Abstract
Researchers performing experiments on animals should always strive towards the refinement of experiments, minimization of stress and provision of better animal welfare. An adequate analgesic strategy is important to improve post-operative recovery and welfare in laboratory rats and mice. In addition, it is desirable to provide post-operative analgesia using methods that are minimally invasive and stressful. This study investigated the antinociceptive effects of orally administered buprenorphine ingested in Nutella® in comparison with subcutaneous buprenorphine administration. By exposing the animal to a thermal stimulus using a hot plate, significant antinociceptive effects of voluntarily ingested buprenorphine administered in Nutella® were demonstrated. This was evident at doses of 1.0 mg/kg 60 and 120 min post administration (P < 0.01), although antinociceptive effects were not as marked as with subcutaneous administration, and had a later onset. It is advised to administer the oral formulation of buprenorphine in Nutella® in a 10-fold higher dose, as well as approximately 60 min earlier, than with the more commonly employed subcutaneous route of administration.
Surgical procedures in preclinical research using laboratory rats are associated with post-operative stress due to tissue damage from the surgery, anaesthesia and post-operative pain. Although the mammalian stress response is an evolutionary coping mechanism, the stress associated with some recovery procedures may be detrimental to the animal. 1 Minimizing stress and pain in laboratory animals is important for animal welfare reasons, but also because excessive stress may influence experimental outcomes, due to larger inter-individual variations in various biological endpoints. 2 Post-operative analgesics may also modify model outcomes, compared with the same procedure without analgesics, via modifications on stress and immune response.3,4 In some instances, this latter situation may be undesirable, but withholding animals from analgesic treatment cannot be reasonably justified based on unproven observations.
The most common analgesic used for systemic perioperative analgesia in laboratory rodents is the partial μ-opioid receptor agonist buprenorphine. 5 Buprenorphine is known to possess analgesic/antinociceptive effects in most analgesiometric tests and to improve post-operative recovery, even though it may also induce uncommon side-effects, such as pica, sedation, decreased appetite, gastrointestinal distress and hyperlocomotion.6–10 Traditionally, buprenorphine is administered subcutaneously, but interest in finding less invasive routes of administration, such as drinking-water, 11 food-pellets or voluntary ingestion of palatable formulations like flavoured jello 12 or sticky nut paste 13 is increasing. However, administration of specific buprenorphine doses can be difficult to gauge, as animals undergoing surgery may have reduced food and water intake post-operatively, and these effects may be exacerbated by the above-mentioned opioid-induced side-effects. Furthermore, ad libitum administration could conceivably induce analgesic tolerance, 11 although its bitter taste may in fact reduce the palatability and decrease the intake when offered in, for example, water. 14 Recent evidence suggests that the voluntary administration of buprenorphine in a sticky nut paste (such as Nutella®) is an effective analgesic strategy that reduces stress and gives long-lasting serum concentrations to a higher extent than the traditional subcutaneous administration route.15–18 However, the extent and duration for which this mode of administration produces sufficient analgesic coverage have been questioned, and the data published are somewhat inconclusive.14,19–21 The aim of the present study was to compare the antinociceptive effects of buprenorphine administered orally via ingestion in Nutella® with subcutaneous administration of buprenorphine. The nociceptive hot-plate test was used to measure reflex nociceptive responses to noxious thermal stimulation to give a surrogate indication of analgesic effect. In the present study, antinociception and analgesia were regarded as equal entities, but it should be kept in mind that a measured antinociceptive effect at a specific dose might not directly translate to effects on post-operative pain. 22
Materials and methods
Animals
The following study was performed in accordance with the Danish legislation (Law No. 474 of 15 May 2014 and Order No. 88 of 30 January 2013) regulating experiments on animals, which is in compliance with European Directive 2010/63/EU. All experimental protocols involving animals were ethically reviewed and approved by the Danish Council for Animal Experiments.
A total of 100 male Sprague–Dawley rats (Crl:CD (SD)) from Charles River, Sulzfeld, Germany, at age seven weeks at the time of delivery, and approximately eight weeks at the time of experiment, weighing 238 ± 6 g (mean ± standard error of the mean [SEM]) at the time of the experiments were used. Only male rats were used in this study, since it was intended that the study’s findings would be compared with previous studies where only males were used. We acknowledge that this is a limitation to the study, since female subjects may have different baseline pain sensitivities and different responses to analgesic treatment.23–26
Housing
All animals were housed in pairs throughout the study in transparent Tecniplast polycarbonate Makrolon type III, high open cages (38 × 22 × 17 cm) from Scanbur, Karlslunde, Denmark. Environmental enrichment consisting of aspen wood chewing blocks (S-Bricks; Tapvei, Paekna, Estonia), paper-wool shavings (LBS Biotechnology, Horley, UK) for nesting material and red Rat Retreats™ (Bio-Serv, Flemington, NJ, US) for hiding were used. For bedding, aspen chips (Tapvei) were used. Food (Altromin 1324; Brogaarden, Lynge, Denmark) and water were available ad libitum, and water was changed on a weekly basis. The light–dark cycle was 12:12 h with lights on from 06:00 h; the room temperature was set to 20 ± 2℃; and the relative humidity was 30–70%. The animals were acclimatized to their surroundings for at least one week after arrival from the vendor, following which they were moved to the testing laboratory at least three days prior to baseline testing. The animals remained in that room during the entire study.
Hot-plate test
The hot-plate procedure is well described in the literature.27–31 For all hot-plate experiments a hot plate (Ugo Basile Srl, Gemonio, Italy) was used with a pre-set plate temperature of 52.5℃ as recommended for rats. 28 A cut-off time was set at 60 s, and a baseline test was performed one day prior to the experiment. The time from when the rat was placed on the hot plate until the first sign of discomfort from the thermal stimulus was recorded immediately, i.e. licking, shaking, or stepping of the paws, was observed. Bannon and Malmberg 28 have recommended focusing only on hind-paw activity, as (i) fore-paw activity is often associated with grooming behaviour and (ii) constant contact between front paws and the hot plate is rarely present. We accepted fore-paw responses in the current study, with these two criteria in mind; albeit the vast majority of the responses were hind-paw ones. No animals were removed prematurely from either study, and none showed signs of thermally-induced damage to the paws once either study was completed.
Experimental design
Our first experiment determined which dose of subcutaneously administered buprenorphine would provide optimal analgesic coverage for use as a comparator in the subsequent experiment, as well as in a related study investigating analgesic treatments in a surgically-induced rat model of neuropathic pain (not shown here). Fifty rats were divided into five groups (n = 10) by randomly allocating cages to the different groups without observing or handling the rats prior to allocation. Thus, all the cage-mates were given the same treatment. One group received only saline and the other four groups were injected subcutaneously with 0.015, 0.05, 0.1 or 0.3 mg/kg of buprenorphine. Baseline testing was performed on all the animals one day prior to treatment to allow for any compensation in possible baseline differences between groups of naïve animals if required. Antinociceptive effects were assessed one hour after buprenorphine injection. Treatments were blinded, as one person administered the injections and another female observer assessed all of the response latencies.
Subsequently, a second experiment compared different doses of buprenorphine in Nutella®, with the vehicle (Nutella) and the subcutaneous dose determined in Experiment 1. Fifty rats were divided into five groups (n = 10), randomly allocated to treatment in pairs/cage, without observing or handling the rats prior to allocation. Cage-mates were allocated to the same treatment, in order to only expose Nutella®-treated animals to possible stress effects by the habituation procedure, explained below. One group received only Nutella® (vehicle), and the other four received either 0.1 mg/kg of buprenorphine administered subcutaneously or 0.5, 1.0 or 2.0 mg/kg of buprenorphine in Nutella®. The Nutella® treatments and the Nutella® vehicle were administered in a blinded fashion. A baseline test was performed one day prior to treatment and antinociceptive effects were then assessed at 30, 60, 120, 240, 360, 480 min post treatment. The same female observer in Experiment 1 recorded all response latencies in this second experiment.
At the end of each experiment, all animals were euthanized in a purpose-made chamber using a gradual fill with 80% carbon dioxide and 20% oxygen.
Drugs and administration
The preparation for voluntary ingestion of buprenorphine (buprenorphine–hydrochloride powder; Sigma–Aldrich, Inc, Saint Louis, MO, USA) mixed in Nutella® (Ferrero, Pino Torinese, Italy) was carried out according to the recommendations made by Abelson et al. 13 The rats were offered 2 g of Nutella®/kg body weight and were habituated to pure Nutella® ingestion twice a day for three days prior to the actual test day. The Nutella® was placed on a piece of adhesive tape on the cage wall, and the animals were returned in their cages to their familiar surroundings. For the first day, the two rats were habituated together, and on the last two days prior to testing, one animal was moved to another cage while ingesting the Nutella. For each habituation session, some bedding material and the Rat Retreat® hiding material were moved together with the animal in question in order to minimize the stress associated with different environmental cues. They were then administered their individual amount of Nutella® while separated, before being returned to their partner. On the last day as well as the day of the experiment, upon separation, all animals ingested their entire individual offered dose of Nutella® within 3–5 min of separation before being returned to their home cage. All analgesics administered in the subcutaneous injection were diluted in saline (sodium chloride 9 mg/mL; Fresenius Kabi, Bad Homburg, Germany) so that the concentration corresponded with an administration of 5 mL/kg body weight solution.
Statistical analysis
Animals were randomly allocated to treatment groups, and each experiment was performed by the same investigator who was unaware of the treatment, to ensure blinding. The number of animals per group was determined by a power analysis 32 where a 50% increase in mean hot-plate latency after treatment was considered to be biologically relevant; the standard deviation was estimated to be approximately 0.5 times the mean; the level of significance (α) was set to 5%; and the desired power (1 -β) was set to 80%. Input data for means and standard deviation were based on previous experience from our laboratory. Statistical analysis was performed using GraphPad Prism version 5 (GraphPad Software Inc, La Jolla, CA, USA), and SigmaPlot 11.0 (Systat Software Inc, San Jose, CA, USA). Using a Shapiro–Wilk normality test (Graph Pad Prism), all groups in the study passed the normality test. Statistical comparisons of mean latency per group indicating overall treatment effects between groups and primarily comparison to vehicle group at the specific time-point in the given experiments were made using one-way analysis of variance (ANOVA) (Experiment 1) or two-way repeated measures ANOVA (Experiment 2), followed by all pairwise comparisons using Bonferroni’s post-treatment analysis. P < 0.05 was considered to be statistically significant.
Results
Efficacy of subcutaneous buprenorphine
Subcutaneous administration of buprenorphine (0.015–0.3 mg/kg) produced an antinociceptive effect as observed by an increase in the latency response to acute thermal stimulation as shown in Figure 1 (F(4,49) = 9.745, P = < 0.0001, as determined by one-way ANOVA with Bonferroni’s post-test). No statistically significant differences were observed between buprenorphine-treated groups, or between baseline levels for individual groups. The 0.1 mg/kg dose was selected for Experiment 2, since we wanted to ensure a clear antinociceptive effect for comparison with administration via Nutella®.
Effect of buprenorphine on acute nociceptive behaviour in non-injured rats. Rats were administered either buprenorphine (0.015–0.3 mg/kg subcutaneously) or vehicle 60 min prior to measuring the response latency to thermal stimulation (52.5℃) in the hot-plate test. Data are presented as mean ± SEM, and the line indicates pre-injection baseline across groups. All groups: n = 10 rats. *P < 0.05, **P < 0.01, ***P < 0.001 vs vehicle (one-way ANOVA with Bonferroni’s multiple comparison test).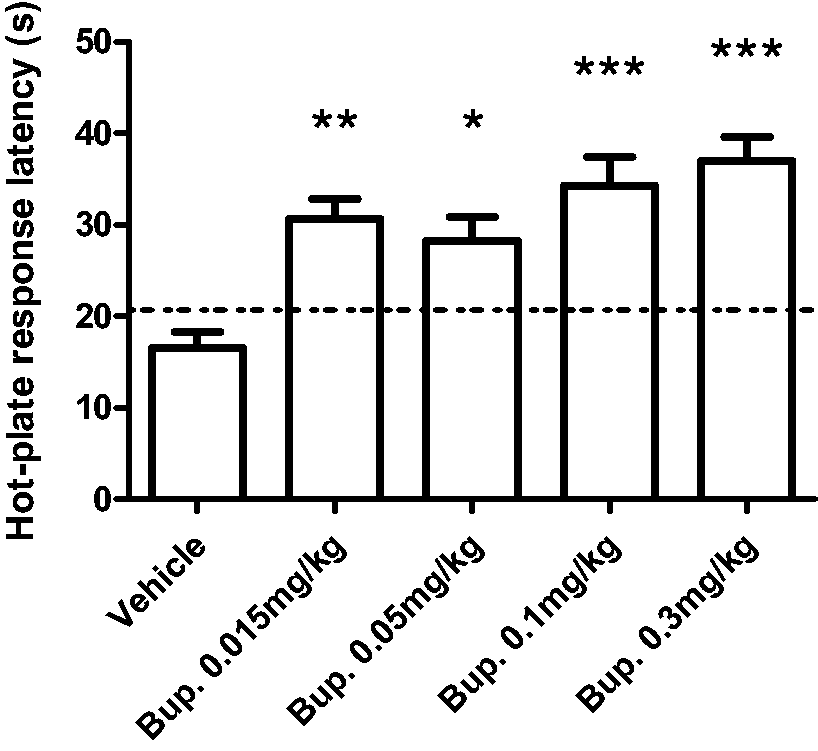
Comparison of buprenorphine in Nutella® and subcutaneous buprenorphine
Figure 2 shows that the latency to respond to noxious thermal stimulation in rats was significantly affected by buprenorphine treatment and by time (F(4,49) = 27.72, P = < 0.0001 and F(4,49) = 5.025, P = 0.002 respectively, as determined by two-way repeated measures ANOVA). More specifically, post-treatment analysis revealed that when 1 mg/kg of buprenorphine was administered in Nutella®, hot-plate latency was significantly increased at 60 and 120 min compared with vehicle (P < 0.01 and P < 0.05, respectively) and with the corresponding baseline latency response. Surprisingly, administration of 2 mg/kg of buprenorphine administered in Nutella® only increased the latency response at 120 min post-treatment (P < 0.05 vs vehicle and P < 0.05 vs the corresponding baseline). By contrast, rats treated with the low dose of 0.5 mg/kg of buprenorphine in Nutella® did not show any differences in response latency compared with the vehicle at any time-point or the corresponding pre-treatment baseline response. Thus, the 1 mg/kg dose was determined as the minimally effective dose. Moreover, no effects of time were measured for the vehicle (Nutella) group, demonstrating that repeated testing using this paradigm did not appear to sensitize the rats to noxious stimulation.
Effects of buprenorphine in Nutella® on acute nociceptive behaviour in non-injured rats. Rats were administered either with vehicle (Nutella®), buprenorphine (0.5, 1.0 or 2.0 mg/kg orally [p.o.] in Nutella®) or buprenorphine (0.1 mg/kg subcutaneously [s.c.]) in saline prior to measuring the response latency to thermal stimulation (52.5℃) in the hot-plate test. Data are presented as mean ± SEM at baseline and then at 30, 60, 120, 240, 360 and 480 min after treatment. Statistical significance compared with vehicle at the given time-point, determined by two-way repeated measures ANOVA with Bonferroni’s post-test. *P < 0.05, **P < 0.01, ***P < 0.001. Statistical significance compared with treatment-specific baseline; +P < 0.05, ++P < 0.01, +++P < 0.001.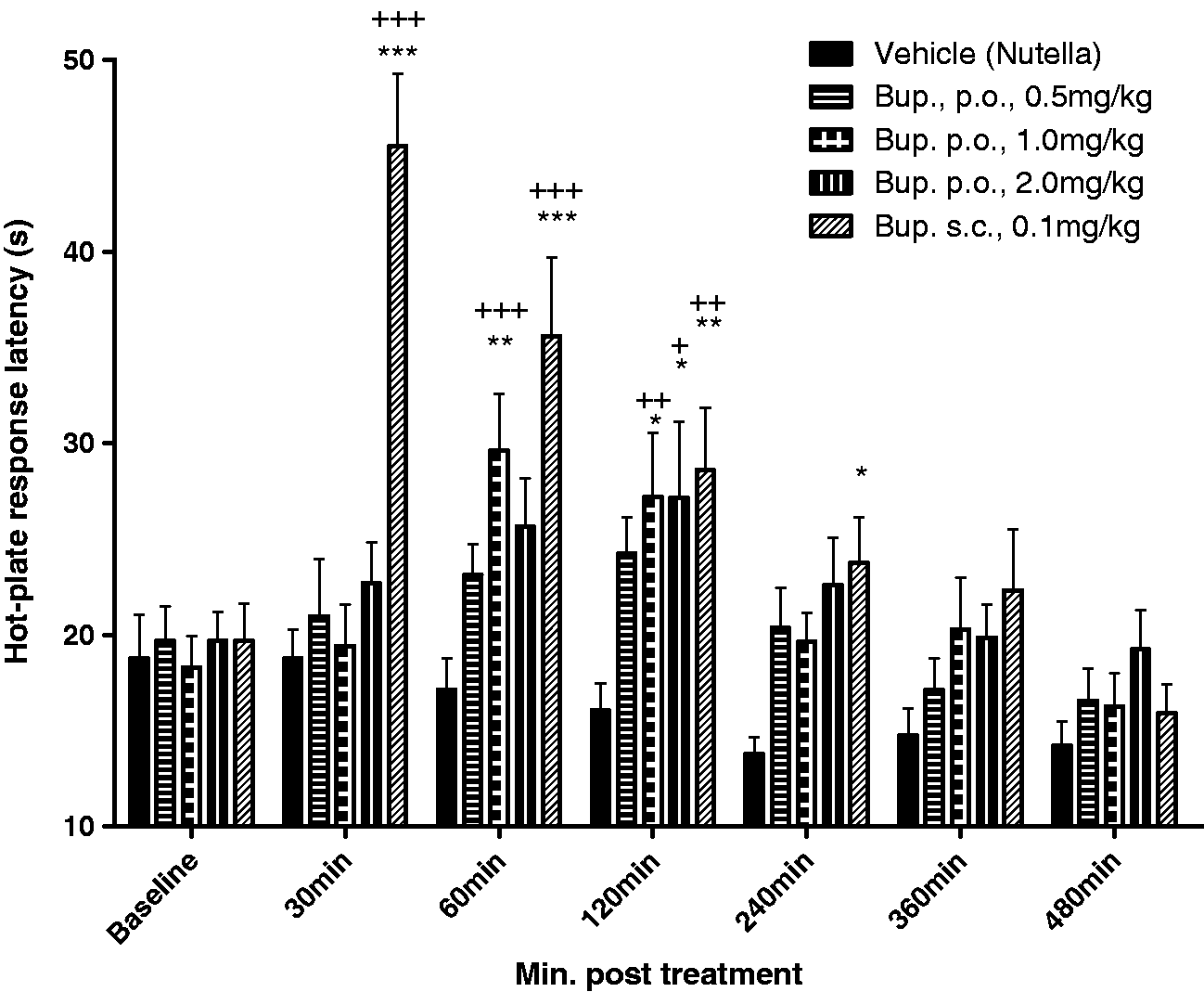
Similar to the first experiment, administration of subcutaneous buprenorphine 0.1 mg/kg produced a robust increase in the response latency within 30 min after injection (P < 0.001 vs corresponding vehicle and baseline, respectively). This effect gradually diminished over time, but still remained significantly different from the vehicle (P < 0.05) 240 min after injection.
Discussion
The hot-plate experiments presented here describe acute antinociceptive effects of the partial μ-opioid receptor agonist buprenorphine in naïve rats, following two related administration protocols. The first experiment was designed to detect a robust and reproducible antinociceptive effect of subcutaneous buprenorphine to be used in subsequent in-house studies using male Sprague–Dawley rats. Accordingly, we found that over the tested range of 0.015 to 0.3 mg/kg, a dose-dependent antinociception was observed. This partially replicated other observations, 33 wherein Kouya and co-workers observed that subcutaneous buprenorphine (0.03–0.3 mg/kg) was similarly dose-dependent, but with a shorter duration of action evident for up to only 60 min post injection. 33 Although we noted that the lowest dose of buprenorphine (0.015 mg/kg) increased the latency response under the conditions tested, we decided to use the 0.1 mg/kg dose in the subsequent experiment based on the following: (i) low doses of opioids may induce hyperlocomotion,10,34,35 with the net result that time taken to reach noxious temperature thresholds for any given limb will take longer to achieve from the moment the rat is placed on the hot plate; (ii) the efficacy obtained with 0.05 mg/kg is not as robust as that from 0.1 mg/kg buprenorphine; and (iii) other groups have recommended subcutaneous doses of 0.1 mg/kg 36 or 0.1–0.5 mg/kg every 8–12 h 22 for optimal analgesia.
In the second experiment, buprenorphine administered orally in Nutella® had a slower onset of action than that administered subcutaneously when both were compared with the vehicle, and achieved a lower level of efficacy than the subcutaneous buprenorphine-treated group. Buprenorphine has a plasma half-life of 2.8 h after intravenous administration in the rat, regardless of the dose. 37 Moreover, it has previously been reported that plasma concentrations of subcutaneous administered buprenorphine (0.05 mg/kg) are high at least one hour after administration, but diminish thereafter and are close to zero after 6 h. 17 By contrast, voluntary ingested buprenorphine gives rise to a peak in plasma concentration at 2 h, and continues at a steady state from 6–14 h, 17 likely due to a slower absorption from the gastrointestinal tract, and potentially also by saturation of the hepatic metabolism of buprenorphine on the first-pass metabolism. 17 Our pharmacodynamic measures obtained in the hot-plate test do not easily reconcile with the pharmacokinetic observations, but it is noteworthy that other studies have also suggested that both the antinociceptive and analgesic effects of oral and subcutaneous buprenorphine may not last for the 8–12 h duration which is often indicated.22,38
Novel sustained–release formulations of buprenorphine can induce analgesic and antinociceptive coverage for a duration of 72 h post injection,39,40 and can also be expected to mitigate the primary shortcomings of the buprenorphine protocols used here (e.g. duration of action). However a study using mice has reported skin lesions and ulcerations in 12 of 15 mice injected with the sustained–release formulation, which was not seen with subcutaneous buprenorphine. 41 Evidently, comparatively high doses of buprenorphine in Nutella® appear to be necessary in order to mediate a significant increase in the nociceptive threshold. This is supported by standard pharmacokinetic indexes which suggest that oral doses of opioid should be in the order of 10 times the parenteral doses as the bioavailability is estimated to be approximately 5–10% following first-pass metabolism when administered by gavage. 42 This is generally consistent with the findings in our hot-plate experiments, where the minimum effective dose of buprenorphine in Nutella® was 1 mg/kg. Indeed, doses of 5–10 mg/kg might be required to produce a more robust antinociception, as some have suggested, 14 but might also decrease the palatability of the oral mixture, making animals more reluctant to ingest it voluntarily. 14 We did not find that palatability of the buprenorphine in Nutella® to be a factor which limited obtainable efficacy over the dose range tested.
Issues of efficacy aside, a perceived advantage of voluntary analgesic ingestion in rodents is clear from a series of studies comparing the effects of buprenorphine in Nutella® with subcutaneous buprenorphine or treatment with local anaesthetics on various post-operative recovery measures including faecal- and plasma-corticosterone levels, water consumption and body weight maintenance, with a dose of only 0.4 mg/kg buprenorphine in Nutella®.15–18 Besides the advantage of being a minimally invasive route of administration, it may also be of benefit to have absorption across the sublingual mucosa, to help avoid first-pass metabolism and to give a more rapid onset of action as compared with absorption only across the intestinal mucosa. 43
It is possible that the general lack of robust efficacy obtained with the administration of buprenorphine either subcutaneously or in Nutella® reflects reports that buprenorphine is less effective in nociceptive tests involving heat stimuli as compared with tests where the stimulus is chemical or mechanical in nature.6,44 The hot plate procedure used here associates the withdrawal latency before and after experimental manipulation to modulation of the noxious thermal threshold, i.e. the antinociceptive effect of a given drug. 45 It is simple to perform and involves minimal handling, and in contrast to mechanical testing, it is objective, repeatable 46 and highly useful in characterizing opioid receptor agonists. 47 However, repeated testing tends to decrease latency responses over time, 48 a trend seen in the vehicle (Nutella® only) group in the second experiment. Notably, this can occur with the use of hot or ambient room-temperature plates, and thereby does not appear to be a result of either noxious learning or thermally-induced sensitization,46,48–50 and it is reported to be less likely to occur when the repeated tests occur during a single session. 50 It should also be noted that the experimental paradigm used here in naïve rats does not induce pathophysiological changes in pain circuits such as those associated with, for example, algogen-induced hind-paw inflammation, 51 which are known to modulate efficacy of opioid agonists. 52 Overall, thermal analgesiometry may not be the most sensitive way of assessing analgesic efficacy of buprenorphine,44,45 which could partly explain the limited time-span of the antinociceptive effect in the current study. Nevertheless, despite these shortcomings it has been possible to compare the efficacy of buprenorphine administered via different routes/formulations. Other methods including pathophysiology relevant to post-operative pain and inflammation could be required to detect more subtle effects.15,16,18
Other methodological considerations that are likely, and that have been reported to influence the antinociceptive or analgesic efficacy and potency of opioids such as buprenorphine in rodents include (i) the strain or stock in question;19,23,25 (ii) whether subjects are male or female;21,23–26 and (iii) vendor supplying the animals. 53 We cannot therefore directly extrapolate the dose findings in the current study, which explored only male Sprague–Dawleys from one vendor, to all rats used in all laboratories; but we can make a principal recommendation that when buprenorphine is administered voluntarily in Nutella® it should be in 10-fold higher doses than those which would normally be administered subcutaneously.
In conclusion, voluntarily ingested buprenorphine administered in Nutella® mediates antinociception with a relatively slow onset of action and needs to be administered at a dose of at least 10-fold higher than subcutaneous buprenorphine. Antinociceptive effects were evident at doses of 1.0 mg/kg in Nutella®, however lower doses may be suggested and may be sufficient in analgesic effect when treating post-operative pain as demonstrated by others.15–18,21 We found that the effective dose and duration of action of buprenorphine in either formulation differed in some aspects from those recommended in the literature, and we highlight a number of facets (e.g. nociceptive testing method, animal strain/sex/vendor) that could contribute to our findings.21,23–26,53 Given the importance of good welfare practice, we would urge other investigators to perform similar pilot studies when implementing a new post-operative analgesic regimen in a given laboratory in order to secure sufficient analgesic coverage. Finally, we would suggest that the choice of analgesic regimen should align with the severity of the procedure being performed. While voluntary ingestion of buprenorphine in Nutella® is clearly a minimally invasive route of administration, if the experimental manipulation affects other behavioural domains such as appetite, inadequate analgesic coverage might subject the animal to other clinical symptoms.
Footnotes
Acknowledgements
The author(s) would like to thank Rie Christensen for technical assistance with the experiments. The present study was supported by the Centre for Applied Laboratory Animal Research (CALAR) and by H Lundbeck A/S, Copenhagen, Denmark.
Ethical statement
All experimental protocols involving animals were ethically reviewed and approved by the Danish Council for Animal Experiments. The protocols were also reviewed by the local animal welfare body at H Lundbeck A/S.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
