Abstract
Ulcerative dermatitis (UD) is an idiopathic, spontaneous and progressive disease typically affecting C57BL/6 aged mice with an unknown aetiopathogenesis. For this study, we evaluated 25 cases of UD in C57BL/6NCrl-Tg(HMGA1P6)1Pg mice. Formalin-fixed, paraffin-embedded skin samples were submitted to morphological investigations. Immunohistochemical analysis was performed to characterize and quantify inflammatory cells using CD3, CD45/B220, CD4, CD8 and IL-17 antibodies. Mast cell-bound IgE was investigated by immunofluorescence, whereas serum and cryopreserved skin samples were collected for molecular analysis. Student's t-test (two-tailed) was performed to assess significant differences between the two groups. Affected skin showed extensive areas of ulceration and diffuse, severe and mixed inflammatory infiltrates. No relevant changes were observed in control mice. Immunohistochemical analysis showed a predominant CD3 + CD4 + leukocyte population with fewer CD45/B220 and IL-17 immunolabelled cells and mast cell-bound IgE. Increases in TNFα, IL-1β and Il-6 mRNA expression were observed in the skin of affected animals compared to controls. Serum TNFα and IL-6 did not vary between affected and control mice. Inflammatory infiltrates and cytokine expression were consistent with both Th2/IgE and Th17 differentiation, a typical pattern of a type I hypersensitivity reaction. Overall, our data suggest an allergic-based aetiopathogenesis of UD in C57BL/6NCrl-Tg(HMGA1P6)1Pg mice.
Introduction
Ulcerative dermatitis (UD) is an idiopathic, spontaneous and debilitating condition found in laboratory mice, typically affecting aged C57BL/6 (B6) mice or genetically engineered mice (GEM) on a C57BL/6 background.1,2 Clinically, UD is characterized by intense pruritus and scratching leading to ulcer formation predominantly on the head, neck or upper thorax.3–5 The intense pruritus and the rapid progression of ulcerative lesions may have severe and devastating effects on animal welfare, often resulting in secondary bacterial infections and formation of scars and skin contractures that may impair species-typical behaviours and mobility.1,2,4,6
UD is speculated to be a multifactorial disease,1,7,8 and several predisposing factors, such as environment,1,2,4,6,9 diet,1,2,10–12 season,1,2,4,6 age at weaning,1,2,13 alopecia,1,2,4,6 sex,1,2,4,9 immune complex vasculitis, 3 and deficiencies in vitamin A metabolism, 6 have been implicated in the pathogenesis of this condition. The detrimental effects on animal welfare and the possible negative consequences on research usually require humane euthanasia of the affected mice. The underlying mechanisms contributing to the initiation, development and progression of UD are still poorly understood. Accordingly, many attempted treatments have been unsuccessful.1,2,4 Recently, Adams et al. suggested that the use of toenail trims may be a highly effective treatment option for UD lesions, 14 resulting in complete lesion resolution with no recurrence in approximately 90% of the mice treated.
To our knowledge, an exhaustive characterization of the inflammatory infiltrate in spontaneous UD is still lacking. Our findings revealed several similarities to the features of a cutaneous type I hypersensitivity reaction, suggesting a possible allergic aetiology for UD in mice. The purpose of this work is to analyse, thorough histological, immunohistochemical and molecular examinations, UD lesions observed in C57BL/6NCrl-Tg(HMGA1P6)1Pg mice. Our ultimate goal was to add some information and missing puzzle pieces regarding the true nature of the inflammatory response associated with this condition and thereby provide new, useful insights into UD pathogenesis.
Animals, materials and methods
The affected mice were chosen from an experimental colony of an inbred strain of C57BL/6NCrl-Tg(HMGA1P6)1Pg mice that were produced by Esposito and colleagues as a part of a study on the role of HMGA1 pseudogenes and cancer. 15
Generation and housing of C57BL/6NCrl-Tg(HMGA1P6)1Pg mice
Briefly, the HMGA1P6 expression plasmid was excised with SalI and HindIII restriction endonucleases by cleaving 10 µg of the plasmid. The fragment was purified from SeaKem GTG agarose (avoiding exposure to UV light) using the Qbiogene Geneclean Spinkit, then dialysed 24 h against 2 l microinjection buffer (10 mM Tris-HCl pH 7.2, 0.1 mM EDTA), and diluted to a concentration of 4 ng/µl. The DNA was injected in three sessions into C57BL/6N-derived zygotes. For this purpose, C57BL/6N female mice (bred at PolyGene from parents obtained from Charles River) were super-ovulated at 28–34 days of age and mated in the PolyGene mouse facility to C57BL/6N breeder males, also originally obtained from Charles River. Pups were biopsied at weaning and analysed for transgene integration by PCR.
All animals were housed in a standardized non-barrier facility at the Istituto Nazionale Tumori ‘Pascale’ (Naples, Italy). Animal care and use as well as all the experimental procedures were in accordance with the Guide for the Care and Use of Laboratory Animals. 16
All animal procedures were reviewed and approved by the Institutional Ethical Committee of the Istituto Nazionale dei Tumori, Fondazione Pascale, Naples, Italy, in accordance with Italian regulations for experimentations on animals. Mice (1–3) of the same sex were housed in wire-topped plastic cages with static filter tops (Tecniplast, Buguggiate, Italy) and a red plastic igloo-style shelter (Tecniplast, Buguggiate, Italy) on aspen wood bedding (Scobistre; Mucedola srl, Settimo Milanese, Italy). The mice were housed on a 12:12-h light:dark cycle and cages were changed twice a week. Plastic cages and bottles were washed in hot (62–82℃) water with detergent–disinfectant solution and were periodically changed to avoid collateral effects on the animals. Mice received a pelleted, autoclavable rodent diet (long term maintenance diet, cat. n° 4RF18; Mucedola srl, Settimo Milanese, Italy) and acidified water (HCl at pH 3.5) ad libitum. Room temperature was maintained between 20.0 and 24.0℃, and relative humidity was maintained between 55% and 65%. Temperature and humidity were constantly recorded (Poseidon2 3266, HWgroup, Casale Monferrato, Italy). The health status of the animals in the facility was monitored annually based on FELASA Recommendations (FELASA Health Monitoring Profile 375M, ENVIGORMS Srl, S. Pietro al Natisone, Udine, Italy). Two 5-week-old Hsd:ICR CD-1 female sentinel mice were exposed to dirty bedding weekly for 10 weeks before being submitted to the commercial vendor (ENVIGORMS Srl, S. Pietro al Natisone, Udine, Italy) for serology, parasitology and necropsy (see Supplemental Table 1 online). The maximal number of cages per sentinel was 24.
Ante- and post-mortem evaluation
Mice were routinely examined by the facility veterinarian (DDB) at least once a week for general condition, and any mice presenting with skin lesions underwent a thorough examination. Data collected from clinical evaluation included sex, age, and a detailed description of skin lesions. In addition, a visual examination of pruritus was performed, as indicated by Hampton et al. 2 UD was diagnosed, when appropriate, on the basis of lesion location (thorax, head and neck) and the presence of typical crusts or areas of ulceration. UD lesions were subsequently scored using a system recently developed by Hampton et al. that considered scratching number, type of lesion, length of lesion and regions affected. 2 Clinical scoring was performed once at the time of diagnosis. When affected mice met endpoint criteria (more than 10% of the total body surface area affected, debilitating scars and/or lesions localized to the face and neck) they were deeply anaesthetized with 5% isoflurane in 100% oxygen (Fluovac Anesthetizing System cod. 726425, Harvard Apparatus, Cambridge, UK). Subsequently, intra-cardiac puncture for terminal blood collection was performed using a 23–25-gauge needle. Following blood collection, euthanasia was ensured by cervical dislocation according to the American Veterinary Medical Association (AVMA) guidelines. 17 The same procedure for blood collection and euthanasia was followed for 10 healthy control animals.
Over an 18-month period, 25 out of 60 C57BL/6NCrl-Tg(HMGA1P6)1Pg mice (41.6%) with lesions compatible with UD were euthanized, and a full post-mortem examination including histopathology and molecular analysis of the dermal lesion was performed. To exclude the possible presence of ectoparasites, the fur of the affected animals was sampled from the scapular area, the ventral cervical region, the axillary area, the inguinal area, and the dorsal rump. The fur was subsequently placed on an adhesive tape and then examined under a light microscope using the 10 × and 40 × objectives.
Histopathology and Immunohistochemical analysis
Standardized skin biopsies from affected animals comprised the area of ulceration and the unaffected adjacent epidermis. Samples were preserved in 10% neutral buffered formalin (code no. 05-01007Q, Bio-Optica, Milan, Italy), dehydrated and embedded in paraffin (code no. 06-7920, Bio-Optica, Milan, Italy). Tissue sections were stained with haematoxylin and eosin (HE) for morphology, toluidine blue (TB) (code no. T3260, Merck KGaA, Darmstadt, Germany) for mast cell count and histochemical stains such as periodic acid-Schiff (PAS) (code no. 04-130802, Bio-Optica, Milan, Italy), methenamine silver (Grocott) (code no. 04-043823, Bio-Optica, Milan, Italy) and Gram (code no. 04-1008002, Bio-Optica, Milan, Italy), the latter two for histological identification of fungi and bacteria, respectively. Special histochemical stains were performed according to manufacturer's instruction. A total of 10 unaffected skin biopsies from C57BL/6JOlaHsd mice (n = 5, all females from ENVIGORMS Srl, S. Pietro al Natisone, Udine, Italy) and from C57BL/6NCrl-Tg(HMGA1P6)1Pg mice (n = 5, 3 females and 2 males) were used as negative controls. The age of B6 healthy mice was 6 months; the mean age of C57BL/6NCrl-Tg(HMGA1P6)1Pg healthy controls was 16 months (13–20 ± 1.49 SD).
The severity of dermal lesions was histologically assessed as follows (see Supplemental Table 2): 2 (1) mild, focal or few foci of inflammation confined to the superficial dermis; (2) moderate, multiple foci of inflammation extending to the deep dermis; (3) severe, regionally extensive foci of inflammation occasionally extending to the subcutis; (4) marked, with inflammatory foci extending to a larger aspect of the subcutis compared to the severe lesions.
Regarding the number of mast cells (toluidine blue), this was calculated as the mean value of five randomly selected high-power fields (magnification 40x) in each group (UD vs healthy controls), as previously described. 18
For immunohistochemistry (IHC), 4-μm-thick sections were processed with the MACH1 Universal HRP Polymer Detection Kit (Biocare Medical LLC, Concord, CA) as previously described. 19 Briefly, skin tissues were mounted on positively charged glass slides (Bio-Optica, Milan). Antigen retrieval pre-treatments were performed using a heat-induced epitope retrieval (HIER) citrate buffer pH 6.0 (Bio-Optica, Milan, Italy) for 20 min at 98℃; peroxide block was applied for 15 min at room temperature, and then the sections were incubated for 30 min with background sniper (Biocare Medical LLC). The primary antibodies were diluted in phosphate-buffered saline (PBS) and incubated overnight at 4℃. Horseradish peroxidase (HRP) polymer was added for 30 min at room temperature. After every step, the sections were washed in 0.01 M PBS (pH 7.2–7.4). The reaction was revealed by using 3,30-diaminobenzidine (DAB) chromogen diluted in DAB substrate buffer. Finally, sections were counterstained in Carazzi's haematoxylin (code n. 05-06012/L, Bio-Optica, Milan, Italy). Primary antibodies included rabbit monoclonal to CD3 (ab16669,Abcam, Cambridge, UK) diluted 1:200, rabbit polyclonal to CD45/B220 as a marker for B cells (ab10558 Abcam, Cambridge, UK) diluted 1:1000, 20 rabbit polyclonal to CD4 (NBP1-19371, Novus Biological, Littleton, CO, USA) diluted 1:200, rabbit polyclonal to CD8 (ab203035, Abcam, Cambridge, UK) diluted 1:100, and goat polyclonal to IL-17 (Santa Cruz Biotechnology, Santa Cruz, CA) diluted 1:200. For IL-17 antibody, the slides were developed using the Vectastain Elite ABC goat IgG kit (Vector Laboratories, Burlingame, CA) following a previously described protocol. 18 To test the specificity of staining, two negative controls were simultaneously performed: incubating one section with PBS, omitting the primary antibody, and the other one with an irrelevant and unspecific IgG.
To identify the predominant lymphocyte subtype, the immunohistochemical scoring was independently performed by two pathologists (DDB and OP) with a concordance rate of 95%. The number of immune-labelled cells was calculated for each section and each antibody in both groups (UD vs healthy controls) and is expressed as the mean value of 10 high power fields.
For immunofluorescence staining, the pre-treatment steps were the same as those used for immunoperoxidase labelling following a previously described procedure.21,22 The primary monoclonal rat anti-mouse IgE antibody (RMGE00, Invitrogen, Rockford, USA) was diluted 1:100 in PBS and incubated overnight at 4℃. Slides were washed three times in PBS and incubated with a fluorescein isothiocyanate (FITC)-conjugated pure goat anti-rat secondary antibody (Alexa Fluor® 488 conjugated, A-11029 Thermo Fisher, Rockford, IL, USA) diluted 1:100 for 2 h at room temperature. Slides were rinsed three times with PBS and coverslipped using Vectashield mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI) (H-1200 Vector Laboratories, Burlingame, CA).
For scanning and photo capture, a laser scanning microscope (DM6B Leica, Wetzlar, Germany) was used. The different frames were scanned separately, with appropriate installation of the optical path for excitation and emission of each scan according to the manufacturer's instructions.
RNA extraction and real-time semi-quantitative PCR
Total RNA was extracted from skin tissues using TRIzol Reagent (Bio-Rad Laboratories) according to the manufacturer's instructions, and cDNA was synthesized as previously described. 23 Each sample contained 1–100 ngcDNA in 2 × QuantiTect SYBR-Green PCR Master Mix and primers, tumour necrosis factor α (TNFα), interleukin 6 (IL6), interleukin 1β (IL1β) and transforming growth factor beta (TGF-β) (Qiagen, Hilden, Germany) in a final volume of 50 μl. The relative amount of each studied mRNA was normalized to GAPDH, and data were analysed, according to the 2−ΔΔCT method. 23
TNF-α and IL-6 determination in serum
Blood samples obtained from mice by terminal intra-cardiac puncture were collected in serum separator tubes. The serum was separated by centrifugation at 10,000g for 5 min at 20℃ (Heraeus Biofuge Pico, DJB Labcare Ltd, England). Serum samples were then aliquoted into Eppendorf tubes and stored at −80℃ until further analysis. To evaluate TNF-α and IL-6 in mouse serum as markers of systemic inflammation, sera were diluted 1:10 with the provided diluent 1 × (e-Bioscience, San Diego, CA), and the enzyme-linked immunosorbent assay (ELISA, e-Biosciences, San Diego, CA) was performed according to the manufacturer's instructions. The results are expressed as pg/ml as previously reported. 24
Statistical Analysis
Data are presented as the mean ± standard deviation (SD). Statistical analyses between the two groups were performed using Student's t-test (two-tailed) using GraphPad Prism (GraphPad Software, San Diego, CA). Statistical significance was set at p < 0.05.
Results
Ante- and post-mortem evaluation
UD was clinically diagnosed in 25 out of 60 C57BL/6NCrl-Tg(HMGA1P6)1Pg mice (41.6%). The mean age of onset was 16 months (13–20 ± 1,49 SD), with no significant predilection for sex (11 males and 14 females). We observed a season-associated increase in UD cases, with a predominance of disease (19/25) during the late spring and summer (see Supplemental Figure 1). Affected animals showed intense pruritus leading to almost uncontrollable scratching and the formation of the characteristic areas of ulceration associated with marked dermal fibrosis. Macroscopically, lesions varied from coalescing crusts to irregularly shaped areas of ulceration extending mostly to the dorsal cervical region, dorsal and ventral thorax (Figure 1). Severe lesions gravely affected the animal welfare, resulting in debilitation and, in some cases, altered posture and mobility. The clinical scoring system for the ulcerative lesions was applied and resulted as follows: 8% mild (2/25), 64% moderate (16/25) and 28% severe (7/25). Final health monitoring of the C57BL/6NCrl-Tg(HMGA1P6)1Pg mouse colony reported negative results for ectoparasites as well as for viral, bacterial and fungal agents.
Ulcerative dermatitis, skin, mouse. Typical presentation of murine ulcerative dermatitis showing focal areas of ulceration with fibrosis on the neck (a), upper and dorsal thorax (b), cervical and posterior areas (c).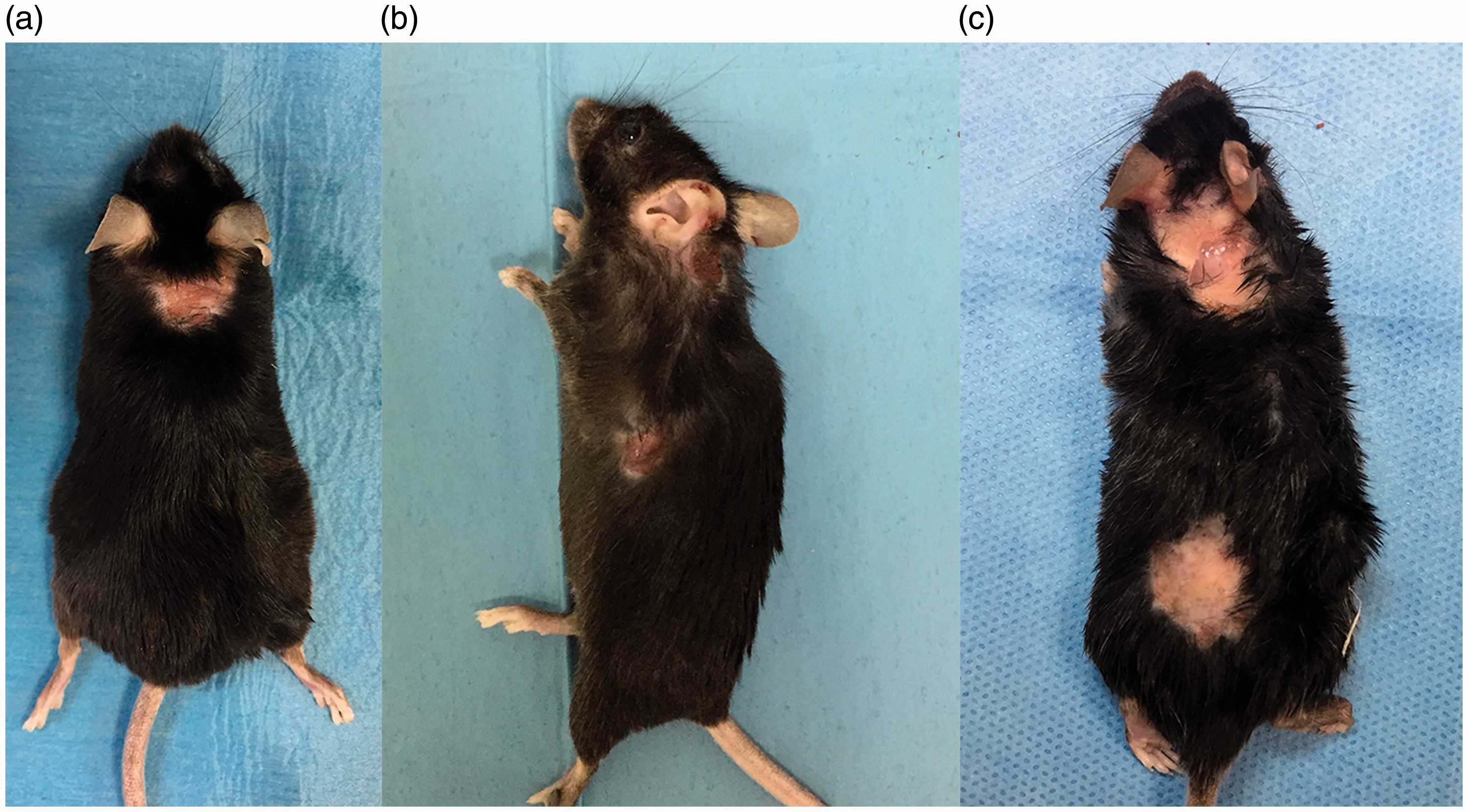
Splenic lymphoma was diagnosed at post-mortem examination and confirmed by means of histology in 32/60 C57BL/6NCrl-Tg(HMGA1P6)1Pg mice, but only in 3/25 of the UD affected animals (12%). No evidence of systemic disease or relevant pathological alterations of tissues or organs were observed in the other affected animals (20/25). Frequently, in the most severe cases, we observed moderate splenomegaly and reactive lymphadenomegaly of the axillary and cervical lymph nodes consistent with Kastenmayer's observations. 4
Post-mortem ectoparasite examination of the affected animals did not show the presence of infestations that could have been responsible for UD lesions.
Histopathology and IHC
Histological examination of formalin-fixed and paraffin-embedded 4-μm sections of clinically healthy mice showed no evident histopathological changes and an absence of dermal inflammatory cells and toluidine blue-positive mast cells (Figure 2(a) and (b)). Conversely, affected skin revealed extensive areas of ulceration and marked hyperplasia with parakeratotic to orthokeratotic hyperkeratosis of the adjacent epidermis. A diffuse and severe inflammation was observed in the dermis, often dissecting through underlying subcutaneous structures. The inflammatory population consisted mostly of neutrophils, toluidine blue-positive mast cells and lymphocytes with macrophages and eosinophils to a lesser extent (Figure 2(c) and (d)). A variable degree of fibrosis was also noticed, admixed with the areas of inflammation. In contrast to other authors,3,6 follicular dysplasia, other follicular abnormalities, and primary vascular lesions were not observed in the affected skin samples. The increase in mast cell accumulation was statistically significant in the affected (14.5 ± 2.7 SD) skin compared to healthy controls (2.04 ± 0.29 SD) (p < 0.05) (Figure 2(e)).
Histological examination of skin, mouse. (a) Healthy controls did not show any pathological alterations and (b) showed rare toluidine blue-positive mast cells. (c) Ulcerative dermatitic skin showed an extensive area of ulceration with an adjacent hyperplastic epidermis and a dermal severe and mixed inflammatory infiltrate. (d) In affected skin, there was a high number of toluidine blue-positive mast cells. (e) Mast cell count revealed a significantly increased number in ulcerative dermatitis cases compared to healthy controls (p < 0.05). Bars = 100 μm.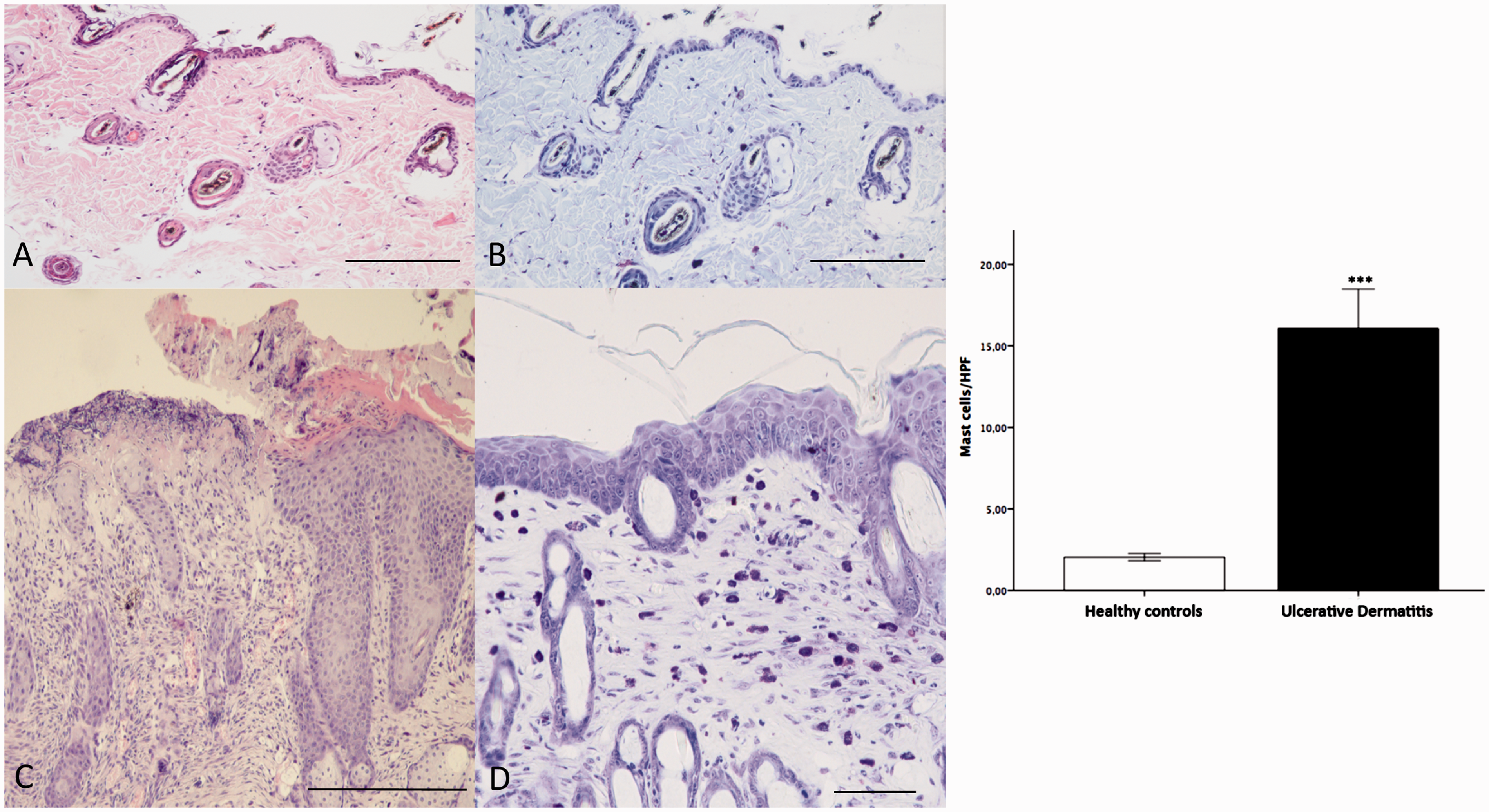
Gram, periodic acid-Schiff (PAS) and methenamine silver (Grocott) stains did not reveal the presence of bacterial or fungal agents associated with dermal inflammation, although crusts were occasionally associated with superficial, Gram-positive, coccoid bacteria (Figure 3). The severity of the lesions in affected animals were histologically graded as follows: 16% mild (4/25), 40% moderate (10/25), 32% severe (8/25) and 12% marked (3/25). The inflammatory population varied between the different histopathologic grades as follows: in mild cases the prevalent population was composed of 40% mast cells, 10% eosinophils, 5% neutrophils, 10% macrophages, 20% lymphocytes and 15% plasma cells; in moderate cases the prevalent inflammatory population was composed of 25% mast cells, 5% eosinophils, 15% neutrophils, 20% macrophages, 20% lymphocytes and 20% plasma cells; in severe cases the prevalent inflammatory population was composed of 15% mast cells, 5% eosinophils, 20% neutrophils, 10% macrophages, 30% lymphocytes and 20% plasma cells; in marked cases the prevalent inflammatory population was composed of 10% mast cells, 5% eosinophils, 25% neutrophils, 5% macrophages, 35% lymphocytes and 20% plasma cells.
Histochemical special stains on ulcerative dermatitic skin samples, mouse. Gram (a), periodic acid–Schiff (PAS) (b) and methenamine silver (Grocott) (c) did not show the presence of bacterial or fungal agents associated with dermal inflammation. Bars = 100 μm.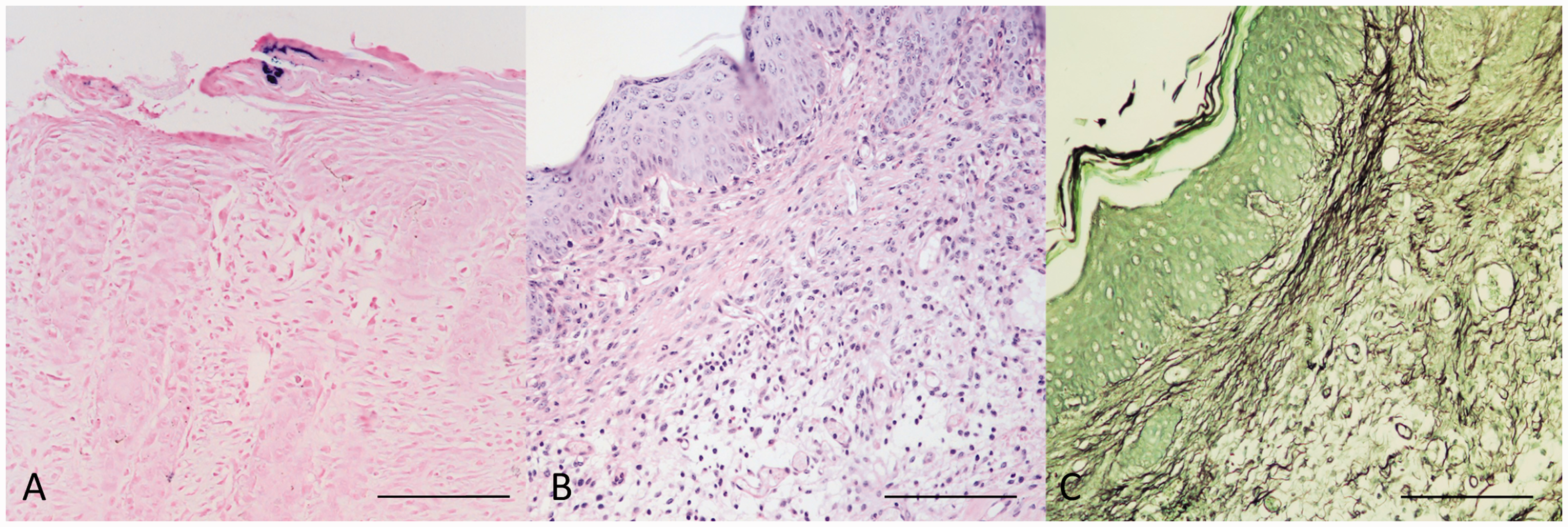
Immunohistochemical analysis was performed to further characterize the inflammatory lymphocyte infiltrates, showing a predominant CD3 + CD4 +leukocyte population with fewer of CD45/B220- and Il-17-immunolabelled cells. We rarely observed scattered, CD8-labelled lymphocytes (Figure 4(a) to (e)). Interestingly, immunofluorescence revealed the presence of numerous IgE-bound mast cells among the inflammatory infiltrates in the dermis and, in marked lesions, around blood vessels of the underlying subcutaneous muscle and adipose tissue (Figure 4(f)).
Immunohistochemical analysis of the inflammatory infiltrate, skin, mouse. Inflammatory infiltrate showed a prevalence of CD3 + (a) and CD4 + (b) cells. Very few scattered CD8-positive lymphocytes were observed (arrows) (c). CD45/B220 and IL-17 (d, e) immune-labelled inflammatory cells. (f) Immunofluorescence showing intense immunoreactivity of mast cells for IgE antibody (arrows). Bars = 100 μm.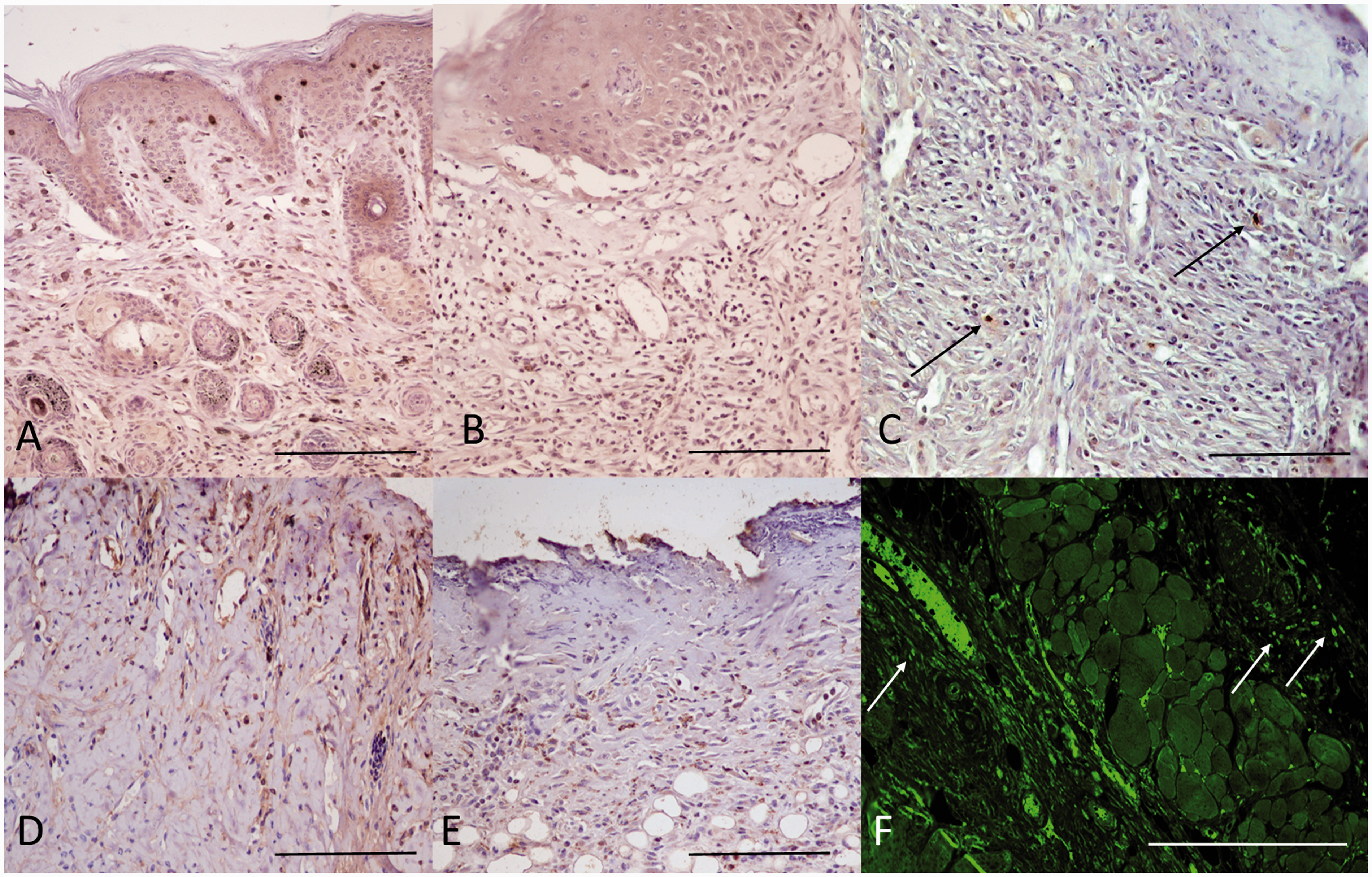
Molecular findings
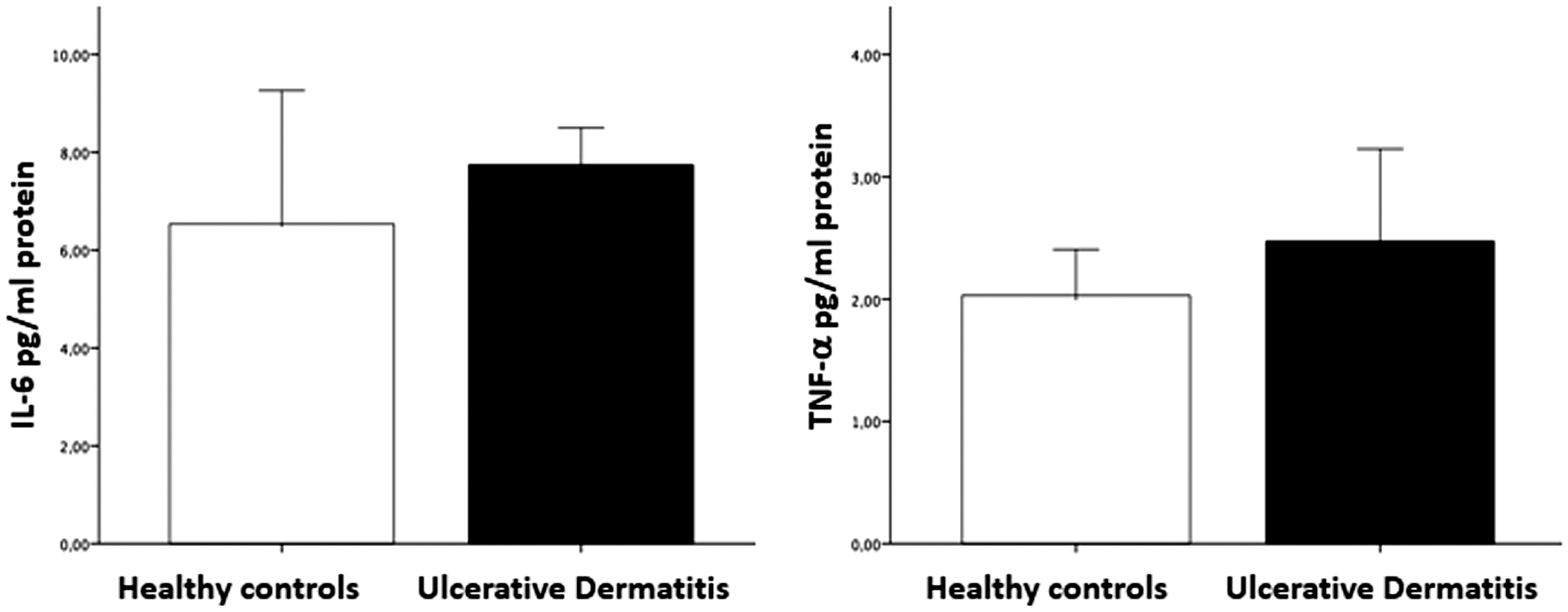
Semi-quantitative RT-PCR detected significantly increased IL-6, TNF-α and IL1β in skin samples from affected animals compared to controls (p < 0.05) while TGFβ mRNA did not differ between affected and control animals (Figure 5). There was no significant difference in serum IL-6 or TNF-α in affected mice compared to healthy subjects (Figure 6).
Cytokine mRNA expression in skin samples. Graphs showing the expression of TNFα, IL-1β, IL-6 and TGFβ in UD mice compared to healthy controls; p-values were determined by independent t-test, and the cytokine levels are presented as the means ( ± SD). All results were considered statistically significant at p < 0.05. Cytokine levels in serum. Graphs showing the serum levels of IL-6 and TNFα in UD mice compared to healthy controls; p-values were determined by independent t-test, and the cytokine levels are presented as the means ( ± SD). All results were considered statistically significant at p < 0.05.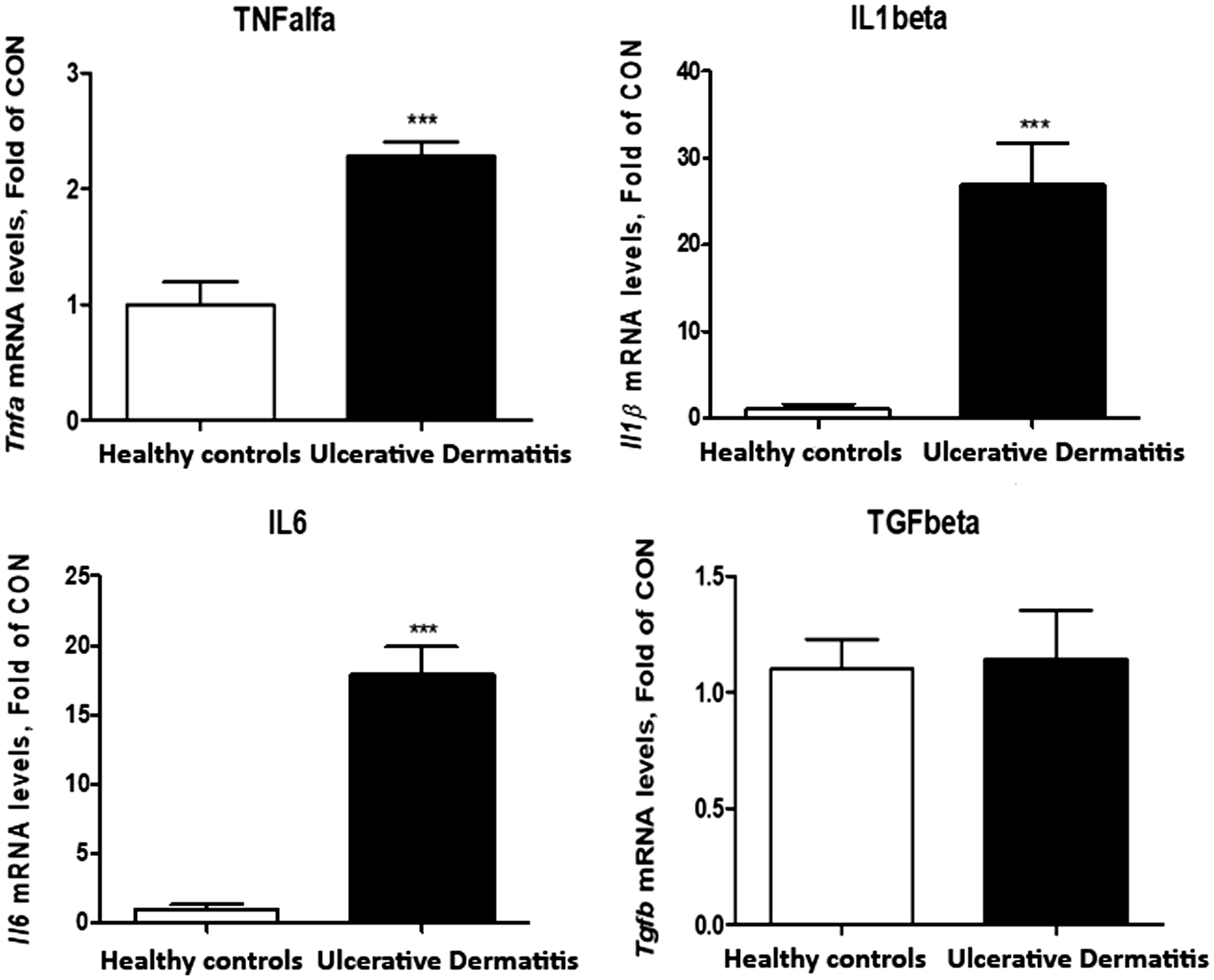
Discussion
The aim of this study was to investigate, for the first time, a potential pathogenic mechanism for UD through the characterization of inflammatory infiltrates and cytokine expression of C57BL/6NCrl-Tg(HMGA1P6)1Pg mice that spontaneously developed itchy and ulcerative skin lesions.
Our clinical observations, histopathological and molecular findings indicate (1) intense pruritus, (2) significant prevalence of the disease in late spring and summer, (3) absence of concurrent infection or parasite infestation and (4) inflammatory infiltrate and an increased cytokine expression consistent with Th2/IgE and Th17 differentiation. These results, in our opinion, share similar features observed in cutaneous type I hypersensitivity reactions, 25 suggesting a possible allergic aetiology for UD in mice.
Immunohistochemical evaluation of dermal inflammatory infiltrates in UD lesions interestingly showed an increase in the number of CD4 + T cells and Th17 + cells and a significant increase in mast cells displaying an intense immunoreactivity for the IgE antibody. Moreover, cytokine analysis showed that affected skin had increased levels of IL-1β, IL6 and TNFα. The type I hypersensitivity reaction is initiated by an environmental antigen (called an allergen) that elicits a Th2 cell-mediated response in susceptible subjects. 25 Since UD incidence has been associated with the seasonal pattern and in particular to changes in humidity,1,2,4 we suggest that the seasonal peak of environmental allergens such as pollen or fungal spores may be responsible, in our case, for the increased number of affected animals, in agreement with Kastenmayer's observations. 4
Th2 cells play a key role in hypersensitivity reaction by producing several cytokines (namely, IL-4, IL-5 and IL-13) that are responsible for the class switching of B lymphocytes to IgE production.25–27 IgE is essential for the activation of mast cells and the release of mediators (histamine, TNFα) that are the principal culprits for the clinical and pathological manifestation (e.g. pruritus)25,28,29 as well as for the influx of Th2 lymphocytes and eosinophils into the skin. 24 During the acute phase of inflammation, eosinophils produce cytokines such as IL-6 and IL-1β, supporting a Th17 differentiation. 29 A pivotal but yet still unclear role of Th17 cells in allergy has been implicated in several allergic skin diseases, such as atopic dermatitis or psoriasis.30,31 Most likely, Th17 cells are involved in a late phase of allergic response, promoting tissue fibrosis, chronicity of the inflammatory process, and the evolution of chronic inflammatory cutaneous lesions. 32 TGF-β expression did not show any noteworthy differences between UD lesions and healthy controls. This finding is not surprising since TGF-β and its signalling pathways have inhibitory effects on B and T cells, IgE production, and mast cell proliferation, and they can also induce apoptosis in eosinophils. 33 It is very likely that, in UD, TGF-β signalling is impaired, so it fails to inhibit the inflammatory process. TNFα and IL-6 are considered mediators of inflammatory acute response and serum analysis of their levels is extremely important for the diagnosis of several systemic conditions, such as inflammation and sepsis. 34 Here, we found no differences in TNFα or IL-6 level in affected animals compared to controls. These findings may indicate that, in the affected animals, there is no sign of systemic inflammation and/or that the lesions observed are consistent with a chronic phase of the allergic response.
A small percentage of UD-affected mice had splenic lymphoma (12%). This finding is not surprising and is consistent with Andrews's observations. 3 The small number of mice with splenic lymphoma does not allow a significant correlation between the presence of this neoplasm and ulcerative lesions.
This work has also some limitations to be addressed. First, intense pruritus and scratching are a distinctive and constant feature of the clinical presentation of UD, so we have to take into account the presence of an inflammatory infiltrate secondary to trauma, such as neutrophils, that may slightly influence the interpretation of the results, especially in relation to triggering events in the development of UD.
Second, we did not evaluate the IL-4, IL-5 and IL-13 levels in skin samples, even though they are responsible for the class switching of B lymphocytes to IgE production. To overcome these issues, we thoroughly characterized the immune infiltrate by IHC, demonstrating the presence of numerous IgE-bound mast cells among the inflammatory infiltrates in the dermis and, in marked lesions, around blood vessels in the underlying subcutaneous muscle and adipose tissue.
In our opinion, the presence of IgE-bound mast cells associated with a Th1/Th2 and Th17 immune response and an increase in IL-1β, TNFα and IL-6, which intervene in the acute phase of the allergic response, are consistent with an allergic eliciting factor for UD development.
Susceptibility to allergic inflammation has a strong genetic component. 25 In a 2-year retrospective study, Kastenmayer et al. reported an overall prevalence of UD of 4.1% (55/1352) in mice with a C57BL/6 genetic background, whereas no UD lesions were observed in non-C57BL/6 mice. 4 Consistent with that, we showed a high prevalence (41.6%) of spontaneous UD lesions in transgenic mice on a C57BL/6 genetic background overexpressing the HMGA1P6 pseudogene. Therefore, it is conceivable that genetic defects are responsible for the development of this disease. Pseudogenes are DNA sequences that have high homology to the corresponding functional gene but have lost their initial functions to code for proteins due to the accumulation of various mutations. 35 HMGA1P6 has a well-established oncogenic action by preventing apoptosis and enhancing cell proliferation and migration.14,36 Overexpression of HMGA1P6 increases the growth rate and migration of different cell lines, contributing to tumour development.14,36,37
The assessment of genetic alterations in UD-affected mice compared to controls was not the principal purpose of this study. De Martino et al. analysed the whole transcriptome of WT and HMGA1P6-transgenic mouse embryonic fibroblasts (MEFs) by RNA-seq to identify the genes regulated by HMGA1P6 expression. 38 The genome-wide RNA expression profile revealed that about one hundred fifty transcripts (32 upregulated and 116 downregulated) were regulated by HMGA1P6 expression with a significant fold-change variation (FDR adjusted p-value of 0.05). Among these genes, suppression of tumourigenicity 2 (ST2) was upregulated in transgenic mice (data not shown). The ST2 gene and his ligand, interleukin 33 (IL33) have critical roles in inflammatory processes, particularly regarding mast cells, type 2 CD4 + T-helper cells and the production of Th2-associated cytokines, promoting the pathogenesis of Th2-related disease such as asthma, atopic dermatitis and anaphylaxis.39,40 These data need to be further and appropriately investigated to confirm a genetic component in the plethora of mechanisms responsible for UD pathogenesis.
We cannot assert without any uncertainty that the manifestation of UD in this specific genetically engineered mouse line is representative of the disease process in other mutant strains or wild-type B6 mice. It is possible that C57BL/6NCrl-Tg(HMGA1P6)1Pg mice have a predisposition towards an allergic dermatitis that is a separate entity from UD with a similar presentation (pruritus and significant secondary trauma from scratching). However, skin samples from different transgenic mice with a B6 background housed in other facilities and showing clinical signs of UD were reported to our laboratory. Interestingly, the histopathological findings observed in this study matched the ones described in scientific literature. For consistency, we decided to exclude the ‘external’ UD cases from our study for the different housing conditions and experimental sampling.
Although the causative trigger of UD in mice is still not completely understood, our data encourage further investigations into the hypothesis that this disease has an allergic aetiopathogenesis, also suggesting the bases for a possible treatment. The inflammatory Th2 infiltrate and its related cytokines can explain, to a certain extent, the partially positive clinical response associated with the use of the vitamin E-fortified diet. 5 As a matter of fact, vitamin E is a potent antioxidant that inhibits membrane lipid peroxidation, modulates the metabolism of arachidonic acid, 41 inhibits cyclooxygenase enzyme activity, 42 and inhibits the release of interleukin 1β by inhibiting the 5-lipooxygenase pathway. 43 Therefore, topical use of anti-allergic drugs seems strongly recommended to test a potential therapeutic target for the control of this disease, in order to improve the refinement of animal research minimizing the pain and suffering due to the devastating skin lesions of UD, thus relieving the distress of mice and improving their well-being.
Supplemental Material
Supplemental material for Characterization of inflammatory infiltrate of ulcerative dermatitis in C57BL/6NCrl-Tg(HMGA1P6)1Pg mice
Supplemental Material for Characterization of inflammatory infiltrate of ulcerative dermatitis in C57BL/6NCrl-Tg(HMGA1P6)1Pg mice by Davide De Biase, Francesco Esposito, Marco De Martino, Claudio Pirozzi, Antonio Luciano, Giuseppe Palma, Giuseppina Mattace Raso, Valentina Iovane, Stefania Marzocco, Alfredo Fusco and Orlando Paciello in Laboratory Animals
Footnotes
Acknowledgement
The authors would like to thank Dr Arianna Ilsami for her valuable technical support and Dr Giuseppe Piegari for his insight in statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: On behalf of all authors the corresponding author states that there are no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
