Abstract
Central venous and arterial access through minimally invasive techniques has been described in adult pigs. This article demonstrates success in juvenile animals. Using ultrasound guidance and the modified Seldinger technique, 5 Fr/15 cm single-lumen central venous catheters and 20 Ga 4.5 cm femoral arterial catheters were placed in six Yorkshire cross-bred swine. All six cases had no loss of venous catheter patency or infection during the 96-hour follow-up period. Arterial catheters remained patent, and no significant bleeding was noted after removal.
Introduction
Reliable vascular access for sampling and treatment is critical in studies lasting longer than a few hours.1,2 Traditional surgical cut-down procedures may cause confounding sites of pain and risk of infection and may falsely elevate inflammatory markers. 3 The method discussed here has become standard of care in human medicine and decreases both complications and the number of attempts at vascular access. 4 Landmark-based approaches for central venous catheterization have been described in fetal and adult pigs.5,6 Ultrasound-guided central venous and arterial access has been described in larger adult pigs, ranging in weight from 50 to 78 kg, with good success.3,7 We present a cohort of juvenile pigs averaging 40 kg in weight to demonstrate feasibility in smaller animals.3,7
Methods
Animals
Six female Yorkshire pigs (Sus scrofa domestica) with a mean weight of 40 ± 4 kg were used. Given the pilot nature of the study, only one sex of animals was used. No randomization or blinding was performed. Animals were obtained from the University of Florida Swine Unit and Wallisser Pork, Inc. (Wimauma, FL). Animals were housed in a climate-controlled (23 ± 2°C) cage with enrichment toys. A light–dark cycle of 12 hours was utilized during a seven-day acclimatization period. Staff performed daily feeding and wellness checks. The University of Florida IACUC (protocol #201810157) granted ethical and protocol approval. The study was completed according to the National Research Council’s Guide for the Care and Use of Lab Animals and reported per the 2020 ARRIVE guidelines for reporting laboratory animal research.
Anesthesia
Pigs were sedated with intramuscular 0.5–2 mg/kg ketamine, and an 18 Ga temporary ear catheter was placed to allow for induction of total intravenous anesthesia using propofol 2–4 mg/kg and ketamine 0.5–2 mg/kg/min while the animals were intubated and ventilated at 10 mL/kg tidal volumes to a target end-tidal CO2 of 25–40 mmHg (Hallowell 2000; Hallowell EMC, Pittsfield, MA). Animals were placed in dorsal recumbency with limbs restrained. Standard monitoring, including continuous electrocardiography, pulse oximetry, noninvasive blood pressure, waveform capnography, and bodily temperature, was used (Waveline Pro; Avente Health Solutions, San Clemente, CA). The right jugular furrow and left inguinal region were clipped, cleaned with chlorhexidine solution, and draped. Anesthesia monitoring was continued throughout the experimental procedure, and animals were euthanized at standard humane endpoints or at the conclusion of the observational period with an intravenous injection of 150 mg/kg sodium pentobarbital with phenytoin solution at the conclusion of a 96-hour follow-up period.
Target vessel identification
The Butterfly iQ handheld ultrasound (Butterfly Network, Inc. Guilford, CT) was utilized for its portability. The 8 MHz vascular access setting was used. The probe was placed in a sterile cover. Figure 1 shows representative neck vessel anatomy, with the needle tip visible in the transverse view of the external jugular vein. The ultrasound probe was held perpendicular to the skin to obtain a cross section of the vessels. Manual compression confirmed vessel identity, as venous structures collapsed easily while the artery remained full and pulsatile. Skin was anesthetized with 5 mL 1% lidocaine after vessel location on ultrasound.
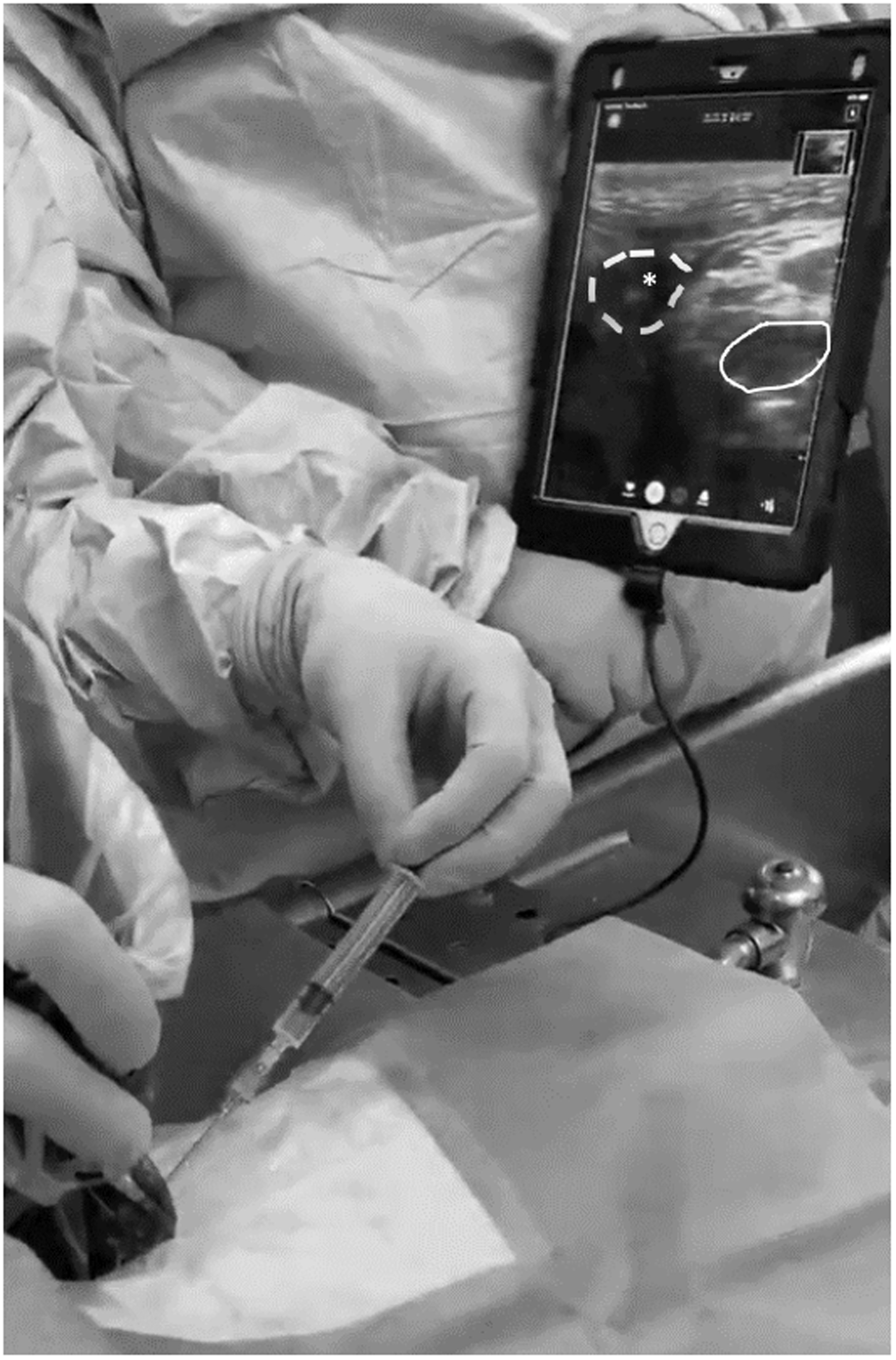
Ultrasound of right external jugular vein and carotid artery. This transverse view shows venous (dashed) and arterial (solid) structures with needle visible within the venous structure (asterisk).
Venous catheter technique
Venous access was established with the modified Seldinger technique using a Cook® 5 Fr/15 cm central venous catheter set and tray (Cook Medical LLC, Bloomington, IN). The 18 Ga introducer needle and syringe were inserted with the bevel facing medially at a 45° angle from the probe and advanced under ultrasound guidance with continuous back pressure to the syringe plunger. Aspiration of venous blood and visualization of needle into the target vessel on ultrasound confirmed correct placement. The syringe was removed while holding the finder needle in place. The 0.0889 cm guidewire was then passed through the introducer needle. The wire position within the vein was confirmed with ultrasound visualization in both transverse and longitudinal views. A 1 cm skin incision was performed to accommodate the dilator. After removal of the finder needle, a rigid soft-tissue dilator was passed over the guidewire to a depth of 2.5 cm in a single passage to separate subcutaneous tissues and dilate the vessel wall. The dilator was removed, ensuring that the guidewire did not lose position, and the 5 Fr/15 cm single lumen catheter was then passed over the guidewire into the vessel to the hub to allow minimal external exposure. The guidewire was removed, and the catheter was attached to a three-way stopcock for medication administration. The catheter was sutured into place with an antimicrobial patch at the site of insertion. Sites were dressed in rolled gauze and clear adhesive. Locking with 1 mL 100 IU/mL heparin was performed, given the small lumen of the catheter. This procedure was completed within 15 minutes in most cases.
Femoral arterial catheter technique
Femoral arteries were identified on ultrasound between the inguinal groove and angle of the knee medially on the hind limb. Femoral artery cannulation was completed using Teleflex 20 Ga 4.5 cm Arrow® Integrated Arterial Catheters (Teleflex, Inc., Wayne, PA) to minimize risk of vasospasm. Arterial catheter placement using the self-contained kit allowed identification of needle tip in the vessel lumen and serial advancement under ultrasound guidance before passage of the guidewire. Arterial blood return was noted, and the guidewire was advanced to allow the catheter to pass into the vessel. This procedure was completed within five minutes in most cases.
Results
Six pigs were cannulated, with a 100% success rate for both jugular and femoral sites. The first two venous and arterial cannulations required a second attempt due to vasospasm and rapid clotting of the needle. When examining vessels transversely under ultrasound, venous structures were noted to have thinner walls and variable shape compared to the more circular and hyperechoic arterial walls. This method provided reliable access during the experiment and subsequent blood draws. No catheter clotting was noted with heparin locking. Animals tolerated the bandage wrap well, with no dislodgements. No insertion site infections were noted. Use of ultrasound allowed both vessels to be cannulated and catheters to be secured within 30 minutes when performed by physicians familiar with procedural ultrasound in human medicine. No cut-down for visualization was required. The self-contained arterial access kits allowed finer control and a higher success rate than described in prior studies.3,7 Arterial catheters had no significant bleeding noted after removal or dislodgement during experimental procedures.
Discussion
Ultrasound-guided vascular access in juvenile pigs is both feasible and timely. Ultrasound access was elected due to the minimally invasive approach having a smaller insult, but the same reliable vascular access though this could vary with provider experience and the need for multiple attempts or hematoma development. Jugular central venous catheterization allows protection from damage or inadvertent removal by the animal. Femoral arterial access allows easier hemostasis after catheter removal. For studies that require multi-day administration of medications or blood draws, ultrasound-guided jugular venous catheters provide reliable access. Femoral arterial cannulation is well tolerated and allows for compression should the animals require anticoagulation. The findings described here demonstrate the applicability of this approach to variously sized animals.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the support and guidance of the University of Florida Animal Care Services staff in the development of this model. This model was developed for research funded by the Society of Academic Emergency Medicine Grant (# RF2019-010) and University of Florida Department of Emergency Medicine.
