Abstract
Experimenter familiarization with laboratory rodents through handling prior to experimentation is an important practice in neurobehavioral research and is implicated in stress, study variability, and replicability. Unfortunately, different handling protocols have not been thoroughly examined. Determining optimal experimenter familiarization protocols is expected to reduce animal stress and thus improve welfare and data consistency. The impact of different handling protocols was determined through behavioral assessments (i.e. elevated plus maze, light/dark box, open field) as well as via analysis of fecal boli counts, ultrasonic vocalizations, and blood corticosterone. Male and female Sprague Dawley rats were distributed among three groups: never handled, picked-up, and handled for 5 min once daily over five days. Handled and picked-up rats spent more time in open arms and less time in closed arms of the elevated plus maze and more time in the center and less time at the perimeter of the open field compared to rats that were never handled, indicating that handled and picked-up rats were less anxious than those that were never handled. Male rats consistently defecated more frequently throughout the handling process and throughout behavioral testing, whereas females showed greater concentrations of blood corticosterone. Female rats were found to emit more 50-kHz calls and fewer 22-kHz calls compared to males. The results observed suggest that picking animals up may suffice as a handling method compared to time-intensive handling procedures, and that there are significant sex differences in response to handling.
Introduction
Rodent research is a necessity: science has not yet reached the point where animals can be substituted with equally informative alternatives. Thus, there remains a responsibility imparted on researchers to provide ethical care for laboratory animals. Although there has been a peak in the interest of animal welfare and its improvement in recent years, issues remain regarding experimental reproducibility,1–4 which is by far the greatest animal welfare concern.
Analyses suggest that genetics are responsible for a minor portion of the variation observed in behavior, suggesting that additional parameters are accountable (e.g. housing conditions, 5 experimenter characteristics, 6 and others). 7 Although some contributors to the lack of replicability occur after (inappropriate statistical analysis, reporting bias) or before (sample size calculation) the experiment, there are several aspects of the experiment itself that are likely contributors, including the relationship between animal and experimenter, especially during behavioral testing. The relationship is commonly initiated with a series of familiarizations. Rats exhibit indicators of stress and fear in response to the presence of and handling by a novel experimenter. 8 Initial handling sessions involve frequent urination, defecation, 9 and escape/freezing behavior until the animal habituates and the aforementioned behaviors subside. 10 Although there is consensus that handling modulates the affective state of laboratory animals,10–12 significant periods of time spent handling are often unreasonable to implement. Protocols demonstrating differences in affective state of animals following shorter handling periods will be important in guiding future study design.
Common paradigms in rodent research used to assess affective state, particularly anxiety, are the open field, elevated plus maze (EPM), and light/dark box—tasks that rely on the animal’s natural exploratory behavior. The open field is the most commonly used test and entails observing the rat’s spontaneous activity within an open maze.13,14 Anxious animals tend to spend more time at the perimeter of the maze, avoiding open, highly vulnerable areas. As the animal becomes more familiar with the maze, time spent at the perimeter will decrease, and time in the central area will increase. 14 The EPM, like the open field, is a test of spontaneous activity; however, there are two “open” (high risk) and two “closed” (low risk) arms in a plus configuration, elevated off the floor. 15 Rodents have a natural tendency to avoid open spaces, particularly when elevated, and are attracted to enclosed spaces. 16 The light/dark box is a rectangular-shaped apparatus consisting of two linked chambers (one black in color and one illuminated by light). Again, animals are presented with the conflict of remaining in the “low-risk” dark chamber and exploration of the “high-risk” illuminated chamber.17–19
It is possible that ultrasonic vocalizations (USVs) may be useful in monitoring handling habituation.15,20 USVs are characterized into two main groups in rats: 50-kHz USVs (positive) and 22-kHz USVs (negative).15,20–26 Physiological indicators of stress/anxiety such as increased corticosterone levels 27 or increases in defecation 8 of an animal may assist in understanding the impacts of various handling regimens.
The influence of experimenter familiarity, among others, is likely implicated in experimental variability and lack of replicability. A greater understanding of the experimenter–animal relationship and establishment of handling guidelines is a significant step toward rectifying issues of replicability and maximizing animal contribution and welfare. To optimize current handling guidelines, we examined the behavior of rats subjected to different handling protocols in the open field, EPM, and light/dark box as well as assessed differences in fecal boli, USVs, and blood corticosterone.
Methods
Animal husbandry
Male (n = 36) and female (n = 36) Sprague Dawley rats (CD) were acquired from Charles River Laboratories and randomly divided into three groups (12 animals per group). Sprague Dawley rats are a general-use model. Animals were 12 weeks old at the beginning of handling—the most frequently used age group in biomedical research. 28 The groups included rats that were never handled, rats that were picked up and placed back into their cage once a day, and rats that were handled for one 5-min interval once a day for five consecutive days prior to testing.
Upon arrival, rats were allowed a one-week acclimation prior to manipulation. Rats were housed within standard cages (266.7 mm × 482.6 mm × 203.2 mm; Ancare Corp., Bellmore, NY) in pairs. Cages were bedded with Hardwood Beta Chips (North Eastern Products, Warrensburg, NY) and changed twice a week. All animals were maintained on an ad libitum diet of Laboratory Rodent Diet 5001 (LabDiet, St. Louis, MO) and water. The colony room was maintained between 19°C and 22°C, humidity 30–52%, and lit using overhead fluorescent lighting at 300–400 lux. The ambient room sound was 70–80 Db. A 12-hour reverse light/dark cycle was utilized, with the lights going off at 0600 hours and coming on at 1800 hours. When it was necessary to enter the room during the dark phase, a headlamp emitting red light was used. All procedures performed during this study were conducted in accordance with the guidelines of the Canadian Council on Animal Care and approved by the University of Prince Edward Island Animal Care Committee protocol #20-024.
Handling protocols
Rats were handled by the same investigator throughout the duration of the study during the dark phase of the light cycle. During handling, the rat was retrieved from the cage with one hand lightly gripping the tail and the other hand, palm up, supporting the rat’s ventral surface. The rat rested in the arms of the handler, with the handler’s arms cradling the rat. There was no caressing of the rat on either the ventral or dorsal surface. A light grip was maintained on the tail of the animal in case the animal attempted to escape. Following handling, the animal was returned to the cage using the reverse process. All animal handling took place in the testing suite to habituate the animals to the protocol used during behavioral testing. The number of fecal boli produced by each animal during handling was recorded each day as an indicator of stress. Additionally, USVs were recorded during handling using a single microphone placed approximately 12 inches above the arms of the investigator holding the rat. USVs were recorded using Avisoft UltraSoundGate (Glienicke/Nordbahn, Germany) and analyzed using Avisoft SASLab Pro v5.2.15 (Avisoft Bioacoustics). The primary measures of interest for USV recordings were numbers of 50- and 22-kHz calls.
Animals in the picked-up group were picked up and put down using the same technique as the handled animals in the testing suite and then returned to the housing room after a period of 10 minutes. Animals that were not handled were habituated to the behavioral testing process by transporting each cage to the testing suite for a period of 10 minutes then returning the cage to the housing room. A period of 10 minutes was used because it represented the duration of the longest behavioral testing paradigm used in the study. Following handling, the experimenter was blind to conditions.
Behavioral testing
Following five consecutive days of handling, rats were assessed in the EPM. Each animal was placed in the open arm of the maze near the center platform, oriented toward the center and allowed to explore the apparatus freely for five minutes. The dimensions of the EPM were: length of arm 111.8 cm, width of arm 10.2 cm, height 30.5 cm, elevation off ground 83.8 cm, center area 10.2 cm × 10.2 cm.
The day immediately after testing in the EPM, each animal was assessed in the light/dark box. Each animal was placed in the light side of the light/dark box and allowed to explore freely for five minutes. The dimensions of both the light and dark chamber were 40.6 cm wide × 40.6 cm long × 39.4 cm tall; the width and height of the opening between the two chambers were 9.5 cm and 8.3 cm, respectively.
The next day the animals were assessed in the open field test. In the open field, each animal was placed in the arena and monitored for 10 minutes. The open field consisted of a circular area with an inner diameter of 91.4 cm and side height of 50.8 cm. All testing was performed between 0900 and 1400 hours. Each testing apparatus as well as holding cages were sanitized with accelerated hydrogen peroxide wipes between trials.
Blood corticosterone
Immediately following testing in the open field, animals were anesthetized using isoflurane and decapitated; trunk blood was collected in K2EDTA tubes (Fisher Scientific, Ottawa, Canada). Samples were centrifuged at 4500 × g, and plasma was collected and stored at –80°C until further processing. Corticosterone was measured in each sample using a commercially available corticosterone ELISA kit (Arbor Assays, DetectX #K014; Cedarlane labs, Ontario, Canada) 29 in duplicate according to the manufacturer’s instructions. Whereas all reagents and materials were brought to room temperature prior to use, the samples were thawed immediately prior to use and kept on ice. The dilution factor for each sample was 200-fold. The coefficient of variance among the duplicates was <10%. The plates were read on a Synergy HT plate reader (Biotek, Winooski, VT) and its Gen5 software, with built-in four-parameter logistic regression used for plotting the standard curve and data extrapolation.
Statistical analysis
Three-way analyses of variance (ANOVAs) were performed for all behavioral tests assessing differences in sex, treatment, and time interval where time spent within each area of the testing apparatus was binned into one-minute intervals to determine whether the time spent within each area changed throughout the duration of the trial. Two-way ANOVAs (sex × time or sex × treatment) were performed for fecal boli counts, blood corticosterone, and USV analysis. Post hoc analyses consisted of paired, two-tailed t-tests with a significance level of 0.05. Statistical analysis for the three-way ANOVAs were performed using Jamovi (Jamovi Project, Sydney, Australia), whereas all other statistical analysis and associated graphs were conducted using GraphPad Prism v6.01 for Windows (GraphPad Software, La Jolla, CA).
Results
EPM
There was a significant sex (F(1, 70) = 7.92, p = 0.005), treatment (F(2, 69) = 5.28, p = 0.006), and time (F(4, 355) = 22.53, p < 0.001) effect for distance traveled in the EPM. Post hoc analysis revealed that picked-up rats traveled a greater distance than handled rats (t(329) = 3.25, p = 0.004). For time spent in the open arms of the maze, there was a significant sex (F(1, 70) = 12.32, p < 0.001) and treatment (F(2, 69) = 8.67, p < 0.001) effect but no time (F(4, 355) = 1.99, p = 0.096) effect (Figure 1(a)); for time spent in the closed arms, there was again a significant sex (F(1, 70) = 7.54, p = 0.006) and treatment (F(2, 69) = 8.32, p < 0.001) effect but no time (F(4, 355) = 1.03, p = 0.394) effect (Figure 1(b)). Further analysis revealed a significant difference between some groups: handled animals, and picked-up animals, spent more time in the open arms (t(329) = 4.12, p < 0.001 and t(329) = 2.55, p = 0.030, respectively) and less time in the closed arms (t(329) = 3.95, p < 0.001 and t(329) = 2.84, p = 0.013, respectively) compared to those that were never handled. Additionally, females spent more time in the open arms compared to males (t(329) = 3.51, p < 0.001).
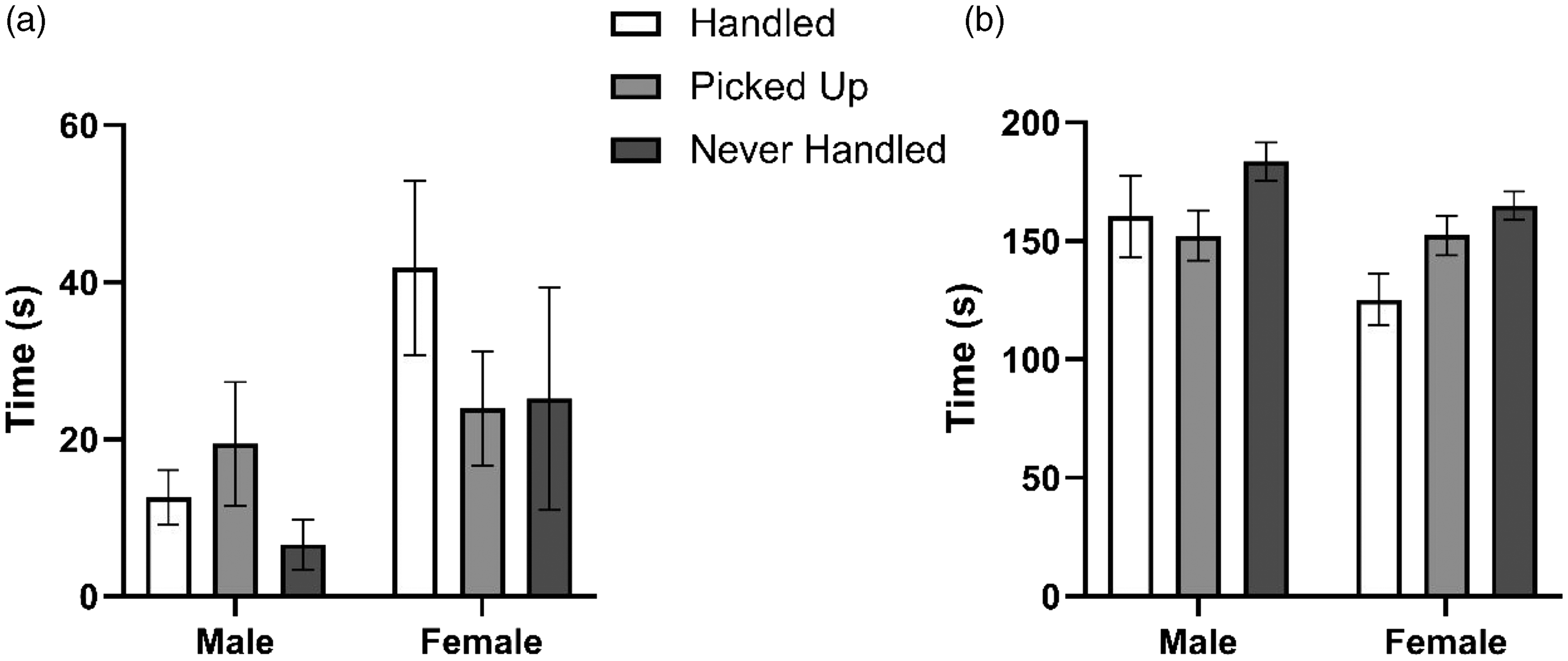
Total time spent in the open arms of the maze (a) versus total time spent in the closed arms of the maze (b) over a five-minute interval. Data shown as mean ± SEM (n = 12).
Light/dark box test
There was a significant sex (F(1, 70) = 47.00, p < 0.001), treatment (F(2, 69) = 5.65, p = 0.004), and time (F (4, 355) = 21.00, p < 0.001) effect for distance traveled in the light/dark box test. Post hoc analysis revealed handled rats traveled a greater distance than never-handled rats (t(329) = 3.33, p = 0.003). When assessing time spent in the light side of the box, there was a significant sex (F(1, 70) = 30.96, p < 0.001) and time (F(4, 355) = 2.82, p = 0.025) effect but no treatment (F(2, 69) = 2.38, p = 0.094) effect (Figure 2(a)), and for time in the dark side of the box, there was a significant sex (F(1, 70) = 26.38, p < 0.001) effect but no treatment (F(2, 69) = 1.21, p = 0.300) or time (F(4, 355) = 2.26, p = 0.063) effect (Figure 2(b)). Post hoc analysis revealed that females spent more time in the light side of the box (t(329) = 5.56, p < 0.001) and less time in the dark side (t(329) = 5.14, p < 0.001) compared to males.
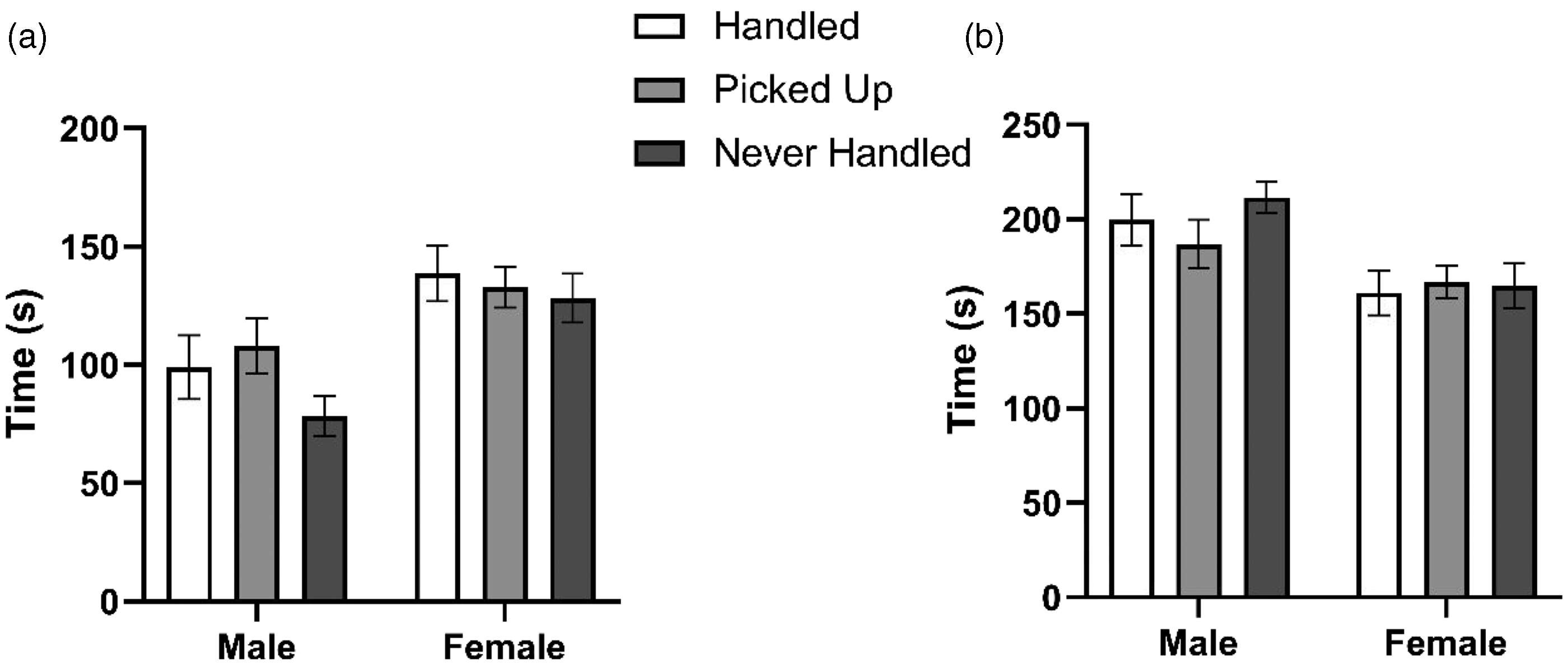
Total time spent in the light side of the maze (a) versus total time spent in the dark side of the maze (b) over a five-minute interval. Data shown as mean ± SEM (n = 12).
Open field test
There was a significant treatment (F(2, 69) = 10.51, p < 0.001) and time (F(4, 355) = 44.23, p < 0.001) effect but no significant sex (F(1, 70) = 2.91, p = 0.088) effect for distance traveled in the open field test. Post hoc analysis revealed handled rats (t(650) = 4.21, p < 0.001) and picked-up rats (t(650) = 3.66, p < 0.001) traveled a greater distance compared to never-handled rats. There was a significant sex (F(1, 70) = 4.06, p = 0.044), treatment (F(2, 69) = 7.34, p < 0.001), and time (F(4, 355) = 2.56, p = 0.007) effect (Figure 3(a)) for time spent in the center of the open field and a significant sex (F(1, 70) = 4.06, p = 0.044), treatment (F(2, 69) = 7.34, p < 0.001), and time (F(4, 355) = 2.56, p = 0.007) effect (Figure 3(b)) for time spent at the perimeter. Post hoc analysis revealed that males spent more time in the center of the maze (t(650) = 2.02, p = 0.044) and less time at the perimeter (t(650) = 2.02, p = 0.044) compared to females. Handled animals, and picked-up animals spent more time in the center of the maze (t(650) = 3.16, p = 0.005 and t(650) = 3.45, p = 0.002, respectively) and less time at the perimeter of the maze (t(650) = 3.16, p = 0.005 and t(650) = 3.45, p = 0.002, respectively) compared to those that were never handled.
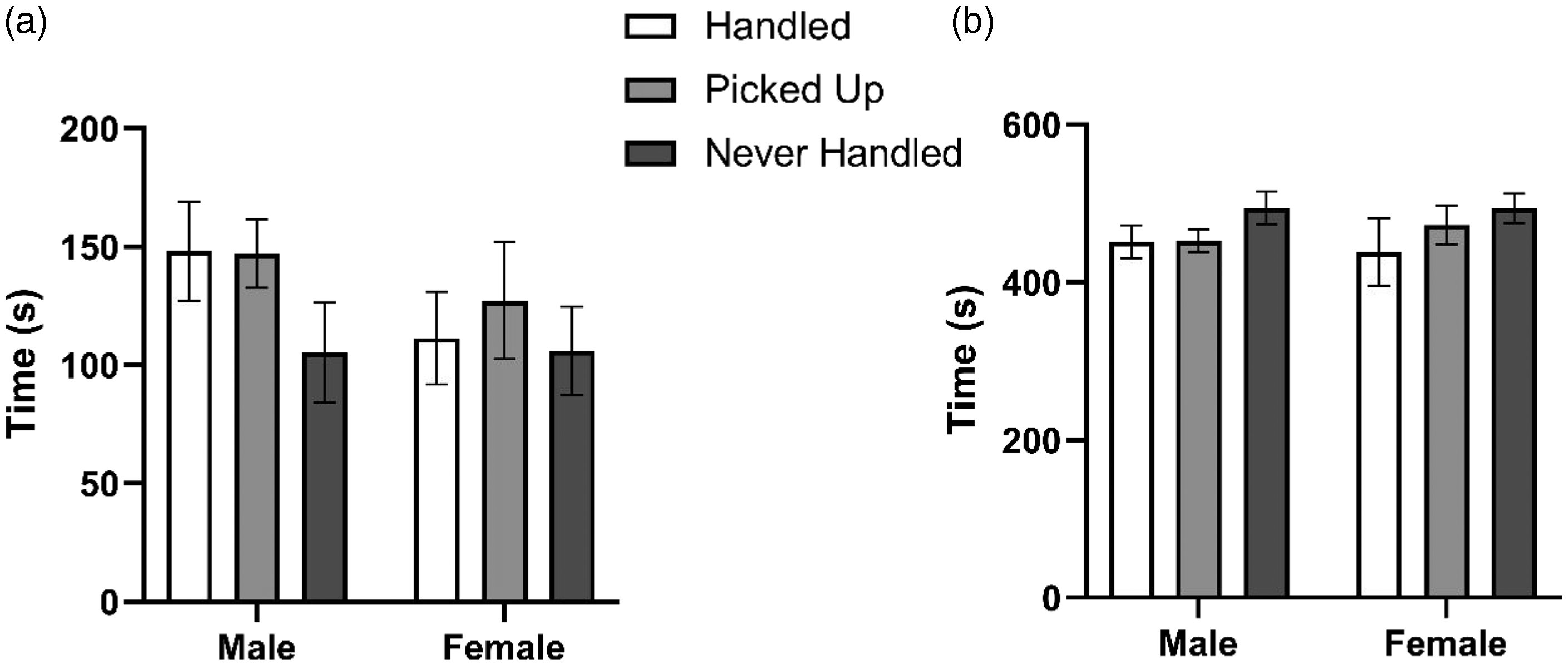
Total time spent in the center of the maze (a) versus total time spent at the perimeter of the maze (b) over a five-minute interval. Data shown as mean ± SEM (n = 12).
There was no significant difference in time spent in the center of the maze (t(650) = 0.25, p = 0.965) or at the perimeter of the maze (t(650) = 0.25, p = 0.965) between picked-up and handled animals.
Fecal boli
There was a significant sex effect (F(1, 22) = 12.75, p = 0.002) but no time effect (F(4, 88) = 2.41, p = 0.055) for the number of fecal boli produced during handling over a five-day period (Figure 4(a)) and a significant sex effect (EPM: F(1, 66) = 7.23, p = 0.009; light/dark box: F(1, 66) = 12.66, p < 0.001; open field test: F(1, 65) = 16.38, p < 0.001) but no treatment effect (EPM: F(2, 66) = 0.49, p = 0.614; light/dark box: F(2, 66) = 0.196, p = 0.823; open field test: F(2, 65) = 1.38, p = 0.258) for the number of fecal boli produced during the three behavioral tests (Figure 4(b), 4(c) and 4(d)).
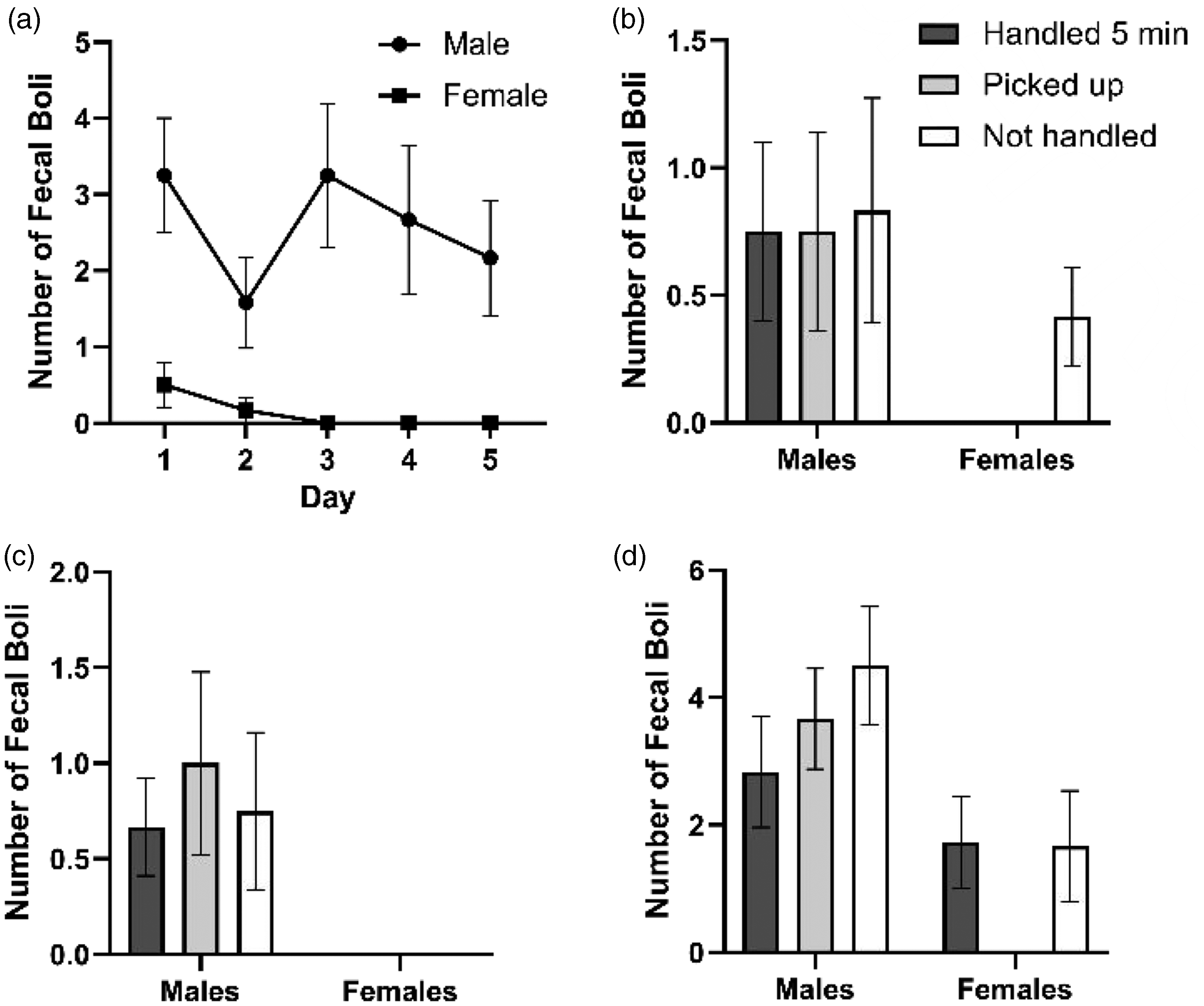
Number of fecal boli produced per animal during a five-minute handling period over a duration of five days (a), during the elevated plus maze (b), light/dark box test (c), and open field test (d). Data shown as mean ± SEM (n = 12).
Blood corticosterone
There was a significant sex effect (F(1, 66) = 33.62, p < 0.001) but no treatment effect (F(2, 66) = 1.09, p = 0.344) for the concentration of corticosterone (ng/mL; Figure 5), indicating female rats had increased concentrations of blood corticosterone compared to males.
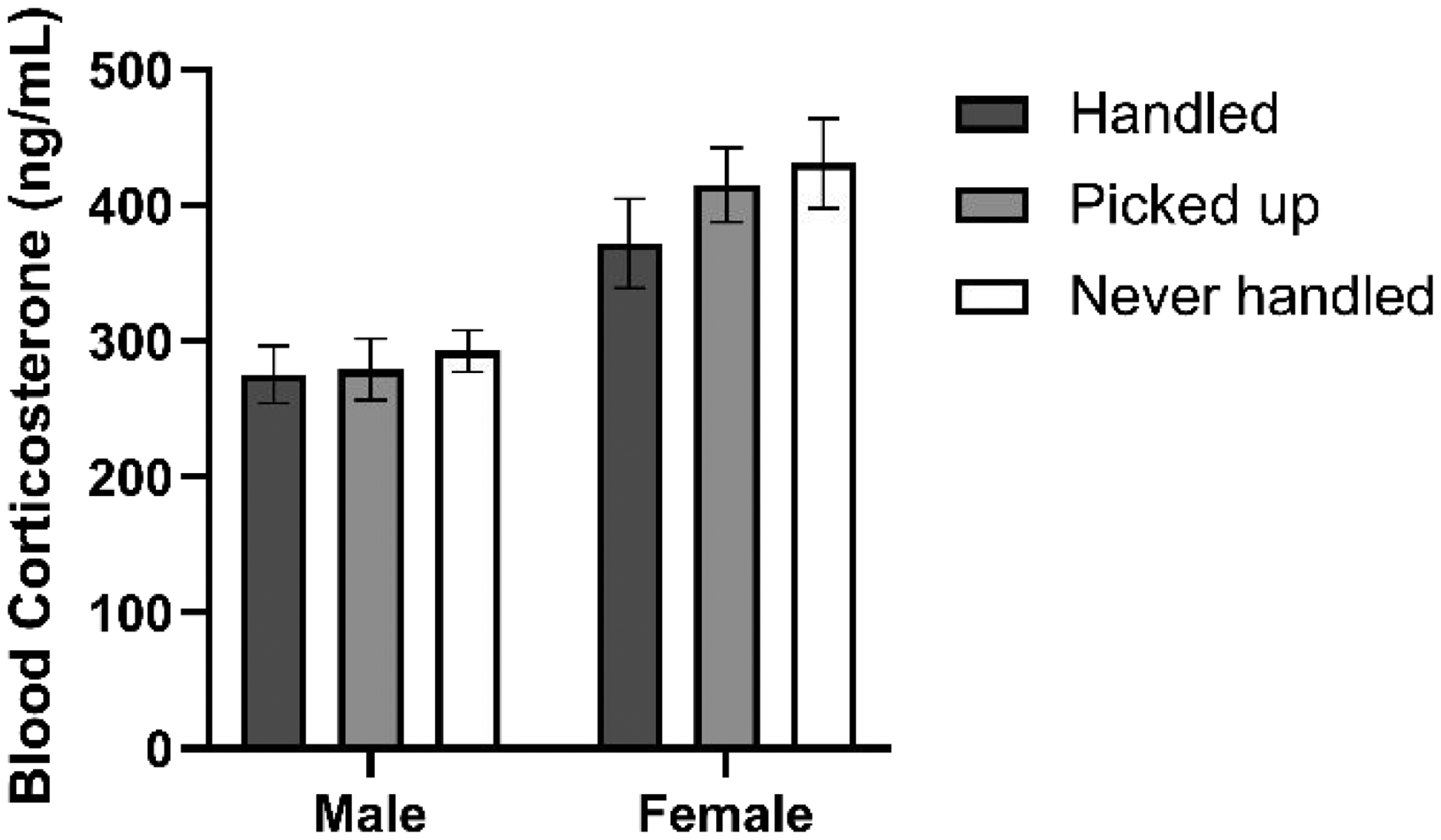
Concentration of blood corticosterone (ng/mL) for male and female rats subjected to varying handling protocols. Data shown as mean ± SEM (n = 12).
USVs during handling
There was a significant sex effect (F(1, 22) = 5.91, p = 0.024) but no time effect (F(4, 88) = 1.50, p = 0.209) for the number of 50-kHz calls per animal over five days, indicating that female rats emitted more 50-kHz calls than males (Figure 6(a)). There was neither a sex effect (F(1, 22) = 2.69, p = 0.115) nor a time effect (F(1, 30) = 1.70, p = 0.205) for 22-kHz calls. Further, it should be noted that only male rats emitted 22-kHz calls (Figure 6(b)).
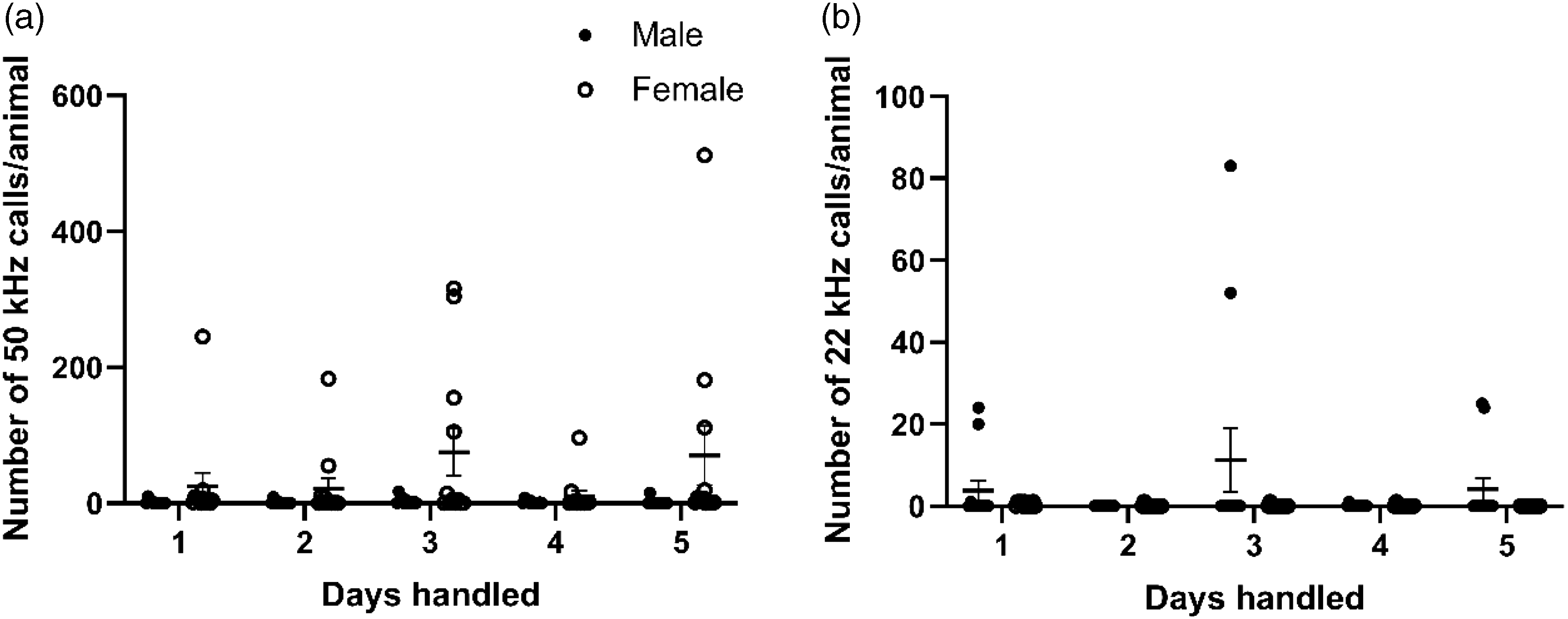
Number of 50-kHz calls per animal (a) and 22-kHz calls per animal (b) within a five-minute interval over five days of handling. Data shown as mean ± SEM (n = 12).
Discussion
Handled and picked-up rats spent more time in the open arms and less time in the closed arms of the EPM compared to never-handled animals. This anxiolytic effect was also found in the open field, with handled and picked-up animals spending more time in the center of the maze and less time at the perimeter compared to animals that were never handled. No group differences were observed in the light/dark box.
The EPM, light/dark box, and open field are the most common tests of anxiety in rodents, and the results of any of these are often used to indicate differences in anxiety between conditions. Some 30 have suggested, however, that it is likely these tests assess distinct aspects of anxiety. Commonly, the results between the three tests are not congruent, 31 leading to the implementation of paradigms that simultaneously incorporate all three tests into one through a physical linkage. 30 A unique profile arises when animals interact with each segment of the integrated maze, such as that observed when animals are tested in each apparatus independently, supporting the idea that each test uniquely assesses emotion, which could explain the observed effects in this study. Exactly what unique aspects of anxiety these tests assess is uncertain.
Regardless of the test utilized, there were significant group differences in distance traveled. Stimuli, such as light, which are considered aversive, reduce activity levels, 14 and rats that show high anxiety levels in the EPM typically have reduced activity in the open field and light/dark box test. 32 In the light/dark box test, where there were no effects for time spent in the light or dark side, handled rats traveled more than non-handled rats. Handled and picked-up rats also traveled a greater distance than non-handled rats in the open field; however, picked-up rats traveled a greater distance than handled rats in the EPM. Thus, based on the distance traveled results, handling rats or picking them up would equally suffice as a measure of habituation.
Although it is possible that each of these tests assesses a unique aspect of anxiety, it does not exclude testing order effects. In the current study, the order of testing was performed from what was considered least to most stressful. The EPM was short and required no exposure to illumination, as in the light/dark box test. The open field was double the duration of the preceding tests. As test-ordering effects have previously been observed by others, 33 precautions were taken to minimize affective state perturbations between tests, such as minimizing time in the colony room, minimizing exposure to red light, not performing cage cleaning throughout the testing series, and limiting access to the housing room to only those experimenters involved in the study.
During assessment of USVs during handling, a small number of vocalizations were produced, and most calls were made by the same rats over multiple days. Previous studies have had success in eliciting USVs from light touch and handling [35]. Interestingly, rats in the aforementioned study only vocalized following the cessation of the stimulus; it may therefore be advantageous to record USVs after returning the rats to their home cage, or prior to handling, as rats also vocalize in anticipation of positive 34 and negative 35 events. Rats vocalize based on the proximity of the threat, usually emitting only when in a place of safety,36,37 possibly explaining the low volume of calls. It is also important to recognize that there may be strain differences in the threshold of vocalization. As mentioned in Brudzynski, 38 some of the rats (Wistar) vocalized in the sonic range when being transferred from the home cage to the testing arena. In our laboratory’s experience, Sprague Dawley (CD) sonic vocalizations have only occurred under distressing circumstances, such as being picked up after long periods of time in an open field or following arduous tasks where the animal is picked up frequently, such as in the attentional set-shifting task. Differences in the rate of calling in response to aversive scenarios has been reported in the literature, 39 thereby limiting the results of USV in response to handling in Sprague Dawley rats. Only handled male rats produced 22-kHz calls during handling, with 33% of male rats vocalizing on at least one of the five handling days, whereas production of vocalizations was more evenly distributed for 50-kHz calls, with 92% of females producing ultrasounds on at least one of the five days of handling compared to 83% for males. In response to a threat, females typically tend to produce more 22-kHz calls compared to males, 36 suggesting females may not find handling as aversive as males do.
Female rodents produced significantly fewer fecal boli throughout the handling period as well as throughout behavioral testing (Figure 4) compared to males. Beyond the first day of handling, there was minimal defecation from females, whereas males continued to defecate regularly. The utility of using female defecation volume as a measure of stress is minimal due to their continual low volume. Additional handling days may have been informative regarding the utility of fecal boli in tracking the habituation process in males, as in the current analysis, there were no statistical indications of change in fecal boli counts over time. Fecal boli comparisons between sexes are further complicated by differences in weight.
There were no treatment differences in corticosterone between the groups. Female rats overall, however, had higher corticosterone levels. Yet, they tended to move more in the open field—an indication of reduced stress. Increases in corticosterone can occur due to an increase in arousal, which can be mistaken for an increase in stress levels. 40 Further, females tend to have a higher baseline corticosterone compared to males. 41 Future studies may explore alternative corticosterone acquisition methods, as anesthesia methods have also been reported to alter corticosterone levels, 42 although this has not been observed in other studies. 43 Future studies should also look at blood corticosterone directly after handling, prior to any experimentation. In this study, the priority was to observe the rats in the behavioral tests prior to any additional manipulation. Further, the blood from these rats was acquired immediately after the last behavioral test, and therefore the corticosterone better represents the rat’s response to behavioral testing following specific handling protocols. Additionally, the rats were sacrificed over a period of several hours, which is enough time for circadian rhythm to influence outcomes considerably. 44
Differences in EPM behavior have previously been observed following handling protocols. Costa et al. 10 found that rats handled for five minutes per day, five times per week, for six weeks exhibited behaviors indicative of reduced stress compared to a non-handled control. Although the handling regimens were dramatically different, including the definition of handling, the study does demonstrate that handling can affect behavior in the EPM; however, handling periods were beyond what is reasonable/feasible and limited to a single sex. The goal of the current study was to define a series of handling protocols that could be reasonably and universally implemented into experimental designs, with the goal of reducing inter-laboratory variability. Limitations in manpower and finances dictate that the familiarization procedures do not occur over several weeks. There is already a reasonable expectation that animals be allowed a habituation period following transportation. 45 The addition of weeks of handling can substantively increase costs to an experiment, making them unpractical to implement.
Comparison of these two studies also brings about the topic of the definition of handling, which was different in both studies, suggesting that observing the influence of different types of handling in rats, in addition to studies interested in the length of necessary handling, will be of significance. Different handling methods in mice have been shown to be of significance. 30
Overall, this study demonstrated that differences in rat behavior in tests of affect can be observed following only five days of handling or picking up, suggesting that picking up rats in a situation such as placing an animal in a testing chamber may be a sufficient means of habituating rats to experimenters. Reducing handling time may reduce stress levels in rats as well as reduce personnel time commitments in future experimental protocols. Determining whether the observed differences persist throughout additional tests of affect and whether the effect of different handling procedures have the potential to affect the outcome of an experimental intervention should be further investigated to improve reproducibility across laboratories.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by the Sir James Dunn Animal Welfare Centre and the Atlantic Veterinary College at the University of Prince Edward Island.
