Abstract
Driven by the longer lifespans of humans, particularly in Westernised societies, and the need to know more about ‘healthy ageing’, ageing mice are being used increasingly in scientific research. Many departments and institutes involved with ageing research have developed their own systems to determine intervention points for potential refinements and to identify humane end points. Several good systems are in use, but variations between them could contribute to poor reproducibility of the science achieved. Working with scientific and regulatory communities in the UK, we have reviewed the clinical signs observed in ageing mice and developed recommendations for enhanced monitoring, behaviour assessment, husbandry and veterinary interventions. We advocate that the default time point for enhanced monitoring should be 15 months of age, unless prior information is available. Importantly, the enhanced monitoring should cause no additional harms to the animals. Where a mouse strain is well characterised, the onset of age-related enhanced monitoring may be modified based on knowledge of the onset of an expected age-related clinical sign. In progeroid models where ageing is accelerated, enhanced monitoring may need to be brought forward. Information on the background strain must be considered, as it influences the onset of age-related clinical signs. The range of ageing models currently used means that there will be no ‘one-size fits all’ solution. Increased awareness of the issues will lead to more refined and consistent husbandry of ageing mice, and application of humane end points will help to reduce the numbers of animals maintained for longer than is scientifically justified.
The study of ageing: scientific background
Human life expectancy is increasing on a global scale. In the UK, since 2000/2002, life expectancy in men has increased from 75.6 to 79.2 years, and in women it has climbed from 80.4 to 82.9 years (UK Office for National Statistics). Increased life expectancy has been accompanied by age distribution shifts, with more people now older than 65 years than younger than 15 years of age. For example, the over 65s are projected to account for 25% of the British population by 2046 (UK Office for National Statistics). 1
This rise in the proportion of elderly people has profound social and economic implications because healthspan – defined here as the period of life free from age-related disease – is not increasing at the same rate as lifespan. Age is the primary risk for many highly debilitating chronic diseases, including cardiovascular disease, Alzheimer's disease, osteoporosis, type 2 diabetes and many forms of cancer. Moreover, these age-associated diseases typically co-occur with multimorbidity, affecting an estimated 60% of people older than 65 years of age. 2 Consequently, more and more individuals will spend a greater proportion of their lives suffering poor health, and therefore there is a significant biomedical interest in advancing our understanding of the ageing process. The hope is that by identifying potential points of intervention, we can ameliorate the ageing process, thereby extending healthspan through the reduction in the incidence of and/or the delaying in the onset of age-related disease.
Considerable understanding of the ageing process has been revealed through the use of genetically tractable, relatively short-lived and easily maintained model organisms such as
To test whether a particular intervention has a beneficial impact on ageing, it needs to be studied in the context of ageing (i.e. using aged individuals). 11 One important caveat when choosing to study a particular chronological age is that profound strain and sex-specific differences exist in lifespan, healthspan and cause of death across different mouse strains. 12 In this regard, a valuable and freely available resource that provides such information for a large number of mouse strains is the Mouse Phenome Database. 13 What is clear is that there is not a ‘one-size-fits-all’ scenario. The particular age classes to be studied need to reflect the ageing profile of that mouse strain appropriately wherever possible.
Studying mice at different ages, whether that be cross-sectional approaches in different individuals or longitudinal approaches over the life course within the same individual, may be necessary to determine whether an intervention acts in an age-dependent manner or an age-independent manner. While significant insights into the ageing process have been identified using progeroid mouse models, 14 these models appear to have limitations in completely recapitulating all aspects of ageing typically seen in conventional mice over their lifespan. Therefore, understanding the fundamental mechanisms underlying ‘natural’ ageing and defining exactly how (and when) a particular intervention acts to modulate age-related traits or processes requires the use of old/aged individuals where the mice may be ‘purposefully aged’ beyond the stage that would be considered as the normal lifespan for this species in commercial breeding or scientific establishments. There are many challenges associated with maintaining cohorts of ageing mice, and the study plan needs to take account of the potential for incidental age-related losses.
The concept of frailty
Before discussing the various aspects of ageing, it is important to consider a closely associated concept: frailty.
Frailty in humans is conceptually defined as a clinically recognisable state of increased vulnerability to adverse health outcomes for people of the same chronological age as a result of ageing-associated decline across multiple physiological systems such that the ability to cope with everyday or acute stressors is severely compromised. 15 Despite much research, the biology of frailty is not well understood, and this is partly due to our inability to evaluate frailty in experimental models. Therefore, the need to capture, account for and measure frailty in our ageing mouse models is not only an ethical and welfare imperative, but also a scientific one.
Operationally, frailty in people has been defined as meeting three out of five phenotypic criteria indicating compromised energetics: low grip strength, low energy, slowed walking speed, low physical activity and unintentional weight loss. 16 The so-called frailty index (FI) goes further by assessing the number of deficits accumulated over time, including disability, diseases, physical and cognitive impairments, psychosocial risk factors and geriatric syndromes. It is proposed as a risk index and is considered a more sensitive predictor of adverse health outcomes.
Eight-item simplified mouse frailty index (FI) assessment. 18

These previous studies all show that it is potentially feasible to develop a reliable system or index for measuring frailty in ageing mice. However, we need not just reliable measures of frailty, but also non-invasive ones that are manageable and do not impose an unrealistic burden on animal care staff time and on resources. Moreover, invasive or stressful measures could easily further compromise already challenged animals by generating welfare issues that may result in a proportion of the cohorts having to be culled earlier than necessary.
Although the risk of frailty increases with age, we consider that its measurement in laboratory mice is not so much an indicator of actual chronological age but rather of the risk of adverse side effects resulting from common stressors such as handling, procedural work and interventional behaviour assessments. A manageable, simplified FI assessment incorporated into the daily or weekly welfare assessment could help predict emerging frailty within aged cohorts, signalling the need for increased monitoring of these vulnerable mice. In addition, widening discussion of frailty in relation to ageing mouse studies can help raise awareness of the need to reduce simple but common potential stressors such as handling and promoting good practice (e.g. the use of tubes or cupping rather than tails when handling mice24,25).
Clinical/phenotypic signs related to ageing
Ageing is a natural process during which structural and functional changes accumulate in an organism as a result of the passage of time and which eventually lead to death. It is a multifactorial process, acting at many levels of the organism's physiological and functional organisation, driven by genetic, epigenetic and environmental factors. 26 Precisely because of its multifaceted nature and causes, there is great heterogeneity in the ageing phenotype, even among members of the same species and even within highly inbred mouse strains. Therefore, producing a comprehensive list of ‘typical’ signs of ageing – even in a given strain – has, of necessity, various caveats and limitations. Moreover, exclusively age-related changes can be difficult to differentiate from disease, as both processes result in impairment. The main difference between the two is that the former is a normal, universal process affecting all individuals, whereas the latter is an abnormal process only affecting a subset. In human clinical practice, a further distinction is made between ageing and age-related frailty. The distinction and what it may mean in a laboratory animal setting – specifically in rodents – is explored elsewhere in this document, but some of the conclusions will necessarily overlap with those outlined in this section. Different mouse strains have different predispositions to develop one or more health conditions as they grow older, and it is critical to know them and how they may manifest themselves. 12 There are several excellent sources that can provide this sort of information and can be supplemented with additional data (e.g. Mouse Phenome Database; The ShARM resource27–30).
It is also important to realise that environmental factors can also be predictive of certain age-related conditions. For example, the generalised practice of ad libitum feeding of laboratory mice, coupled with a lack of opportunity to exercise, can predispose to obesity and obesity-related pathologies. 31 Therefore, a clear knowledge of the animals' past and present environment needs to be taken into consideration as much as possible when assessing age-related health conditions.
As ageing is universally associated with a general and progressive decline in organ systems, there are some clinical/pathological manifestations that can be considered more or less typical in a given species or strain. The welfare relevance of these various clinical/pathological manifestations will depend to a large extent on what organ(s) or system(s) are mostly affected and on how well the animal may cope or compensate for them (see the section on frailty). Manifestations such as hearing loss, impaired vision or generalised hair loss do not normally cause the same level of impairment in laboratory mice as they probably do in their wild counterparts or indeed in humans. By contrast, other manifestations such as neoplasia or degenerative joint conditions can have profound effects on the animals' welfare. As mice age, many conditions develop slowly over extended periods of time, and the body adapts to them. However, often a time is reached such that an animal suddenly shows clinical signs resulting from a decline in function that can no longer be compensated (e.g. progressive heart or kidney failure). At a cellular and tissue level, changes lead to reductions in function, such as muscle strength, epithelial renewal, alignment of the teeth and cardiovascular output. Initial signs of ageing in laboratory mice may include thinning, texture and/or colour change of the coat; reduced self-grooming; body-mass change (middle-age obesity may be followed by loss of body condition); hearing loss; deteriorating eyesight; reduced spontaneous activity and exercise intolerance. Individually, these changes seldom have a significant impact on the animals' general well-being. However, collectively, the animals' decline can eventually become noticeable and can be measured by, for example, grip strength, endurance activity, voluntary movement, speed of walking and reduced rate of wound healing.32,33 In addition to reduced function, the incidence of overtly terminal conditions (e.g. invasive or metastatic tumours) rises dramatically with advancing age.
‘Common’ signs of ageing in mice, grouped by organ/system.

Clinical signs in italics denote those considered by the authors as most commonly encountered in aged mice.
Signs of ageing in mice and recommended action.
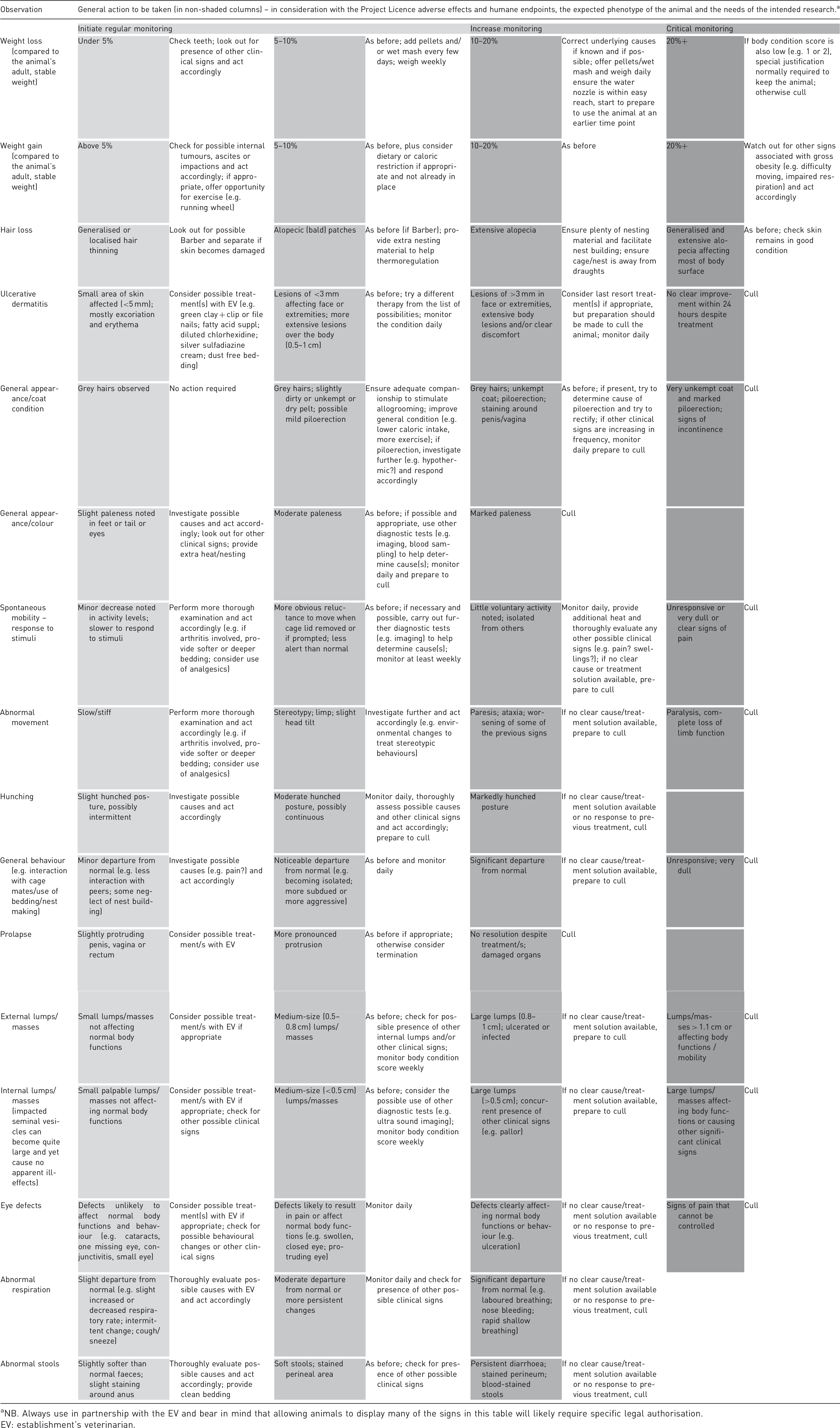
NB. Always use in partnership with the EV and bear in mind that allowing animals to display many of the signs in this table will likely require specific legal authorisation.
EV: establishment's veterinarian.
Example of an enhanced monitoring sheet

NB. Commence enhanced health check, score and weigh ageing animals (unless there is information to suggest otherwise) from 15 months of age on a fortnightly basis. This frequency should be increased if the animal gives any cause for concern. Moreover, there is some evidence to suggest that weekly monitoring after the animals' reach 20 months of age is recommended – at least in some strains – as there can be a steep downward trend in their health. After 30 months of age, it is likely that fewer animals will remain alive and that they may require daily enhanced monitoring. The above monitoring frequencies are only a broad recommendation. As you gather more information on your mouse strains, frequencies may be altered to suit your local needs. Monitoring frequency can be influenced by many factors including – but not limited to – the animals’ immediate housing environment (e.g., cage type), strain and genetic background of mice, gender, disease model and study needs. Table 3 lists a number of changes that can be associated with the ageing process, together with suggestions as to how to monitor and/or act upon them. As observed changes accumulate or increase in ‘intensity’, so the frequency of monitoring increases in proportion. It is important to emphasize that there is no perfect tabulated list of clinical signs that can cover all the various possible pathological conditions that animals may develop as they age. In addition, local practices and experience, together with any limitations that may be described in associated project authorization and study design, will necessarily affect the way these conditions are assessed and acted upon.
Depending on the knowledge of the particular mouse strain, it will be possible to design a customised monitoring regime to suit both the expected adverse effects and the scientific needs of the study. Thus, monitoring for age-related clinical signs may be combined with the potential adverse effects of the experiment, for example related to lameness and swollen joints in an experimental study of arthritis, to give a more comprehensive clinical monitoring and scoring system relevant to any individual study.
Legal considerations when using ageing rodents for scientific purposes
A final consideration when working with ageing rodents is the question of when exactly should the ‘purposeful ageing’ of rodents be considered as requiring authorisation under relevant legislation. What follows is not intended to be an authoritative text on legal requirements, since these vary between different countries and even within the same country. Instead, we aim to raise awareness that regulations exist and should be complied with wherever the research is conducted. We also attempt to describe the criteria to be considered so as to achieve a consistent approach with regard to setting the time point for commencement of enhanced monitoring and determining when legal authorisation may be required. We argue that the criteria for project authorisation cannot be based solely on the age of animals. Instead, it should be based on the potential for the animals to experience adverse effects of pain, suffering, distress or lasting harm (P, S, D or LH) above the minimum threshold, as indeed enshrined by most laws governing the use of animals in scientific research. In Europe, the relevant legislation is the Directive 2010/63/EU (EUD). 36 The EUD defines a ‘procedure’ as any use, invasive or non-invasive, of an animal for experimental or other scientific purposes, with known or unknown outcome, or educational purposes, which may cause the animal a level of P, S, D or LH equivalent to, or higher than, that caused by the introduction of a needle in accordance with good veterinary practice. Keeping animals alive, for a qualifying scientific purpose, beyond this point would require project authorisation. This is important because procedures can only be applied to a protected animal as part of a programme of work described in a project authorisation.
Where the scientific investigation relates to the physiological process of ageing, or alternatively because there is a requirement to investigate a disease mechanism or metabolism of a drug in an ageing animal, perhaps to mimic the human condition more accurately, then keeping mice beyond the age when they would normally be kept in standard efficient breeding programs will be necessary in order to achieve the scientific goals. There are currently many thousands of mice in regular use in scientific studies globally. These include non-genetically altered, wild-type, in-bred and genetically altered (GA; where the genetic alteration may have been either created experimentally or occurs naturally and is potentially expected to be associated with a harmful phenotype). Some of these are very well characterised both genetically and phenotypically, others less so, particularly the phenotypes of ageing strains. Both in EU and the UK, all mice kept for use in scientific procedures are protected animals irrespective of whether they carry a genetic alteration. In the UK, the breeding and maintenance of virtually all GA mice, irrespective of whether they have a harmful clinical phenotype associated with the genetic alteration, requires project authorisation. This is because it is assumed that all experimentally created mouse strains
What about mice that are ‘purposefully aged’? Does the ‘purposeful ageing’ of the animals, in itself, require project authorisation, as defined above? Should consideration be given to the effect – enhancement or diminishment – of the genetic alteration on the ageing process? Will they have different or additional adverse clinical effects as a consequence of the ageing process that may need to be defined in a project authorisation?
As stated above, we consider that the requirement for project authorisation of ageing animals cannot be determined solely on age but should be viewed as a performance standard when any P, S, D or LH manifests as ageing-associated clinical signs and has a detrimental impact on the animal and the animal is not immediately culled. This is consistent with the definition of a procedure for scientific purpose (see above). Given that there is already published evidence of age-related ‘watersheds’ in some mouse colonies, 37 when no information is available from establishment health records or from the published literature on a particular mouse strain or new genotype, then we consider it reasonable to propose that there should be a point for enhanced monitoring of any ageing mice. Working with the scientific and regulatory communities in the UK, comprised of a wide range of scientists and experienced animal care technologists caring for large colonies of aged mice used for different purposes, we propose that this age-related trigger point for enhanced monitoring should be 15 months. From this age onwards, mice should undergo an enhanced regime of monitoring with additional husbandry or veterinary intervention steps aimed at identifying, recording and mitigating any ageing-associated adverse clinical signs. This general recommendation does not preclude the establishment of earlier points for enhanced monitoring when this is seen as a prudent decision, such as when expected progeroid mouse models are generated or when new GA models start showing signs of frailty or ageing earlier than expected.
Where there is information available on the phenotype of the mouse and the strain is already well characterised, with details available through, for example, the Mouse Passport Scheme, 38 establishment health records or published literature, then the trigger point for enhanced monitoring and consideration given to project authorisation should coincide with the onset of any known adverse ageing-associated clinical or phenotypic sign(s). Obviously, this timing – for both enhanced monitoring and project authorisation – can vary widely, depending on the onset of expected age-related clinical or phenotypic sign(s) in mice of that particular strain and need not coincide with the 15-month trigger point mentioned above. Consideration of project authorisation will involve a harm–benefit analysis (HBA) weighing the potential ageing-related harms that the animal is expected to experience against the potential scientific benefits. Such a HBA will need to be conducted on a study-by-study basis, relying on sound information about the scientific objectives, the likelihood of achieving them, their beneficial impact, the potential welfare implications, their mitigation and so on.
In all cases, information on the background strain must be taken into account, as this is known to have an influence on the onset of age-related clinical signs. For instance, the question may be asked whether it is necessary to implement enhanced monitoring of mice from 15 months of age where no adverse phenotype is expected. We are of the opinion that if there is evidence (e.g. from health records) to show that no adverse clinical or phenotypic signs are expected, then an informed decision can be taken that there is no need for enhanced monitoring because no signs of P, S, D or LH are anticipated. For example:
It is intended to maintain a well-known and well-characterised wild-type strain of mouse with no genetic alteration for up to 24 months for a study on ageing. The evidence from many years of health records shows that these mice do not display any adverse clinical signs before 18 months of age. The researcher intends to kill the mice humanely as soon as the first clinical signs of ageing are observed. Therefore, in this case, the P, S, D or LH trigger point for enhanced monitoring would be reached when the mice became 18 months old. As the researcher does not intend to maintain the mice beyond the point where they develop adverse clinical signs, project authorisation would not be required. A GA mouse strain is already bred under project authority because it carries a genetic alteration associated with a phenotype featuring reduced lifespan. The genetic alteration is known to induce premature ageing, and the mice are expected to display age-related clinical signs from six months onwards. If the intention is to keep these animals alive beyond six months of age, then they should be subject to enhanced monitoring against a predetermined set of criteria and at an agreed frequency from just before this age, for example five months, to minimise adverse effects experienced by the mice and potentially refine the model.
Conclusions
Human lifespan is increasing globally, whereas healthspan – the period of life free from age-related diseases – is not increasing at the same rate. One consequence is the fact that we are witnessing a concerted research effort into the causes of ageing and the control of its consequences, and this global effort has resulted in a marked increase in the number of research groups that use ageing rodents – particularly mice – that are ‘purposefully aged’ beyond the stage that would be considered normal in commercial breeding or scientific establishments. Hence, there is an urgent need to ensure that the husbandry and management of these ageing animals is approached in a consistent way, thereby optimising their welfare and, at the same time, underpinning the achievement of good quality, reproducible scientific results.
An obstacle for optimising the welfare of ageing rodents is the fact that age and frailty-related health problems and their manifestations can vary significantly, depending on a range of factors, including genotype, sex, housing and environment. The interface between ageing and frailty itself is complex and not easy to tease apart. For models of accelerated ageing using GA animals, for example, it could prove particularly helpful to include the signs of frailty in any enhanced monitoring regime by combining parameters from the various tables presented. Clearly, there is a significant advantage to animal welfare through identification and application of extra vigilance for those animals identified as frail by the scoring criteria. As mentioned previously, there is no ‘one-size-fits-all’ answer, and the monitoring and interventions need to be tailored to the mouse strain, study design and scientific objectives without causing additional harms to the animals and without being too labour intensive.
We believe that a significant way forward towards the achievement of improved animal welfare and better science is the adoption of enhanced clinical monitoring of ageing mouse colonies intended for scientific use. Working with the scientific and regulatory communities in the UK, we advocate that such an enhanced monitoring regime is adopted from (a) the expected onset of one or more age-related adverse clinical signs where the mouse (wild type or GA) is well characterised, (b) an age-related time point of 15 months where no information is yet available to suggest that there will be adverse age-related clinical signs prior to this time, or (c) a combination of these parameters.
There is no provision within current EU legislation to ascribe an age-related ‘engineering’ threshold to regulation. Instead, the threshold is based on the performance standard related to the potential of any procedure – including purposeful ageing – to have the effect of causing the animal a level of pain, suffering, distress or lasting harm equivalent to, or higher than, that caused by the introduction of a needle in accordance with good veterinary practice. Accordingly, throughout this paper, we have described a trigger point for enhanced monitoring based not solely on the age of the animals, but also on the likelihood of clinical signs being evident at a particular age. This will also assist in determining whether and when legal authorisation is required for particular strains.
As a first step to addressing the enhanced monitoring of ageing animals, this paper has focused throughout on mice, as this is the most commonly used species in scientific research in Europe, accounting for 61% of all of the animals used in scientific procedures in Europe in 2017. However, the principles described herein are applicable to other species and other scientific fields. By raising awareness of the issues around the use of ageing mice and the adoption of staged, progressive monitoring systems, we aim to improve the welfare of ageing mice kept for scientific purposes and consequently refine the research models that use them.
The above monitoring frequencies are only a broad recommendation. As you gather more information on your mouse strains, frequencies may be altered to suit your local needs. Monitoring frequency can be influenced by many factors, including but not limited to the animals' immediate housing environment (e.g. cage type), strain and genetic background of mice, sex, disease model and study needs.
Table 3 lists a number of changes that can be associated with the ageing process, together with suggestions as to how to monitor and/or act upon them. As observed changes accumulate or increase in ‘intensity’, so the frequency of monitoring increases in proportion. It is important to emphasise that there is no perfect tabulated list of clinical signs that can cover all the various possible pathological conditions that animals may develop as they age. In addition, local practices and experience, together with any limitations that may be described in associated project authorisation and study design, will necessarily affect the way these conditions are assessed and acted upon.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
