Abstract
Over the past 50 years, image-guided procedures have been established for a wide range of applications. The development and clinical translation of new treatment regimens necessitate the availability of suitable animal models. The juvenile Göttingen minipig presents a favourable profile as a model for human infants. However, no information can be found regarding the vascular system of juvenile minipigs in the literature. Such information is imperative for planning the accessibility of target structures by catheterization. We present here a complete mapping of the arterial system of the juvenile minipig based on contrast-enhanced computed tomography. Four female animals weighing 6.13 ± 0.72 kg were used for the analyses. Imaging was performed under anaesthesia, and the measurement of the vascular structures was performed independently by four investigators. Our dataset forms a basis for future interventional studies in juvenile minipigs, and enables planning and refinement of future experiments according to the 3R (replacement, reduction and refinement) principles of animal research.
Over the past 50 years, the number of procedures in interventional radiology has risen, and image-guided procedures have been established for a wide range of applications.1,2 Because they are less invasive than surgical interventions, image-guided procedures are suitable for infants. 3 For example, image-guided transcatheter interventions, such as balloon atrial septostomy, have evolved to become suitable alternatives to surgery in the field of paediatric cardiology.4–10 Applications are transposition of the great vessels, 9 mitral valve stenosis, 8 haemodynamical significant atrial septum defects 9 and critical pulmonary valvar stenosis. 9 Apart from paediatric cardiology, there are other applications including congenital and infantile vascular malformations,11,12 embolization of tumours, and bleeding. 11 Future applications for image-guided transcatheter interventions are expected in regenerative medicine, especially in the field of cell transplantation; for example, liver cell transplantation has been successfully performed via a femoral artery catheter into the spleen in adults. 13 Because liver cell transplantation is primarily effective in congenital metabolic liver disease, 14 it is expected that trials in infants will follow. Further potential applications are stem cell infusions into damaged tissues, e.g. in type 1 diabetes.
The development and clinical translation of new treatment options necessitate the availability of well-suited animal models. The use of pigs as clinically-sized experimental models has increased in various experimental fields. 15 Due to the many similarities between human and porcine biology, especially that of the cardiovascular system,16,17 pigs have been used in preclinical trials of paediatric transcatheter interventions such as the testing of new stents,18,19 catheters 20 and intravascular occluder devices. 21
Special challenges are faced by neonates and young infants due to their small intravascular volume, the inability to control their body temperature and the small size of their arterial system. 22 Along with ethical difficulties with evaluations in humans, an experimental model should be chosen in which these problems can be simulated. The juvenile Göttingen minipig presents a favourable profile as a model for human infants; at 11 weeks old, it has approximately the same weight as that of a human infant at birth. 23 One advantage of the Göttingen minipig over the Landrace pig is a slower linear growth, 23 which leads to a higher resilience of the circulatory system. 24 Other advantages are a more stable heart frequency,25,26 higher haemoglobin levels,27–31 and lower systemic blood pressure.25,32 These and other advantages predispose these animals to cardiovascular research. 33 For these reasons, the juvenile Göttingen minipig seems to be a suitable preclinical model for human infants.
Although extensive information is available on the vascular system of pre-adult and adult minipigs, 33 no information on the vascular system of juvenile minipigs can be found in the literature. Such information is imperative for planning the accessibility of target structures by catheterization. By providing these data, we hope to facilitate the planning of preclinical trials and basic research in the aforementioned fields.
Materials and methods
Experimental setting/design
Four female Göttingen minipigs, aged 8–12 weeks, were used for the imaging studies (Ellegaard, Dalmose, Denmark). The animal weights were 5.5, 5.6, 6.1 and 7.3 kg. Computed tomography (CT) images for the specification of the vessel size were obtained as part of another experimental protocol approved by the State Office of Health and Local Affairs (LAGeSo, Berlin, Germany; Reg. No. G 0260/13), and animals were controlled by the local animal welfare officer. The animals were kept in the animal care facility of the Department of Experimental Medicine of the Charité – Universitaütsmedizin Berlin (certified by ISO 9001:2008). The housing conditions were conformed to the recommendations of FELASA and GV–SOLAS. The piglets were housed in pairs or with an adult minipig dedicated for companionship.
Anaesthesia for CT investigations
All experiments were performed under anaesthesia. Premedication was administered with an intramuscular injection of 3 mg/kg azaperone (Stresnil®; Janssen-Cilag, Neuss, Germany), 25 mg/kg ketamine (Ursotamine®; Serumwerk, Bernberg, Germany), 3.5 mg/kg xylazine hydrochloride (Rompun®; Bayer Vital GmbH, Leverkusen, Germany) and 0.03 mg/kg atropine sulfate (Atropin sulfate®; B Braun Melsungen, Melsungen, Germany). The animals were orally intubated (ID 4.0 with cuff, Mallinckrodt, Mallinckrodt Medical, Athlone, Ireland) and kept under general anaesthesia with spontaneous breathing using repeated administration of 4–6 mg/kg propofol (Fresenius Kabi AG, Bad Homburg, Germany) and/or 15–25 mg/kg ketamine (Actavis Group PTC EHF, Munich, Germany). Mechanical ventilation was performed when required to stabilize blood gases and pH homeostasis. A total of 500 mL of balanced crystalloids (Jonosteril®; Fresenius Kabi AG) were administered continuously.
Computed tomography
CT angiography was performed by the Department of Radiology of the Charité – Universitätsmedizin Berlin. A 64-row CT scanner (LightSpeed VCT; GE Healthcare, Little Chalfont, UK) was used for the imaging with a slice thickness of 0.625 mm and an image matrix of 512 × 512. The window level (WL) and the window width (WW) were adjusted individually using the preset for vessel measurements with a WL of 200 Hounsfield units (HU) and a WW of 600 HU. The contrast medium used was iopromide with an iodine concentration of 370 mg of I/mL (Ultravist 370; Bayer, Berlin, Germany).The contrast agent was applied at 1 mg/kg body weight, with 0.7 mL/kg of normal saline, and a flow rate of 1 mL/s. The measurements consisted of three phases: the arterial phase via bolus-tracking of the abdominal aorta, the portal–venous phase starting directly after the arterial phase, and the venous phase starting after 60 s.
Image analysis
The images were accessed via the Visage 7.1 (Visage Imaging GmbH, Berlin, Germany) imaging platform. Measurements of the vascular structures of each minipig were performed independently by four trained senior researchers of the Department of Surgery, Experimental Surgery and Regenerative Medicine. The measuring protocol was developed by a senior radiologist. Each individual researcher had no access to the measurement results of the other researchers when performing the task. When the results differed largely, the deviating measurements were controlled and revised eventually. In cases of revision, the deviating measurements were discussed with all the researchers involved and a consensus was reached. The vessel diameters were measured three times per session by each examiner, once horizontally, once in a vertical direction and once by defining a circular region of interest along the wall of the vessel, and determining the diameter of the inlying area. Endpoints of the horizontal and vertical measurements were defined as the edge of the contrast agent signal. To ensure accuracy of measurement, the section planes were adjusted to cut the vessel orthogonally (Figure 1). Due to technical issues regarding the imaging of one pig, some vessels were only measured in three of the animals, as noted in the Results section.
Measuring process with adjusted planes. Planes have been adjusted in partitions b and c. This results in an orthogonal cut in picture a.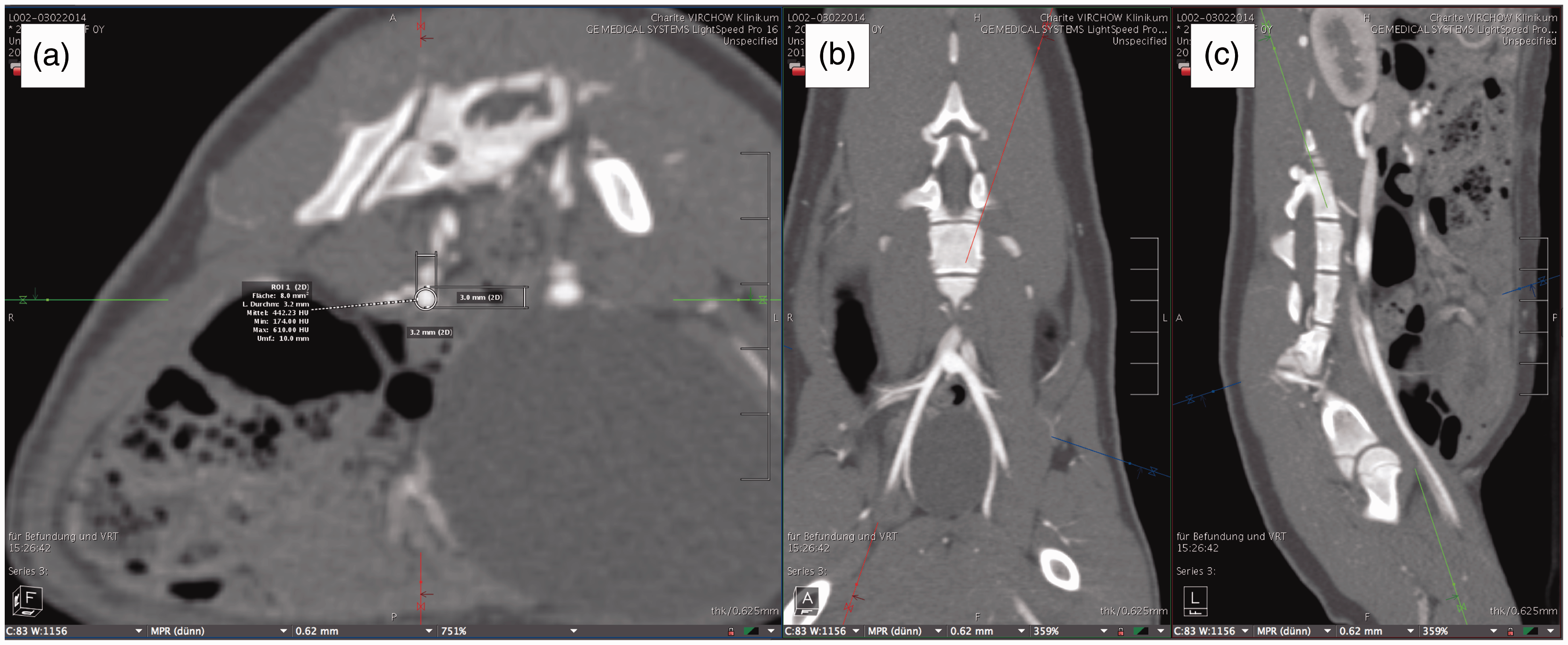
The measuring points were derived from ‘Macro- and micromorphometric studies of the vasculature of the Göttingen minipig® using contrast enhanced computer tomography’ by Müller, 33 with adjustments made due to the smaller size of the animals.
Statistical analyses
All analyses were performed using Prism 6 software (GraphPad Software Inc, La Jolla, CA, USA). The mean, median, standard deviation, and coefficient of variation were determined.
Results
Statistical analysis of all measurements taken.
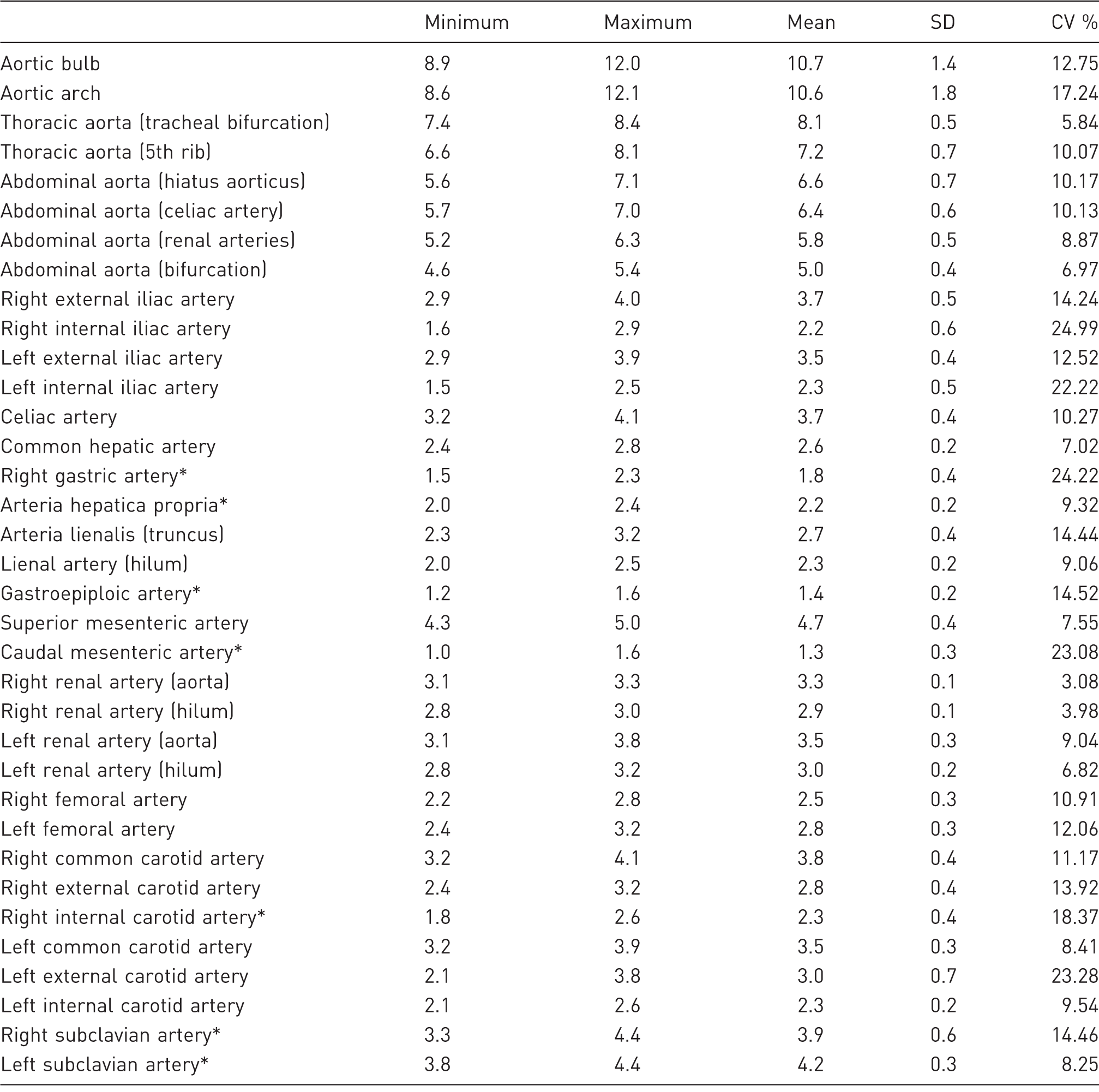
Some vessels are measured at various points. These measuring points are shown in brackets. Vessels marked with an asterisk contain only three animals in the dataset. Data are given in mm. SD: standard deviation, CV: coefficient of variation.
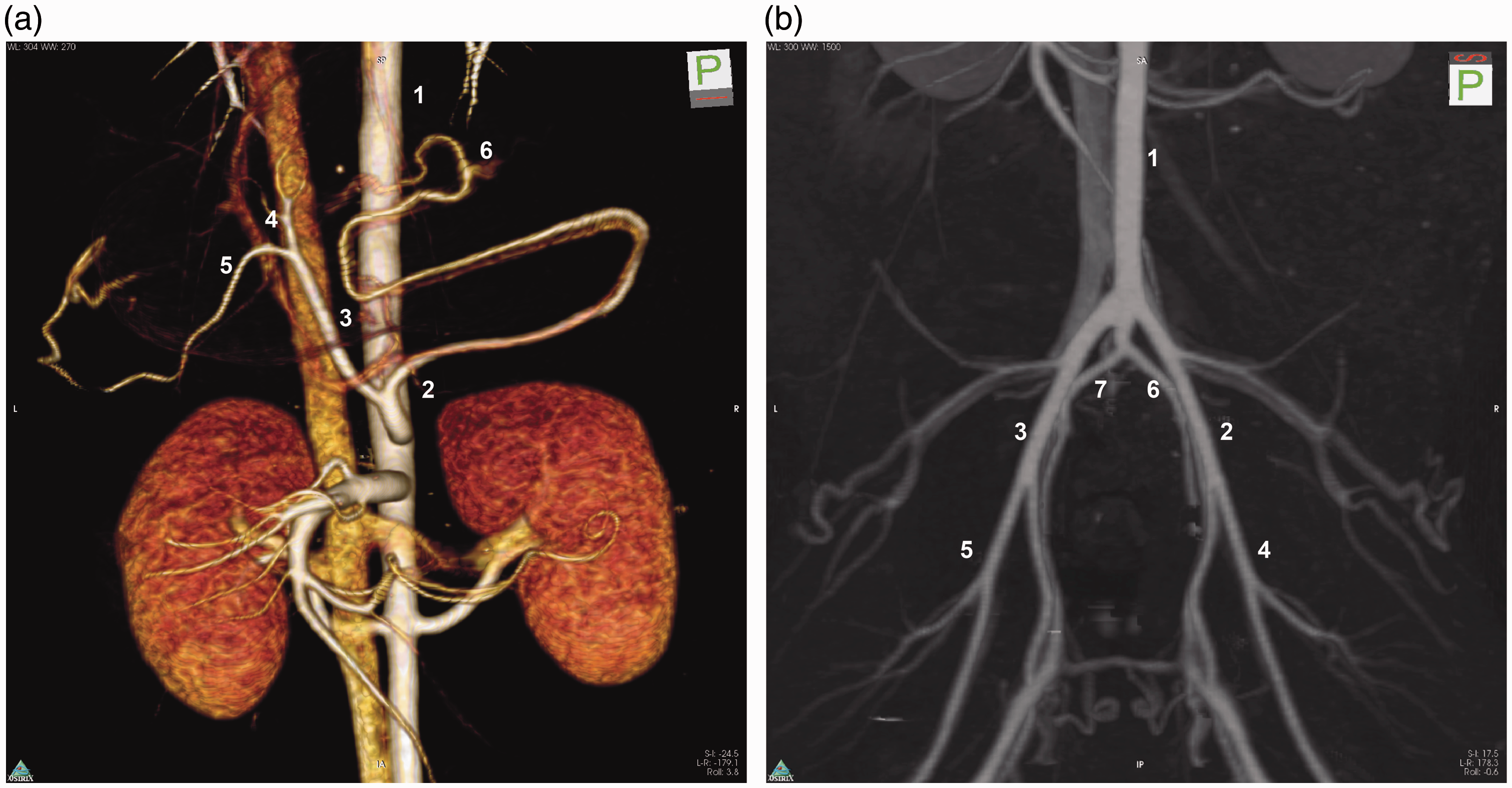
(a) Three-dimensional reconstruction of the aorta, celiac artery and branches, superior mesenteric artery and renal arteries. 1: aorta; 2: lienal artery; 3: common hepatic artery; 4: right gastric artery; 5: proper hepatic artery; 6: gastroepiploic artery. (b) Mapping of the pelvic and lower extremity arteries. 1: aorta; 2: left external iliac artery; 3: right external iliac artery; 4: left femoral artery; 5: right femoral artery; 6: left internal iliac artery; 7: left internal iliac artery.
Aorta
The aorta was measured at a mean of 10.7 mm at the widest point, the aortic bulb (Table 1, Figure 3a). It subsequently narrowed until the bifurcation. The aortic arch was measured with a mean diameter of 10.5 mm. The thoracic descending aorta started at a mean diameter of 8.1 mm at the tracheal bifurcation and narrowed to 7.2 mm at the height of the 5th rib. The abdominal aorta started with a mean diameter of 6.6 mm and narrowed to 6.4 mm at the origin of the celiac artery, 5.8 mm at the renal arteries and 5.0 mm at the aortal bifurcation.
Boxplots of the aorta (a), the arteries of the abdomen (b) and of the lower torso (c). Each point represents the measurements of one pig.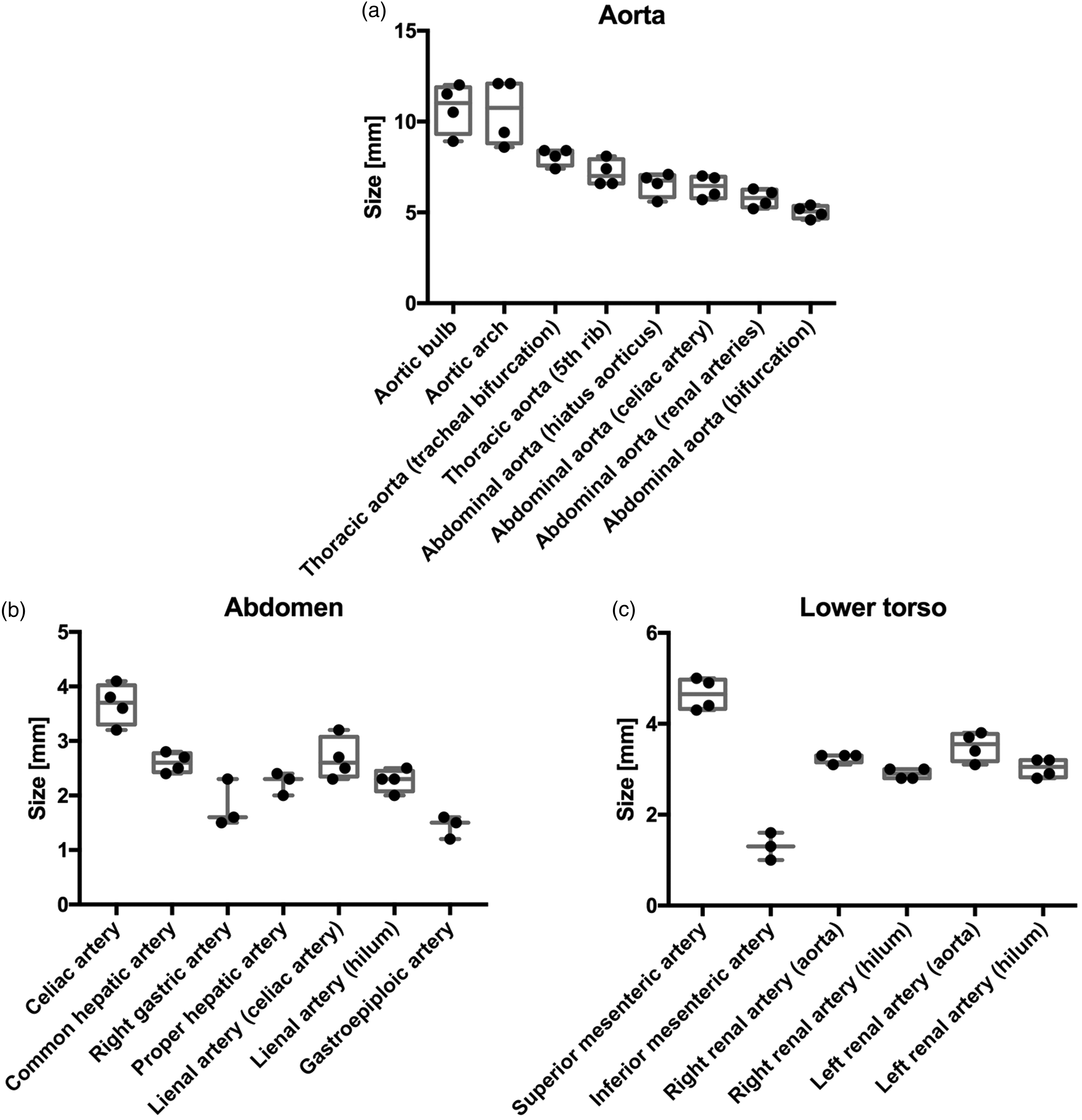
Abdomen
The mean diameter of the celiac artery was measured at 3.7 mm (Table 1, Figure 3b). The common hepatic artery was measured at a mean of 2.6 mm. At its departure from the common hepatic artery, the right gastric artery was measured at a mean of 1.8 mm. Subsequently, the proper hepatic artery narrowed to a mean of 2.2 mm at the hilum.
The splenic artery was measured at a mean diameter of 2.7 mm at its root and narrowed to 2.3 mm at the hilum. After passing the spleen, the gastroepiploic artery was observed to emerge with a mean diameter of 1.4 mm.
Lower torso
The superior mesenteric artery had a mean diameter of 4.7 mm, and the inferior mesenteric artery diameter was 1.3 mm, with a large coefficient of variation of 23% (Table 1, Figure 3c).
The right renal artery was measured at a mean diameter of 3.3 mm at the aorta and narrowed to 2.9 mm at the hilum. The left renal artery was almost the same size, 3.5 mm at the aorta and 3 mm at the hilum. The variation between animals was small, with coefficients of variation from 3% at the base of the right renal artery to 9% at the base of the left renal artery.
Pelvic and lower extremity arteries
The external iliac arteries had mean diameters of 3.7 mm on the right and 3.5 mm on the left (Table 1, Figure 4a). The internal iliac arteries had mean diameters of 2.2 mm on the left and 2.3 mm on the right. However, the internal iliac arteries had large coefficients of variation, 25% on the right and 22% on the left.
Boxplots of the arteries of the pelvis and lower extremities (a) and of the neck and head (b). Each point represents the measurements of one pig.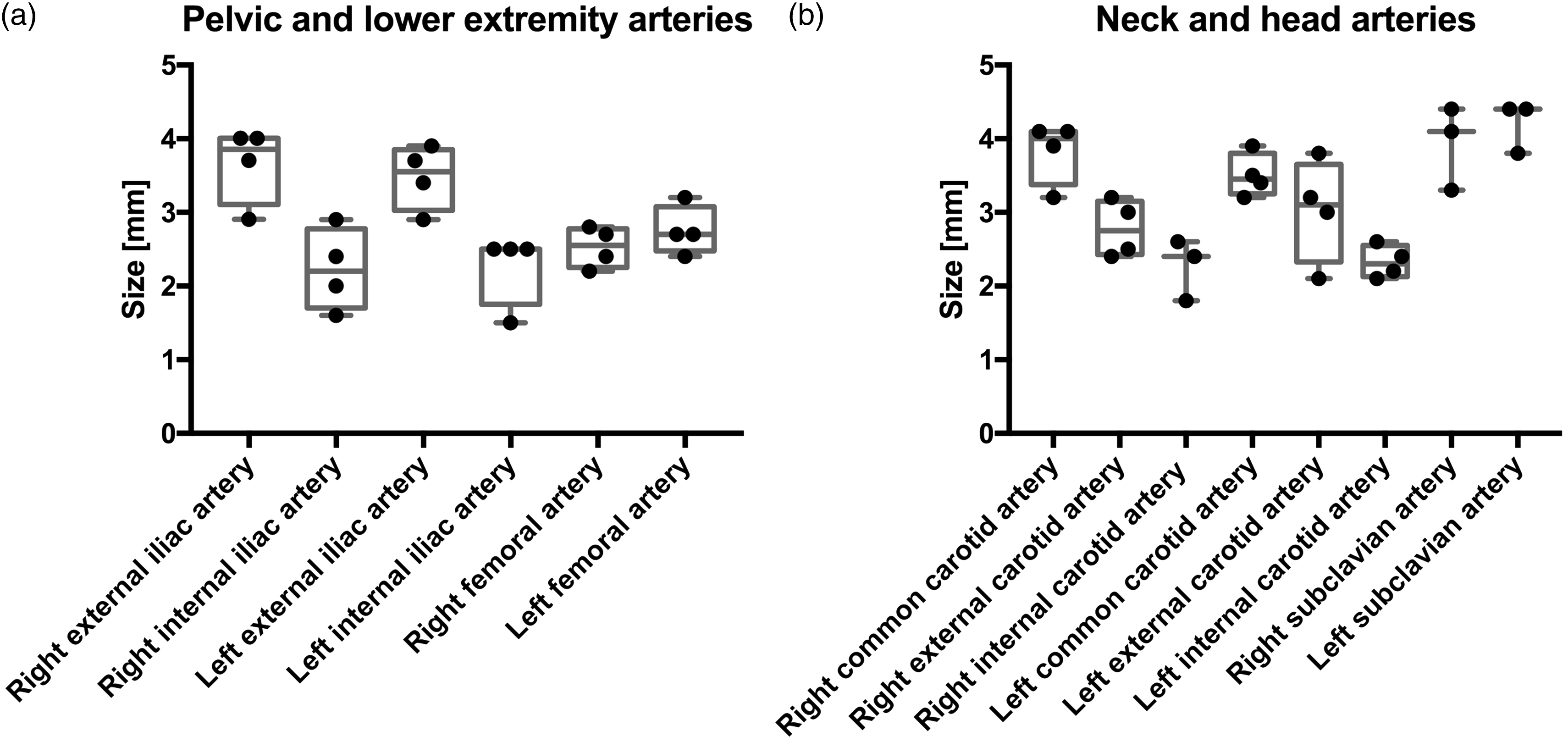
The common femoral arteries had mean diameters of 2.5 mm on the right and 2.8 mm on the left, with coefficients of variation of 11% and 12%, respectively.
Neck and head arteries
The mean diameters of the common carotid arteries were measured at 3.8 mm on the right and 3.5 mm on the left (Table 1, Figure 4b). After division of the common carotid artery, the mean diameters on the right were measured at 2.8 mm for the external carotid artery and 2.3 mm for the internal carotid artery, compared with 3 mm for the external and 2.3 mm for the internal carotid artery on the left side. The coefficients of variation were some of the largest in the study, 23.3% on the left external carotid artery and 18.4% on the right internal carotid artery.
The mean diameters of the subclavian arteries were measured at 3.9 mm on the left and 4.2 mm on the right.
Discussion
The pig model is by far the most commonly used large animal model for scientific research in Germany. 34 Because of the growing importance of the minipig model, detailed mapping of the vascular anatomy of pre-adult and adult animals has been performed. 33 However, data on juvenile minipigs are lacking.
The prime objective of this study was to evaluate the accessibility of important target structures by catheterization in juvenile Göttingen minipigs. Therefore, the vascular circumference of the access vessels and the hilar arteries are of interest. A commonly used access vessel for interventional catheter placement is the femoral artery. The mean diameter was measured as 2.5 mm in the left femoral artery and 2.8 mm in the right femoral artery. This should allow for standard 21-gauge entry needles to be used in the femoral artery. In a recent study, balloon angioplasty was performed using a 4 F introducer sheath. 35 With further advancement in microcatheter technology, the juvenile Göttingen minipig should be a suitable preclinical model for transfemoral catheterization.
If larger access vessels are required, other options should be explored. For instance, the smallest recommended sheath size for intravascular occluder devices (e.g. Amplatzer occluder; St Jude Medical, St Paul, MN, USA) is 6–7 F. 36 This may not allow for transfemoral insertion. However, by assessing the data, other possible access routes can be evaluated. The iliac artery has a mean diameter of 3.5 mm on the left and 3.7 mm on the right. Additionally, the subclavian artery could be considered, with diameters of 4.2 mm on the left and 3.9 mm on the right. Therefore, both of these vessels may likely to be suitable.
Apart from the heart, other possible targeted organs are the liver and the spleen. At its departure from the celiac artery, the lienal artery measures 2.7 mm; however, at the splenic hilum it narrows to 2.2 mm. In our experience, this size is sufficient to use customary microcatheters such as the Cook Medical MicroFerret-18 Zeta Infusion Catheter (Cook Medical Inc, Bloomington, IN, USA). After passing the spleen, the lienal artery becomes the gastroepiploic artery. This artery has a diameter of 1.4 mm. Because in pigs the gastroepiploic artery supplies the greater curvature of the stomach, which differs from humans, it has to be taken into consideration that liquids or particles inserted into the splenic artery may transcend into the gastric wall and cause unwanted effects.
Our CT-based dataset of the arterial vasculature of juvenile minipigs provides reliable and precise imaging. However, in small vessels and structures, the reliability of measurements is limited by the resolution. There are variations in the size of the arteries measured between the animals. The largest variations in size between the animals are observed in the internal iliac arteries, the right gastric arteries, the inferior mesenteric artery and the right external carotid artery, which might be due to the fact that these vessels have relatively small diameters compared with the other vessels analysed in our survey. The aforementioned suggested entry and target vessels have smaller coefficients of variation in our analyses, which were performed by four independent investigators. The number of animals used in our study was limited to four. Moreover, additional data on animals of varying weights and gender would be necessary to generate a more complete set of data of the various growth stages in the juvenile Göttingen minipig. The anaesthetics used in our protocol may also have had an effect on the vascular diameters of the animals. Xylazine, an alpha-2 adrenoceptor agonist, and atropine, a muscarinic antagonist, are known to cause vasodilation. Propofol also causes vasodilation due to inhibition of the sympathetic vasoconstrictor nerve activity. 37 We could only speculate on the effect size of our anaesthetic protocol on the vascular diameters of the juvenile minipigs. Since anaesthesia is necessary in the majority of interventional studies in minipigs, we believe this circumstance has little practical relevance. Of note, locally injected lidocaine, which acts by blocking of the voltage-gated sodium channels in the neuronal cell membrane, might lead to different results with presumably slightly greater vessel diameters. 38
However, we believe that our data represent a realistic image of the vascular anatomy of an eight-week-old juvenile Göttingen minipig, which is a suitable preclinical model for the human newborn. Therefore, we have provided elements for refinement in future experiments, according to the 3R (replacement, reduction and refinement) principles of animal research. 39 For interventional studies in animals, possible access routes and the right choice of catheter could be assessed based on these data. These enhancements in planning could lead to a reduction in the number of animals needed for experiments. For our experiments, a pilot study using one animal had to be performed prior to a transcatheteral interventional study accessing the femoral artery (unpublished data). This could have been avoided if the data in this study had been available.
In conclusion, we present a CT-based mapping of the arterial diameters of juvenile Göttingen minipigs weighing 6.13 ± 0.72 kg. Our data show that the anatomy of the juvenile minipig allows for many procedures to be carried out in ways similar to those used in human infants. However, not all access routes are available for all procedures, and the arteries of the target organs may pose obstacles for certain types of catheters. Our data may serve as a tool for assessing and forecasting these problems, and may therefore lead to more efficient planning resulting in the use of fewer laboratory animals.
Footnotes
Acknowledgement
We would like to thank Visage Imaging GmbH for providing us with the software used in the image analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Nathanael Raschzok and Dr. Benjamin Strücker are in the BIH-Charité Clinician Scientist Program funded by the Charité – Universitätsmedizin Berlin and the Berlin Institute of Health. The other author(s) did not receive any funding.
