Abstract
Anaesthesia is used daily in fish experimental procedures; however, the use of an inadequate anaesthetic protocol can compromise not only the animal's welfare but also the reliability of results. The use of zebrafish (
The use of zebrafish (
Although the use of anaesthetics is important to ensure zebrafish welfare, these drugs can also have side-effects; 11 and it is essential to establish an anaesthetic regimen (doses, combinations) that suits each research procedure so as to minimize collateral effects. This review summarizes the anaesthetic and analgesic drugs that are used in laboratory adult zebrafish, anaesthesia protocols, anaesthesia depth, and recovery. Also, during all experiments it is of major importance to monitor zebrafish welfare, and for this, we propose a score sheet to monitor distress and pain.
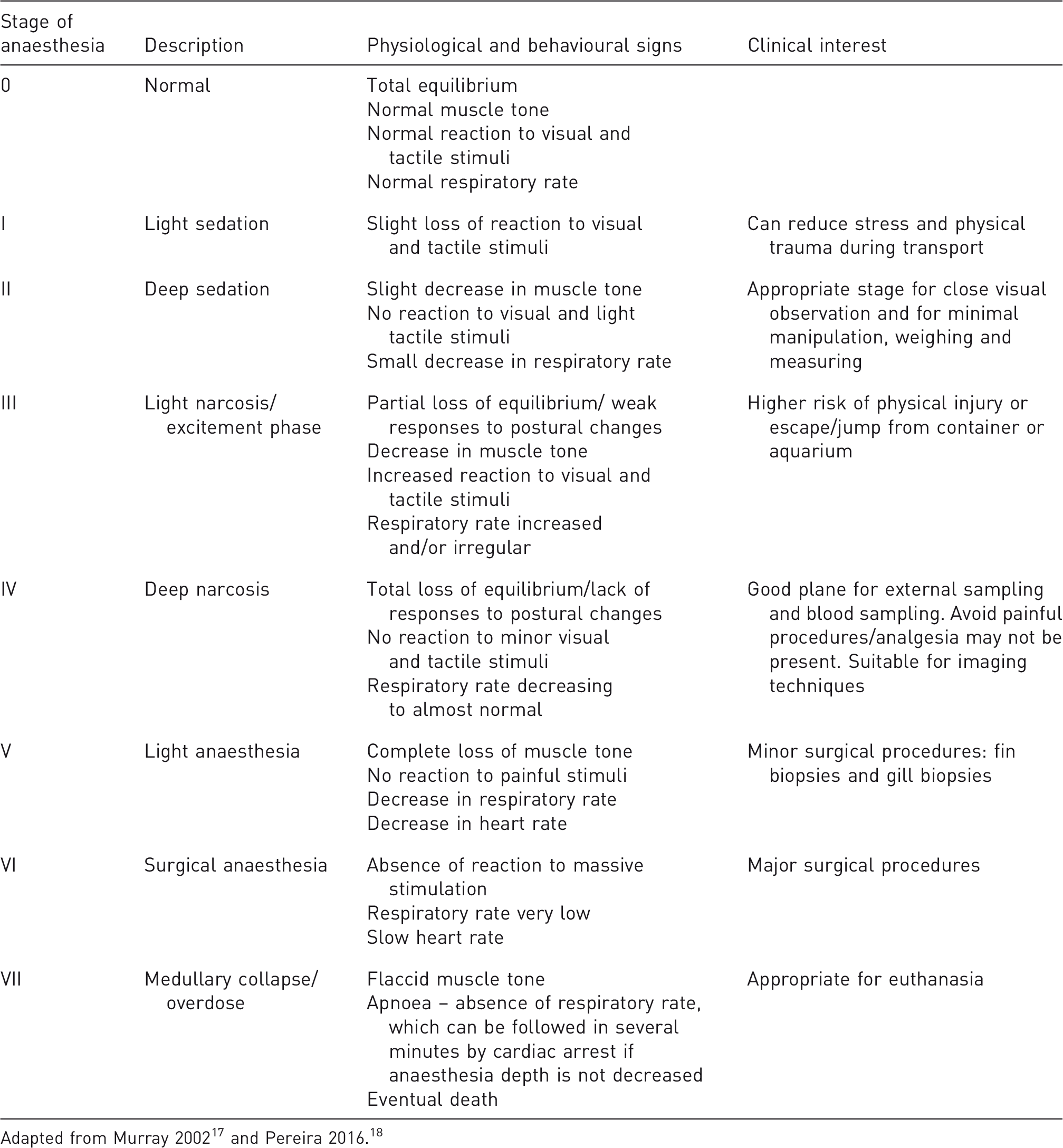
Anaesthesia
Anaesthesia in fish may be achieved by diluting the anaesthetics in water (inhalation anaesthesia), which induces a general anaesthesia with the drug being absorbed mainly through the gills but also through the skin in a few species.3,13,14 In fish, anaesthesia effects may vary depending on the administration route, pH, temperature, salinity, oxygenation, nitrogenous compounds and other water conditions. 3 Furthermore, anaesthesia depth and recovery depend on its duration, anaesthetic concentration, animal's body weight and metabolism, gill surface, fish health status, strain, age, and on the different particularities of fish species.3,11,15 Thus, anaesthesia trials with small numbers of fish, i.e. pilot studies, must be performed to determine the optimal dosage and exposure time prior to the establishment of protocols. In addition, proper training and supervision of fish anaesthesia are essential to avoid complications that can lead to death. Not only should anaesthesia be carefully monitored but also complete fish recovery,2,16 which has been disregarded in the literature.
Anaesthetic agents used in fish
The ideal anaesthetic agent should (i) be easy to administer and effective at low dose or exposure; (ii) be able to induce sedation or anaesthesia in less than 3 min with a minimum of stress; (iii) provide immobilization and effective analgesia throughout the procedure; (iv) induce a quick recovery from the anaesthetic stage within 5 min; and (v) induce no or minimal changes in physiology and behaviour during or after anaesthesia. 15 Ideally, the anaesthetic should be affordable, easily available, practical to use, and safe for the operator.
A recent international survey showed that around 93% of the surveyed participants used tricaine methanesulfonate (MS-222) for zebrafish anaesthesia. The use of 2-phenoxyethanol, benzocaine, clove oil, isoeugenol, etomidate, and lidocaine was also referred. 19 In the following, several anaesthetic agents are discussed.
MS-222
MS-222, classified as a local anaesthetic, is the most used inhalant anaesthetic in fish. MS-222 is highly absorbed through the gills and is administrated by bath, inducing general anaesthesia. Overall it is a safe anaesthetic for fish, 20 although there are some concerns regarding risks of overdose in deeper stages of anaesthesia and long duration procedures, mainly in small animals such as zebrafish.2,14,20,21 The anaesthetic solution of MS-222 should be buffered before use due to its acidic nature, which may cause aversion, epidermal and corneal lesions, and physiological alterations in the fish.14,22,23 Also, MS-222 can be toxic to humans.14,24
Clove oil
Eugenol is the major constituent (70–90% by weight) of clove oil extracted from the plant
Metomidate and etomidate
Metomidate and etomidate are non-barbiturate hypnotic drugs, and both are used as inhalant anaesthetics in a water bath in fish. These agents induce quick anaesthesia induction and recovery but they should only be used for minor procedures, as they do not induce a surgical anaesthetic stage or analgesia.3,15 Also, they alter fish physiology by suppressing cortisol production.11,15
Lidocaine
Lidocaine hydrochloride, a local anaesthetic and analgesic, 15 is used as an immersion anaesthetic in fish. It induces anaesthesia within one minute and the recovery is also rapid, taking about three to four times the induction period. 15 A high dose of lidocaine seems promising as an anaesthetic agent for surgical procedures but has a low margin of safety in zebrafish. 20 Thus, the addition of another agent, such as propofol, may potentiate this effect and reduce the dosage. 2 Moreover, perioperative analgesia with lidocaine seem to improve zebrafish welfare. 26
Propofol
Propofol is a sedative-hypnotic anaesthetic drug used for the induction and maintenance of general anaesthesia. 27 Propofol can either be injected or used in an anaesthetic bath. It is rapidly metabolized, thus lacking any cumulative effects. Although propofol is highly lipophilic, it can induce anaesthesia in a rapid and smooth way. Moreover, the recovery from anaesthesia is also quick and complete.2,28 Although the preliminary results seem promising 2 it may not be fully soluble in the water and more research is needed.
Ketamine
Ketamine is an injectable agent often used in mammals and induces a dissociative anaesthesia with some analgesia.29,30 In fish, it can either be injected or used in an anaesthetic bath. The use of ketamine in fish depends largely on the species, since it could cause incomplete anaesthesia, apnoea, prolonged recovery and excitement in salmonid species. 3 Ketamine has been revealed to be neurotoxic to zebrafish larvae,31,32 and to interfere with the development of embryos. 33
Isoflurane
Isoflurane is a hypnotic volatile drug routinely used in mammal anaesthesia. Studies evaluating volatile drugs in fish are scarce, since the anaesthetic depth is difficult to control, causing an overdose.3,20 Furthermore, anaesthetic preparation and anaesthesia should be conducted in a chemical fume hood for scavenging waste gas, and reducing the risk to the operator. These characteristics and the observed clinical effects render volatile anaesthetics less practicable in fish anaesthesia.3,20
Anaesthesia in laboratory adult zebrafish
Laboratory zebrafish has emerged as a powerful vertebrate model system in research.1,34 Despite this interest, research on welfare and refinement of procedures in this species are scarce. Pain and unexpected mortality due to incorrect anaesthesia in zebrafish can constitute a serious animal welfare issue, which increases data variability, imposing an important scientific and economical cost on research.
Anaesthetic and analgesic agents tested in laboratory adult zebrafish.

N/A: not applicable/not evaluated. All anaesthetic agents were administered by immersion, with exceptions: IM: intramuscular injection. *Paul Schroeder, personal communication, 19 July 2016.
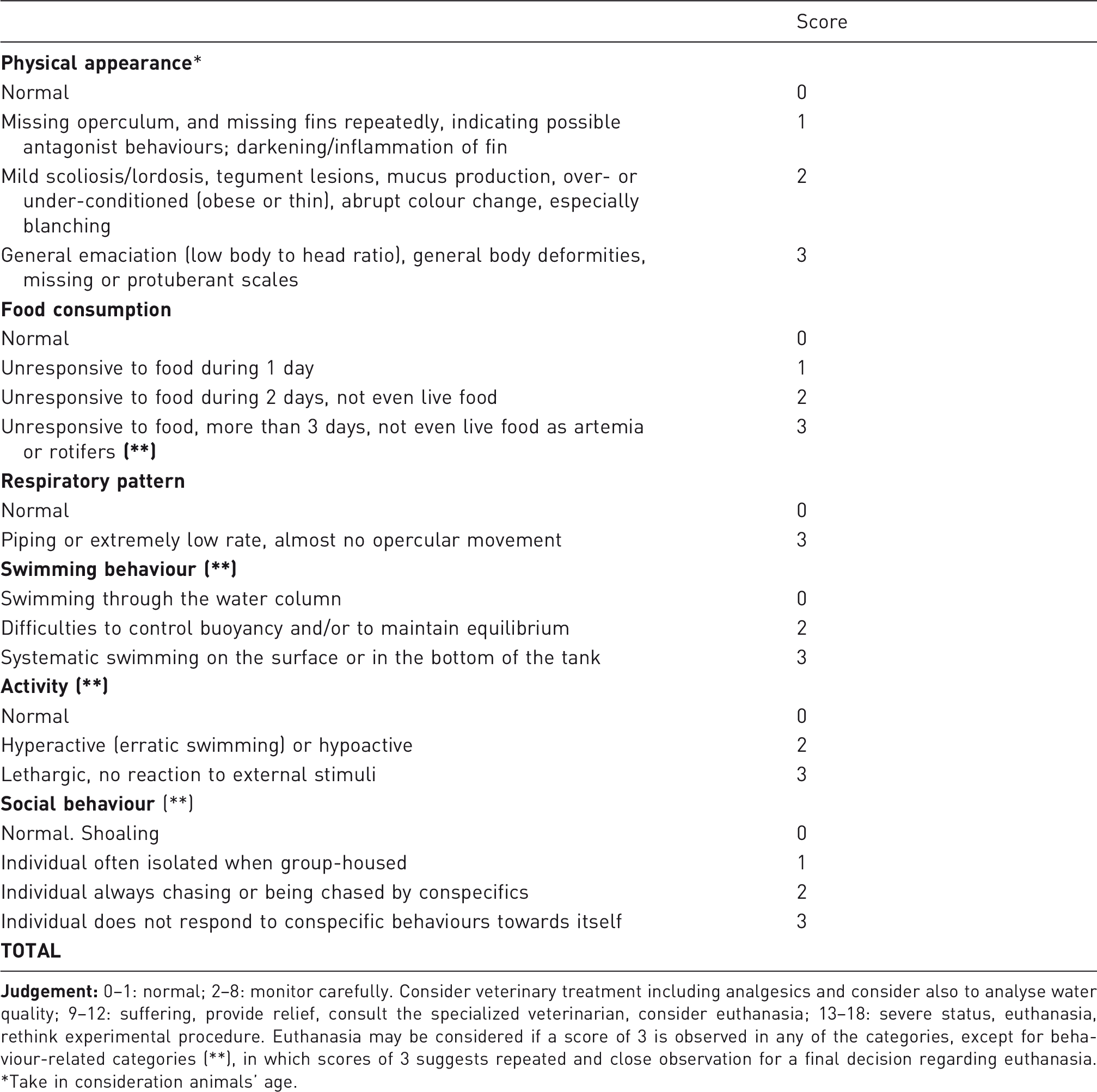
Monitoring zebrafish welfare
In addition to the importance of a good anaesthetic protocol to ensure fish welfare during experimental procedures, animals should also be monitored for distress, discomfort and pain throughout the experimental protocol and maintenance. Fish have demonstrated to react consistently to noxious chemical stimuli and present reliable phenotypes of stress, fear, and anxiety.40,41 Thus, the use of analgesic drugs in fish during and after painful experimental procedures should be considered, especially due to the fact that not all anaesthetic agents available for fish have proved to have adequate analgesic properties. 3 The information concerning the use of analgesics in zebrafish is extremely scarce (Table 2). Furthermore, the available analgesics tested in fish are administered intramuscularly and/or by the intraperitoneal route, 11 which make it difficult to perform in small fish, such as zebrafish, or in a large number of animals. 26 Lidocaine has recently been described as a promising analgesic for zebrafish, 26 which is an important step in the development of analgesic protocols to be administered in a water bath.
Proposal of a pain and distress score sheet for laboratory adult zebrafish.
Concluding remarks
The use of a suitable anaesthetic protocol able to produce anaesthesia with effective analgesia is an important refinement for painful procedures. Studies addressing the effects of anaesthetics in zebrafish are variable and lack important information such as the time during which the anaesthetic stage can be maintained without secondary effects, a clear description of the anaesthesia stage achieved with a certain dose, and data about recovery. This lack of information can have a negative impact on zebrafish welfare when investigators are not experienced in the use of zebrafish anaesthesia. In addition, analgesia in zebrafish is another topic that needs further investigation in order to refine analgesic protocols during experimental procedures and for postoperative pain management, thus ensuring zebrafish welfare and reliable data collection.
In general, MS-222 is a good anaesthetic, justifying its wide use in zebrafish, 19 but further refinement could be valuable when long duration procedures are required.11,21 Furthermore, two studies have described zebrafish aversion towards this anaesthetic.22,36 Thus, finding other anaesthetic protocols are advisable, for example, the use of anaesthetic combinations to decrease individual concentrations and the risk of unwanted side-effects. Also, lidocaine seems to be the most promising analgesic to be used in zebrafish, however, its efficacy needs to be tested in different painful procedures and experimental situations.
Footnotes
Acknowledgements
The authors would like to thank Hugo Santos (BOGA, CIIMAR – Interdisciplinary Centre of Marine and Environmental Research), Ana Cristina Borges, Maysa Franco e Liliana Vale (IGC – Instituto Gulbenkian de Ciência), Petra Pintado e Fábio Valério (CEDOC – NOVA Medical School), and Sandra Martins (Champalimaud Foundation) for their critical comments on the scoring sheet table.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Postdoctoral Fellowship SFRH/BPD/103006/2014 funded by FCT, and by the Fellowship BI/CITAB/UTAD/VET/2015 supported by: European Investment Funds by FEDER/COMPETE/POCI – Operacional Competitiveness and Internacionalization Programme, under Project POCI-01-0145-FEDER-006958 and National Funds by FCT – Portuguese Foundation for Science and Technology, under Project UID/AGR/04033/2013.