Abstract
In this study, the effect of four anaesthetic protocols that included the combination of xylazine (X) and ketamine (K) with acepromazine (A) and opioids (methadone (Me), morphine (Mo) or tramadol (T)) was evaluated in laboratory rats of both sexes. Ultrasonic vocalization (USV) was used as an indicator of pain during the recovery period. The objective was to evaluate the physiological parameters and the analgesic effect of each protocol to determine which protocol was the safest and fulfil the requirements of a balanced anaesthesia. The better protocols were the XKA protocol for both sexes and the XKMe protocol for females because the combinations achieve surgical plane of anaesthesia in rats. However, pain assessment during the formalin test revealed that rats anaesthetized with XKA produced more numbers of USV, suggesting that it is not a good protocol for the control of immediate postoperative pain. All protocols produced depression in body temperature and respiratory and heart rates, and had important effects, such as micturition and maintenance of open eyes. Only rats anaesthetized with XKA protocol did not present piloerection. These results demonstrated that good monitoring and care during anaesthesia must be included to prevent complications that compromise the life of the animal and to ensure a good recovery. The inclusion of analgesia in anaesthesia protocols must be used routinely, ensuring minimal presence of pain and thus more reliable results in the experimental procedures.
Rats are commonly used in research, and the use of anaesthesia is required and recommended for many experimental procedures. Well-controlled anaesthesia is essential to reduce potential surgical complications and ensures safety and success in the experimental procedures. 1 Anaesthesia in laboratory rodents is particularly challenging due to the problems related to their size, for example, accelerated metabolism, easy development of hypothermia and difficulty in determining cardiorespiratory functions. 2 Therefore, the goal is to establish a balanced anaesthetic protocol that includes a combination of several drugs to obtain good control of unconsciousness, analgesia and myorelaxation to minimize anaesthetic risks. 3
The anaesthetic protocols that mix two or three components have better effects compared to a single anaesthetic because one product cannot produce all the effects that are sought in anaesthesia (hypnosis, muscle relaxation and analgesia). 4 Ketamine is one of the most commonly used parenteral drugs in laboratory rats and is normally combined with other drugs such as xylazine.5,6 The use of this combination with acepromazine has been described in the literature,3,5 but it is not commonly used in many laboratories. In addition, there is not much information on anaesthetic protocols used in laboratory rodents that include the combination of these anaesthetics with opioids, such as tramadol, morphine and methadone. The preoperative administration of opioids simultaneously with the anaesthetic protocols has advantages such as the possibility of reducing the required dose of anaesthetics to achieve surgical anaesthesia as well as reducing the need for postoperative analgesics. 7 In view of these benefits the present study is important to clarify the interaction of the combination of XK with different opioids in an injectable anaesthetic protocol.
Vocalization serves as an indicator of emotional and aversive states in rats. 8 Juvenile and adult rats emit basically two types of ultrasonic vocalizations (USV) that are distinguished on the basis of the frequency: the 22 kHz vocalizations produced in response to aversive and dangerous situations and the 50 kHz vocalizations that are produced in response to appetitive situations, such as copulation and play behavior. 9 Jourdan et al. also demonstrated that monitoring rodent USV is a potential method of measuring the negative affective component of pain by a brief electrical pulse applied to the tail. 10
The effects of opioids have been extensively studied using the formalin test, and it has been shown that morphine, methadone and tramadol influence pain control in rats.11,12,13 However, there are no studies that have been performed yet with the opioids that were used in this experiment in the context of anaesthesia in rats. For this reason, it is important to evaluate intraoperative pain and recovery from anaesthesia.10,14,15 The objective of this study was to evaluate the effect of different anaesthetic protocols on the physiological parameters (heart and respiratory rate, temperature and clinical signs) and their intraoperatory analgesic power to determine the most effective and safe protocols for surgical procedures and to ensure the well-being of laboratory rats.
Materials and methods
Animals
In total, 64 SPF Wistar-Han rats (Rattus norvegicus), 28 females and 36 males aged from eight to 12 weeks were used. Animals were obtained from the animal facility of the Institute of Biomedical Science of the University of São Paulo, and they were free of ecto- and endoparasites, Mycoplasma pulmonis, Pasteurella pneumotropica, Bordetella bronchiseptica, Helicobacter spp., Klebsiella oxytoca, Klebsiella pneumoniae, Pasteurella multocida, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus β-hemolytic spp., Streptococcus pneumoniae and Salmonella spp., Kilham Rat Virus, Pneumonia Virus of Rat and Reovirus. At most, 4 animals were housed per cage on corn-cob bedding (Granja R.G., SP, Brazil) in polypropylene cages (41 × 34 × 16 cm) that were changed once a week. Paper rolls were added as environmental enrichment. All the animals were maintained under climate-controlled conditions of 12:12-h light/dark cycle, temperature range from 22 to 24℃, relative humidity of 45–65%, ad libitum access to drinking water, and a standard food-pellet diet (Nuvital®-Quimtia, PR, Brazil). Before the beginning of the experiments, the animals were housed in the animal facility of the Department of Pathology of the School of Veterinary Medicine and Animal Science, University of São Paulo, where they stayed for at least one week for adaptation and handling before the start of the experiment.
All animal procedures were performed in accordance with the guidelines of the Ethics Committee of the School of Veterinary Medicine and Animal Science of the University of São Paulo, Brazil (no.9635260116).
Anaesthetic procedure
Four different protocols were established for this study16–18 (see Supplementary Material 1).
For males:
XKA: Xylazine 7.5 mg/kg (Anasedan®-Ceva, 20 mg/ml, SP, Brazil) + Ketamine 60 mg/kg (Dopalen®-Ceva, 100 mg/ml, SP, Brazil), +Acepromazine 2 mg/kg (Acepran®-Vetnil, 2 mg/kg, SP, Brazil); XKMe: Xylazine 5 mg/kg + Ketamine 60 mg/kg + Methadone 5 mg/kg (Mytedom®-Cristália, 10 mg/ml, SP, Brazil); XKMo: Xylazine 7 mg/kg + Ketamine 60 mg/kg, + Morphine 1 mg/kg (Dimorf®-Cristália, 10 mg/ml, SP, Brazil); and XKT: Xylazine 7.5 mg/kg + Ketamine 65 mg/kg, Tramadol 5 mg/kg (Tramadon®-Cristália, 50 mg/ml, SP, Brazil).
For females:
XKA: Xylazine 5 mg/kg + Ketamine 60 mg/kg + Acepromazine 1 mg/kg; XKMe: Xylazine 5 mg/kg + Ketamine 60 mg/kg + Methadone 5 mg/kg; XKMo: Xylazine 5 mg/kg + Ketamine 60 mg/kg + Morphine 1.5 mg/kg; and XKT: Xylazine 5 mg/kg + Ketamine 60 mg/kg, Tramadol 5 mg/kg.
Rats were previously weighed to determine the exact dose of each drug. To avoid any interference of circadian rhythm, the experiment was always performed from 6.00 a.m. to 12.00 p.m. The administration of the anaesthetic protocol was done by intraperitoneal route. The drugs were mixed in a sterile plastic tube immediately prior to the administration and were given in one injection to minimize handling stress with a maximum injection volume of 0.5–0.7 ml. Then, the rat was placed in a cage alone to observe its behaviour, and when the animal lost the righting reflex, it was laid in dorsal recumbency on a preheated thermal blanket (between 35 and 37℃) to minimize the loss of body temperature. Sterile ocular lubricant (Vidisic®-Gel-Bauch+Lomb, 2 mg/kg, SP, Brazil) was administered to both eyes.
During the anaesthesia, the following periods were recorded:
Induction time: the period from the administration of the anaesthesia until the loss of the righting reflex. Non-surgical anaesthesia: the period between the loss of the righting reflex and the loss of most or all reflexes (blink, pedal and tail withdrawal reflex). Surgical anaesthesia: the period between the loss of all the reflexes (considering surgical tolerance the loss of the pedal withdrawal reflex) and their recuperation. Recuperation time: the period between the recuperation of all the reflexes and the return of the ability to walk.
The total duration time of the anaesthesia, the period from the administration of the anaesthetic protocol until the withdrawal of the ability to walk, was also measured.
The rats initially breathed room air but 7 minutes after losing the righting reflex, they were supplied with 100% oxygen using a nose cone until the end of the anaesthesia (Supplementary Material 2).
After the induction time, a pulse-oximeter (NT1A-V®-Solaris Medical Technology, Inc., CA, USA) was positioned on the pad of the left hind limb, and then the physiologic parameters and reflexes were measured every 10 minutes until the righting reflex return. The heart rate was determined by the pulse-oximeter and the respiratory rate was counted by observing thoracic or abdominal movements. The body temperature was measured by a digital thermometer placed into the animal’s rectum. The reflexes were evaluated as follows: the pedal withdrawal reflex by pressing the paw with an atraumatic forceps, alternating both hind limbs; the palpebral reflex by a slight touch with the atraumatic forceps in the margin of the eyelid; and the tail pinch reflex by a compression of the tail with the atraumatic forceps; 19 any negative or positive response was classified as the loss or the return of the reflex, respectively.
Pain evaluation by ultrasonic vocalization
When the rats recovered the pedal reflex or their whiskers started to move, a subcutaneous injection of 60 µl of formalin solution (formaldehyde 37%, wt/wt, diluted to 10% in 0.9% saline wt/wt) 8 was performed in the dorsal surface of the right hind paw of each rat. 14 After 5 minutes of the formalin administration, the USV were recorded using a USV detector monitor (Ultravox 2-0, Noldus Professional System for Automatic Monitoring of Ultrasonic Vocalization, USA). Each rat was tested individually by placing the USV detector 2 cm above the head of the animal in dorsal recumbency without any kind of restriction. The USV detector was set to detect frequencies of 22 kHz with an amplitude filter setting of 4 to minimize background noise. To avoid environmental noise, the experiment was performed in a soundproof room.
The total number and duration of USV for each rat were recorded during four sessions, starting 5 minutes after the application of formalin, for a total period of 25 minutes or until the righting reflex recovery. Each session consisted of 3 minutes of recordings with 2-minute lapses between them. During the recording, the rats received a stimulus every 20 seconds by compressing the area where the formalin was injected using a bulldog clamp. The intensity of the clamp tightening was always the same, letting it completely close for a second and immediately open.
At the end of the study all animals were euthanized by an overdose of X (30 mg/kg) and K (300 mg/kg) IP.
Statistical analysis
A power calculation was carried out with a total power of 0.8 and a two-sided significance of <−0.05. Estimates of variability for power calculation were based on data from a pilot project. The study was appropriately powered to detect a mean difference of 20% in the total duration of surgical anaesthesia, since this was considered clinically relevant. In males, we used up to 10 rats because the administration of formalin caused an immediate recovery from anaesthesia in some animals, and this did not allow the USV recording of them. The rats of each treatment were distributed randomly in different cages by sex.
Linear mixed effects models (LMMs) were used to test the effect of drug treatment and time (predictor variables) on the response variables: body temperature, cardiac frequency and respiratory frequency in male and female rats. The rat identity was used as a random factor to account for repeated measures in each rat. Linear models (LMs) were used to investigate the effect of drug treatment on the response variables: induction time, non-surgical anaesthesia, recovery and total duration of anaesthesia. Generalized linear mixed models with Poisson distribution were conducted to investigate whether the number of vocalizations (response variable) differed according to the predictor variables: drug treatment and time. The random factor was rat identity. A candidate set of models were constructed for each analysis previously mentioned and used an information-theoretic model selection (I-T) to determine which model(s) was best fit and well supported by the data. 21 The I-T model selection provides a strength of evidence for a set of a priori models based on Akaike’s Information Criterion values (AIC) and do not rely on null hypothesis testing. 21 The best fit model of each candidate set was determined based on AIC adjusted for small sample size (AICc); and delta AICc (ΔAICc) and Akaike weights (AICcw) were used to evaluate the support of the models (for a complete treatment of I-T, see Burnham and Anderson). 20 Here, we considered a model having the lowest AICc, ΔAICc <2 and AICcw approaching 0.90 or higher the best fit and well-supported model for the containing predictor(s). We took the best fit model in each case and reported the effects of predictor(s). When a main effect was detected we conducted post-hoc pairwise comparisons. Specifically, we used mean difference and Cohen’s d effect size for conducting post-hoc tests on LMMs and LMs, respectively, and 95% confidence intervals of estimates (95% CIs) to measure precision and ‘statistical significance’. If 95% CIs overlapped, 0 indicates ‘statistical non-significance’ in a pairwise comparison. The rules of thumb defined in Sawilowsky 21 were used for interpreting Cohen’s d values. We used descriptive statistics to describe the patterns of vocalization duration due to small size to fit a statistical model. The data are shown as the mean ± SE, unless otherwise stated. We decided not to report statistical significance (p values) in order to focus attention on the biological relevance of effect sizes. 22 Statistical analyses were conducted in Rv.3.3.2 (R Development Core Team 2017).
Results
Physiological parameters
The best fit and well-supported model for explaining variation in the body temperature and heart and respiratory rates in male and female rats was the time only model (Supplementary material 3). The significant differences between the parameters over time using mean differences and 95% CIs can be observed in Supplementary material 3–5.
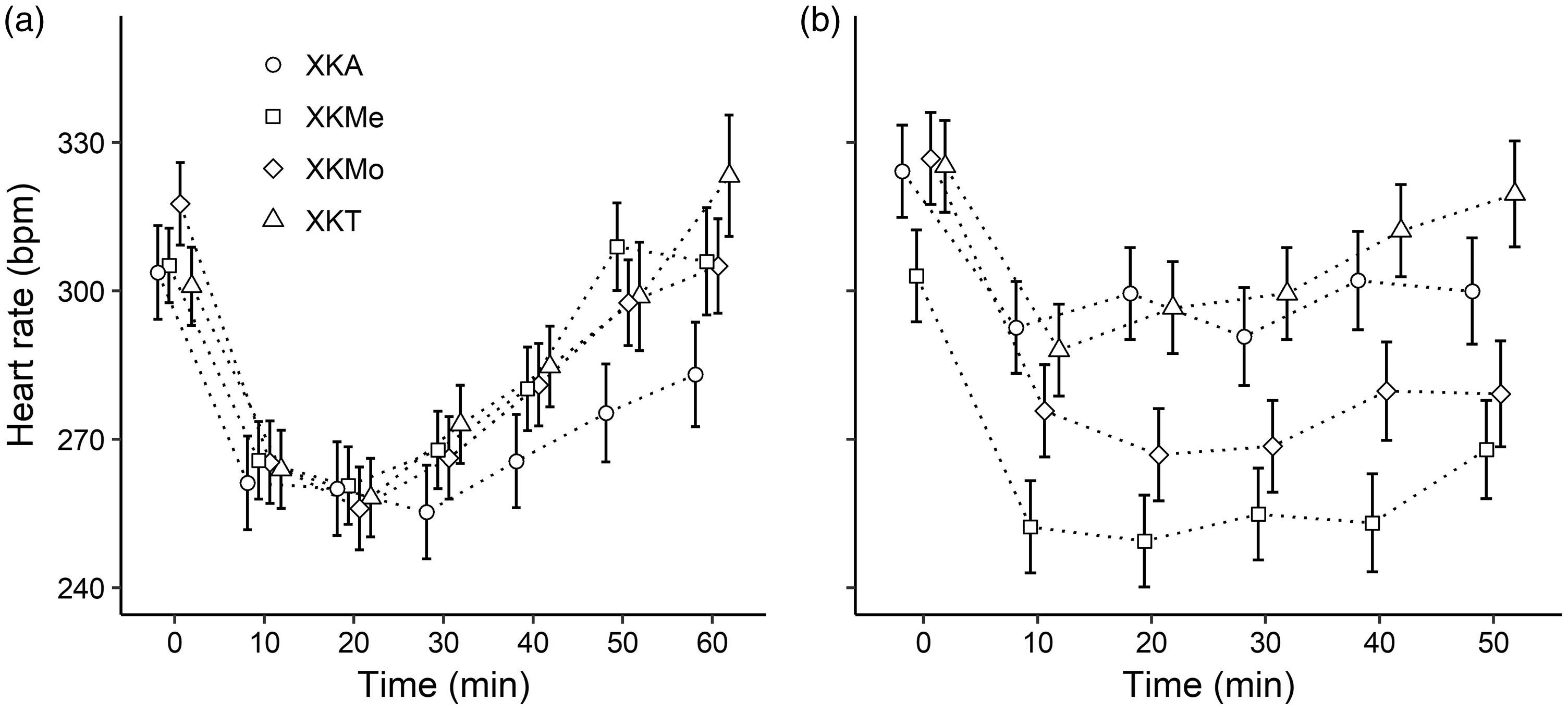
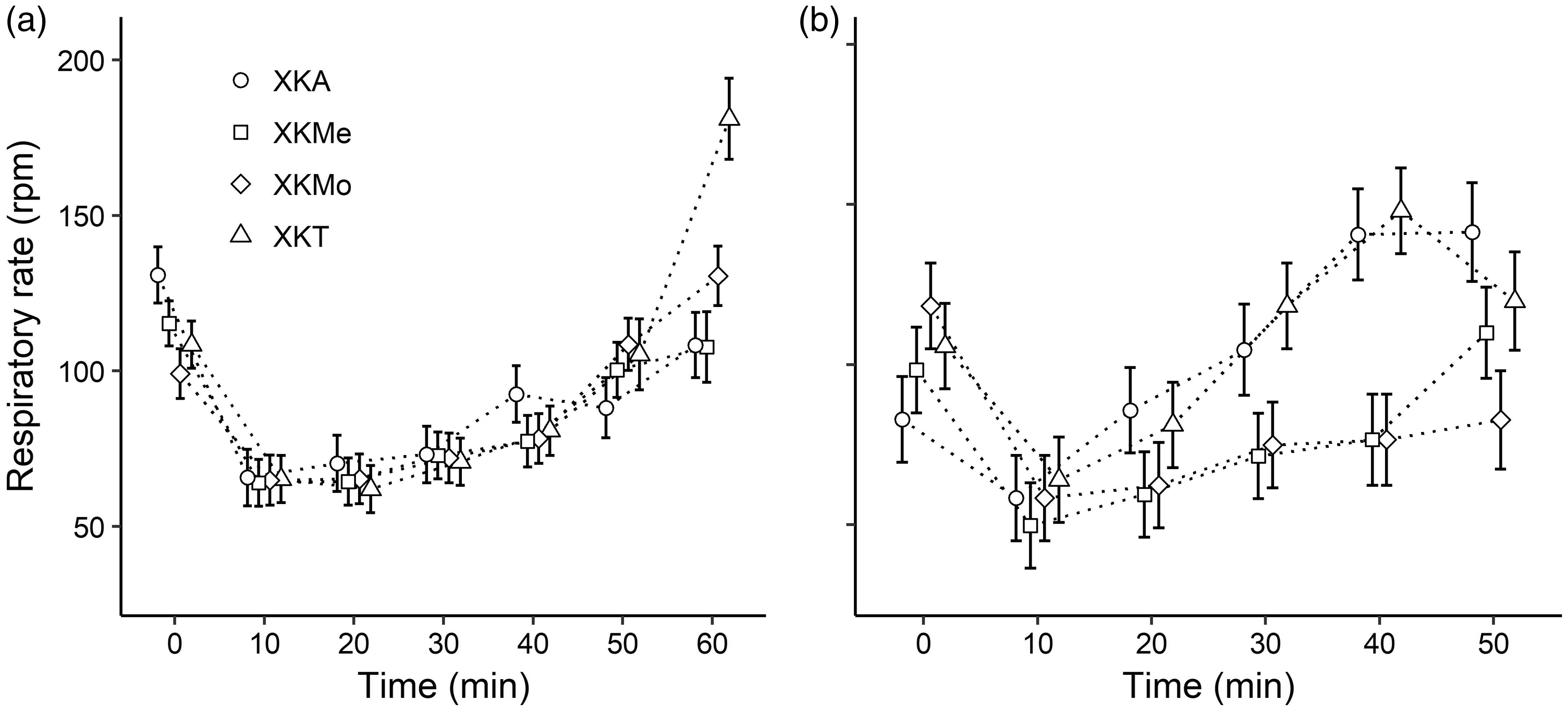
Considering all the anaesthetic protocols, the mean body temperature of the males ranged from 35.66 ± 0.19℃ (at 10 minutes) using XKA to 37.62 ± 0.24℃ (at 60 minutes) using XKMe (Figure 1(a)). In females, the mean body temperature ranged from 36.30 ± 0.07℃ (at 10 minutes) using XKMe to 37.78 ± 0.54℃ (at 50 minutes) using XKMo (Figure 1(b)). In males, all protocols presented a decrease in the mean heart rate, which reached the lowest rate at minute 20 (approximately 258 bpm); after this time, there was an increase in the mean heart rate in the four anaesthetic protocols (Figure 2(a)). In the case of the females, in which the protocol of XKMe was administered, after 10 minutes, the mean heart rate remained approximately 250 bpm until minute 40 (Figure 2(b)). All protocols for both sexes presented means lower than 70 rpm at minute 10 in the respiratory rate. However, females had a marked increase in respiratory rate after 20 minutes, except in the XKMo and XKMe protocols (Figure 3). In both males and females, there was a decrease in all the physiological parameters evaluated in the first 10 minutes.
Variation in body temperature (℃) in male (a) and female (b) rats submitted to different anesthetic associations. (X: Xylazine, K: Ketamine; A: Acepromazine; Me: Methadone; Mo: Morphine; and T: Tramadol.) The points represent fits to the model’s predicted values, and the lines indicate the SE of model fits. Variation in heart rate (beat per minute (bpm)) in male (a) and female (b) rats submitted to different anaesthetic associations. (X: Xylazine; K: Ketamine, A: Acepromazine, Me: Methadone, Mo: Morphine, and T: Tramadol.) The points represent fits to the model’s predicted values, and the lines indicate the SE of model fits. Variation in the respiratory rate (breaths per minute) in male (a) and female (b) rats submitted to different anaesthetic associations. (X: Xylazine; K: Ketamine; A: Acepromazine; Me: Methadone; Mo: Morphine; and T: Tramadol.) The points represent fits to the model’s predicted values, and the lines indicate the SE of model fits.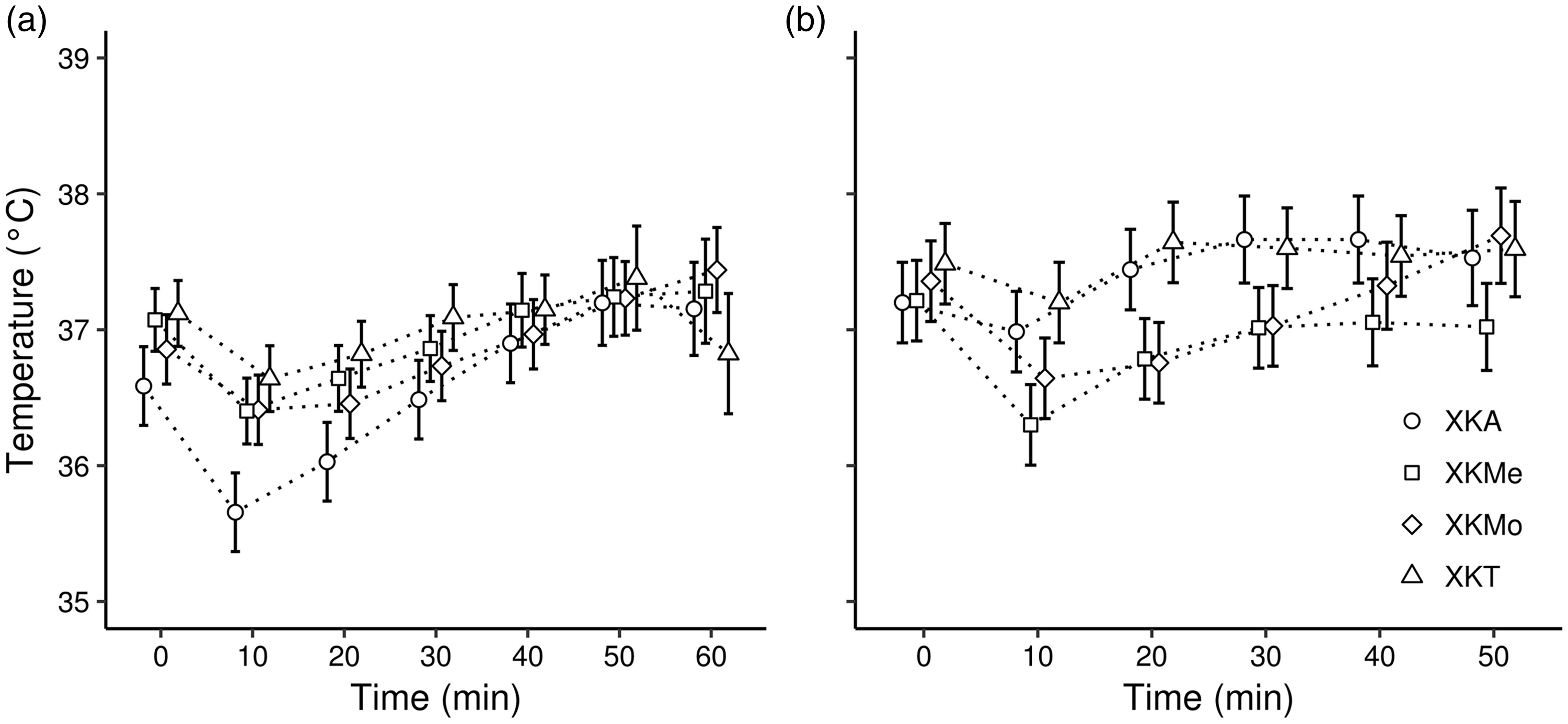
Clinical signs, reflexes and anaesthetic times
Comparison of safety margin, clinical signs, reflexes and anaesthesia times (minutes) of male and female rats submitted to different anaesthetic associations.
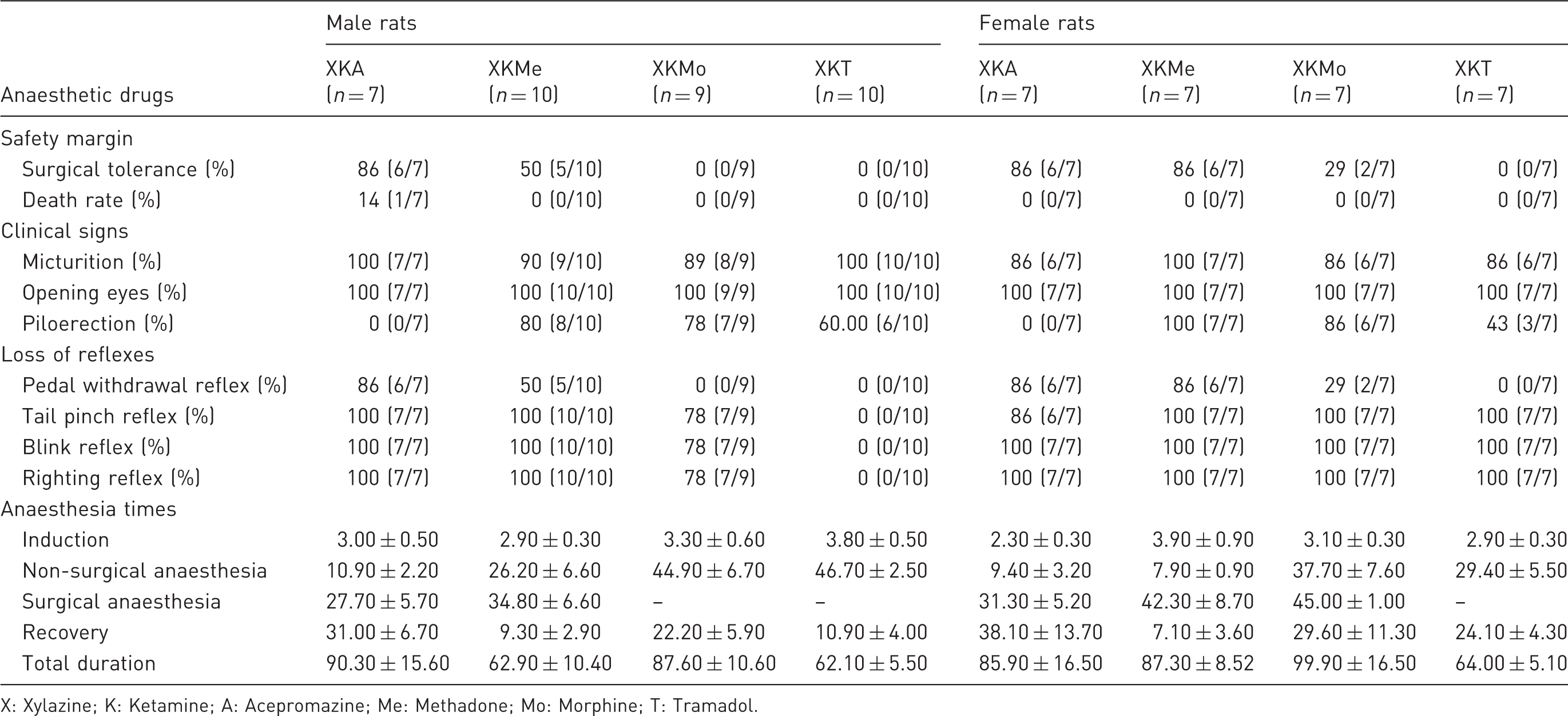
X: Xylazine; K: Ketamine; A: Acepromazine; Me: Methadone; Mo: Morphine; T: Tramadol.
The protocol in which acepromazine was used had better surgical tolerance in both sexes, and females also had good tolerance with the protocol XKMe. The rats from both sexes from protocol XKT did not achieve surgical tolerance, as well as males from protocol XKMo (Table 1).
The drug treatment model was the best fit and well supported model for explaining variation in the duration of nonsurgical anaesthesia and recovery of male rats (Supplementary material 7). Male rats anaesthetized with XKA showed a lower duration of non-surgical anaesthesia compared to the other protocols (Table 1), and differences were large between them according to Cohen’s d effect sizes (Supplementary material 8). In relation to duration of recovery, there were large and medium differences between protocols, except between XKT and XKMe, where the difference was negligible (Supplementary material 8). Regarding duration of anaesthesia, the model with drug treatment effect was not well supported but provided the best fit (Supplementary material 7). The protocols XKA and XKMo showed a higher duration compared to XKMe and XKT, and their differences were large according to Cohen’s d (Supplementary material 8).
In females, the duration of non-surgical anaesthesia differed between protocols (Supplementary material 7). The protocols XKA and XKMe showed a lower duration compared to the protocols XKMo and XKT (Table 1). Drug treatment models were not well supported but provided the best fit to examine the duration of recovery and total duration (Supplementary material 6). Recovery was the lowest in rats anaesthetized with XKMe (Table 1) and showed some significant differences with other protocols according to Cohen’s d effect sizes (Supplementary material 8). Total duration was the lowest in female rats anaesthetized. with XKT (Table 1) with some significant medium and very large differences with other protocols (Supplementary material 8).
Ultrasonic vocalization
For the number of male vocalizations, the best fit and well-supported model of the four possible models examined through AICc criteria was the drug treatment by time interaction model; the same result was observed in female rats (Supplementary material 9). These interactions indicate that the effect of the protocol on the number of times rats conducted a vocalization was not constant through time. The painful stimulus increased the number of vocalizations with a longer duration pattern in the XKA protocol for both sexes (Figure 4 and 5). In the case of male rats, after the application of formalin some animals recovered the righting reflex before the USV recordings started, so to evaluate the formalin test, the number of animals was increased in order to reach at least seven animals per group. Only in the 20–23 interval, in males, was an increase in the number of vocalizations observed with the XKMo protocol, but with a lower duration pattern in comparison to the XKA protocol (Figure 4(a)).
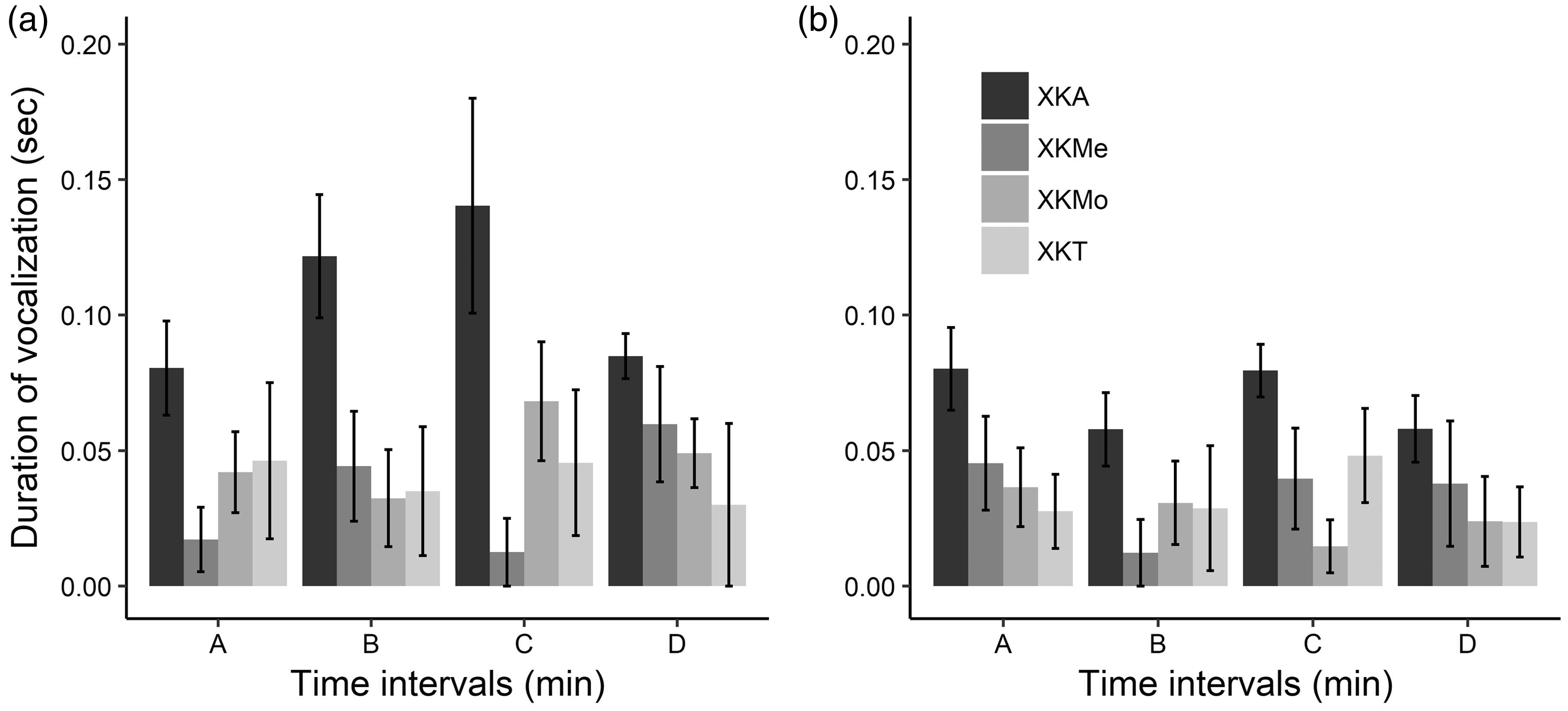
Number of ultrasonic vocalizations during the formalin test in male (a) and female (b) rats at different time intervals (A: 5–8 minutes; B: 10–13 minutes; C: 15–18 minutes; and D: 20–23 minutes), submitted to different anaesthetic associations. (X: Xylazine; K: Ketamine; A: Acepromazine; Me: Methadone; Mo: Morphine; and T: Tramadol.) The bars represent fits to the model’s predicted values, and the lines indicate the 95% confidence interval of model fits. Duration of ultrasonic vocalizations (seconds) during the formalin test in male (a) and female (b) rats at different time intervals (A: 5–8 minutes; B: 10–13 minutes; C: 15–18 minutes; and D: 20–23 minutes), submitted to different anaesthetic associations. (X: Xylazine; K: Ketamine; A: Acepromazine; Me: Methadone; Mo: Morphine; and T: Tramadol.) The bars represent mean values, and the lines indicate ± SE.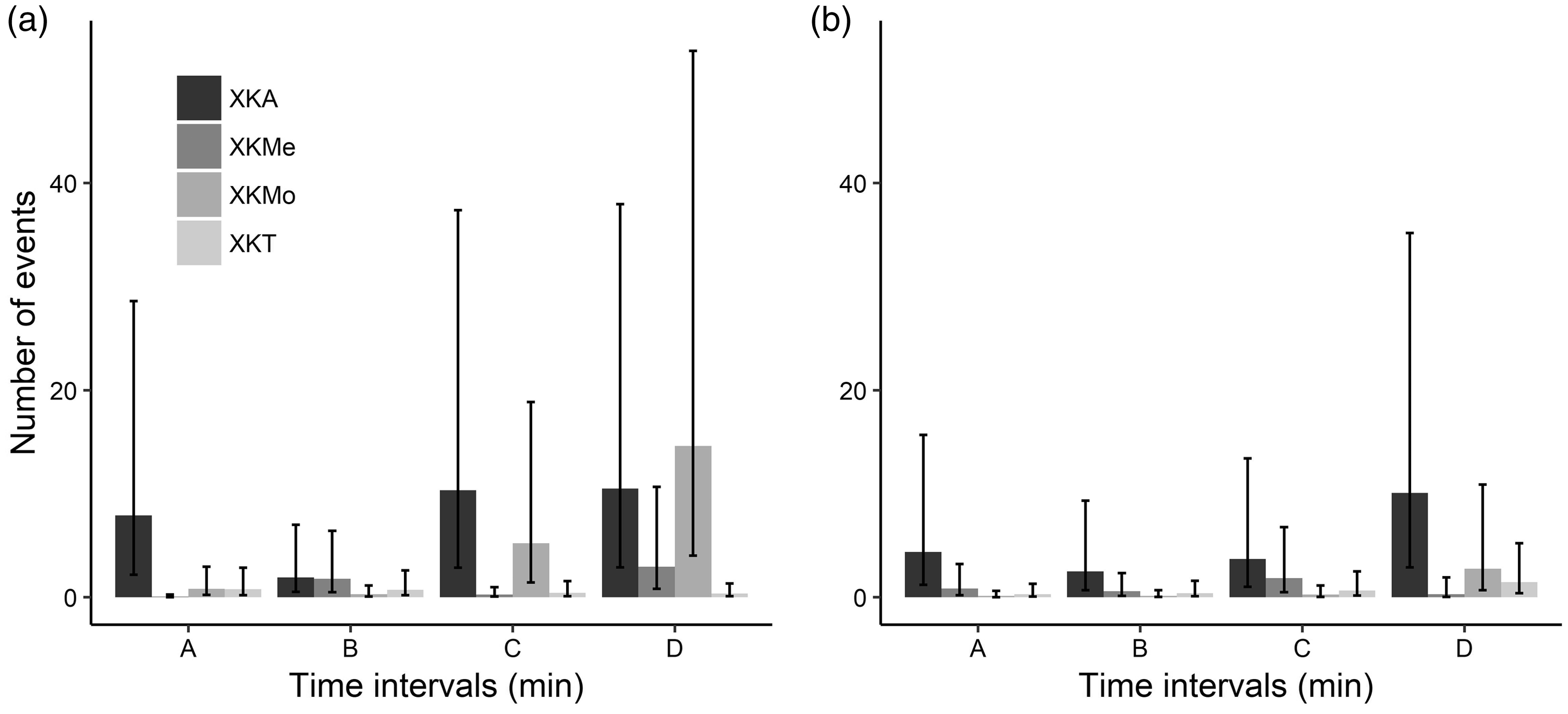
Discussion
The doses recommended in the literature for ketamine and xylazine are very varied and depend on whether they are applied alone or in combination with other drugs. Because the combinations of XK with opioids used in this study were not described in the literature, the doses were based on a pilot study conducted prior to this experiment (unpublished data). There is evidence that rats female and male can have differences in the response to the same anaesthetics, 23 so the sex of the animal is a variable that should be considered for the establishment of the doses to be used. Although the doses were not identical for both sexes, the similar behaviour in the dynamics of the physiological parameters of each protocol allows us to conclude that the doses used in this study were adequate. For this same reason, the primary objective was not to compare the effect between sexes.
Physiological parameters
The reduction in all physiological parameters in the first minutes observed in all anaesthetic protocols has been previously described with the XK protocol.6,24,25 The reduction in body temperature observed in rats may be related to the depressant effect of XK on thermoregulatory mechanisms, in addition to the fact that small rodents easily lose heat when anaesthetized, given the high proportion of body surface in relation to their weight.25,26
The heart rate depression observed in all protocols in male rats and in the XKMe protocol in females can be produced mainly by the action of xylazine and methadone. Ketamine usually causes a stimulation of cardiovascular function causing an increase in heart rate and blood pressure. Thus, the reduction in heart rate may be a side effect of α2-agonists as a result of an increase in vagal reflex activity and decrease in norepinephrine release in the sympathetic nervous system. 26 Methadone can cause calcium channel blockage producing bradycardia, 27 an effect that was observed in both sexes with the XKMe protocol.
In addition to cardiac depression, respiratory depression was observed in all protocols. Opioids can produce this effect in high doses or in combination with other CNS depressants. 17 Opioid receptors are abundant in the respiratory control center. Moreover, μ-opioid receptor agonists, such as morphine and methadone, bind to these receptors, activating them and causing respiratory rate depression. 28 Xylazine can also cause CNS depression by stimulation of α2-adrenoreceptors. 29
All protocols in this study follow the same pattern of physiological parameter decrease at the first 10 minutes. The clinical support of the animal is critical mainly in this period of time. A better recovery and a reduction of mortality can be enhanced by monitoring, heating and oxygenating the animals.
Clinic signs, reflexes and anaesthetic times
A marked micturition (86% or more) was observed in all protocols, which was also recorded in rats treated with xylazine.25,26,30 This drug produces inhibition of the antidiuretic hormone release, which is the reason why the use of combinations with xylazine are not recommended in animals with urinary tract obstruction, dehydration or hypovolemia. 31 Only the rats that were anaesthetized with XKA did not present piloerection. Xylazine can cause piloerection as a side effect, but when combined with acepromazine, which has relaxant properties, this effect can be counteracted. 32
All animals anaesthetized in this study kept their eyes open during anaesthesia; besides the loss of blink reflex, the combination of XK induces a mild proptosis of the globe and the retraction of the eyelid. 33 According to Turner and Albassam, it is possible that the blink reflex is depressed to a greater degree in rats anaesthetized with the combination of XK versus other injectable or inhaled anaesthetic combinations. 34 Corneal lesions predisposed by the use of this anaesthetic combination may occur due to trans-corneal water loss due to aqueous humour alteration when continuous corneal exposure occurs, 35 or by the vasoconstriction of the ciliary and iridial vessels, and the local and systemic hypoxemia caused by xylazine, which may culminate in cell death. 34 For this reason, the use of protocols with XK is not recommended for ocular studies in which corneal evaluation is important. However, if there is no other option to use a different anaesthetic combination, eye drops, 100% oxygen supplementation and the administration of an adequate dose of yohimbine (to reverse the action of xylazine) are recommended to minimize the incidence of corneal lesions.
The XKA protocol resulted in a better anaesthetic effect in males. This protocol allows procedures with a duration time of approximately 25 minutes with a good margin of safety. Based on Guedel’s stages of anaesthesia, the loss of the pedal withdrawal reflex indicates that the animal achieved the second plane of the third stage of anaesthesia. 36 This means that those rats that lost this reflex achieved surgical tolerance. The XKMe protocol had a longer duration of surgical anaesthesia compared to XKA protocol having a recovery time three times faster; however, fewer animals achieved this level of anaesthesia. None of the animals achieved surgical anaesthesia with the XKMo and XKT protocols. Therefore, the use of these doses for surgical procedures is not recommended. For females, XKMe and XKA were the most effective anaesthetic protocols. The XKMe protocol had a longer surgical anaesthetic duration and a much faster recovery compared to XKA. Since methadone is an opioid that has great analgesic effect, the XKMe combination may be a good option for procedures that are invasive and painful.
When using the XKMo protocol, only two out of seven animals achieved surgical anaesthesia and the recovery period was very prolonged. These results suggest that this protocol is not the best recommendation for surgical procedures. As in males with the XKT protocol, no female reached anaesthesia, which indicates that in this dose interval, it is not a good protocol for use in surgery either.
The only animal that died in the experiment (with the XKA protocol), presented hyperthermia and increased heart rate. This could be associated to an individual reaction to anaesthesia.
Ultrasonic vocalization
Although there was great individual variability among the treated individuals, in both sexes, the XKA protocol presented the highest number of ultrasonic vocalizations with a frequency of 22 kHz with the longest duration, but these vocalizations did not exceed 0.3 seconds classified as short call. Two biologically significant subtypes of 22 kHz vocalizations have been identified, both expressing negative emotional sates: long calls (more than 300 ms) that serve as alarm calls and signal external danger or potential danger; short calls that express a state of discomfort or distress without external source of danger. 37 Thus, although XKA protocol seemed to be a safe anaesthetic protocol, we showed that these rats may be feeling pain.
The great difference of vocalization observed from the protocol XKA with the other protocols can be because acepromazine is a phenothiazine that can produce moderate sedation but has no analgesic effect alone. 27 On the other hand, opioids are analgesics that interact with μ-opioid receptors that inhibit pain. It has been demonstrated that the emission of 22 kHz USVs by rats subjected to experimental pain can be attenuated by the administration of drugs that possess clinically relevant analgesic properties. 8
The increase in the number of vocalizations in the 20–23-minute interval with the XKMo protocol in males could be because morphine can generate an excitatory effect in rats 39 that could generate the animals vocalizing more.
Contrary to what was described by Wallace et al., 40 these results suggest that the USV can be used as a complementary behavioural parameter in experimental models of pain. Nevertheless, this study was a pioneer in using USV to assess pain in anaesthetized rats in a recovery phase, so additional research is required to verify possible anaesthesia influences on the USV pattern of rats.
In conclusion, the XKA anaesthetic protocol was the best protocol for male rats and was also suitable for females, considering only the intraoperative period. Indeed, the analgesic effect of this protocol is low, and the use of an intraoperative analgesic is recommended. The XKMe protocol resulted in better results for females due to its high analgesic effect that can be used for more invasive surgical procedures. USV measurement was revealed to be a useful tool to evaluate the potential analgesic effect of drugs used in rats subjected to anaesthesia for surgical procedures, and it could be used in other behavioural models used in neuropharmacology.
Supplemental Material
Supplemental material for Combination of ketamine and xylazine with opioids and acepromazine in rats: Physiological changes and their analgesic effect analysed by ultrasonic vocalization
Supplemental Material for Combination of ketamine and xylazine with opioids and acepromazine in rats: Physiological changes and their analgesic effect analysed by ultrasonic vocalization by Jilma Alemán-Laporte, Luciana A Bandini, Mariana SA Garcia-Gomes, Dennis A Zanatto, Denise T Fantoni, Marco A Amador Pereira, Pedro E Navas-Suárez, Thiago Berti Kirsten, Randall R. Jimenez, Gilbert Alvarado and Claudia Cabrera Mori in Laboratory Animals
Footnotes
Acknowledgements
We thank Maria Martha Bernardi, Aline Magalhães and Yamê Miniero for earlier discussions about the topic. We thank the anonymous reviewers for their thoughtful comments that improved the manuscript. We would like to thank Carol Valenzuela for the English language review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brazil (CAPES) Finance Code 001.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
