Abstract
Oral administration of perioperative analgesia to laboratory mice is beneficial compared with administration by injection. The mice become less stressed when allowed to voluntarily ingest the drug in a palatable feed item and it results in high and long-lasting serum concentrations of the drug. We have previously demonstrated sticky nut and chocolate paste to be well-liked by mice and readily ingested in most cases. However, a disadvantage with nut and chocolate paste is its high content of fat and sugar, which may have undesirable effects in some experimental models. Alternatively, a delivery system using an aqueous gel may serve as a supplementary source of fluid post-operatively and as a vehicle for analgesic drugs. In the present study, we investigated the willingness of the mice to ingest a commercially available gel, by measuring the duration from introduction of the gel to first ingestion, as well as the amount ingested overnight. Furthermore, buprenorphine in two different concentrations (5 and 15 µg/mL) was mixed in the gel and the resulting serum concentrations of buprenorphine were investigated. The aqueous gel was ingested by the mice, but their willingness was low and did not increase over time. The serum concentrations of buprenorphine were similar to, or higher than, those following a subcutaneous injection (0.1 mg/kg body weight), but the variation was considerably higher. In conclusion, aqueous gel may serve as a relevant vehicle for the voluntary ingestion of buprenorphine in mice, but the willingness of the mice to ingest the gel needs to be improved.
Keywords
The use of adequate pain relief in laboratory rodents subjected to surgery or other invasive procedures is essential. Insufficient analgesia may be a source of compromised welfare for the animals, leading to unnecessary suffering, 1 and a lack of efficient analgesia during a painful procedure will induce a physiological stress response.2–5 A stress response has a major effect on physiological and endocrine functions, which impairs the recovery of the animals and alters allostatic mechanisms. This, in turn, may be a significant confounder of experimental data, leading to inaccurate results as well as increased inter- and intra-animal variations.4–6 Analgesic drugs can be administered to animals in numerous ways. Parenteral administration via subcutaneous (SC) or intraperitoneal injection is commonly used. 1 However, injectable analgesia requires handling and restraint of the animal in addition to the injection itself, all of which inevitably inflicts stress on the animal.7,8 Allowing animals to voluntarily ingest these drugs should thus be beneficial in comparison. We have demonstrated the beneficial effects of voluntary ingestion of analgesics in several studies, where rats and mice were less stressed and had high and long-lasting serum concentrations of the drug when allowed to voluntarily ingest the opioid analgesia buprenorphine in a sticky nut and chocolate paste (Nutella®).3,4,6,8–13 The nut paste was well-liked by the rodents and was ingested in almost all cases. We have therefore implemented it as a standard vehicle for oral delivery of buprenorphine in the animal facilities at our department. However, a disadvantage with nut and chocolate paste is its high content of fat and sugar, which may have unknown and undesirable effects in some experimental models.14–17 Thus, there is need for alternative vehicles.
Other studies have investigated the applicability and analgesic effect of oral administration through flavored jelly 18 and drinking water. 19 These routes of administration have been shown to provide analgesia, with some limitations such as reduced willingness of the animals to ingest the jelly or a reduction of water intake.19–21. Another option could be the use of a gel delivery system, where a gel consisting of approximately 98% water (MediGel) is presented to the animals to serve as a post-operative supplementary source of fluid and as a vehicle for the analgesic.
The first aim of the present study was to investigate the willingness of laboratory mice to ingest MediGel, measuring the duration from introduction of the gel to first ingestion, as well as the amount ingested overnight. The second aim was to investigate whether the gel would be suitable for oral administration of pre-emptive buprenorphine, by presenting the MediGel–buprenorphine mix in the late afternoon, and measuring the serum buprenorphine concentration, from the morning of the day after.
It was hypothesized that mice would be reluctant to ingest the gel when it was first provided, but that their willingness would increase over time. It was further hypothesized that pre-emptive administration of buprenorphine via the Medigel on the afternoon of the day before a planned procedure would result the following morning in serum levels of buprenorphine corresponding to those obtained after SC injection.
Materials and methods
Animals
All animal experiments in the present study were performed with the approval of the Danish Animal Experiments Inspectorate (license no. 2012-DY-2934-00018) under the Ministry of Food, Agriculture and Fisheries, and after review of the local animal welfare body of the faculty. Fifty male BomTac:NMRI mice from Taconic, Ry, Denmark were used in the study.
Housing conditions
The animals were housed in an animal facility fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALAC) in accordance with the Guide for Care and Use of Laboratory Animals. 22 Prior to the experimentation, the mice were acclimatized for one week. Mice were singly-housed in Makrolon type III individually ventilated cages (IVCs) (800 cm2 floor area, 15 cm high; Tecniplast, Buguggiate, Italy). Wooden chips (Tapvei Oy, Kortteinen, Finland) were used as bedding material. Bite bricks (Tapvet®; Tapvei Oy), Enviro-dri® nesting materials (Shepherd Specialty Papers, Quakertown, PA, USA), and cardboard houses (Brogaarden, Gentofte, Denmark) were used for environmental enrichment. A diurnal rhythm was maintained with a 12:12 h light–dark cycle and with artificial light from 06:00 h. Cage temperature was kept at 21–22℃, relative humidity at 50 ± 10%, and air was exchanged 75 times per hour. Mice were fed ad libitum with Altromin 1319 pellets (Brogaarden), and acidified tap water (pH 3.0) was available at all times.
Study design
The study was divided into three experiments. In the first experiment, 11 mice were presented with a limited amount (0.5 mL) of unmedicated MediGel (MediGel Sucralose; ClearH2O, Portland, ME, USA) placed in a plastic petri dish in the afternoon. The gel was presented before noon, and the mice were filmed until the lights went out (approximately 6–7 h). The time from presentation to the start of ingestion was recorded, as was the time at which each animal had ingested the entire amount provided.
In the second experiment, a MediGel container with 20 mL of unmedicated MediGel was placed in the cage at 17:00 h, and the amount of MediGel that had been ingested by 08:00 h the following morning was recorded. This experiment involved 15 mice and was repeated for three consecutive days. These mice were also used in the last experiment.
Thirty-nine mice (15 mice with experience of ingesting MediGel from the second experiment, together with 24 naïve animals) were used in the last experiment. Buprenorphine (Temgesic; Schering-Plough Europe, Brussels, Belgium) was mixed in the gel in concentrations of 15 µg/mL (Group A) and 5 µg/mL (Group B). An amount of 20 mL of the medicated gel was placed in the cage at 17:00 h, and the animals were allowed to ingest the gel overnight. Blood was collected during restraint without anaesthesia by puncture of the facial vein starting at 08:00 h the following morning, then at 14:00 h, 20:00 h and finally at 08:00 h, 24 h later. Each mouse was sampled twice, and ∼100 mL of blood was sampled at each occasion. Sampling occasions for each mouse was randomized according to a block design to ensure that all treatments were represented at all time points. In addition, blood was sampled at the same time points from animals treated with 0.1 mg/kg body weight buprenorphine injected subcutaneously one hour prior to first sampling (Group C). Animals were randomly distributed in the three groups described above, in order to ensure good representation of experienced and naïve animals in all groups. Thus, any bias from the mice’s previous experience of MediGel was avoided.
The serum concentration of buprenorphine was quantified in duplicate by using DRG–Diagnostics Corticosterone ELISA (EIA4164; DRG Instruments, Marburg, Germany) in accordance with the manufacturer’s instructions.
Since it was suspected that part of the gel would evaporate in the ventilated cages, the extent of evaporation was tested by placing containers with 20 mL of unmedicated MediGel in empty cages from 17:00 to 08:00 h. Containers were placed in three cages (one per cage), and the test was repeated three times, producing nine evaporation samples in total.
Statistical analysis
Data from Experiment 1 are merely descriptive, and no statistical testing was made on these. Data in Experiments 2 and 3 were tested for Gaussian distribution using D'Agostino–Pearson omnibus normality test. Data in Experiment 2 were found not to follow a Gaussian distribution, and these data are presented as medians with range, and statistical analysis was performed with Friedman’s test followed by Dunn’s multiple comparison test. Data in Experiment 3 did pass the normality test, and these data are presented as mean ± standard error of the mean (SEM), and statistical analysis was performed with a two-way analysis of variance (ANOVA) followed by Bonferroni’s post-test. Statistics were calculated using GraphPad Prism 5.0 (GraphPad Software Inc, La Jolla, CA, USA).
Results
The time from the presentation of unmedicated MediGel until the start of ingestion and the finishing of the provided portion (Experiment 1) is presented in Figure 1. It was found that six out of 11 mice began to ingest the MediGel within the first 5 min. One mouse started to ingest 5–15 min after presentation, and one 60–120 min after, while the remaining three mice did not start to ingest the gel until more than 180 min after presentation. After the first 5 min, two of the animals had ingested the entire amount presented. Two mice had finished the portion after more than 180 min, while the remaining seven mice did not finish the portion during the time of observation.
The percentage of animals that started to ingest and finish the portion, respectively, of a 0.5 mL portion of MediGel, during the specified time intervals.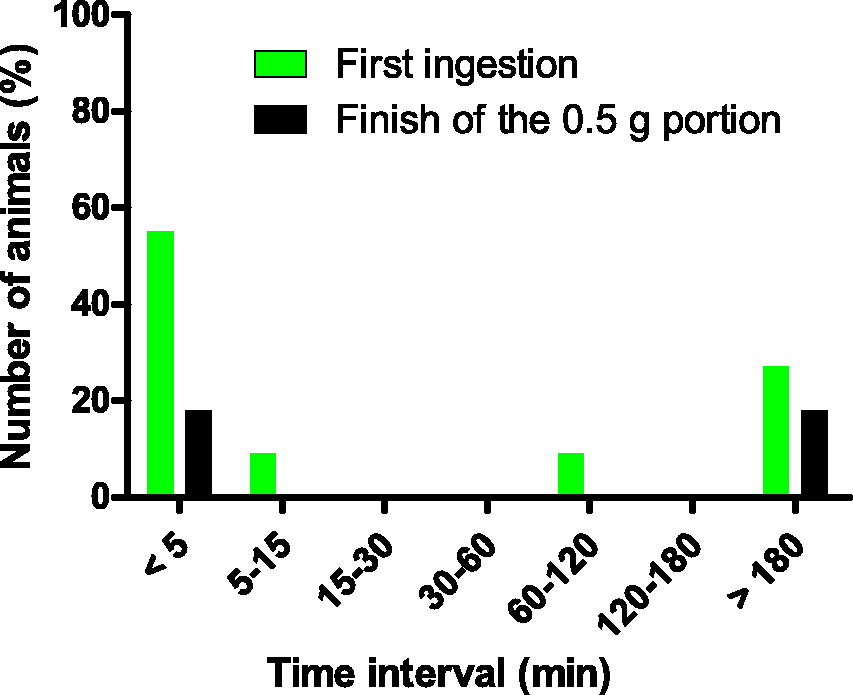
The amount of unmedicated MediGel ingested overnight for 15 individual animals over three days (Experiment 2) is presented in Figure 2. Twenty millilitres of unmedicated MediGel was presented to each mouse. The evaporation of MediGel in the IVCs during the period from 17:00 to 08:00 h was found to be 6.1 ± 0.3 mL (mean ± SEM), which meant that the maximum amount of gel available was 14 mL per mouse. The evaporation was accounted for in all calculations of ingested gel. It was found that the median amount of MediGel ingested during the first period (Day 1) was 5.4 mL, ranging from 2.4 to 13.9 mL. In the second period (Day 2), the median amount ingested was 3.4 mL, ranging from 0 to 13.9 mL. In the third period (Day 3), the median amount ingested was 3.9 mL, ranging from 0 to 13.9 mL. The ingested amount was significantly lower on Day 2 than on Day 1, as determined with Friedman’s test and with Dunn’s multiple comparison test.
The amount MediGel ingested overnight by 15 individual mice, repeated over three consecutive days.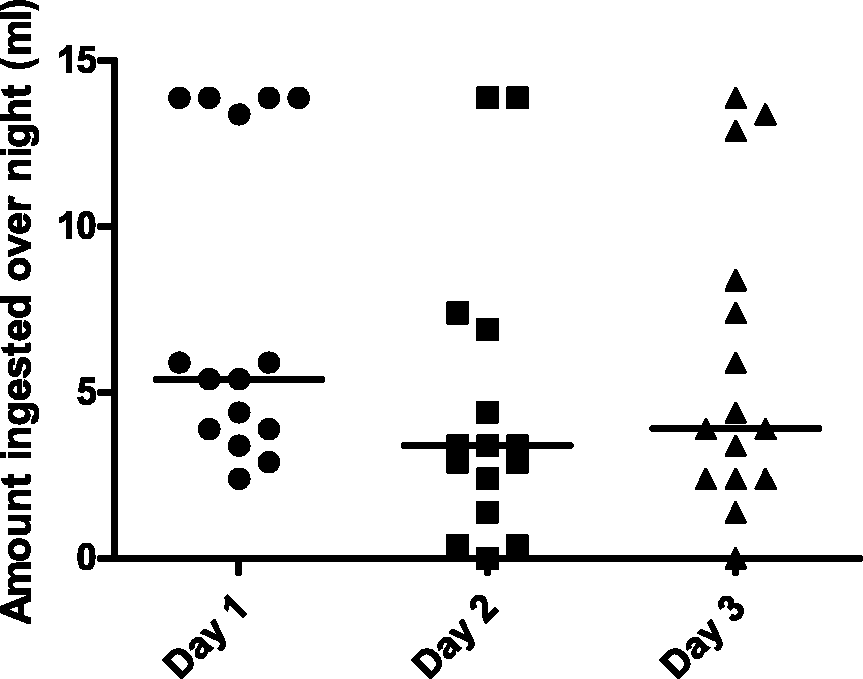
The serum concentration of buprenorphine after ingestion of medicated MediGel in Group A (15 µg/mL) and Group B (5 µg/mL) or SC injection in Group C (0.1 mg/kg) is presented in Figure 3. At the first sampling occasion (08:00 h), 15 h after presentation of medicated MediGel or one hour after SC injection, the serum concentration of buprenorphine in Group A was 7.18 ± 2.29 ng/mL (mean ± SEM). This was significantly higher than the concentrations in Group B (2.98 ± 0.93 ng/mL) and Group C (1.53 ± 0.23 ng/mL), as determined with a two-way ANOVA with Bonferroni’s post-test. At 14:00 h, the serum concentrations of buprenorphine were 3.15 ± 0.71 ng/mL for Group A, 0.99 ± 0.38 ng/mL for Group B, and 1.05 ± 0.16 ng/mL for Group C. At 20:00 h, the concentrations were 1.49 ± 0.46 ng/mL for Group A, 0.40 ± 0.21 ng/mL for Group B, and 0.53 ± 0.05 ng/mL for Group C. At the final sampling occasion (08:00 h the following morning), the concentrations were 0.87 ± 0.17 ng/mL for Group A, 0.09 ± 0.06 ng/mL for Group B, and 0.55 ± 0.10 ng/mL for Group C.
The serum concentration of buprenorphine 15–39 h after presentation of the MediGel–buprenorphine mixture ingestion or 1–25 h after subcutaneous (SC) injection.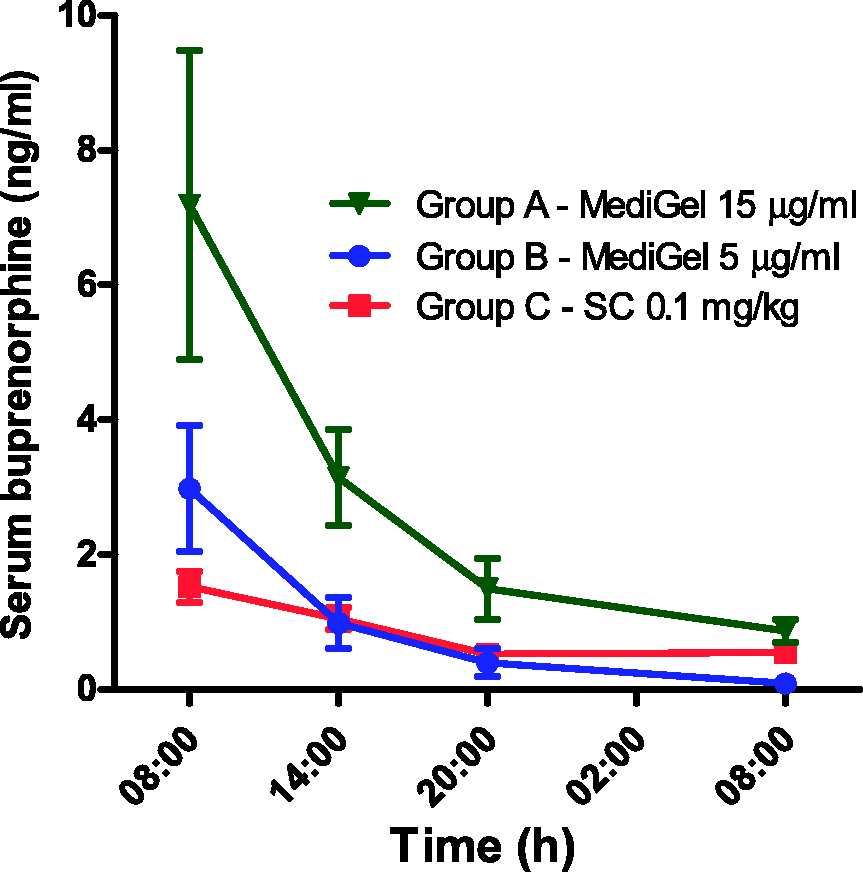
Discussion
Oral administration of buprenorphine by voluntary ingestion requires a vehicle that is well-liked by the animals and is thus readily ingested when presented. In our previous studies, we found that Nutella works well as a vehicle for administration of buprenorphine. In 2012, Jacobsen et al. demonstrated that both male and female C57BL/6 and BALB/c mice that received Nutella ingested the entire portion within one hour. 11 This meant that the dose of buprenorphine administered to each individual animal was readily controlled. Furthermore, the portions were ingested even when containing concentrations corresponding to 3 mg buprenorphine per kg body weight. In other studies, where buprenorphine has been mixed in drinking water, it has been shown that animals tend to decrease their daily water intake, probably due to the bitter taste of the drug, 19 which was not the case with Nutella. Thus, Nutella is a highly preferable vehicle for oral administration of buprenorphine. However, ingestion of Nutella may be problematic in certain circumstances, since it involves a slight alteration of the diet composition, which may affect metabolism and other physiological functions. Although this problem can be circumvented in many cases by providing control animals with the same treatment, there may be animal models where the ingredients of Nutella may have unwanted effects on the experimental data, or where the precise composition of diet is crucial for the model outcome. Therefore, the present study investigated whether MediGel could be applied as an alternative vehicle to Nutella.
The first aim was to investigate the willingness of laboratory mice to ingest MediGel, regarding the duration from introduction of the gel to first ingestion, as well as the amount ingested overnight. As expected, the mice were rather unwilling to ingest the gel at the first presentation. Mice are known to be neophobic, 23 and habituation to novel food items is often necessary. Unexpectedly, however, their willingness to ingest did not seem to increase over time. By contrast, the amount ingested decreased from Day 1 to Day 2. Furthermore, the variation between individual animals was very large, where some animals did not ingest any MediGel and other individuals ingested all of the presented gel. This issue is further discussed below.
The second aim was to investigate whether the gel would be suitable for oral administration of pre-emptive buprenorphine, by presenting medicated gel in the late afternoon the day before blood sampling. When subjecting a mouse to a surgical procedure or other painful invasive procedure it is beneficial to provide the analgesia pre-emptively.1,24 It is common practice to administer buprenorphine subcutaneously approximately 30 min, or orally one hour, prior to the procedure, to obtain sufficient serum concentrations of the drug.3,8,10,12 However, a mouse presented with buprenorphine mixed in a food item shortly before the procedure may not immediately ingest the item. Therefore, presenting the buprenorphine mix the afternoon before the day of the procedure should increase the likelihood that the mice will ingest the mix. Jacobsen et al. 11 demonstrated that mice presented with Nutella containing buprenorphine in doses of 1–3 mg/kg body weight had serum concentrations in a range considered to provide analgesia 17 h after presentation. 12 The same strategy was applied in the present study: providing the animals with a container of medicated gel with 5 or 15 µg/mL of buprenorphine for ingestion overnight corresponds to a dose of 1–3 mg/kg body weight (based on the mean intake of the mice), which should be an appropriate dosing regimen for mice. Subsequent serum concentrations were indeed similar to or higher than those after SC injection. Furthermore, previous studies have demonstrated that serum concentrations above 0.5 or 1.0 ng/mL are sufficient to give analgesia to mice;25,26 and based on this, the medicated gel used in the present study should in theory provide sufficient analgesia for 8–12 h after an intervention that was to occur 15 h after initiation of ingestion. However, as discussed above, the ingested amount varied largely between individual animals, and the time for ingestion was not fully controlled. In addition, not all of the gel missing the following morning had necessarily been ingested, since some animals might have spilled or discarded some of the gel. The subsequent serum concentrations at the time of the planned procedure may thus vary considerably (Figure 3) in comparison with those from SC injections. Another drawback is that individual animals that had ingested large amounts of gel with buprenorphine in a short time window may have overdosed, which could have a negative impact on the well-being of the animals as well as complicate the anaesthetic procedure and the subsequent recovery of the animals. However, no signs of overdosing, such as pica behavior or increased activity, 27 were observed in the animals. Thus, the intake of medicated MediGel was likely to have occurred over an extended period of time during the night.
The present investigation was made in male, outbred mice. The rationale for choosing outbred mice was to study the applicability of the gel delivery system in heterogeneous animals, representing a broader population. However, it is possible that inbred strains would behave differently, and therefore future studies should be undertaken to investigate this. The same is valid for sex differences, and so future studies should also include female animals.
In conclusion, MediGel was ingested by the mice, but general willingness was low and did not increase over a three-day period of time. Serum concentrations of buprenorphine 15 h after presentation of the buprenorphine–Medigel mix were similar to or higher than those after SC injection, but the variations were considerably larger. MediGel may thus be considered as an option for the oral administration of analgesics, but further studies are needed in order to improve the willingness of mice to ingest the gel and to control the amount of gel ingested before the vehicle is implemented for supplying perioperative analgesia.
Footnotes
Acknowledgements
The authors wish to thank the lab technicians Trine Marie Ahlman Glahder and Helle Porsdal for their helpfulness with the buprenorphine quantification. We would also like to thank Otto Kalliokoski for his valuable review of the manuscript.
Declaration of conflicting interests
The authors declare no competing interest. This research received no specific grants from any funding agency in the public, commercial or not-for-profit sectors.
