Abstract
Rabbits are commonly used for sciatic nerve injuries larger than 1.5 cm. This report provides insight into risks and benefits associated with using rabbit models in sciatic nerve injury models and proposes interventions that researchers can use to prevent experimental complications. Fifty-six rabbits from a sciatic nerve injury study that involved a 40 mm sciatic nerve injury were analyzed to examine postoperative complication rates. Autophagy of the phalanges and plantar pressure ulcer development were the most common and serious complications faced. These complications led to 23.2% (n = 13) of rabbits not being used for data in the original experiment due to euthanasia outside of intended postoperative time points. This increased the cost needed to complete the experiment by $25,038.44. It is our recommendation that alternative models be used instead of rabbits for sciatic nerve injuries. If rabbits must be used, a treatment protocol for preventing autophagy and pressure ulcers is outlined below.
Introduction
Rabbits are the third most commonly used animal for sciatic nerve injury studies. 1 Rabbits are generally used for experiments that require prostheses or for sciatic nerve injuries larger than 1.5 cm. 1 Their ease of handling, size, docility, short life-span, and sensitivity to implants makes them a superb model.2–4 This sensitivity presents a double-edged sword that must be considered when using a rabbit model for sciatic nerve injuries. The benefit of gaining insight into possible immunologic responses to surgical implants may be offset by experiment-related complications that could increase the cost of experimentation and worsen pain for the rabbits. The objective of this report is to give insight into risks and benefits associated with using rabbit models in sciatic nerve injury models and propose interventions that researchers can use to prevent experimental complications.
Research with animal models has vastly extended our knowledge of treating diseases, understanding biochemical mechanisms, and optimizing biomedical technologies. Both in vitro and in vivo testing is necessary to evaluate the safety and reliability of these new treatments. Implant technologies have benefited from animal models because of their advantages with assessing implant toxicity, evaluating biocompatibility, and improving implant designs. 3 According to the most recent United States Department of Agriculture (USDA) report, rabbits are the second most researched USDA species, with 133,634 rabbits used for research in 2018. 5
Rabbit sensitivity can make maintaining the rabbits very strenuous and costly. The cages must be made of a nontoxic material that would not harm the rabbit if chewed or licked, designed so that the rabbit is unable to hurt itself with sharp edges, and is able to be decontaminated easily and repeatedly. 6 Rabbits do not generally show clear behaviors indicating pain, making distress from pain difficult to identify. 4 Distress from pain can cause the rabbits to die within 36 hours after surgery. 2 Self-mutilation of the toes is a major concern that can occur days or weeks after surgery. 7 This is especially worrisome in studies affecting the sciatic nerve, since the rabbit may focus attention on the limb with nerve damage.7,8 Development of pressure ulcers can affect rabbits without proper housing arrangements and damage the rabbit’s limbs.9,10 These pressure ulcers can lead to difficulty with ambulation and ulcerative pododermatitis on the injured limb. If severe enough, these complications can lead to early endpoint criteria for the rabbit before the experiment’s completion. 11
The following is a report of procedures done during our original experiment testing 40 mm sciatic nerve gap repairs in rabbits with diffusion tensor imaging published elsewhere. 12 Here, we document a series of unexpected complications as well as interventions to ensure experimental progression with humane treatment of the rabbits. A cheap and effective postoperative care plan was implemented to prevent self-mutilation and deter development of pressure ulcers in rabbits with injured sciatic nerves.
It is our hope that this description of the complications and our strategies to address them will help reduce pain for rabbits in future experiments and persuade others to replace rabbits with a different model for sciatic nerve injury.
Methods
Fifty-six New Zealand white rabbits of both sexes were purchased through Vanderbilt University Medical Center to a USDA-certified vendor. Our corresponding Institutional Animal Care and Use Committee (IACUC) approved all procedures in accordance to the Guide for Care and Use of Laboratory Animals to reduce pain and suffering. The mean weight of the rabbits before surgery was 3.2 ± 0.19 kg.
Operative technique
Rabbits were given a mixture of ketamine (40 mg/kg) and xylazine (9 mg/kg) intramuscularly once on the non-experimental leg for sedation. A volume of 1.9–2.7 mL was installed according to the weight of each animal. Following intubation of the trachea, anesthesia was maintained with isoflurane 2% in oxygen with the flow rate set at 3 mL/minute. All surgery was performed by the same surgeon (S.A.) using standard surgical techniques. A longitudinal 5 cm incision was performed over the lateral surface of the left thigh. Dissection was carried down by planes and using a split-muscle technique, exposing the left sciatic nerve at its extension. At this point, we harvested 40 mm of sciatic nerve graft using sharp scissors. For the purposes of our original study, rabbits were grouped into the following three categories: cut without repair, autologous nerve repair in which the nerve section was reversed 180° and sewn immediately to the remaining stumps using 9-0 Ethilon sutures (Ethicon, Somerville, NJ), and repair with a sterile nerve conduit that was sewn into the defect also using 9-0 Ethilon sutures (Ethicon). All incisions were approximated layer by layer using a 3-0 Vicryl interrupted suture pattern (Ethicon) for the muscular plane and a 5-0 Monocryl running suture pattern (Ethicon) for skin closure.
Postoperative care
Rabbits were monitored by study investigators after surgery until full recovery and until they were ready to be transferred to our animal facility in accordance with IACUC protocol. All therapeutic interventions (injections) were performed in the contralateral hind limb. Our analgesia protocol consisted of the rabbits receiving buprenorphine hydrochloride at a dose of 0.02–0.05 mg/kg subcutaneously or intramuscularly every eight hours for the first 24 hours. Additionally, they were treated with meloxicam subcutaneously at a dose of 0.1–0.3 mg/kg/24 hours for 72 hours as needed, based on a rabbit grimace scale. 13
Allentown cage rack systems for rabbits were used for single housing. Under the racks, a non-contact Techboard was used to manage excrement and urine. The subjects were switched to clean cages every two weeks. All rabbits were fed ad libitum by automatic feeders. A high-fiber diet and water were replenished every day. The rabbit room in the animal facility was set up to have automatic 12-hour light/dark cycles.
Rabbits were monitored daily by animal care staff and every week by the investigators. The animals were inspected thoroughly for complications, and their weight was recorded weekly. When the rabbits fulfilled their time periods (3, 6 or 12 weeks), they were euthanized using sodium pentobarbital injected intravenously at a dose of 125 mg/kg. Samples of the nerve were collected for either immunohistochemistry (IHC) or magnetic resonance imaging (MRI) for the purposes of our original study.
Exclusion criteria
Our complication analysis includes only those related to sciatic nerve injury postoperatively. Rabbits were euthanized before fulfilling their assigned time period if they experienced any of the following: self-mutilation of any limb with underlying osseous display, wound infection, full-thickness ulcer on the experimental limb, failure to thrive by refusing water or diet, >20% loss of preoperative weight, or immobility despite adequate pain control.
Statistical analysis
Student’s t-tests were performed between groups (pre and post intervention) using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp, Armonk, NY). A p-value of <0.05 was considered significant.
Results
Complication classification
Autophagia was the most frequent postoperative complication encountered (Figure 1). A total of 20 rabbits suffered some sort of self-mutilation. The majority of the affected rabbits focused on chewing the phalanges of the injured limb. Other areas involved included the surgical site, the dorsum, and the web-spaces of the experimental foot.
Distribution of complications based on the total population of subjects enrolled in our project. Elizabethan collar (left) and donut collar (right) used in this experiment.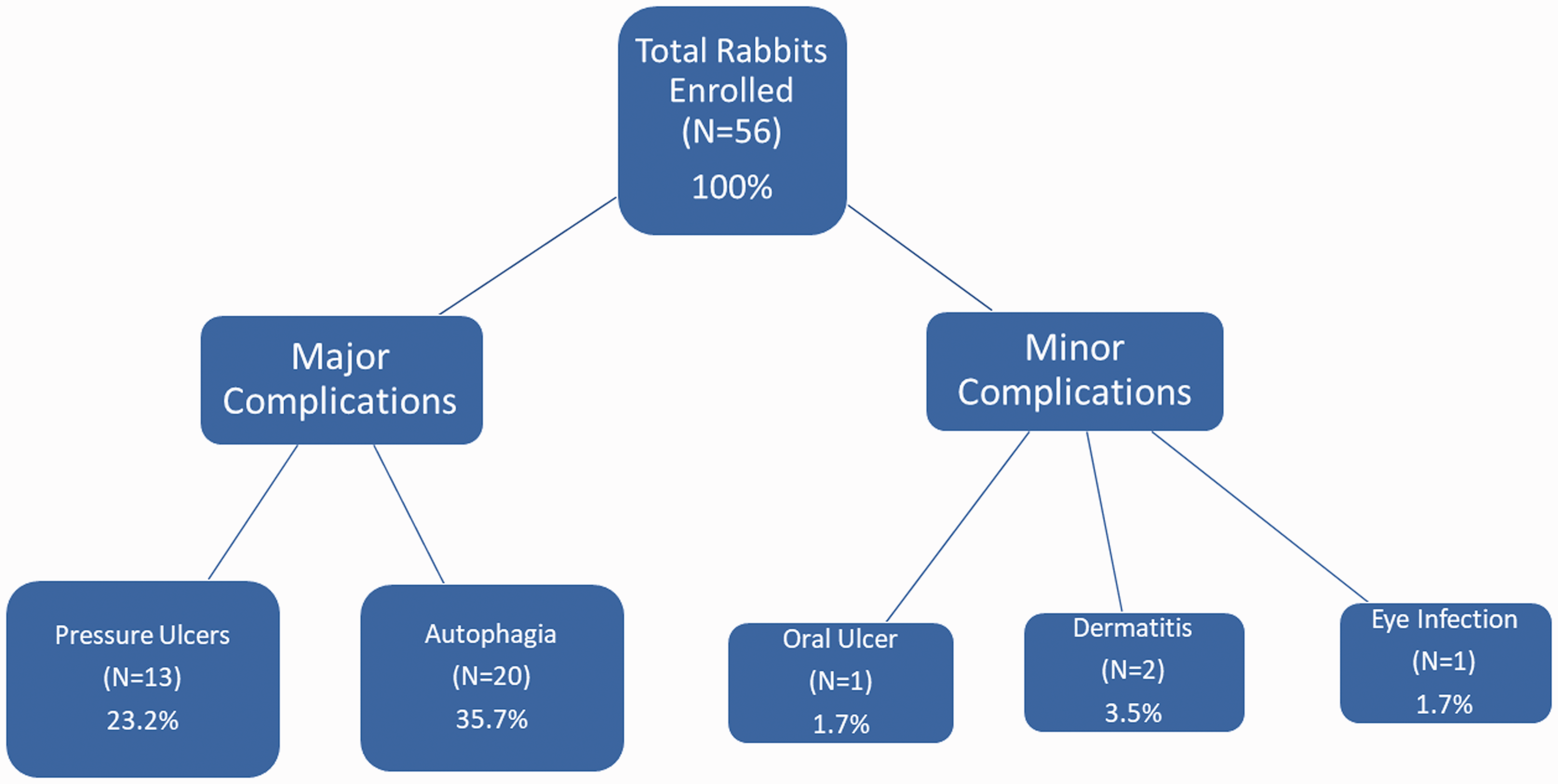

Pressure ulcers were the second most frequent postoperative complication (Figure 1). A total of 16 rabbits suffered from this morbidity. Most of the ulcers were located on the plantar aspect of the experimental foot, close to the heel. These usually started as an erythematous plaque devoid of fur near the heel, which evolved into an eschar. These fully progressed into deep defects where the base reached the calcaneus at a variable conversion rate between the subjects.
Rabbits that needed to be euthanized one week or more prior to their intended postoperative endpoint were considered outliers (about 23.2% of the total number of rabbits). Data collected from the outliers was not used for the original study due to the variation in postsurgical time. The data of these outliers were included for the purposes of this report.
Intervention plan
A plastic Elizabethan collar with soft edges was used to deter autophagia. These collars were adjustable and ranged from 6″ to 9.5″ in diameter (Figure 2). One person safely held the rabbit while the other gently placed the collar onto the rabbit. The incidence of hind-limb autophagia ranged from 12.29% before the intervention to 2.52% following the intervention (p = 0.0093). Euthanasia related to autophagia decreased from 7.50% to 1.04% (p = 0.00164).
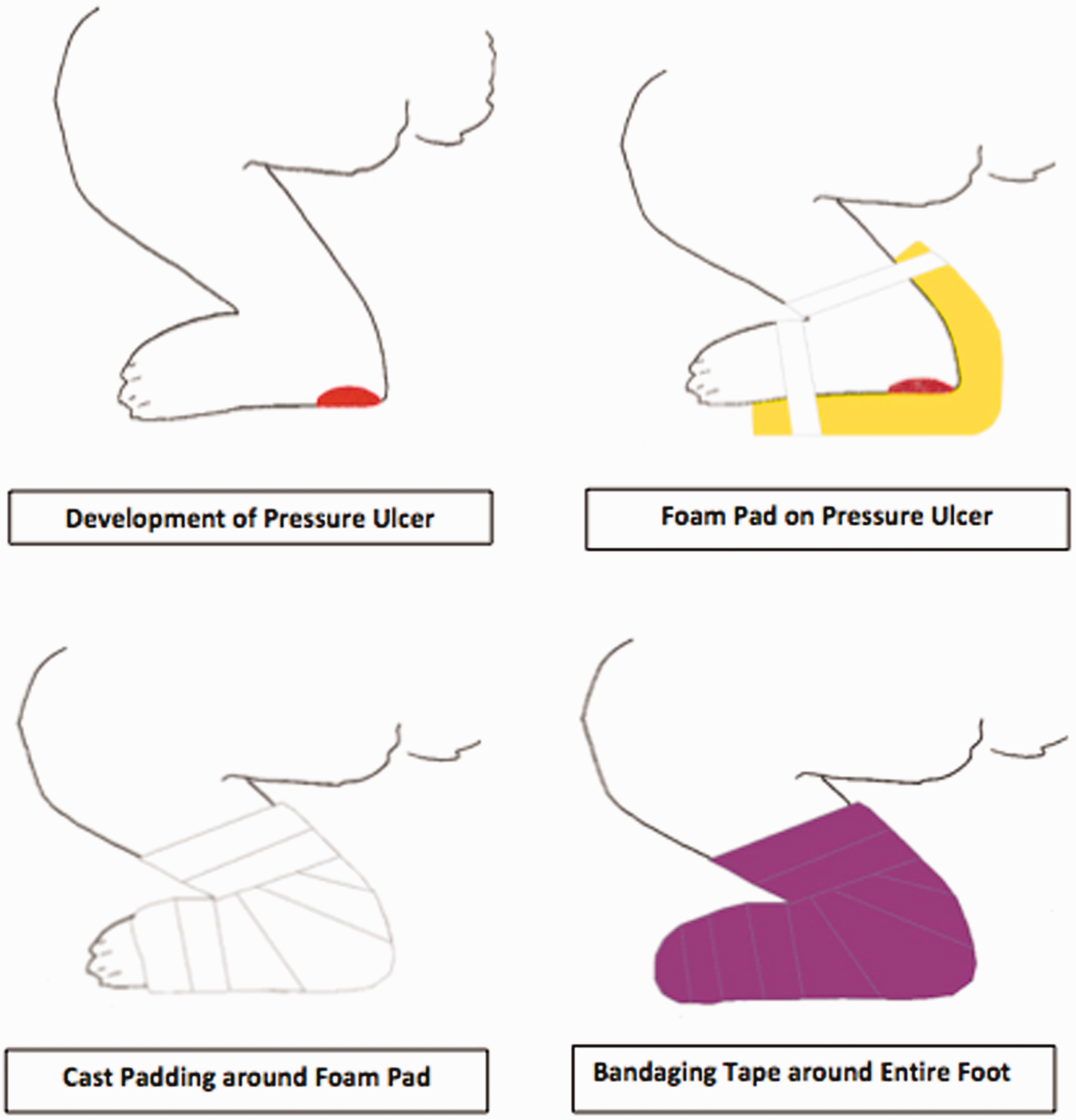
An “ulcer cushion” was developed that consisted of a custom-cut piece of foam sheet in contact with the bottom of the rabbits’ feet (Figure 4). The rabbits were held safely by one person, while another person gently applied the ulcer cushion. The foam was then wrapped with cast padding followed by veterinary bandaging tape along the rabbit’s entire foot (Figure 4). Moderate pressure was applied to the last layer of bandaging tape to ensure the bandage did not slip while making sure it was not too tight to affect circulation to the foot.
Cost analysis
Cost analysis of experiment.
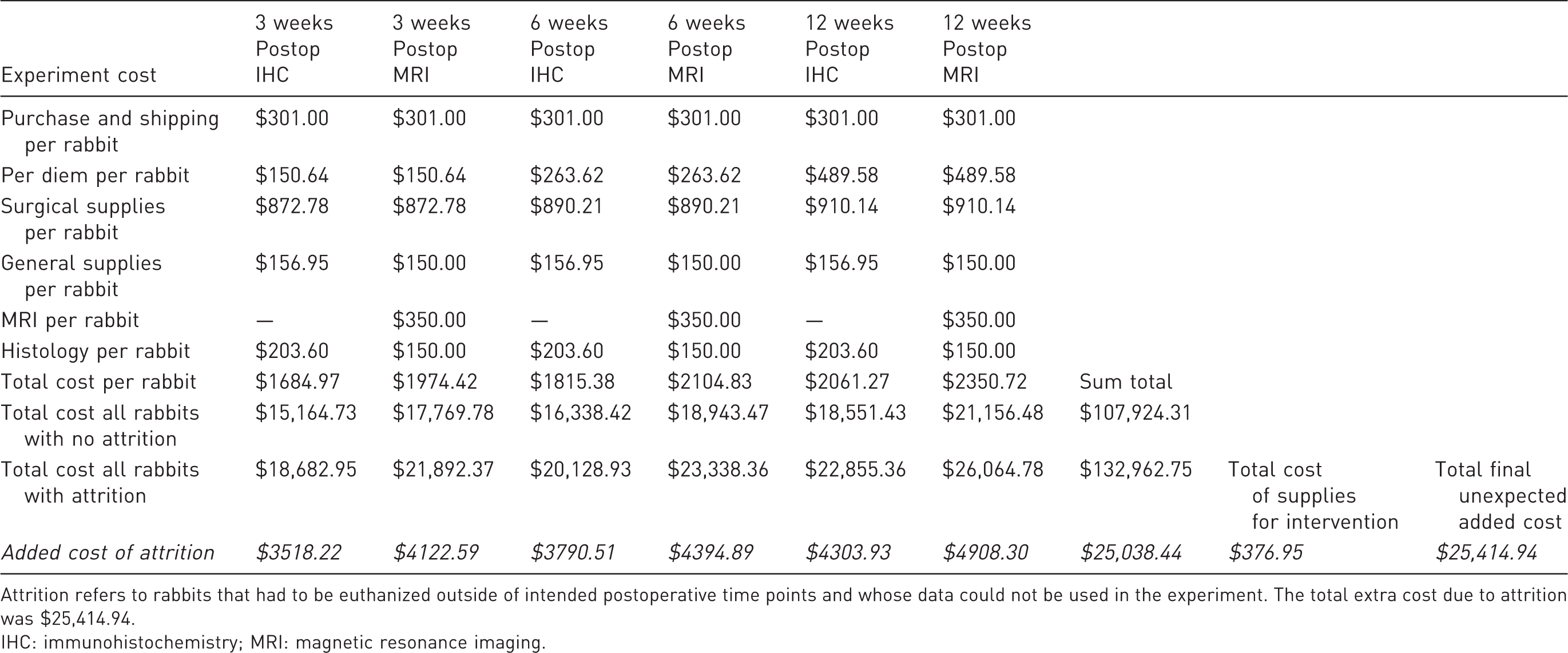
Attrition refers to rabbits that had to be euthanized outside of intended postoperative time points and whose data could not be used in the experiment. The total extra cost due to attrition was $25,414.94.
IHC: immunohistochemistry; MRI: magnetic resonance imaging.
Discussion
Studying peripheral nerve injury is a main focus of our laboratory. We designed this animal model in order to study the effects of larger implant sizes on peripheral nerve regeneration. Our original study was compromised due to the high incidence of unexpected complications. 12 In order to maintain the well-being of our subjects, we had to develop a strategy to improve their quality of life. In this report, we share the incidence of complications experienced, as well as our approach to manage and prevent them for future researchers.
One of the most frequent adverse effects encountered was autophagia. We concluded that this phenomenon was mostly due to the presence of axonal regeneration producing dysesthesia and not dependent on any intramuscular injections. This conclusion was supported by the fact that all of the intramuscular injections in our study were performed in the non-experimental hind limb. In previous studies, Vachon et al. demonstrated the correlation of autophagia of rabbits after the injection of ketamine and xylazine. This was related rather to inflammation and subsequent fibrotic tissue encapsulating the sciatic nerve. 7 Beyers et al. also encountered significant perineuritis on the ipsilateral hind limb injected with the same sedative combination. 14 Other studies have described that this phenomenon in other species could be related to environmental stressors such as temperature, isolation, and even diet.15,16 A recommendation regarding the total amount of volume has also been implemented to avoid this issue. 7 Despite the amount injected into muscle in our study, no self-mutilating injuries were encountered in the non-experimental limb, and we did not discover any histopathologic changes that could suggest that this could be the culprit.
Once autophagia occurred in the experiment, the rabbits were initially placed into foam donut collars immediately postoperatively. These collars were chosen because of their ease of application, ease of cleaning, softness, and overall lack of toxicity for the rabbits. This was insufficient for the intended purpose as the rabbits were still able to reach their toes. We quickly changed to a plastic Elizabethan collar with soft edges due to their extended range around the rabbit’s head. This collar was more effective at restricting the rabbit from reaching its toes, resulting in a decreased incidence of autophagia. We purchased two sets of 10 collars and developed a system to avoid delay in treatment. The benefits of these devices were that they were exchanged immediately, while the soiled ones were sent for cleansing. Through data analysis, we determined the improvement that our developed therapeutic intervention had in our studied population. We found a direct positive effect on the number of animals surviving until the endpoint and an inversely proportional effect on self-mutilating behavior after our intervention was applied.
Run chart demonstrating the effect of changing rabbit collar-type on rates of hind limb autophagy (green squares) and subsequent euthanasia (red circles) within the first 21 days after sciatic nerve injury. Blue triangles show total number of live rabbits enrolled in experiment at the recorded time point. Postintervention demonstrated statistically significant reduction in hind limb autophagy and euthanasia due to autophagy (p=0.0093 and p=0.0164, respectively). Illustration demonstrating how to apply the proposed ulcer cushion.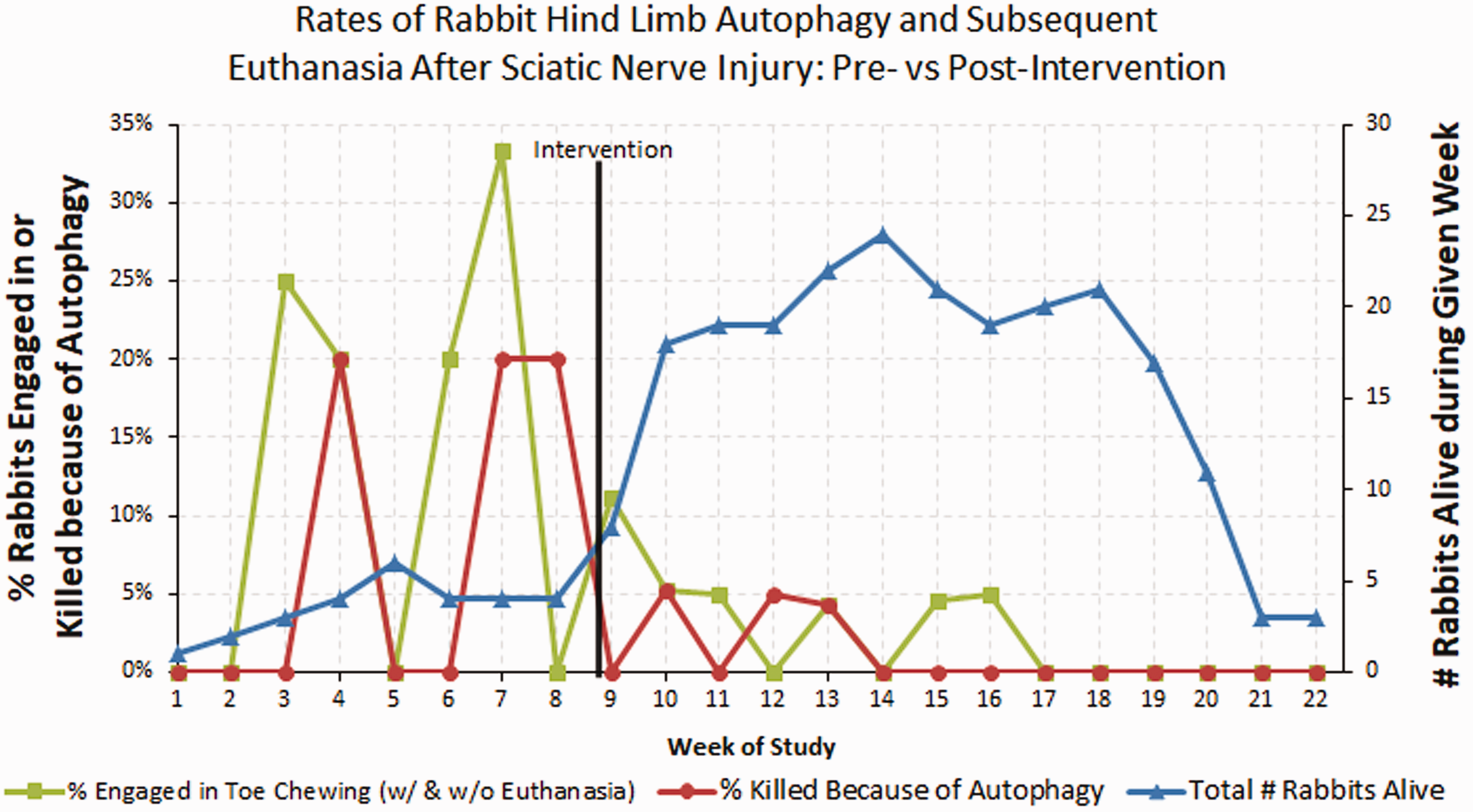
The collars were frequently found to be smeared with feces. Therefore, they were checked daily and changed regularly to maintain sanitary conditions and to avoid eye infections. It is also imperative to check the rabbit's necks where the collars are attached in order to decrease excessive moisture. If water accumulates around the neck, this can lead to fungal dermatitis. Certain rabbits tried to chew off the soft parts of the collars on the rims. If the rabbit chews the collar, it is necessary to replace the soft edges with something the rabbit cannot chew off from the collar, such as hypoallergenic surgical tape.
To treat pressure ulcers, the ‘ulcer cushions’ were based on what is used clinically to avoid this morbidity in human patients. Currently, there is not a commercially available device to treat this condition in animals, and therefore we manufactured our own. Caution should be taken to ensure the wrapping is not too tight so that circulation to the hind limb is compromised. We believe that our treatment not only aided in preventing pressure, but also added an extra layer of protection precluding autophagia. Statistical analyses on the effectiveness of the ulcer cushions were not performed, as they would be confounded by the recovery in motor function that the rabbits experienced over time, allowing them to lift their legs and relieve the pressure from their foot.
When we summed all the expenses that our therapeutic intervention required (collars and ulcer cushion), we were able to demonstrate how inexpensive this was compared to the cost of ordering more rabbits and their corresponding expenses ($376.95 vs. $25,038.44). In this study, we demonstrated that with a small amount of money invested in supplies, we could have save thousands of dollars and limited the number of animals used in the experiments.
Our interventions were able to treat and prevent these unexpected complications, but other measures could also help this morbidity. Applying the knowledge we have of treating human pressure sores, we could use inflatable beds in the bottom of the rabbits’ cages. These would have to be custom made because they are commercially unavailable. We also believe that adding other medications to our multimodality pain regimen such as gabapentin could improve the well-being of future animals involved in sciatic nerve injury models by treating neurologic pain.
The most important limitation of these interventions is their effect on recording behavioral recovery in the rabbits. If ambulatory behavior needs to be recorded postoperatively for an experiment, the added bulk of the cast and the limited range of motion from the collar can affect how the rabbit positions its legs while moving. The rabbit may drag the leg with the cast or overcompensate and raise the leg with the cast higher than the normal leg. This could lead to a lack of accurate behavioral data postoperatively. Since behavioral recovery is a common goal in sciatic nerve injury studies, this limitation must be considered before applying our interventions on rabbits. The high maintenance required to sustain treatment for the rabbits presents a second limitation. The casts and collars must be monitored and changed frequently. We recommend having at least two members of full-time staff with rabbit-handling experience capable of managing these treatments on a daily basis. If these requirements cannot be met, other models may offer a less time-intensive and costly option. Mini pigs, dogs, cats, and guinea pigs have been successfully used in previous experiments involving sciatic nerve injury and may serve as a better and more resilient model for preclinical experimentation that requires large sciatic nerve injuries.1,17
Conclusion
The rabbit model for sciatic nerve injury experiments appears viable based on past research. There is, however, an enormous gap in the literature describing common postoperative complications resulting from these experiments. Based on our experience, we recommend that if the issues described in this article cannot be effectively handled by full-time staff, then another animal model should be considered for sciatic nerve injury experiments. Each model holds risks and benefits. Thus, knowing about possible complications can help scientists choose an animal model most suited for the research team’s experimental question and resources. The apparent low cost of the purchasing rabbits is vastly offset by the high complication rates coupled with the intense amount of time, energy, and money needed to prevent and manage the complications. If rabbits absolutely must be used, the interventions described here, if applied immediately postoperatively, will reduce future complications and allow experimental progression.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One of the authors (J.M.C.) is supported by NIGMS of the National Institutes of Health under award number T32GM007347. The content in this report is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
