Abstract
Olfaction plays a crucial role in mouse communication, providing information about genetic identity, physiological status of conspecifics and alerting mice to potential predators. Scents of animal origin can trigger physiological and behavioural responses that could affect experimental responses and impact positively or negatively on mouse welfare. Additionally, differing olfactory profiles could help explain variation in results between laboratories. A survey was sent to animal research units in the UK to investigate potential transfer of scents of animal origin during routine husbandry procedures, and responses were obtained from animal care workers and researchers using mice in 51 institutions. The results reveal great diversity between animal units regarding the relevant husbandry routines covered. Most [71%] reported housing non-breeding male and female mice in the same room, with 76% reporting that hands were not washed and gloves not changed between handling male and female mice. The most commonly reported species housed in the same facility as mice was the rat (91%), and 41% of respondents were aware that scents from rats could affect mice. Changing of gloves between handling mice and other species was reported by 79% of respondents. Depending on the aspect considered, between 18 and 33% of respondents believed human and non-human animal odours would strongly affect mouse physiology, behaviour or standardization, while approximately 32–54% believed these effects would be weak. This indicates uncertainty regarding the significance of these factors. Understanding and controlling these practices could reduce unwanted variability in experimental results and maximize welfare.
Mice have adapted to cope with a dangerous world. In nature they compete for resources with their own kind (conspecifics) and avoid being predated by other species, relying mainly on olfaction to do so.1,2
Mice use olfactory cues in deciding whether or not to interact with the signaller, for avoiding predators or seeking a mate; transfer of scents may also occur in the laboratory setting, where multiple species, or both genders of mice are housed in the same building. Transfer of animal-derived scents is likely to occur in shared areas and through staff members. Odours from previous animals can linger on anaesthetic, behavioural and other equipment and influence animals that are subsequently exposed.
Scents provided by a conspecific are called pheromones 3 and can trigger physiological reactions that will depend on the identity of the signaller. For example, territory marking by urine is known to provide both non-variable genomic information like species, sex and identity, and metabolic information such as health, social hierarchy and nutritional and reproductive status. 4 These endocrine and behavioural changes can be mediated via non-volatile and volatile metabolites.5–8
Social signals are produced by both males and females and produce effects in both genders. Male urine may elicit aggression, fleeing behaviour or submission in the recipient male depending on its position in the social hierarchy 9 (but less so if the males are of the same inbred strain as each other 10 ). In females, male urine induces changes in reproductive physiology such as the oestrus synchronization or ‘Whitten Effect’.11–13 Female urine can initiate fights in group-housed male mice,14,15and suppress oestrus in other females in a phenomenon known as ‘the Lee-Boot Effect’. 16
Species-specific odours called ‘kairomones’ derived, for example, from the fur, urine, faeces and anal gland secretions of other species, 17 enable predators and prey to detect each other at a distance. Laboratory-bred mice retain the capacity to detect predators to which they have never been exposed. 18 Exposure to predator scents induces neuroendocrine changes that affect patterns of activity (e.g. freeze or flee), decrease feeding rate and hamper reproductive performance. 19 This phenomenon is even used in modelling human emotional disorders such as anxiety and panic.20–22
Interaction with humans may also affect the mouse’s behaviour, immune response and physiology, according to the nature of the contact. The identity, including gender, of the handler is reported to produce different effects23–26 and stressful situations (handling or painful procedures) prompt mice to produce urine containing alarm pheromones (APs) that can be detected by other individuals of both sexes. 27 Exposure to APs has been linked to an inhibition of the immune system and produces active behaviours (e.g. increased locomotion, air sampling) similar to those caused by predators.27–32
The olfactory environment specific to the experimental situation could explain some of the inconsistency of laboratory results.33,34 Scents that are idiosyncratic to a particular place or person could be a source of systematic error that could reduce external validity of the results. Additionally, variations in the olfactory environment could increase random errors requiring larger sample sizes in order to find significant results. With this in mind, husbandry procedures should be designed to minimize the transfer of pheromones and kairomones to mice by adequate changes in handling routine and personal protective equipment (PPE).
In summary, conspecific and allospecific odours can have behavioural and physiological effects on mice, but little is known about the variability and propensity for these effects to occur across laboratories. We therefore carried out a UK-wide survey of animal units to learn about routine husbandry practices with the potential to transfer animal scents.
Materials and methods
The questionnaire was emailed to individuals involved in laboratory animal work in the UK in February 2012. These contacts were obtained through the professional network of the Royal Veterinary College (RVC) Named Veterinary Surgeons Group. A second round was sent in May 2012 to maximize UK coverage and the survey closed in June 2012. During the second round, the survey was distributed using specialist mailing lists (Vets on Line [VOLE] and Institute of Animal Technology [IAT]) and it was advertised in the Laboratory Animal Science Association (LASA) Spring Forum magazine. To safeguard anonymity no personal details were asked and respondents were given the option not to disclose the name of their organization. A cover letter accompanied the questionnaire communicating the purpose of the study and providing instructions on how to complete the survey. Participants were also reassured that anonymity would be observed for individuals and for institutions.
The questionnaire was created using Survey Gizmo (www.surveygizmo.com), an online application that allowed respondents to enter free text and to select a predetermined answer from a list. Colleagues with knowledge of the field (veterinarians and animal technicians) completed a pilot run of the questionnaire and their feedback was used to improve its design.
The survey consisted of 34 questions covering five main topics: glove use and cleaning products,35 other animals, staff policies and personal opinions (Supplementary material S1, see http://lan.sagepub.com). There were 23 multiple-choice questions and 11 open questions. Open questions were intended to provide further details on multiple-choice questions, or were used to leave an opinion or a comment. The language of the questionnaire was English. The questions relevant to the current study covered:
Demographic information: role, age and sex of the respondent, type of facility, and type of rodent caging. If strains or sexes were kept separately from other mice, and if so, why. Whether gloves, gowns or clothes were changed, and/or hands washed between mice of opposite sexes, and/or between handling other species and mice. Whether anaesthesia chambers or behavioural apparatus were cleaned between individual mice and at the end of the day. Whether respondents had noticed any effects of male mouse odour on female mice, or vice versa. What species were housed in the same room as mice, or elsewhere in the facility. Which species the respondents believed affected mice (giving details of effects). Ownership of pets by staff members. Respondents’ opinions on the relative importance of odours from other mice or other species with respect to standardization, mouse health and physiology, and mouse behaviour.
Ethical approval for the survey was granted by the RVC Ethics and Welfare Committee (URN 2012 0052H).
Statistical analysis
Most data were analysed using descriptive statistics. Respondent beliefs about the importance of odour influences on mice were scored between 0 (odours perceived as likely to have little effect on all four factors asked about) and 16 (odours perceived as definitely affecting all four factors). Respondent actions were also scored by totalling the numbers of possible husbandry procedures enquired about that were carried out to potentially avoid scent transfer. Spearman rank correlations were then used to investigate relationships between beliefs, actions and certain respondent demographics.
Results
Demographics
There were a maximum of 80 responses to any one question, with 23 respondents dropping out before reaching the end of the questionnaire and 57 respondents going through all the questions but, as questions were not compulsory, they did not necessarily answer them all. Only 41 respondents volunteered their institution name. However, Survey Gizmo unexpectedly provided the internet provider (IP) addresses for the respondents, which were solely used to verify the independence of responses, and helping to exclude duplicate responses; this revealed that the 80 usable responses originated from 51 institutions (15 of which apparently had more than one mouse unit and thus generated two or more independent, noticeably different responses). Most respondents (50%) were named animal care and welfare officers, followed by unit managers (19%), animal unit staff (13%), named veterinary surgeons (11%) and scientists (7%). The highest represented type of establishment was academic research institutions (63%), followed by government scientific research institutes (20%), pharmaceutical industries (14%) and contract research organizations (4%).
Sources of conspecific odours
The majority (71%) of respondents indicated that non-breeding male and female mice were housed in the same room as each other in their animal facility. Although 70% of respondents always used gloves when handling mice and 30% occasionally using bare hands, the majority 76% did not wash their hands or change their gloves between handling males and females (Figure 1). Among the 21% of respondents that took some precautions (Figure 1), four specified that they sanitized their gloves between animals with either alcohol (1), Trigene™ (1), hand foam (1) or an unspecified product (1). Although 45% and 35% of respondents reported noticing an effect of female mice on male mice and vice versa respectively, there was no significant correlation between these beliefs and the above practices ( Number of respondents that wash hands or change gloves between working with male and female mice. The number of responses is given on the top of each bar (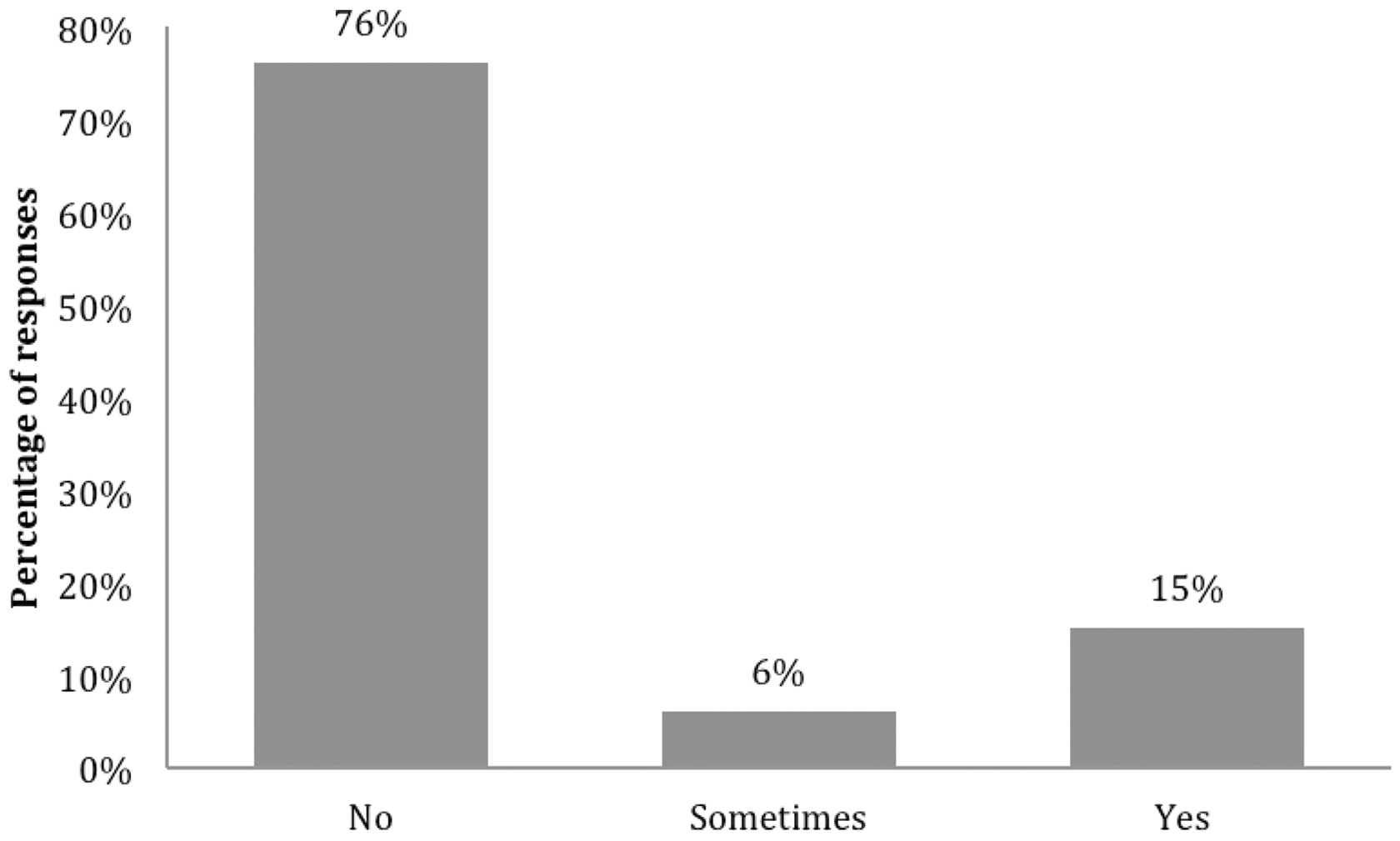
The most common (49%) caging system was a combination of barrier cages (including individually-ventilated cages [IVCs], isolators and filter tops) and open cages, but 33% and 17% reported using solely barrier cages or open cages, respectively. When using anaesthesia or euthanasia chambers, 65% of respondents used a product to clean the equipment between each mouse and 30% wiped them down with water and 8% rarely cleaned them.35
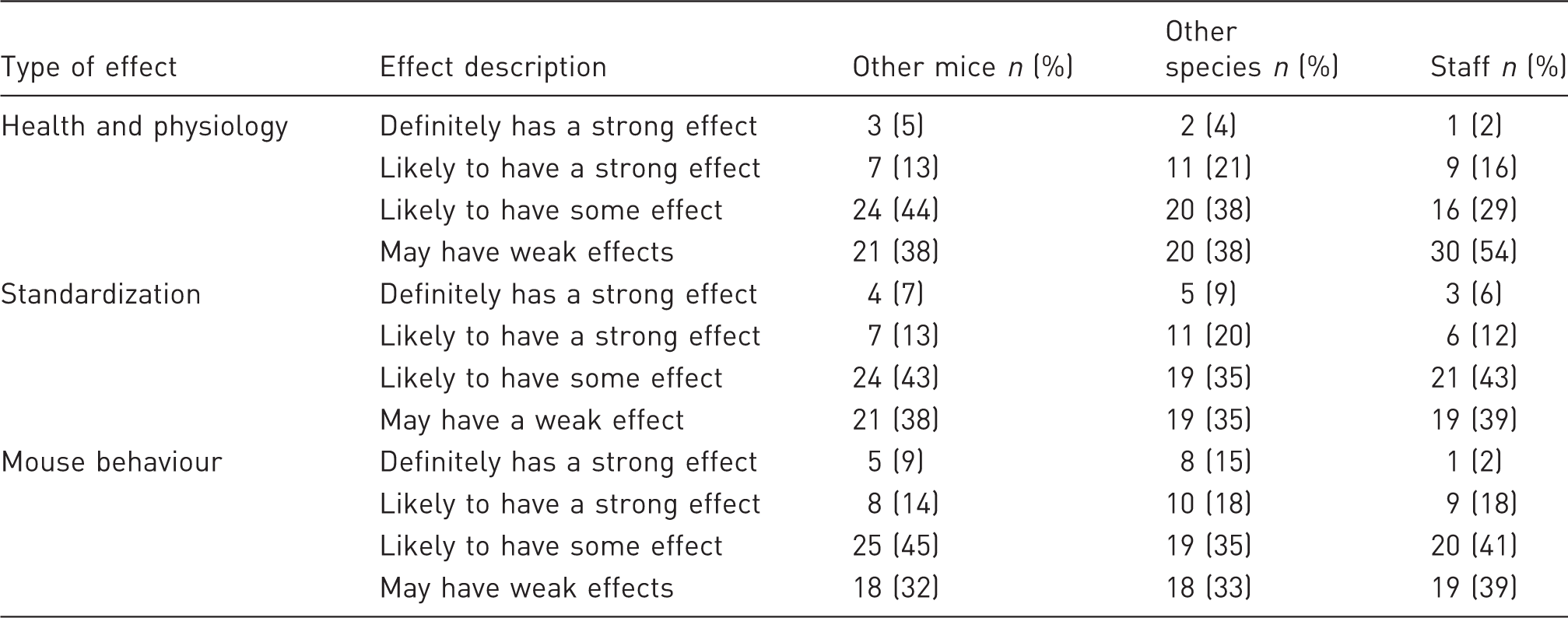
Opinion of respondents regarding the effect of other mice, other species and staff on mouse health and physiology, standardization of experiments and mouse behaviour.
Sources of allospecific odours
Fifty-four respondents from 40 different institutions reported housing some predatory species in the same facility as mice, with rats being the most common (41) followed by ferrets (9), primates (6), cats (5) and dogs (3) (Figure 2). When participants were requested to provide their opinion about which species in their facility could produce odours that might affect mice, 12/29 (41%) suggested rats, 5/29 (17%) ferrets and 3/29 (10%) humans. Other responses were cats (1), ‘predators’ (1), other mice (3), primates (1) and ‘miscellaneous smells’ (3).
Responses indicating what species are housed in the same facility as mice. The number of responses for species is given on top of each bar (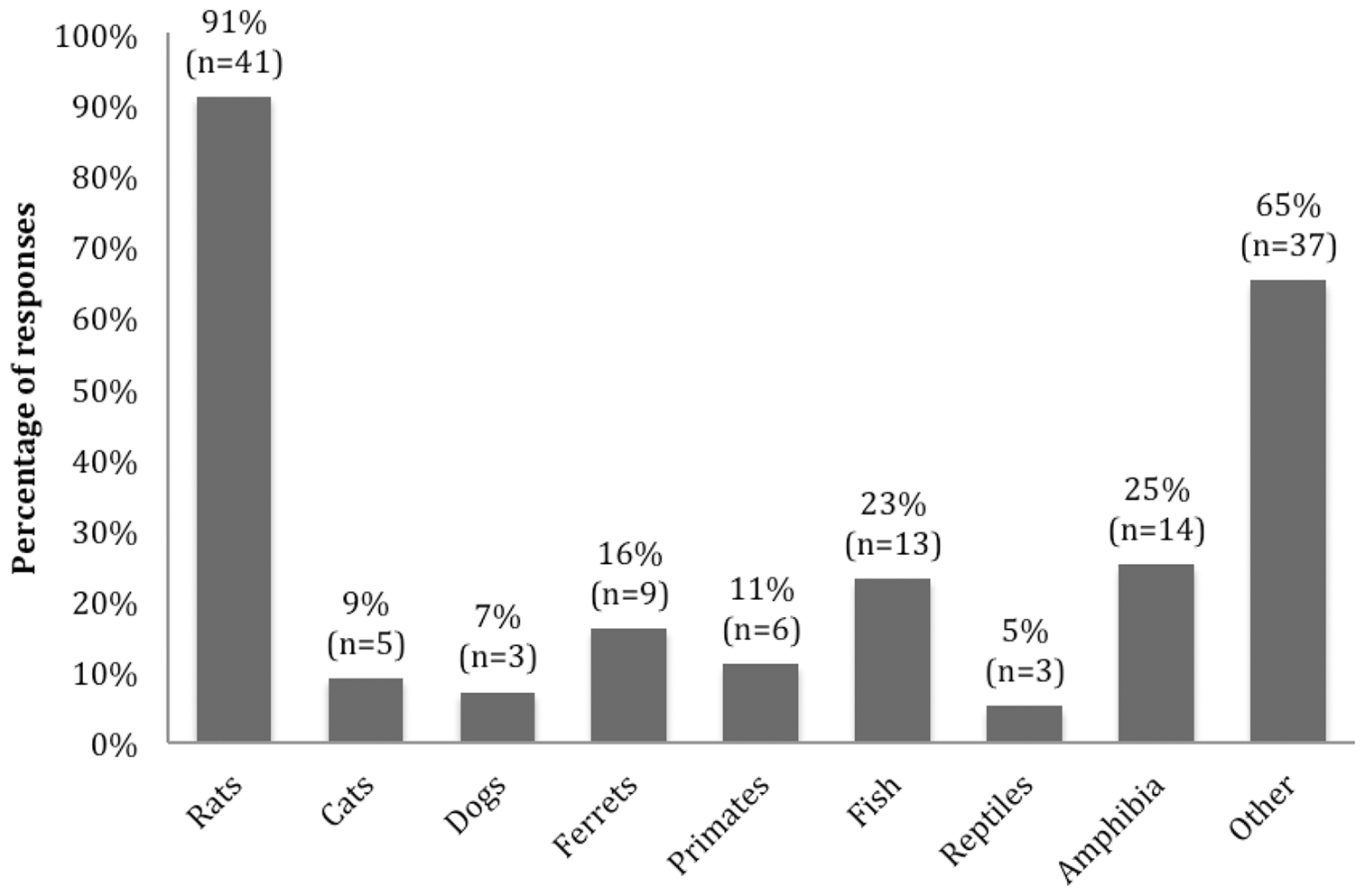
Regarding practices that could restrict the transfer of scents between other species and mice, most respondents (79%) reported changing their gloves and 46% washed their hands, but it was not clearly specified if this was done when handling other species followed by mice, or vice versa. Other less common practices were changing gowns 39% or changing all clothes (15%) and 9% of respondents did not report taking any precautions when handling different species (Figure 3). Additionally, gowns were most commonly shared between staff (63%), washed weekly (32%) and only 32% had species-specific gowns. No statistically significant correlation was found between these beliefs and the practices carried out to minimize inter-species scent transfer ( Current practices for changes of PPE between working with mice and other species. The number of responses for each practice is given on the top of each bar (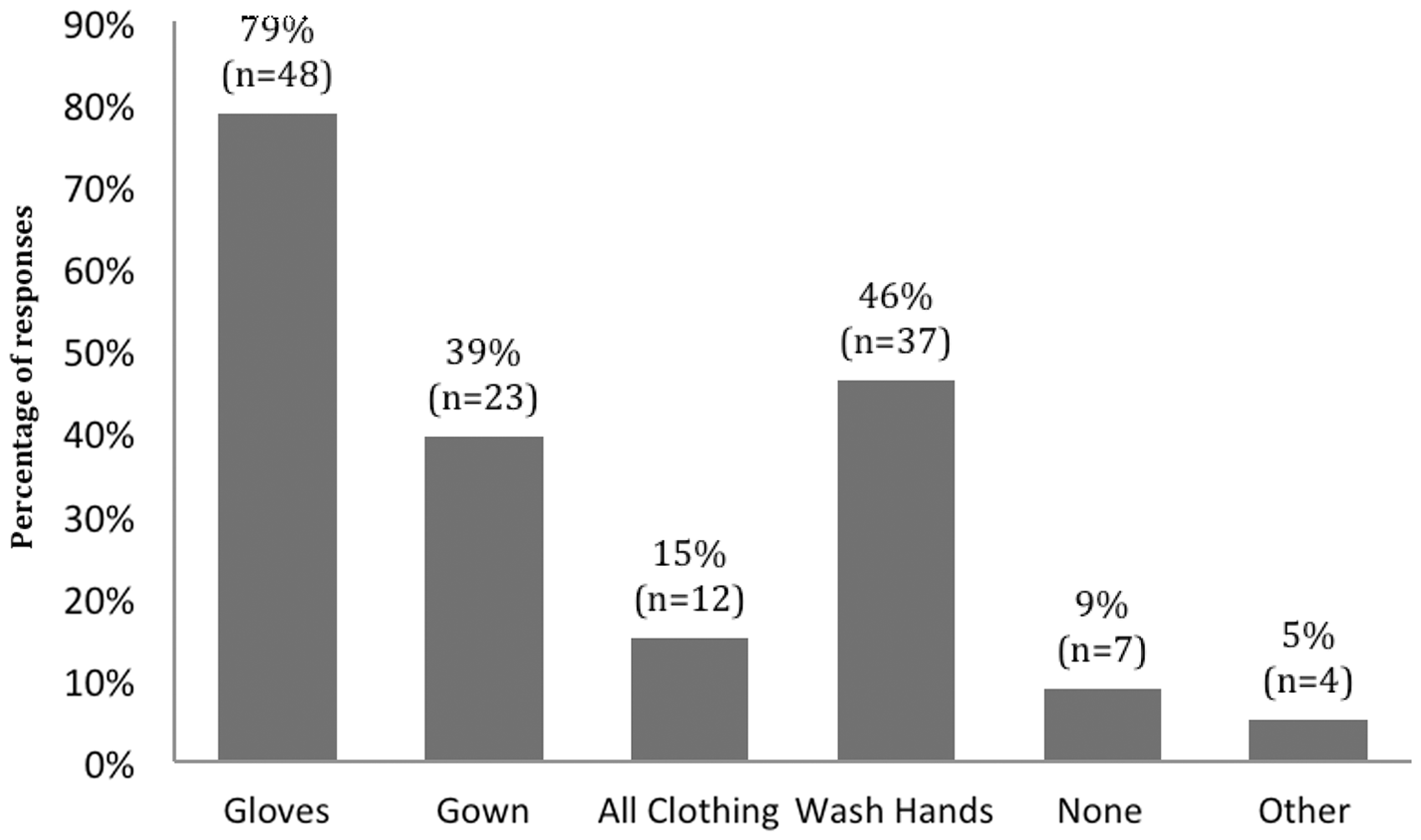
With respect to facility policies, 57% of respondents indicated that their place of work had a policy regulating staff pet ownership. These policies were mainly aimed at controlling the possession of rodents: keeping rodents was not allowed (10), limited (1), discouraged (1), or required a shower before entering the animal unit (2). Cats and dogs were the most common (74%) species kept as companion animals. Other predatory species kept as pets were rats (16%) and ferrets (9%).
Once again, respondents perceived mouse behaviour as being most strongly affected by scents of other species (33%) or staff (20%). By contrast, the weakest effects were reported on health and physiology (38% and 54%, respectively). Interestingly, human odours were generally considered as having less of an effect than other animal odours (Table 1).
Discussion
The results of the survey have revealed that a variety of routine husbandry procedures could allow the transfer of scents of animal origin, such as odours of conspecifics of different sexes and strains, and other species including mouse predators. The critical factors, with arguably the biggest risk of scent transfer, are the lack of changes of PPE, and the methods of cleaning anaesthesia/euthanasia chambers and behavioural apparatus between animals. The survey also provided the opportunity for those most involved in the daily care of laboratory mice to raise any concerns and give their opinion regarding the importance of this type of scent transfer.
With both sexes of mice sharing the same room in the majority 71% of cases, pheromones of conspecifics of the other sex can be transferred if gloves are not changed and hands are not washed when handling both sexes, as reported by 76% of participants. The effects of pheromones on behaviour and reproductive physiology have been extensively studied and are widely addressed in the management of mouse-breeding programmes. Although the use of barrier cages could reduce the exposure to pheromones in the home cage (but not necessarily during procedures), conventional open cages are still used in many establishments. Additionally, the non-volatile nature of pheromones limits the capability of barrier cages to prevent pheromone exposure via hands or gloves, relying on adequate hand sanitizing practices to prevent the transfer of sexual cues. The transfer of pheromones between mouse cages could add a source of physiological variation and possibly of stress or frustration. However the lack of exposure to conspecific scents could be considered as a type of sensory deprivation and might have a negative impact on mouse welfare. Furthermore, mice perform differently on various behavioural tests depending on the type of cage they are housed in, adding a source of variation to experimental results.1,2,4,36–40
The survey revealed that rats are commonly (91%) housed in the same animal unit as mice and that a large (41%) percentage of participants believed that rat odours might affect mice. When predator and prey species are housed in the same facility, it is important to ensure that husbandry procedures prevent the transfer of kairomones from the former to the latter. Kairomones present in urine and fur can trigger stress-associated physiological and behavioural changes, but scents associated with the fur of predators could have a particularly high impact as, in nature, they would indicate high proximity to the threat.5–7,27–32,41 The survey also revealed that changing gloves and washing hands are moderately (76% and 46%, respectively) common practices when handling different species (although the direction of species change was not specified) but species-specific gowns are used by only 32% of respondents. This may not be an issue if repeated exposure to predatory scents leads to habituation, but strong stimuli might be resistant to this. In some instances, if the stimulus is withdrawn and later reintroduced, responses can be recovered and even potentiated.3,10,42 For example, mice exposed to cat urine for 10 days failed to show habituation and continued to show inhibition of sexual behaviour and over a three-fold elevation of plasma corticosterone throughout this time.9,14,15,19,43 However, the effect of rat scents on mouse habituation has not been studied, and further research on the onset, length and magnitude of this phenomenon would be beneficial.
Despite the effects of cat and ferret odours on mice being well researched, and indeed these odours are explicitly used as stressors in some experiments,1,2,4,44–47 people’s awareness of their effects on mice is low (cats: 3%, and ferrets: 17%). This may be a relevant concern, since 74% of respondents reported the keeping of cats as pets by staff at their institutions, and 9% of institutions housed cats; and 16% housed ferrets.
Studies on human allergies have identified clothing and human hair as common sources of cat and laboratory animal allergens.5–7,27–32,48–50 Because allergens, like pheromones, derive from urine and other animal secretions, not changing clothes or failing to cover hair (particularly if predatory species are kept as pets in the household) could introduce olfactory cues with a noticeable effect on behaviour and a possible impact on mouse welfare.3,10,18,43–45,47,51–61 Indeed, respondents seemed most concerned about the effects of other species’ odours on mouse behaviour, health and physiology and standardization of experiments, with the highest proportion (33%) believing that these effects would be strong. However, there is considerable uncertainty, because approximately 40% believed that any effects would likely be weak, so research is required to resolve the importance of these effects in an applied context.
Transfer of human scents to mice can occur during husbandry procedures, particularly when it involves handling the animals with bare hands (30%). Recently published studies9,14,15,25,43 have shown that male and female mice exposed to human males or their scents (who share a similar androgen-dependent pheromonal make-up as males of other species) display behaviours associated with stress (increased thigmotaxis in the open field and increased defecation), signs of pain inhibition (decreased facial grimacing after injection of inflammatory agents), and physiological changes indicating hypothalamic activation (increased plasma corticosterol levels and hyperthermia). Additionally, rodents seem to be able to perceive differences between the olfactory identities of the people who interact with them. The presence of unfamiliar experimenters can cause fear-like behaviours such as higher anxiety scores in the elevated plus maze16,26 or increased locomotion and place preference.8,17,62,63 These types of behavioural tasks are routinely used to assess mouse models of neuropsychiatric disorders;18,22,64–71 drug screening and toxicology;19,68–70,72 and for phenotyping genetically-modified and mutant mice.27,69,71,73–77 The effects of the housing and test environment, and factors such as the sex of the experimenters, and familiarity of the animals with them, should be considered in the experimental design of animal studies.2,32–34
As routine husbandry procedures might have a negative (or positive) impact on the welfare of laboratory animals, and could add a source of variation to experimental results, they could also affect reproducibility between laboratories. The survey highlights discordance between respondents as to whether scents of animal origin have strong versus weak effects on mice. Further studies to investigate these effects are required to facilitate recommendations for best practice.
Footnotes
Acknowledgement
We are grateful for all the responses from the respondents who participated in this survey.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Noelia López-Salesansky was supported by a Went Scholarship at the RVC. This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sectors. RVC publication approval reference PPH_00934.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
