Abstract
Percutaneous catheterization is currently utilized for central venous access in adult and adolescent porcine models; however, neonatal models require a cut down to gain venous access. This approach requires general anesthesia and can result in systemic inflammation, which can confound studies investigating other inflammatory triggers. Here we present the first successful series of percutaneous, ultrasound-guided, durable central venous access in newborn piglets weighing 1 kg with a novel method of tunneling the catheter subcutaneously using a needle. Catheters (3–5 Fr, single- or double-lumen) were successfully placed in the right jugular vein of eight piglets weighing 1.3 ± 0.4 kg (mean ± standard deviation) with an average duration of catheter patency of 4.5 ± 2.1 days. There were no adverse events from catheter placement, though one catheter was inadvertently removed. This technique is a safe, minimally invasive method for obtaining secure central venous access in a neonatal porcine model.
Introduction
Central venous access is commonly required for animal research involving repetitive blood sampling or medications that can only be administered centrally such as total parenteral nutrition (TPN) and many chemotherapeutics. Typically, central venous access in animals is gained via surgical cut down. Invasive surgical procedures pose an increased infection risk to the animal 1 and require general anesthesia, postoperative pain control, and longer recovery times. Furthermore, surgery is an inflammatory process that can confound data in studies analyzing inflammatory markers. 2 Percutaneous access decreases perioperative animal care, is associated with decreased rates of surgical site infections, 1 can be performed under moderate sedation, and may minimize the inflammatory response. Ultrasound guidance has been shown to improve success rates, decrease complications, and decrease number of attempts when compared to landmark-based vascular access.3,4
Percutaneous, ultrasound-guided central venous access has been successfully demonstrated in pigs as small as 4 kg. 5 Newborn piglet models, however, are critical for the study of pharmacotherapies 6 as well as conditions specific to premature newborns or neonates, such as neonatal acute respiratory distress syndrome (NARDS) 7 and short bowel syndrome (SBS). 8 Central venous access is beneficial in these studies to allow for administration of irritant or vesicant medications as well as regular blood draws for cellular and immunologic analysis. However, percutaneous central venous access has never been described in a neonatal piglet model.
Here we present the first successful series of percutaneous, ultrasound-guided, durable central venous access in newborn piglets weighing 1 kg. This includes a novel method of creating a subcutaneous tunnel with a needle with the goal of providing positional stability with minimal additional trauma to the small piglets and protecting the line from damage during long-term studies. The purpose of this article is to report on the details of the technique and review our initial experience.
Animals, materials, and methods
Animal subjects
The catheterization method was performed on eight crossbred piglets (Yorkshire × Landrace; five males, three females) from multiple litters weighing 1.3 ± 0.4 kg (mean ± standard deviation; range: 0.7–1.9 kg). These piglets were part of a larger study on the effects of TPN on the intestinal bacterial flora. This study is still ongoing; thus, the results are not reported here. The piglets were delivered vaginally at an outside location. The umbilical cord was clamped and cut, and the piglets were immediately transported 2.5 h to the surgical facility. During transport, the animals were kept in a portable incubator supplemented by heated water bottles and blankets and the ambient temperature was monitored. The piglets were approximately 4 h old at the time of catheter placement. They received no oral feeding prior to the procedure. The animal protocol was approved by the University of Michigan Institutional Animal Care and Use Committee (#PRO00008016).
Surgical technique
Isoflurane 3% was administered to the piglets through a mask with 100% FiO2 until the piglets were not responsive to mild noxious stimuli but were spontaneously breathing and had no changes in capillary refill, mucous membrane color, or oxygen saturation. Piglets were placed on an operating table with a forced-air heating pad used to maintain core temperature. The following physiologic parameters were continuously monitored: heart rate using electrocardiogram, temperature using a rectal probe, and oxygen saturation using a pulse oximeter placed on the ear of the piglet. Capillary refill and mucous membrane color were evaluated every 15 min. They were placed in dorsal recumbency position with the neck extended (dorsiflexed) and the front legs retracted caudally. The right neck was prepped sterilely using chlorhexidine. An ultrasound machine (Zonare Z.One SmartCart, Mountain View, CA) with an L14-5sp transducer was used to obtain anatomic images of the vasculature of the right neck (Figure 1(a)). The skin around the planned puncture sites was infiltrated with bupivacaine 0.5%. A 21-gauge needle was then placed into the external jugular vein under direct ultrasound guidance (Figure 1(b)). Upon needle entry into the vein, a 0.018-inch cope (soft-tipped) wire was passed through the needle into the vessel. The needle was then removed from the vein. The 21-gauge needle was then inserted on the dorsolateral aspect of the piglet’s neck, just caudal to the ear, 3 cm from the initial puncture site. The needle was passed ventrally through the subcutaneous tissue and exited from the same hole as the previous puncture, adjacent to the wire. The free end of the wire was then passed retrograde through the needle and out the dorsolateral aspect of the piglet’s neck. The needle was removed, leaving only the wire in place. Intravascular position of the wire was confirmed with ultrasonography (Figure 1(c)). A skin nick was made with a scalpel where the wire exited the skin. A 3–5-Fr catheter on a dilator, 5–8 cm in length, was then passed over the wire, through the subcutaneous tunnel, and into position in the external jugular vein. The wire was removed and the line was flushed with normal saline. The line was sutured in place at the base of the catheter where it exited the subcutaneous tunnel (Figure 2). The catheter was then connected to an intravenous line that was passed through a custom tethering system, which included a jacket worn by the piglets. This protected the catheter while allowing full mobility of the piglets within the cage throughout the study. At procedure end, isoflurane sedation was stopped and the piglet was awoken and transported to the intensive care unit.
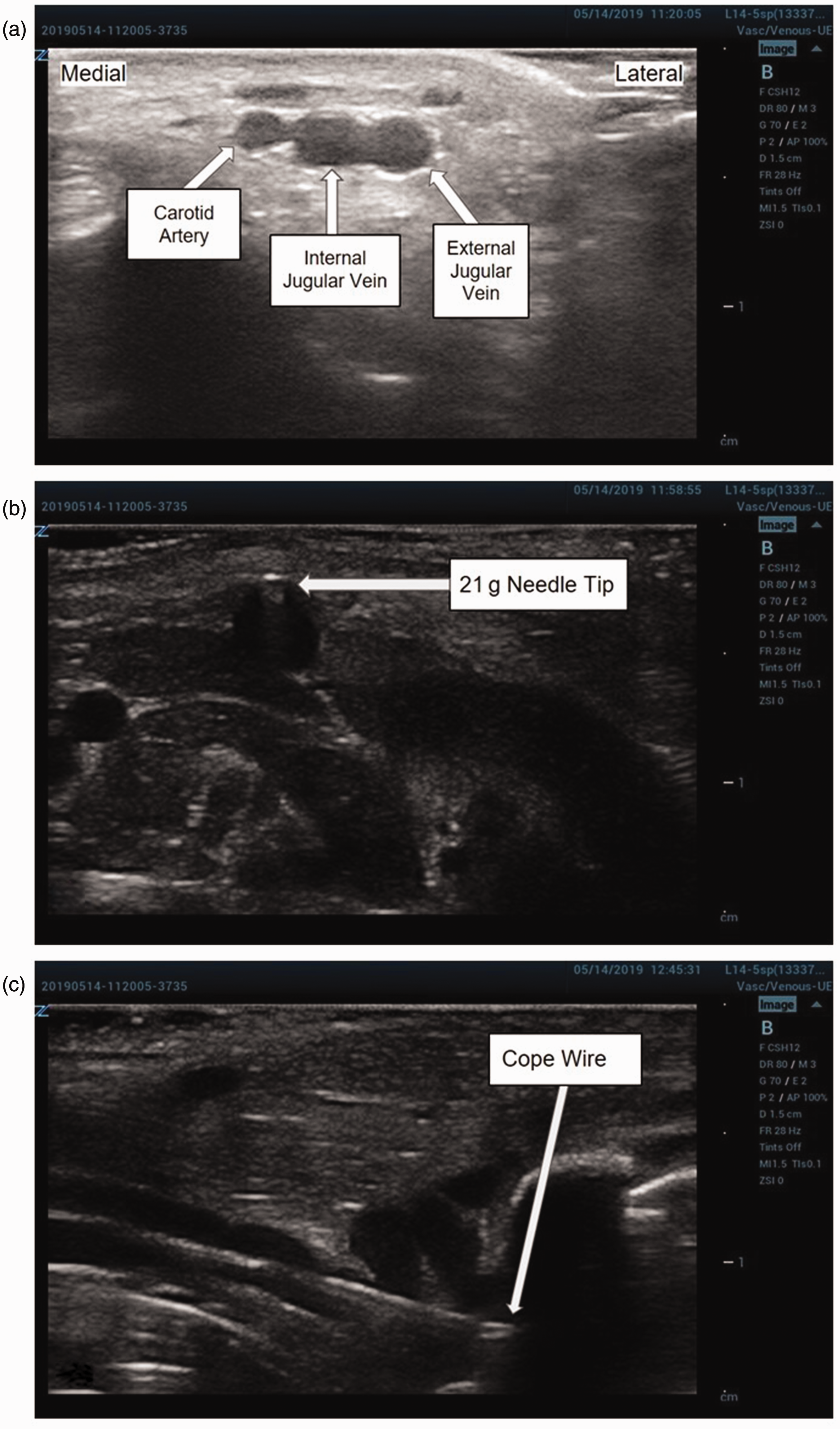
Ultrasound-guided vascular access: (a) position and depth of the target right external jugular vein in cross section confirmed via ultrasonography prior to puncture; (b) the external jugular vein, seen in cross section, is punctured with a 21-gauge needle and (c) a 0.018-inch cope wire is passed through the needle into the external jugular vein, seen in longitudinal view.

The newborn piglet with the tunneled catheter in place in the right external jugular vein, secured in place behind the ear.
Post-operative care
The piglets were housed in individual cages with enrichment and in view of other animals when possible. The temperature of the animal was monitored hourly and the cage temperature was adjusted using heating lamps and forced-air warmers to maintain normothermia. The piglets were continuously monitored for up to seven days with hourly recording of respiratory rate, heart rate, urine output, defecation quantity and quality, activity levels, and vocalizations. Results of this examination were used to calculate a post-operative pain score (Table 1). The intravenous catheter for each piglet was connected to a continuous infusion of either 0.225% sodium chloride with 5% dextrose or TPN, depending on study protocol. Piglets receiving infusion of 0.225% sodium chloride with 5% dextrose were supplemented with milk by mouth ad lib. The duration of follow-up and indications for termination were dictated by the overarching study protocol, the details of which are beyond the scope of this technique paper. The position of the external catheter hub relative to the puncture site was examined every 4–8 h to check for catheter displacement. The catheters were considered patent if fluid was infused with normal pressure reading on the infusion pump (Heska, Loveland, CO). Intermittent blood sampling was conducted throughout the experiment. If additional pain control was needed according to the pain score (Table 1), buprenorphine 0.01 mg/kg was administered as needed every 12 h. The FELASA/ECLAM/ESLAV Working Group definitions were used to classify the severity of the procedure. 9
Post-operative animal pain score.
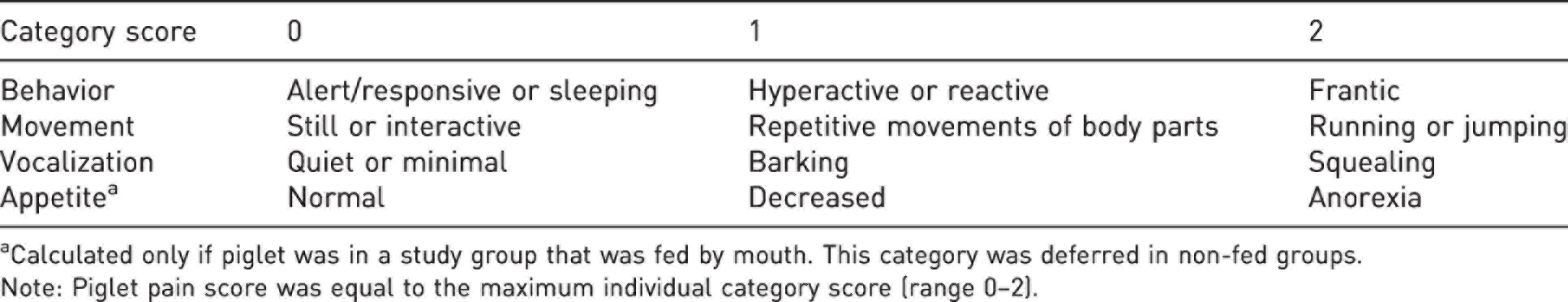
aCalculated only if piglet was in a study group that was fed by mouth. This category was deferred in non-fed groups.
Note: Piglet pain score was equal to the maximum individual category score (range 0–2).
Results
Catheters were successfully placed in eight piglets. Procedure time was 62 ± 29 min, which included the initiation of and recovery from sedation. Average duration of catheter patency was 4.5 ± 2.1 days. The animals demonstrated no significant impairment in well-being after recovery from procedural sedation (average post-operative pain score 0.1 ± 0.4), and only required one dosage of buprenorphine given intramuscularly post-operatively, making this a mild-severity procedure. 9 There were no instances of catheter thrombosis or occlusion, or infection. One piglet had an inadvertent removal of the catheter. Necropsy showed no significant swelling, hematoma, or infection and appropriate positioning of the tip of the catheter on all other piglets.
Discussion
This study demonstrates a safe, mild-severity method for achieving secure, long-term central venous access under ultrasound guidance in newborn piglets weighing 1 kg. To the authors’ knowledge, this is the first published technique for minimally invasive central catheterization of newborn piglets of this size. Though the effectiveness of ultrasound-guided central venous access has been demonstrated in adult and juvenile porcine models,4,5 it has never been demonstrated to be feasible in piglets weighing less than 4 kg. Previous approaches to central venous access in neonatal piglets have used an open cut-down technique. 10
Even such minor surgical interventions as a neck cut-down alter stress-related endocrine profiles in animal subjects for several days, whereas cortisol levels are only mildly elevated 1 h after percutaneous catheterization and return to baseline 2 h afterward. 11 Though hormone levels and inflammatory markers were not collected in these piglets nor was an open cut-down comparison group included, the piglets in our study demonstrated no impairment in well-being post-operatively, suggesting that our percutaneous technique similarly causes minimal stress to the animals. This optimizes animal welfare and post-procedure recovery. Minimizing animal stress during catheter placement may also limit confounding factors from surgical technique in studies of immunologic or inflammatory processes such as SBS or NARDS, though further study is needed to test this hypothesis.
Implementation of this technique may be limited by several factors. First, instrumentation was performed by pediatric surgeons (MWR) and surgical trainees (MDJ, BPF) with advanced training in ultrasound-guided procedures. The size and fragility of the vessels make this a technically challenging procedure, which may require extensive experience with ultrasound-guided procedures. Though the follow-up period in this series was relatively short, it was likely adequate to capture most potential complications of the procedure, as most central venous catheter complications typically occur at the time of placement. 12 Additionally, high-resolution ultrasound equipment, which greatly facilitates catheter placement, is expensive and may not be available in all laboratories. Finally, due to the technical difficulty, operative time may increase as researchers are gaining experience with the technique. 13
In conclusion, this technique is a minimally invasive and reliable method of obtaining long-term central venous access in neonatal porcine models.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
