Abstract
With their highly sensitive olfactory system, the behaviour and physiology of mice are not only influenced by the scents of conspecifics and other species, but also by many other chemicals in the environment. The constraints of laboratory housing limit a mouse’s capacity to avoid aversive odours that could be present in the environment. Potentially odorous items routinely used for husbandry procedures, such as sanitizing products and gloves, could be perceived by mice as aversive or attractive, and affect their behaviour, physiology and experimental results. A survey was sent to research institutions in the UK to enquire about husbandry practices that could impact on the olfactory environment of the mouse. Responses were obtained from 80 individuals working in 51 institutions. Husbandry practices varied considerably. Seventy percent of respondents reported always wearing gloves for handling mice, with nitrile being the most common glove material (94%) followed by latex (23%) and vinyl (14%). Over six different products were listed for cleaning surfaces, floors, anaesthesia and euthanasia chambers and behavioural apparatus. In all cases Trigene™ (now called Anistel™) was the most common cleaning product used (43, 41, 40 and 49%, respectively). Depending on the attribute considered, between 7 and 19% of respondents thought that cleaning products definitely, or were likely to, have strong effects on standardization, mouse health, physiology or behaviour. Understanding whether and how these odours affect mouse welfare will help to refine mouse husbandry and experimental procedures through practical recommendations, to improve the quality of life of laboratory animals and the experimental data obtained.
Non-regulated routine husbandry procedures, such as certain methods for handling1–3 cage-cleaning 4 and ear biopsies used for identification marking 5 have some impact on mouse well-being. Moreover, although UK establishments are expected to comply with the minimum provisions set out by the Home Office 6 these only govern certain aspects of husbandry, and different animal units might still differ in the way they carry out some of the tasks. Reports about the influence of laboratory environment on the outcome of behavioural genetics in mouse experiments7–9 have raised questions about which environmental factors are most relevant. 10 To help identify sources of variation that could affect results, researchers are encouraged to provide more thorough descriptions of all aspects of the experiment, including the apparatus, procedure, strain, environment and husbandry. For example, the National Centre for the 3Rs (NC3Rs) has developed the Animal Research: Reporting In-Vivo Experiments (ARRIVE) 11 guidelines that have been adopted by many journals and research funding bodies. However, perhaps due to our inherent sensory limitations as humans, the olfactory environment that mice are exposed to is generally omitted.
Although humans and mice share the same five senses, there are important differences in their perceptual sensitivity. Mice rely on the olfactory system as a major sensory modality 12 whereas humans rely more on vision and have largely lost their olfactory sensitivity. 13 Some seemingly innocuous or imperceptible olfactory cues could thus cause physiological and behavioural changes in mice, potentially confounding experimental data and/or increasing the severity of procedures. For example, toluene, an organic solvent used in many products such as paints, printing ink, rubber and disinfectants, is a potent stimulant of the trigeminal system, which functions to detect irritants and potentially noxious chemicals. In mice, exposure to toluene causes aversion, measured by a significant decrease in the time spent in an area containing this substance when compared with water. 14 Similarly, when rats are presented with a capped or uncapped permanent marker pen in a two-choice Grice aversion test, they show increased latencies, spend less time and visit less often the box containing the uncapped one, suggesting aversion to the odour or solvent released. 15
Effects of essential oils on anxiety-related behavioural tests in mice.

We carried out a survey to gain information on the diversity of the olfactory environment laboratory mice were exposed to during routine husbandry procedures. Although the survey was aimed at the UK, one response from Ireland was also included. The survey focused on the type of glove materials used to handle mice and cleaning products employed to sanitize different areas. We also asked participants specific questions about their personal experience and opinions on the subject. The results can be used to guide researchers as to the most common products currently used to aid standardization efforts in the short term, and to stimulate research into best practice over the longer term. It should be noted that, while the focus of this questionnaire was on potential olfactory effects on mice, each product could affect mice in other ways too, e.g. toxicity, psychoactive effects, or tactile effects.
Materials and methods
The survey was first launched in February 2012. A request to complete the questionnaire was sent by email to individuals involved in laboratory animal work in the UK. These contacts were obtained through the professional network of the Royal Veterinary College’s (RVC’s) Named Veterinary Group. A second round was sent in May 2012 to maximize UK coverage, and the survey officially closed in June 2012. During the second round, the survey was distributed using specialist mailing lists (Vets on Line [VOLE] and Institute of Animal Technology [IAT]) and it was advertised in the Laboratory Animal Science Association (LASA) Spring Forum magazine. To safeguard anonymity no personal details were asked and respondents were given the option not to disclose the name of their organization.
The questionnaire was created using Survey Gizmo (www.surveygizmo.com), an on-line application that allowed respondents to enter free text and/or to select predetermined answers from lists. A pilot run of the questionnaire was completed by colleagues with knowledge on the field (veterinarians and animal technicians), and their feedback was used to improve its design before the survey was launched.
The survey consisted of 34 questions covering five main topics: glove use, cleaning products, other animals (covered in López-Salesansky et al. submitted to this journal), staff policies and personal opinions. A full copy of the survey can be found as supplementary material in Lopez-Salesansky et al. (submitted to this journal). There were 23 multiple-choice questions and 11 open questions. Open questions were aimed at providing further details on multiple-choice questions or were used to leave an opinion or a comment. The language of the questionnaire was English. The questions of relevance to sources of non-animal scents included:
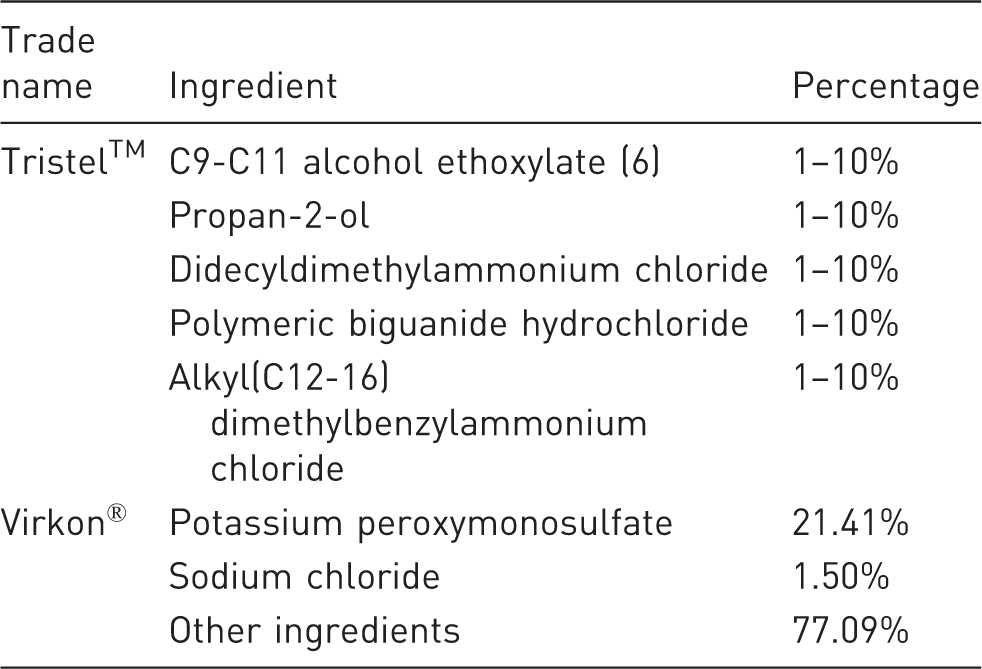
Demographic information including role of the respondent, type of facility, and type of rodent caging. Whether mice were handled with gloves and what glove material was used. Whether gloves of different materials left a smell on human hands after use. What products were used for washing hands in their facilities. What cleaning products were used to clean mouse cages, surfaces/floors, anaesthesia/euthanasia chambers, behavioural apparatus and surgical equipment after each mouse and at the end of the day. The particular products inquired about were Virkon®, Trigene™, alcohol, iodine and chlorhexidine, with free text for other products (see Table 2 for composition information). Whether they thought that any products (used currently, or in the past) might adversely affect mice, data quality, or human workers. Whether there were policies in their place of work regulating the use of perfumes and deodorants or personal hygiene products. What perfumes and deodorants they knew of that were used by people working in the facility. How frequently gowns were washed and with what product. Respondents’ opinions on the relative importance of odours from cleaning products used to wash mouse cages and specialist equipment with respect to standardization, mouse health and physiology, and mouse behaviour. Composition/information of cleaning product ingredients.
Ethical approval for the survey was granted by the RVC Ethics and Welfare Committee (URN 2012 0052H).
Statistical analysis
Descriptive analysis of multiple-answer, binary and scale questions was done through frequency distribution descriptive statistics using Microsoft Excel.
When the survey was returned partially completed, all questions that were answered were included in the analysis. If more than one individual responded from the same institution (anonymized but distinguishable from each other by internet provider [IP] addresses), the answers were compared by eye and, if the information provided was clearly different, they were used separately in the analysis, because each institution may have more than one mouse unit. Only one response was discarded due to likely duplication.
Results
Demographics
Although 57/80 respondents reached the end of the questionnaire, questions were not compulsory and some of them failed to answer all of them, with some respondents skipping certain questions even if they reached the end. Therefore a maximum of 80 responses to each question were obtained from 51 different animal institutions within the UK. Responses from non-UK institutions were discarded, except for one response from Ireland. Named animal care and welfare officers (NACWOs) and unit managers provided most of the responses, summing up to 70% (50 and 19%, respectively), followed by animal unit staff (13%), named veterinary surgeons (11%) and scientists (7%). The age of respondents was mainly between 35 and 54 years old (70%) and both genders were almost equally represented (females 54%, males 46%). The most common (63%) type of organization surveyed was academic research institutions.
Gloves and hand washing
Most (70%) of respondents reported always wearing gloves when handling mice. The remaining 30% occasionally used bare hands, and one out of the 69 used forceps.
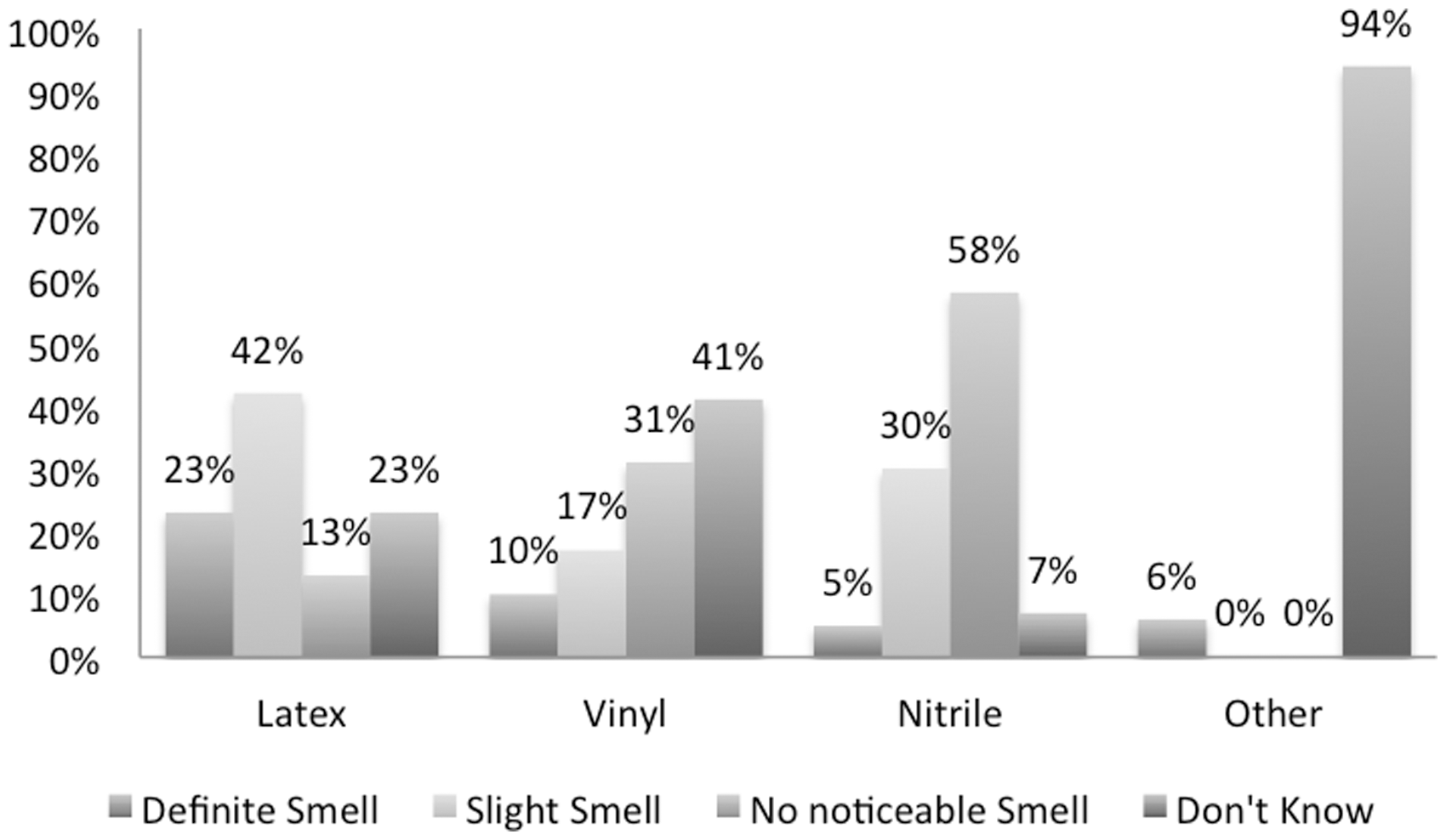
The most common glove material used for mouse handling was nitrile (94%) (Figure 1) and 25/80 participants used more than one type of glove in the same institution. Participants reported that latex gloves seemed to leave the most noticeable smell on their hands compared with other materials, with 23% and 42% of respondents reporting a definite smell and a slight smell, respectively (Figure 2).
Glove use. Type of glove used, each slice is labelled as the glove type, followed by the percentage of respondents using it. Glove smell. Respondent’s perception of a residual hand smell with the use of different types of gloves.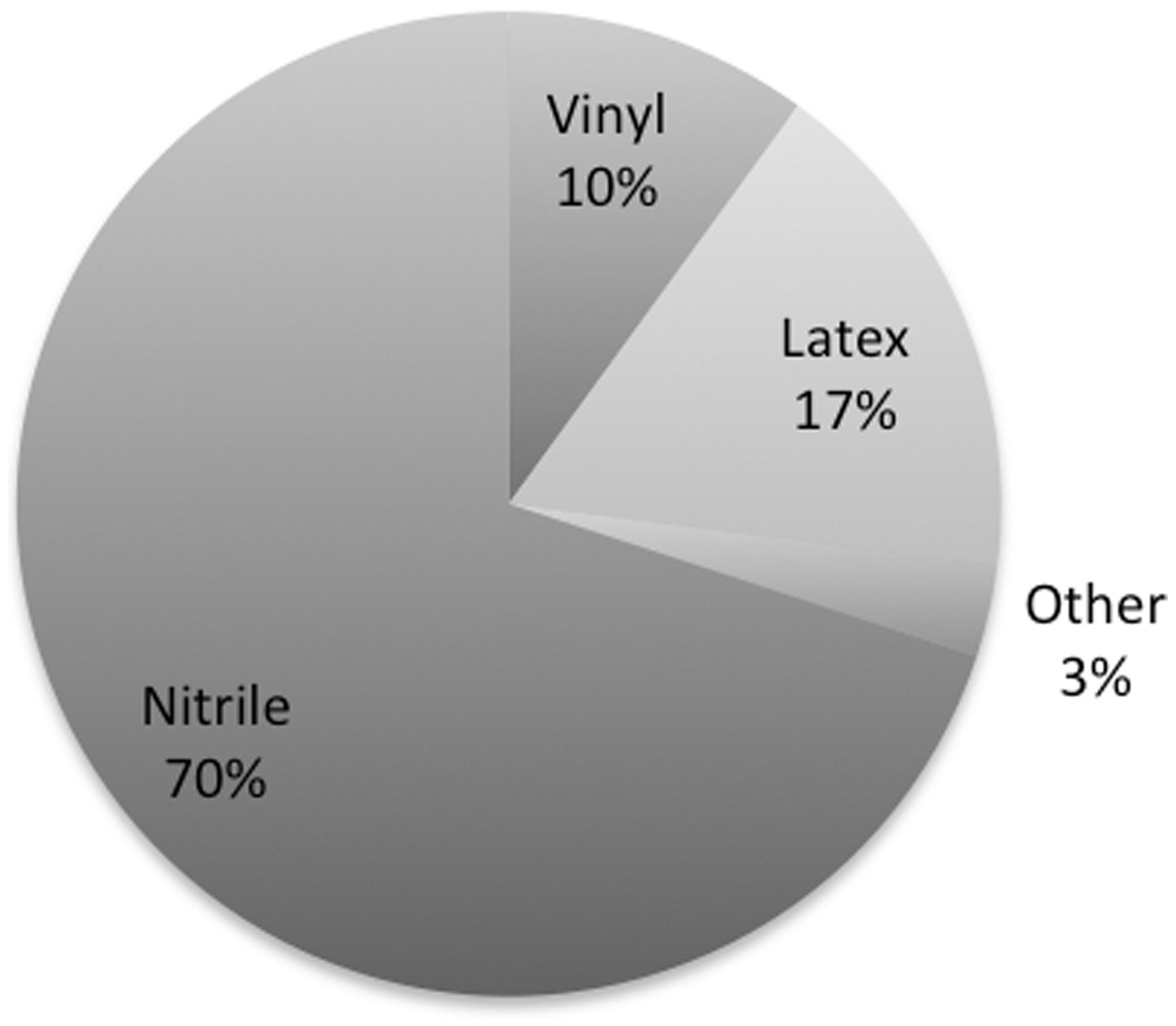
There was a high variation in hand washing practices, with 40/76 of respondents using more than one product for sanitizing their hands. Both antibacterial products and soaps were used either on their own, or in combination. The general term ‘soap’ was used by 35/76 respondents whereas 39 respondents provided a specific commercial brand including Carex™ (9), Deb (9), NewGenn™ (6), Gojo® (5) and Purell (4). Some of these brands have standard soap and antibacterial varieties, so it was not possible to determine to which product they were referring. With regards to antibacterial use, 18/76 respondents specified using Hibiscrub™ (chlorhexidine), 9/76 specified using alcohol and 10/76 did not provide a specific name. Thus over seven different hand sanitizers were named.
Cleaning practices
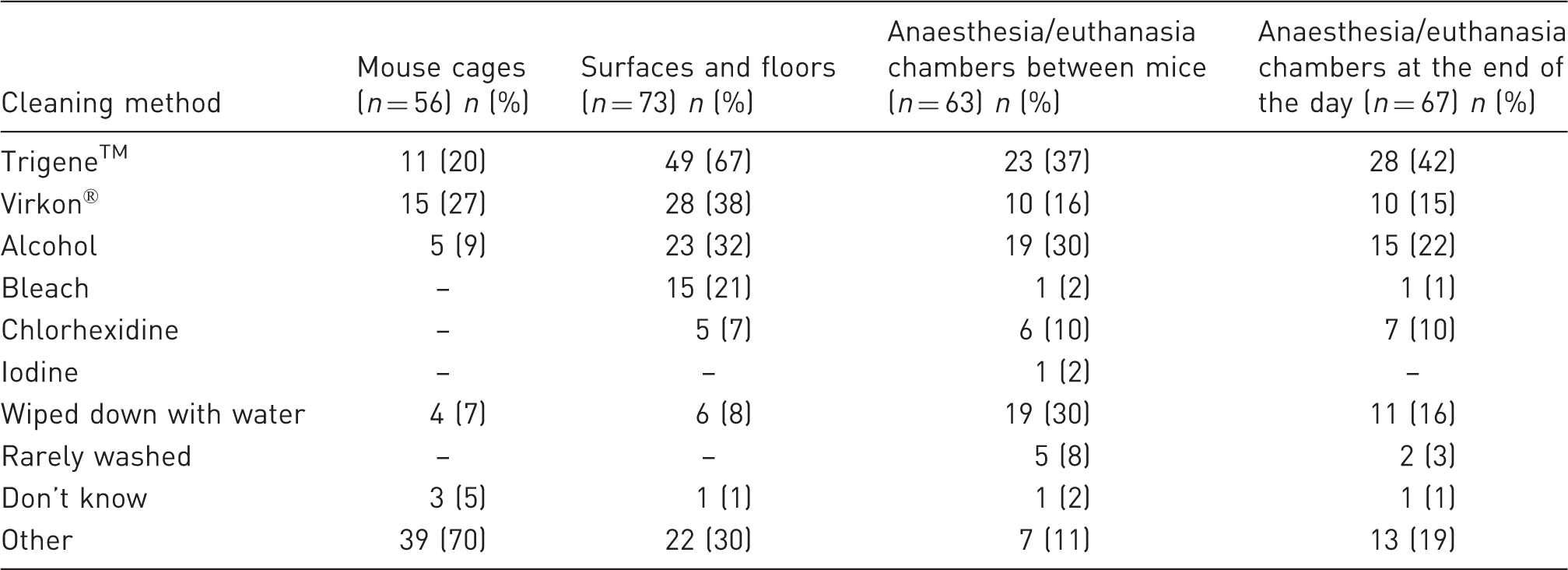
Method for cleaning mouse cages, surfaces and floors and anaesthesia/euthanasia chambers (between each mouse and at the end of the day).
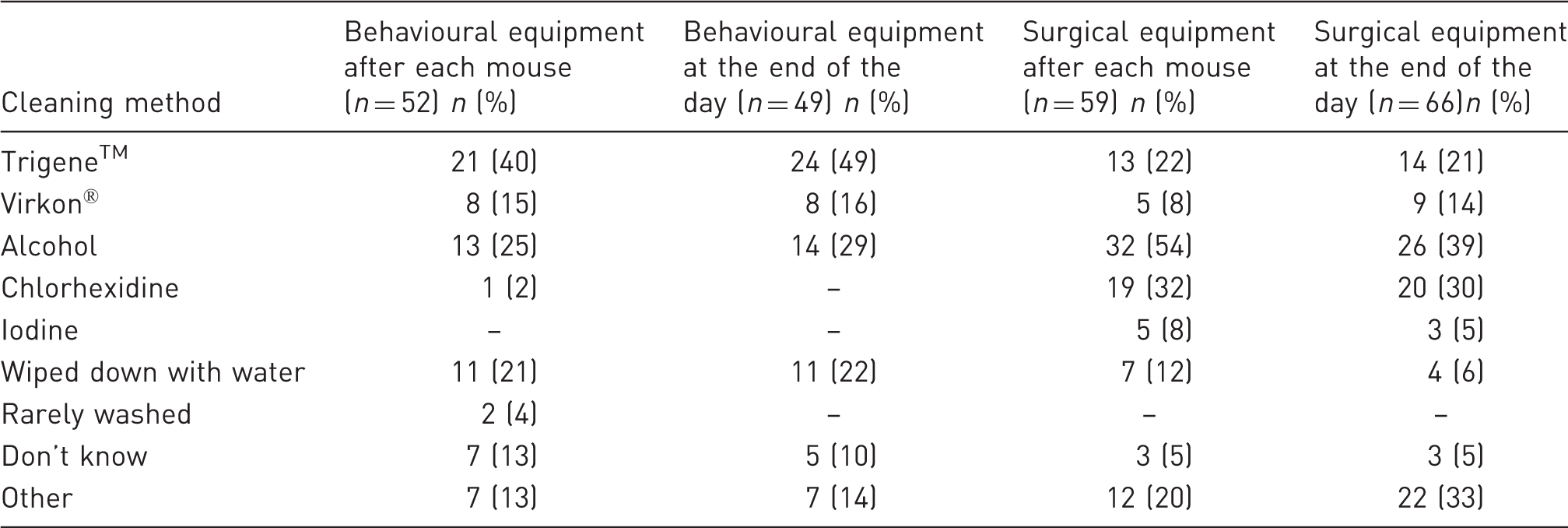
Method for cleaning behavioural and surgical equipment (between each mouse and at the end of the day).
Most (70%) of the respondents selected the option ‘other’ for cleaning mouse cages. In the free text, they explained that cages were put through the cage washer or washed with a specific cage washer product.
Although Trigene™ (renamed Anistel™ in April 2012) was by far the most commonly employed cleaner for surfaces and floors (67%), various commercial and off-the-shelf cleaning products were also used for this purpose, including Terminator™ one-step disinfectant (1), Sanifex™ (2), (3) Flash™ , Novacross™ (1), ‘generic pine’ (1), Grime-go™ (2) and Super Q™ (2). Thus, in total 13 different products were listed for cleaning surfaces and floors (Table 3).
Trigene™ was again the product most frequently used to sanitize anaesthesia and euthanasia chambers (37% and 42%, respectively), and to sanitize behavioural apparatuses between each mouse and at the end of the day (40% and 49%, respectively).
Alcohol was the main product used to clean surgical equipment between each mouse (54%) and at the end of the day (39%); and between 20% and 23% of respondents selected the option ‘other’ for cleaning this type of material, specifying in the free text that washing, autoclaving and hot bead sterilizers were the methods used to sanitize these items.
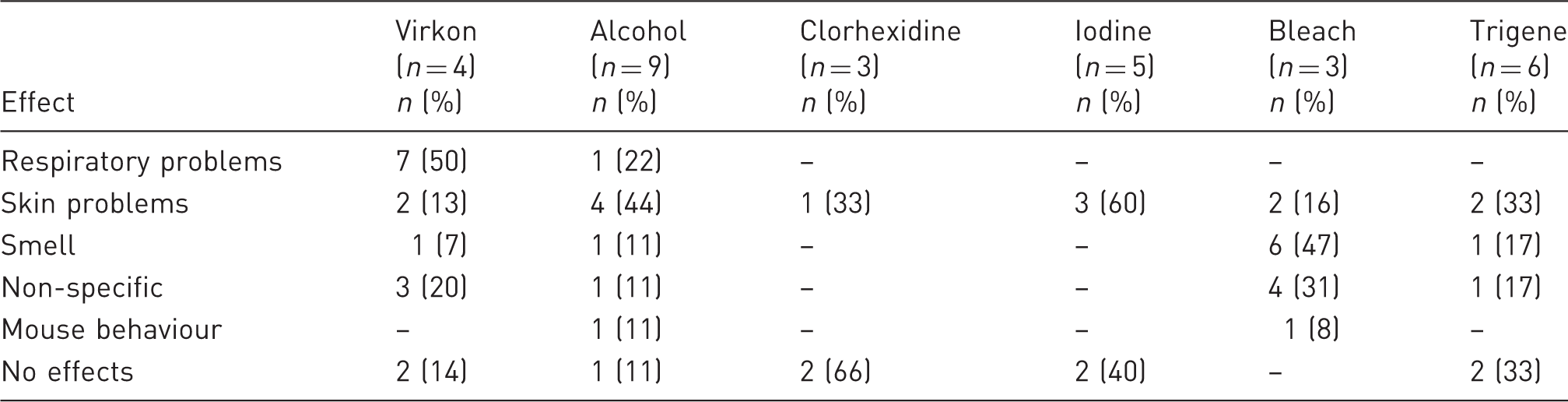
Opinion of respondents regarding the adverse effect of products on mice, data quality, or human workers.
None of the survey participants reported the use of air freshener in the animal facilities.
Staff policies
Half (7/14) of the respondents providing information regarding the existence of in-house policies for staff reported the existence of restrictions on the use of perfumes. The policy discouraged strong perfumes (2), did not allow them (4), or required consistency of the brand for neurobehavioural studies (1). Participants (4/14) also reported restrictions on the use of deodorants or personal hygiene products. In this case, original source products were not allowed (1), and unscented/‘not smelly’ products were encouraged (2), and in one case consistency of the brand was required for neurobehavioural studies. Across all 14 respondents various common brands available commercially were listed as hygiene products used by staff, including among others, Sure™, Sanex™, Dove™, Lynx™, Impulse™ and Mitchum™.
Gowns were most commonly washed weekly (20/63) or after each use (16/63), and 7/63 reported washing them whenever they were found to be dirty (Figure 3). When asked about the product used to wash gowns, Persil™ (11/56) and Ariel™ (6/56) were the two main commercial products used, but other brands were also reported. Additionally, 10/56 did not know what product was used because gowns were sent to an external laundry for washing.
Gown use. Frequency of gown washing.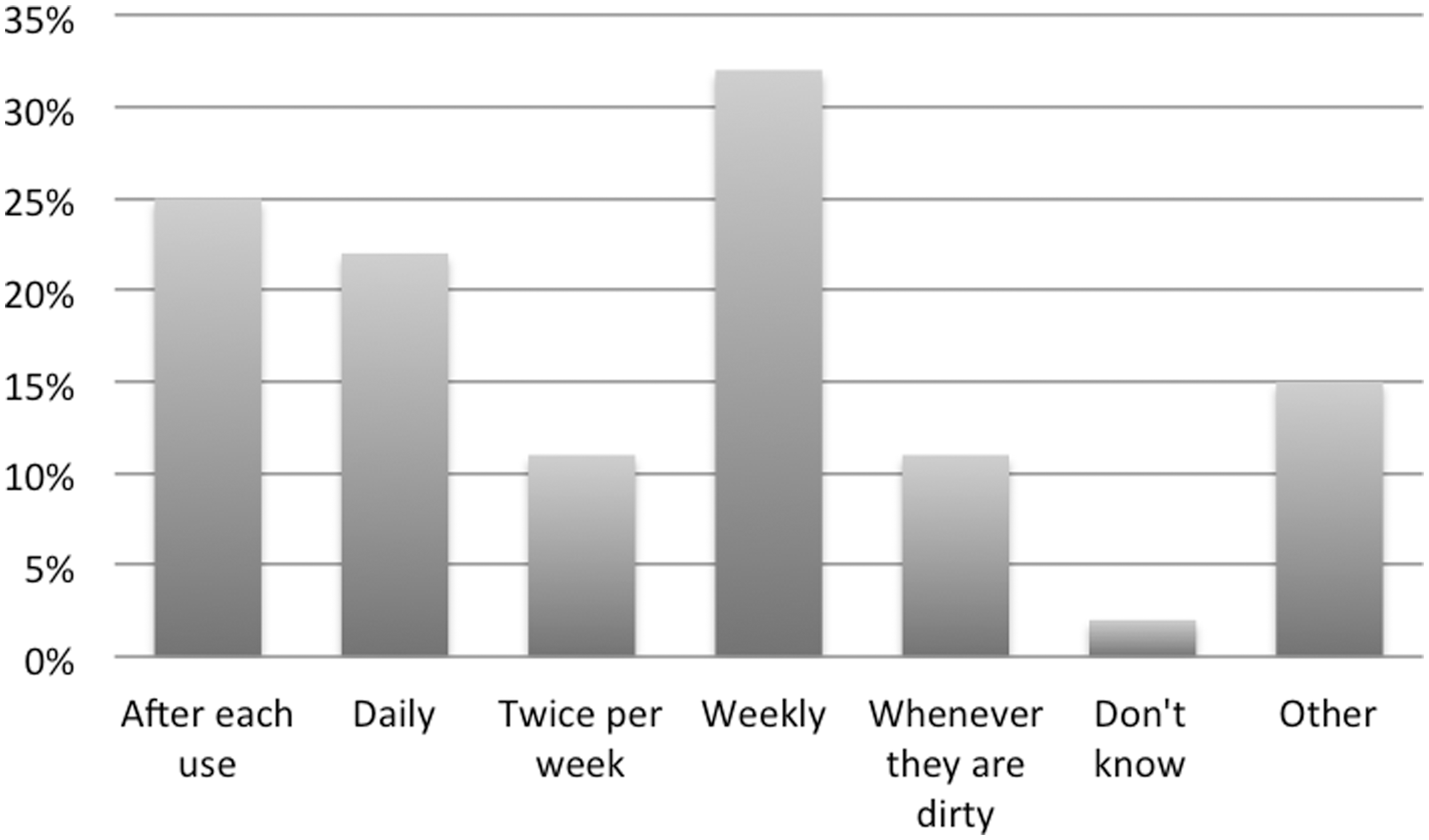
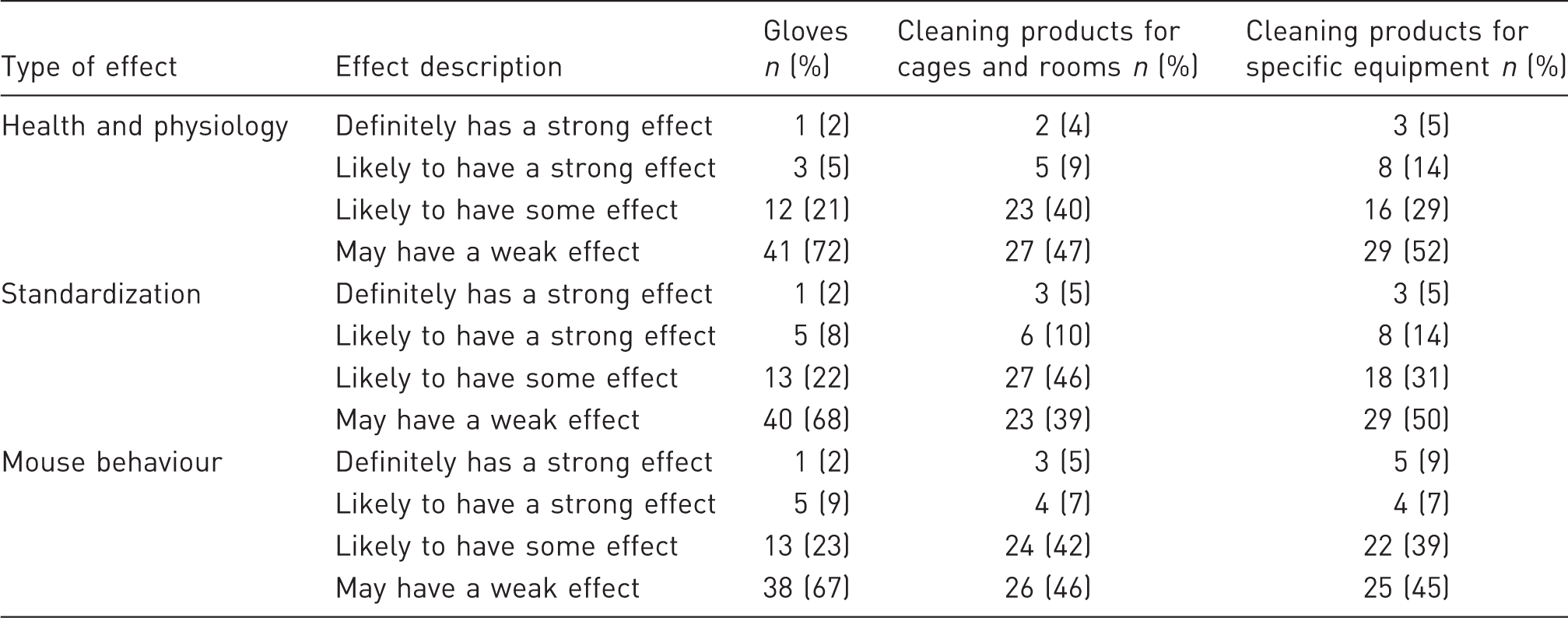
Opinion of respondents regarding the effect that gloves and cleaning products might have on mouse health and physiology, standardization and mouse behaviour.
Discussion
The results of the survey show a large variation in the way husbandry procedures are carried out across animal units in the UK. The variation is likely to be even greater internationally, as different commercial products are available in different countries. The survey also allowed people closely involved in the care of laboratory animals to raise any concerns about how certain practices could affect mice and staff members, and showed that opinions varied widely. This reveals a data deficit concerning aspects of husbandry with potential for improved standardization and refinements.
Although the majority of respondents wore gloves for handling mice, there was a large proportion (30%) who also used bare hands. This was a surprising finding, and is mainly due to the usual requirement to use personal protective equipment (PPE) imposed by health and safety regulations to prevent the development of laboratory animal allergies. 22 Consequently, within the same units, animals might not only be exposed to the smell of different types of glove materials determined by the handler’s preference, but also to the scent of the handlers themselves, as well as the hand sanitizing products used to wash hands or gloves. For example, it is common practice to avoid the transfer of pathogens between individually-ventilated cages (IVCs) by disinfecting gloves (and surfaces) between cages under the laminar flow cabinet. This is generally carried out with alcohol-based sprays or rubs or other disinfecting products. Consequently, the inherent and acquired scents of the handlers could explain differences in results obtained when different handlers carry out the same experiments. 23 Sorge et al, demonstrated that olfactory exposure to male handlers or their scents induces physiological (increased plasma corticosterone, hyperthermia and decrease in Fos protein-positive neurons) and behavioural (reduced facial grimacing and nocifensive behaviour, increased defecation, and increased thigmotaxis in the open field) changes that suggest stress-induced analgesia and increased anxiety. 24 However the impact of handling mice with or without gloves has not yet been tested.
Cleaning of mouse cages was mainly through the cage washer, using available products sold by the cage manufacturers. Again, there may be variations in these products that could not be identified here. Mouse cages provide the most immediate and unavoidable environment for the mouse, and the scent profile of the detergent used could have a chronic behavioural and physiological impact on the animal. The same could be said about the cleaning practices of all the other areas of the animal unit, although more remote. Surfaces and floors are sometimes cleaned with products that are reported as strong smelling to humans, and with commercially available scented products. Although animals in IVCs might be less exposed to these odours, they are directly exposed to disinfectants used to clean surfaces to avoid cross contamination between cages.
The most common product used for surface and equipment cleaning was Trigene™ (that changed its name to Anistel™ in 2012, maintaining the same formulation). This product is available in a 500 mL ready to use spray with a lavender scent, or in a 5 L concentrated formulation that can be ‘unscented’ or have an apple, lavender, eucalyptus or citrus scent. 25 Unfortunately, the survey did not question whether the Trigene™ formulation was unscented or which type of scent was used. This leaves the possibility that variation between animal units was even greater than described here. It also makes standardization to the most common product difficult; if a diversity of Anistel™ formulations is used, then the single most common products could actually be Virkon for surfaces and floors, or alcohol for behavioural equipment.
The method used for cleaning anaesthesia and euthanasia chambers could have an especially important impact on the welfare of the mouse being subsequently anaesthetized or euthanized. Because mice can detect alarm odours from stressed individuals, 26 it is possible that wiping down the chamber with the wrong product between mice, might not adequately remove these olfactory cues, possibly increasing the amount of anaesthetic required and making the experience potentially more harmful for the animal. Moreover, 13/67 people reported either wiping down with water (11) or rarely washing (2) anaesthesia and euthanasia chambers.
On the other hand, it is possible that cleaning these chambers with alcohol could have an impact on the pharmacology of anaesthetic agents and on the speed of euthanasia. Alcohol acts as a central nervous system depressant in a similar way to anaesthetic drugs. 27 In an experiment where rats were given an intraperitoneal injection of alcohol before inhalation anaesthesia with halothane, the concentration of the anaesthetic was reduced by 50% at the onset of anaesthesia and by 20% when cardiac arrest occurred. 28 A similar effect was found in mice, with a reduction of up to 70% in the anaesthetic required for surgical anaesthesia at the highest intraperitoneal dose of alcohol (4 mg/kg). 29 Additionally, both studies found an increased tolerance to anaesthesia when alcohol was ingested chronically. It should be noted, however, that the concentrations of alcohol inhaled from a wiped surface are likely to be much lower than those administered in those studies.
When mice are used in behavioural experiments it is desirable that the behaviours displayed are not misleadingly affected by extraneous cues in their environment, so as to improve replicability. As with anaesthesia and euthanasia chambers, different products were used to clean behavioural equipment between mice and at the end of the day, but Trigene™, alcohol and water were mainly used. Mouse models of alcoholism have shown a variety of behavioural consequences to its acute or chronic administration and to its withdrawal, including increased aggression 30 and memory deficits. 31 Although the concentrations that might be inhaled following handling or equipment cleaning will be in orders of magnitude less than in the above studies, they may still lead to subtle variations in behaviour that have not been studied.
Out of 20 responses, six participants indicated that their place of work had a policy restricting the use of perfumes and four indicated a restriction in personal hygiene products. Approximately 30 different brands of perfumes and deodorants were used, all of which have very different olfactory profiles. Most of these products are formulated with plant EOs, reported as having various effects on rodent physiology and behaviour18,32–36 (Table 5). Regulating the use of personal hygiene products might prove particularly challenging, due to the different preferences of people, and little is known on the particular effects that they could have in an animal unit.
In summary, the survey revealed that the olfactory environment of the laboratory mouse is highly variable between institutions. The use of different cleaning products and the variability of husbandry procedures could impact mouse welfare and influence mouse behaviour, potentially affecting the scientific data obtained. Standardized husbandry practices will help refine experimental protocols reducing variability and consequently the number of animals. Further research is required to develop guidelines for best husbandry practices but, in the meantime, it will be important for researchers to report any potentially relevant details in their publications as these could affect the interpretation of their results.
Footnotes
Acknowledgements
We are grateful to all the respondents who participated in this survey. Thank you also to my colleagues at the Named Veterinary Surgeons Department, who gave constructive comments on the pilot versions of the survey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NL-S was supported by a Went Scholarship at the RVC.
