they can directly change the structure of cells, molecules, membranes, and may also affect the protective system of plants. Furthermore, there may be unintended effects of NPs based on their properties, solubilization and generation of the reactive compounds. 35 NPs stimulate production of reactive oxygen species (ROS) that concern growth and development of plant (Figure 2, Table 1).32,36

Phytotoxic effects of nanoparticles on development and growth in plant. 32
Summary of phytotoxicity of nanoparticles in different plants.

At elevated concentrations, NPs may change the structure and functional processes of plants. High dose of NPs in root hinders seed germination, changes nutrients and water uptake, inhibits development of root and reduces biomass production and leaf development. Nanomaterials based toxicity also involves oxidative burst that results in membrane disruption, disorganization of chloroplast, decreased photosynthetic activity, damage to the cells and changes in gene expression.
Effects on physiological growth parameters of plants
NPs can have considerable negative impacts on plants including inhibition of germination, abolishment of shoot elongation and may even cause death of plants. Seeds germination is crucial for productivity and there are number of reports highlighting toxicity of NPs induced seed's germination inhibition.29,51 Seeds germination in corn and ryegrass was significantly decreased after treating with ZnO NPs and zinc NPs (at 2000 mg/L), respectively. 18 Another study reported a decrease in seed's germination process when TiO2 NPs were exposed at 0.01, 1, and 10 mg/mL concentrations.52,53 Wu et al. compared toxicity of selected ZnO NPs and zinc ions (Zn) on plants using transcriptional and physiological studies. Both zinc NPs and zinc ions inhibited primary growth of plants, with former inhibiting the development of zones of elongation and the latter having toxic effects toward cells of the meristem. 54 Hao et al. observed the toxicity of several concentrations of mesoporous carbon NPs on seedlings of Oryza sativa. The multi-walled carbon nanotubes (MWCNs) resulted in significantly impaired root and shoots lengths. MWCNs having a shorter diameter (MCN2: 80 nm) had pronounced inhibitory effect on the length.
The phytotoxic effects of different metal oxide nanoparticles were tested by incubating seeds of cucumber, lettuce and radish, with metal oxide NPs (NiO, CuO, Fe2O3, TiO2, Co3O4) significant inhibition of germination of seeds was noted in all three plants when exposed to NiO and CuO NPs. This deleterious impact was attributed to NPs adsorption on seeds surface subsequent to the release of free metallic ions. 55
NPs not only impede germination of seeds but may also have negative impacts on other growth parameters of plants including shoot and root elongation, leaf number and biomass in plants. For example, oxidized multiwalled carbon nanotubes (MWCNTs) significantly decreased biomass production and growth in mustard. 56 Similar toxic effects were reported in Glycine max and Zea mays when exposed to multiwalled CNTs and C60 fullerenes and a significant decrease of 36.5%–45% was reported in plant biomass. 57 Mirzajani et al. observed that silver NPs (Ag NPs) were not able to enter roots cell of Oryza sativa (rice) when their small concentration was used (30 μg/mL) but caused toxicity and destruction of the structure of the cells at higher dose. 39 Shaw and Hossain, also described toxicity of CuO NPs in Arabidopsis seedlings as substantial decline in root length and Arabidopsis seedlings weights was observed. 58
Lin and Xing studied the impact of Zn/ZnO NPs on elongation of root in six plant species, that is, lettuce, rape, cucumber, radish, ryegrass and corn. 18 Their results demonstrated that at concentrations of 2000 mg/L, roots elongation in plants practically ceased. A different study observed the phytotoxicity of metallic oxide NPs for instance La2O3, CeO2, Yb2O3 and Gd2O3 NPs on elongation of root in seven plants (lettuce, cabbage, radish, cucumber wheat, tomato and rape). 59 Their results revealed that impacts on the growth differed considerably among plant species and NPs. CeO2 NPs at concentration of 2000 mg/L had no effect on root elongation in six plants excluding lettuce, whereas at this concentration, Gd2O3 NPs, La2O3 NPs and Yb2O3 NPs had severely reduced elongation of root in tested plant species. Oryza sativa treated with Ag NPs (0.5 mg/L concentration) resulted in significant decrease in the shoot and root biomass and leaf area, carotenoids and chlorophyll contents were altered. 34 ZnO NPs also executed toxic effects in corn plants by damaging epidermis, vacuoles, roots and cortical cells, when applied at 1000 mg/L concentration. 60
Toxic effects on phytohormones
Plant growth hormones are extremely significant contributors influencing the growth and crop yield in different environmental circumstances such as stress. The activity and amount of phytohormones are crucial toxicity indicators in the plants. 61 NPs had a substantial impact on phytohormones production. It was observed that when rice seeds were subjected to the carbon nanotubes, phytohormone's concentration was reduced. 62 Bleecker and Kende studied that silver ions reduced production of ethylene, which resulted in a weakened interaction between ethylene and indole acetic acid (IAA). 63
Different concentrations of exogenously applied Fe2O3 NPs had adverse impact on root hormone concentrations in both the Bt-transgenic cotton and the usual cotton that resulted in impaired development of plant. After exposure to Fe2O3 NPs, the abscisic acid (ABA) and gibberellic acid (GA) contents in leaves were significantly reduced in conventional cotton in comparison with transgenic one that led to a rise in the GA content of leaves without affecting the ABA content. 64 Gui et al. 65 found an increase in IAA and ABA contents of roots appearing in both transgenic and non-transgenic rice when subjected to γFe2O3. 64 In contrast to these observations, Van et al. showed that CeO2 NPs posed no substantial impact on GA, IAA and ABA in leaves of usual and Bt-transgenic cotton comparing to control groups. However, conventional cotton was subjected to 500 mg/L cesium oxide NPs (CeO2 NPs), there was a 25% decrease in trans zeatin riboside (t-ZR) in leaves as compared to the control. 66
In Arabidopsis plant, increase in ZnO NPs dose resulted in a decline in the synthesis of phytohormones like auxins and cytokines in apices. On the contrary, when 20 and 100 mg/L concentration of NPs were utilized, cis zeatin, a hormone released in stress conditions, was increased by 28 and 59% in the roots, respectively. 44
Toxic effects on crops quality
Within environment, NPs can immensely change yield and quality of soil-based food crops. 67 Rico et al. and others noted toxicity of CeO2 NPs in rice plants by altering amino acids, fatty acids, phenolics and nonreducing sugars.26,68 Similar toxic effects were observed in transgenic cotton (Bt 29317) after application of CeO2 NPs and a decrease in the content of micronutrients, that is, magnesium, zinc, iron and phosphorous was observed.17,64 Au NPs also altered reducing sugars in mustard seeds. 69 Engineered nanoparticles (ENPs) have been reported to substantially affect plant's carbohydrate contents, 56 protein contents,70,71 phenolic content, radical scavenging ability72,73 and nutrient elements.74,75
Genotoxicity of NPs
NPs due to their smaller size have the potential to enter the cells and can cause differential gene expression. After exposure to NPs, these changes can be observed in various plants such as errors during cell division, DNA damage and chromosomal abnormalities and deregulation of micro-RNA. Exposure to various NPs such as NPs for instance, ZnO, CeO2, TiO2 and CuO induced genotoxic effects in different plant species of tobacco, buckwheat, onion and radish.42,76–81 Upon exposure to Ag NPs, cytologic changes were observed in barley and wheat. Ag NPs provoked chromosomal aneuploidy, micronuclei formation, deletion of chromosomes, chromosomal aberrations, binucleate cells, sticky chromosomes and chromosomal fragments deformation. 82 These NPs reduced mitotic index in faba beans root tips. 81 CuO NPs also induced alteration in genes responsible for root growth in the rice. They have been further found to be involved in DNA damage in ryegrass and radish.76,83 CuO NPs decreased mitotic index in onion roots and raised abnormality index. 84 It was reported that TiO2 NPs and CeO2 could alter gene 142 and 204 in Arabidopsis thaliana. 85
Factors influencing the phytotoxicity of NPs
NPs have been shown to exert various effects on plants that are mainly affected by various attributes such as physicochemical features of nanomaterials (stability, type, size and concentration), coating materials for NPs, type of plant, growth stages and growth medium for plants. 86
Physicochemical features of NPs
One of the most decisive factors for phytotoxicity of NPs is the concentration of nanomaterials. In most cases, a dose-dependent effect is observed and a rise in concentrations of NPs takes a sharp boost in threshold of toxicity. In four rice cultivars, exposure of Fe NPs in the concentration range of 4–9 mM altered lateral roots and shoot lengths, internal CO2 concentrations and pigments. 87 Similarly, a better dose of CuO NPs also exhibited toxicity (reduction of roots) by ion coverage on plants,23,83,88,89 Cucumis sativus, exposure of Fe3O4 engineered NPs, at 100–5000 mg/L concentration reduced the root development, whereas, in A. thaliana an increased growth rate was observed upon exposure to nanoscale zero-valent iron (nZVI) at 0.5 g/L concentration. 90
Because of their reduced size that usually results in an increased reactivity, NPs exhibit greater phytotoxicity in comparison to their bulk counterparts. For example, leaf necrosis in tobacco was dependent on size of particle; and 18 nm-sized Au NPs did not stimulate necrosis, whereas smaller NPs with a size of 3.5 nm induced necrosis of leaf. 91 However, in contrast to these findings, phytotoxic effects of NPs were enhanced by increasing their size. Yasur et al. observed that Ag NPs had not any substantial influence on castor growth, but significant inhibition of growth was observed when silver was used in bulk form. 92 In another study, TiO2 NPs significantly improved the rates of seed germination in tomatoes; but bulk TiO2 had inhibited seed germination. 93
Nanoparticles coating materials
When coating materials are used for NPs, they not only alter the functions of different nanomaterials but also change their solubility and toxicity. To modify toxicity mechanism induced by NPs, surface modifications can be performed that change cellular interactions of NPs. The studies revealed that toxic potential of CuO NPs was induced by solubilization of NPs in the toxic metal ions. The toxicity of NPs was altered by surface modifications in Lemna gibba. The modification resulted in enhanced ROS generation even at low concentrations of NPs that ultimately decreased the plant growth. 94 Similar to these observations, another research revealed that suspension of Ag NPs increased the rate of germination in 5 out of 11 species. However, exposure to gum Arabic-coated silver NPs (GA-AgNPs) increased the rate of germination in only one plant species. The increased rate of germination was due to the manifestation of a co-ligand in the soil which reduced toxicity and the bioavailability of Ag+. Due to the presence of coated material, toxicity of GA-Ag NPs was long lasting in comparison to AgNPs. 95
Growth medium and other environmental conditions
The environmental conditions including temperature, growth medium, pH and organic matter content of the soil can significantly alter the phytotoxicity of NPs. Majumdar et al. analyzed the effect of organic matter content on toxicity of CeO2 NPs in kidney bean plants. They observed that the plants subjected to CeO2 NPs in minimal organic matter soil for 52 days usually retained higher concentrations of Ce when compared to tissues gathered from soil enriched in organic matter. 96 In other studies, CeO2 NPs induced different levels of phytotoxicity in the lettuce seedlings grown in three different types of media. 65
Light can also affect various physiological responses in plants and alter NPs-induced phytotoxicity. Zou et al. reported the impact of post and pre-illumination conditions on the exposure of Ag NPs in Wolffa globosato.97,98 Their results revealed that Ag NPs toxicity was induced by the presence of light mainly by changing the protein content and photosynthetic pigments. Moreover, Ag NPs toxicity in soil microbes was reduced by increasing the pH and clay content; however, it was not affected significantly by organic content of soil.97,98
Seed types and growth stages
Phytotoxicity of NPs is also affected by different growth stages in plants. For example, in cucumber seedlings, there was a significant inhibition in leaf size after treating with 200 mg/L of CuO NPs and CeO2 NPs in comparison to untreated seeds. However, exposure to these NPs at the same concentrations did not affect the mature plants indicating that toxicity induced by NPs would vary with different growth stages. 99 Similarly, different cultivars and seed varieties can also exhibit a different response toward the same NP. For instance, in a study three different rice varieties (high amylose (HA), medium amylose (MA) and low amylose (LA)) were grown in soil improved with CeO2 NPs. 100 The results revealed that no significant difference in cerium content was examined between the control and HA varieties. Nevertheless, cerium content was increased in MA and LA varieties when compared to the control group. 100
Mechanism of NPs induced phytotoxicity
Some nanomaterials after diffusion get accumulated into the plants thereby leading to phytotoxicity. The mechanism of NPs induced phytotoxicity involves (a) the increased generation of ROS during the interaction between plants and NPs thus leading to oxidative damage, 101 (b) transcriptional response induced by NPs 102 and (c) NPs induced genotoxicity due to the interaction of NPs with chromosomal or mitochondrial DNA. It has been observed that oxidative stress may indirectly induce genotoxicity, such as chromosome aberration and micronucleus formation that may alter the gene expression and the level of plants biological components. 103
Generation of ROS and initiation of oxidative stress is a critical biomarker for NPs induced toxicity. 104 Plants produce a wide array of ROS species in reaction to NPs or heavy metal stress.105–109 ROS are reduced or activated forms of oxygen such as superoxide anion O−2, hydrogen peroxide H2O2, hydroxyl radical HO and singlet oxygen O2. These species of oxygen are highly reactive and toxic and can damage cellular components. 86 Physicochemical characteristics of NPs and test species both can affect the generation of ROS. Phytotoxicity of NPs induced by ROS generation might be dependent on factors as shape, particle dissolution, size and solubility of NPs, metal ions out from metal and metallic oxide NPs, light and biotransformation.107–109 Different consequences have been observed in plants due to ROS generation such as induction of damage due to oxidation, peroxidation of lipids, 110 change in ion transportation through cell membranes, 111 intracellular and extracellular harm to cell membrane. 3 Moreover, ROS generation in plants owing to the exposure to NPs also damage DNA by breaking its strands or point mutations resulting in expression of death receptor genes. 86 It results in leakage of intracellular Ca2+ ions followed by disruption of mitochondria and certain proteins to be encoded are malfunctioned.112,113 In plants, the oxidative stress disturbs different biochemical reactions and produces a decline in gas exchange and photosynthetic activity due to generation of extensive amount of ROS.114,115 It is examined that exposure of plant to very little amount of NPs consequence in the generation of excessive amount of ROS. 116
Plants have detoxification machinery to cope with this stress, but undue generation may take to programmed cell death or apoptosis (Figure 3). Nair and Chung, reported the NPs induced ROS generation in mung bean seedlings when CuO NPs were used at concentrations of 20, 50, 100, 200 and 500 mg/L. They observed a significant increase in the hydrogen peroxide content and lipid peroxidation in these plants. 88 Similarly, iron oxide nanoparticles-mediated phytotoxicity in Lemna minor was also stated by observing rise in ROS production and malondialdehyde content in a dose-dependent way. 117
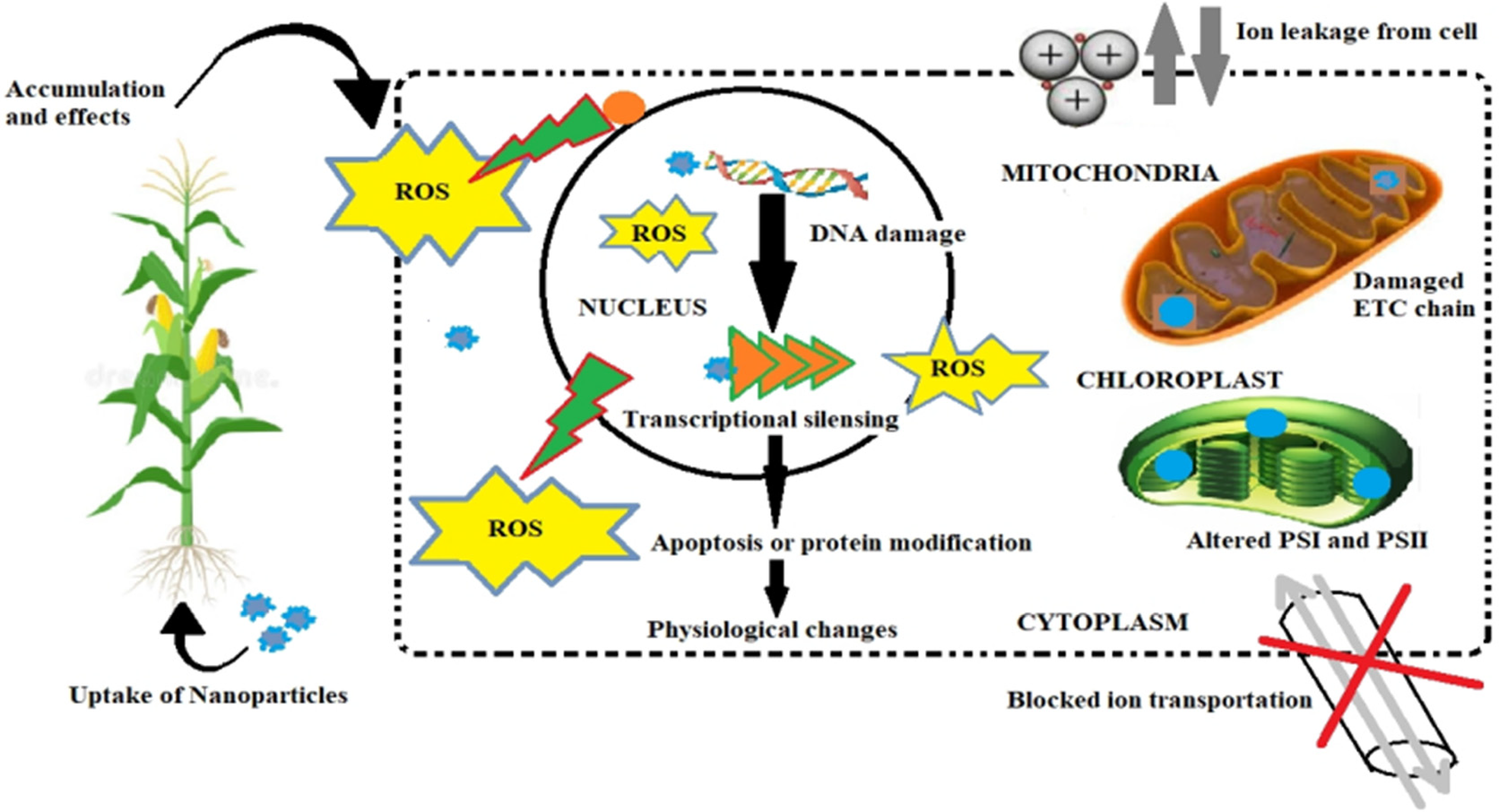
General mechanism of nanoparticles uptake in plants and ROS-mediated toxicity (Adopted from Khan et al. 118 ).
Methods to assess the phytotoxicity of NPs
To measure the phytotoxic effects of NPs, different parameters can be considered such as physicochemical attributes, timings of exposure, developmental phase of plant, as well as route of administration. There are different strategies for the exposure of plants to the NPs including NPs injection directly in plant tissues, 119 applying of NPs into plant's part, 120 administration of NPs directly into soil and plants irrigation with suspensions of NPs, 121 and infecting seeds or cellular pollens.95,122
For phytotoxicity analysis, different growth media can be used. Usual medium for growth is water, other options contain agar or soft gels that well represent soil. Finally, the soil can be used itself. 123 NPs apt to adsorb to the matrix of soil and aggregate in organic environment thus lower the bioavailability and mobility. Moreover, when alternative substrates are studied to check the interactions of NPs with plants, they do not get in account possible interface between NPs with soil and related water phase. 124 For instance, NPs in soil affects growth of soil microbes that may incidentally influence plant growth.
Plant's detoxification pathways
Plants possess antioxidant system/ROS scavenging pathways to survive different stresses. 125 The antioxidant system in plants is composed of antioxidant enzymes and secondary metabolites. The antioxidant enzymes include SOD (superoxide dismutase), APOX (ascorbate peroxidase), DHAR (dehydro-ascorbate reductase), GR (glutathione reductase), GPX (glutathione peroxidase), GST (glutathione-S-transferase) and CAT (catalase) that assist in reducing damage instigated by oxidative stress and in neutralizing the highly toxic ROS species. The non-enzymatic agents include thiols, phenolics and glutathione (GSH). SOD is a highly effective intracellular enzyme that performs a crucial function in antioxidant defense system by promptly converting O2•− to relatively harmless H2O2 that begins antioxidant pathways thus facilitating detoxification pathways in plants. CAT among all antioxidant enzymes was initially discovered and plays an imperative role in detoxification process. There are three steps for the detoxification of H2O2, during the first pathway, CAT enzyme catalyzes transfer of H2O2 into H2O and O2; whereas in the second pathway, APOX converts H2O2 to H2O via oxidation of ascorbate into mono dehydro-ascorbate (MDA) and dehydro-ascorbate (DHA). The third reaction is catalyzed by GPOX that produces oxidized GSH and GR. 126
Exposure of CuO NPs increased activities of CAT, APOX, GPX, and SOD in rice, A. thaliana and soyabean. 127 In a study by Da Costa and Sharma 127 a significant boost in SOD levels was observed in tomatoes after exposure to 0–1000 mg/L NiO NPs. Rico et al. examined that exposure to CuO NPs in Elodea densa plants altered the photosynthetic activity by increasing the CAT content, which ultimately managed to an expansion in SOD activity thus eventually breaking down the oxygen molecules. 100 In contrast to these observations, NPs exposure resulted in reduced activity of APX in Indian mustard 88 and CAT in alfalfa. 128
At the moment, it is hard to authenticate the impact of NPs (either helpful or harmful) on the antioxidants-based defense system in plants. Therefore, additional studies are required to represent a correlation between NPs toxicity and antioxidant enzymatic systems of plants.
Conclusion and future perspectives
Use of nanoparticles in agriculture has resulted in high yield and better quality of crops, however, there are several potential risks related to NPs upon their exposure to plants and environment that should be addressed. NPs can pose serious threats to the plants at both physiological (decrease in biomass, inhibition of root and shoot length, delayed growth) and cellular (damage to DNA and cell membrane, disruption to chlorophyll and decrease in pigment production) levels. Exposure to NPs and other stresses result in the generation of ROS as a stress response, nonetheless, there is a dire need to identify the molecular mechanism of plant's response to oxidative stress stimulated by NPs. To unveil the plant response at DNA levels, various molecular techniques (such as DNA microarrays, proteomics and metabolomics) must be employed. Furthermore, existing toxicity assessment methods of NPs exposure to plants are restricted to controlled laboratory conditions. Therefore, there is an imperative need to study the exposure of NPs and their effects in the field conditions for more holistic and realistic approach on the yield and growth of plants. The use of nanotechnology in agricultural field holds promise for various applications, but it also comes with certain limitations and challenges like potential toxicity, impact on environment and unknown ecological interactions. Limitations of the nanoparticles uses also include their bioavailability, sometime lack of standardization, and public acceptance.
Footnotes
Author's contributions
All author(s) contributed to the study's conception and design. SM and SH conceptualized and supervised the work. AA, MHS and BA prepared the outline. RA, MA, MF and IR contributed to the drafting of the manuscript. All authors were involved in the writing, reviewing and editing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Dr. Saima Muzammil is an Associate Professor in Institute of Microbiology. Her area of research is towards use of nanoparticles in the field of agriculture.
Dr. Asma Ashraf is a professor and working on the use of plant extract for the antimicrobial activities.
Dr. Muhammad Hussnain Siddique is Assistant professor in Department of Bioinformatics and Biotechnolgy. His research area is the industrial nanotechnology.
Dr. Bilal Aslam is an associate professor and working on one health. He is working on antibiotic resistance.
Dr. Ijaz Rasul is Assistant professor in Department of Bioinformatics and Biotechnolgy. His research area is the plant and industrial Biotechnology.
Rasti Abbas is PhD student in Institute of Microbiology and working on metallic nanoparticles uses in different fields.
Dr. Muhammad Afzal is Assistant professor in Department of Bioinformatics and Biotechnolgy. His area of research is plant biotechnology.
Dr. Muhammad Faisal is Assistant Professor in MNS-University of Agriculture, Multan. He is working on plant genomics.
Dr. Sumreen Hayat holds a PhD in plants biology. Her area of research is nanobiology and industrial microbiology.
