Abstract
Gradual filling of a chamber with carbon dioxide is currently listed by the Canadian Council on Animal Care guidelines as a conditionally acceptable method of euthanasia for rats. Behavioural evidence suggests, however, that exposure to carbon dioxide gas is aversive. Isoflurane is less aversive than carbon dioxide and may be a viable alternative, though objective data are lacking for the period leading up to loss of consciousness. It has been shown that during negative states, such as pain and distress, rats produce ultrasonic vocalizations. The objective of this study was to detect ultrasonic vocalizations during exposure to carbon dioxide gas or isoflurane as an indicator of a negative state. Specialized recording equipment, with a frequency detection range of 10 to 200 kHz, was used to register these calls during administration of each agent. Nine female Sprague–Dawley rats were exposed to either carbon dioxide or isoflurane on two different occasions. All rats vocalized in the ultrasonic range (30 to 70 kHz) during exposure to carbon dioxide. When exposed to isoflurane, no calls were detected from any of the animals. The frequent occurrence of ultrasonic vocalizations during carbon dioxide exposure suggests that the common practice of carbon dioxide euthanasia is aversive to rats and that isoflurane may be a preferable alternative.
With over 2.5 million animals being used annually in Canada and the European Union, rats are one of the most common species in biomedical research.1,2 The great majority of these animals will be euthanized using an overdose of carbon dioxide gas. Despite evidence from behavioural studies showing that carbon dioxide gas is aversive to rats,3,4 the practice remains popular because it is cheap, effective, widely available, and poses a minimal health risk to personnel. Euthanasia of rats with isoflurane as the sole agent 5 or as part of a two-stage process 6 has been deemed acceptable. Current national guidelines are largely based on evidence from approach avoidance studies, with the unavoidable limitation that data cannot be collected between the onset of aversion and loss of consciousness.4,7 Vocalizing in the ultrasonic range is a strategy rats have developed to adapt to high predatory pressure. Therefore ultrasonic vocalization (USV) allows communication with conspecifics but is inaudible to many predators. 8 USVs in rats have been shown to reflect a negative state (such as pain or distress) 9 and may provide a valuable tool for identifying pain and distress during euthanasia. 10 In general, lower frequency USVs (18–32 kHz, so-called ‘22 kHz calls’) have been associated with negative states, and higher frequency USVs (32–92 kHz, so-called ‘50 kHz calls’) with positive states. Lower frequency USVs act as alarm calls and have been associated with pain, distress, and fear.9,11 For these reasons, the recording of USV has been suggested as a measure of pain and fear in laboratory animals.12,13 Oliveira and Barros assessed USV as a behavioural measure of pain and recorded a significantly increased number of low frequency USVs from rats during the formalin test. 9 During exposure to carbon dioxide with rats, at a rate of 17.25% chamber volume per minute, Niel and Weary showed an increase in the occurrence of USVs. 10 By contrast, high frequency USVs (50 kHz calls) have been recorded during purported positive states such as tickling and mating.14,15 We conducted a pilot study to evaluate the application of USV recording as a reflection of pain or distress (or both) experienced by rats during exposure to carbon dioxide or isoflurane, building on previous work by Niel and Weary. 10
Nine female Sprague–Dawley rats (Health Sciences Animal Resource Centre, University of Calgary, Calgary, Canada) between the ages of 7 to 9 weeks old and weighing 195–312 g were used in this experiment. The animals were housed in groups of two or three in a standard rat cage (47 × 25 × 21 cm) with commercially available wood shavings (Aspen chip, NEPCO, Warrensburg, NY, USA) and plastic tubes for enrichment. The rats received water and food (Prolab 2500 Rodent 5P14, LabDiet, PMI Nutrition International, St Louis, MO, USA) ad libitum and were kept on a 12 h light–dark cycle (lights off at 19:00 h). All the experiments were performed between 15:00 h and 18:00 h with a minimum of 24 h between treatments to allow the rats to recover.
Six animals were exposed to each gas on different occasions. The order of these treatments was determined by a random draw. Three other rats received only carbon dioxide as a treatment. Each animal was tested individually and exposed to the gas in a purpose-made closed Perspex test chamber (3000 mL volume) while remaining within sight of its cage mate(s) throughout the experiment. The test chamber had three openings fitted for instrument connection (microphone, gas analyzer and gas inflow tube). The following standardized protocol was used: 5 min acclimatization period in room air, then 5 min with oxygen inflow at one litre per minute (L/min), which equals 30% chamber volume per minute (CV/min), and finally (once oxygen concentration had returned to 21%) exposure to the treatment agent. Carbon dioxide (100%) or isoflurane (2.5% carried in oxygen) was delivered at a flow rate of 1.0 L/min (30% CV/min). Sound recordings were performed using an ultrasound microphone (Condenser ultrasound microphone and UltraSoundGate CM16/CMPA, Avisoft Bioacoustics, Berlin, Germany) from the time the animal was placed in the test chamber. Carbon dioxide and oxygen were delivered with agent-specific calibrated flowmeters, and the isoflurane and oxygen concentrations were monitored with a calibrated gas analyzer (Datex Ohmeda s/5 monitor, GE Health Care, Waukesha, WI, USA). The experiment was terminated and the animal was allowed to recover following confirmation of a loss of righting reflex by tilting the test chamber until the animal was in dorsal recumbency. This was performed once gross purposeful movement ceased and the animal became recumbent in the test chamber. Vaginal swabs were taken during recovery and smears were prepared for cytological examination (slides were air-dried and stained with Diff Quik) to determine if there was a correlation between the stage of oestrous cycle and the presence or absence of USVs. All recordings were visually inspected twice for USV identification by a blinded observer. Vaginal smears were also evaluated by a blinded observer. Descriptive statistics are reported and data are shown as median and range. The decision to use female rats was driven by a wish to reduce animal use. It was preferable to use surplus stock from our institutional colony rather than order in animals from an external source. Consequently, this limited availability to female animals.
This experimental protocol was reviewed and approved by the Animal Care Committee at the University of Calgary, Canada, which operates under the auspices of the Canadian Council on Animal Care.
Control recordings made during the acclimatization period and oxygen inflow (performed before each gas exposure) resulted in one rat vocalizing once during both oxygen exposures. Data from this animal were not included in the analysis as conclusions could not be drawn from the presence or absence of any calls during the subsequent treatment (isoflurane or carbon dioxide) phase. It would be impossible to interpret such behaviour as being due to the different treatments and not the sound of gas inflow. Exposure to isoflurane did not elicit USVs from any rat (0 out of 6 animals). By contrast, during exposure to carbon dioxide, we recorded USVs from all animals (8 out of 8 animals). Of these, a median of two calls per rat (range 1 to 8) were recorded. The frequency ranged from 30 to 70 kHz (median 51 kHz) with a median duration of 0.05 s (0.014 to 0.26 s). The onset of USVs ranged from 81.9 to 152.1 s (median 133.8 s) after carbon dioxide gas flow began. With the 1 L/min flow rate employed and chamber volume of 3 L, this equated to a calculated chamber concentration change of greater than 95% (more than three time constants). The results are summarized in Table 1 and a typical USV is shown in Figure 1. Vaginal cytology revealed that the vocalizing rats were at various stages in the oestrous cycle and that there were no associations with the occurrence of USVs.
Example of a typical ultrasonic vocalization emitted by a female rat exposed to 100% carbon dioxide at a fill rate of 30% chamber volume per minute. All rats exposed to carbon dioxide gas (8 out of 8 animals) produced ultrasonic vocalizations. No rats (0 out of 6 animals) exposed to isoflurane (2.5% carried in oxygen at a fill rate of 30% chamber volume per minute) produced ultrasonic vocalizations. Occurrence and properties of ultrasonic vocalizations during exposure to room air, oxygen alone, isoflurane (2.5% carried in oxygen) and carbon dioxide (100% carbon dioxide at a fill rate of 30% chamber volume per minute) in female rats. Frequency and duration are reported as median (range). NA = not applicable.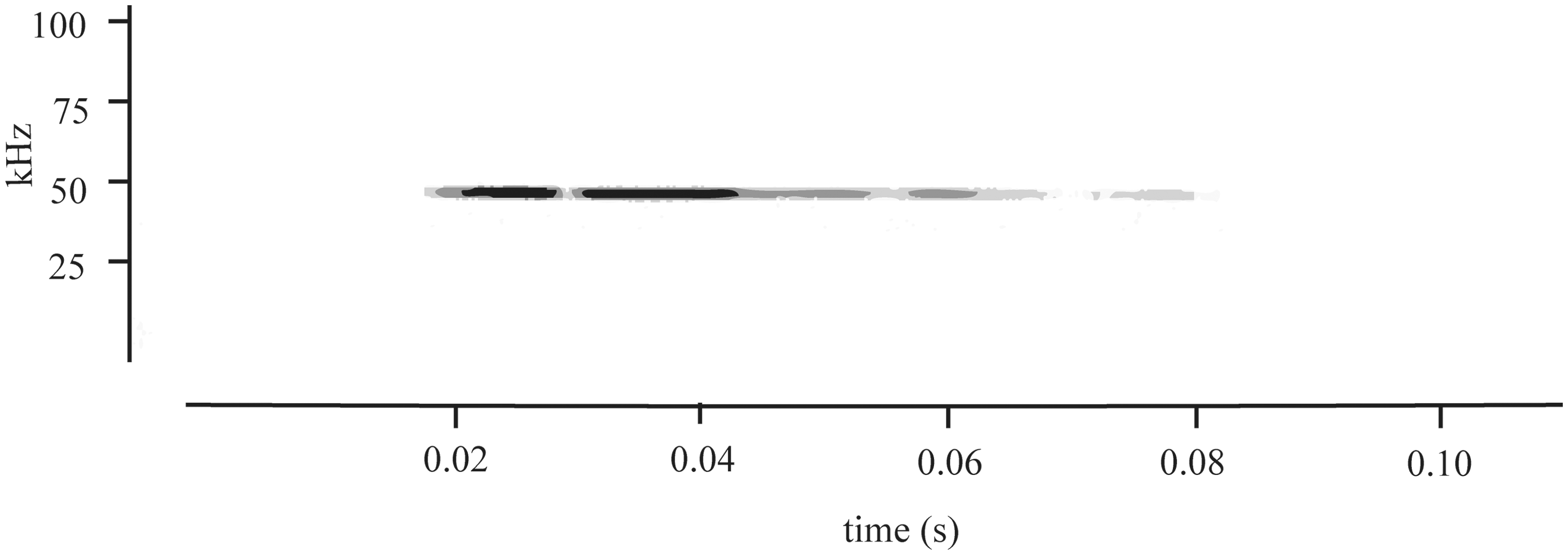

To our knowledge, these preliminary data show, for the first time, that female rats vocalize during exposure to carbon dioxide but not when exposed to isoflurane. Though 22 kHz calls are usually associated with negative states such as distress or pain, our findings in the context of evidence from approach avoidance studies indicate that USV in a higher frequency range may also be reflective of these states. Work by Niel and Weary using adult male Sprague–Dawley rats, has reported findings similar to our data, with 50 kHz calls being identified in response to carbon dioxide exposure. 10 Additionally, a series of experiments by Wöhr et al. have shown that 50 kHz calls are not strictly attributed to positive experiences but are also emitted when rats are separated from a cage mate, during an open field test, elevated plus maze test and introduction to a novel cage. 16 These data indicate that 50 kHz calls are not restricted to positive states. Taken together with the results of approach avoidance studies showing that isoflurane is aversive3,4 our data indicate that it may be a preferable alternative to carbon dioxide. This work does not allow generalization to male animals or other rodents, and further work is necessary to address these issues in addition to determining whether different administration techniques or alternative agents provide more humane alternatives. As millions of rodents are euthanized by carbon dioxide each year the implications are widespread.
Footnotes
Funding
This study was funded by the Margaret Gunn Endowment for Animal Research from the University of Calgary.
