Abstract
Fasting of mice is a common procedure performed in association with many different types of experiments mainly in order to reduce variability in investigatory parameters or to facilitate surgical procedures. However, the effects of fasting not directly related to the investigatory parameters are often ignored. The aim of this review is to present and summarize knowledge about the effects of fasting of mice to facilitate optimization of the fasting procedure for any given study and thereby maximize the scientific outcome and minimize the discomfort for the mice and hence ensure high animal welfare. The results are presented from a number of experimental studies, providing evidence for fasting-induced changes in hormone balance, body weight, metabolism, hepatic enzymes, cardiovascular parameters, body temperature and toxicological responses. A description of relevant normal behaviour and standard physiological parameters is given, concluding that mice are primarily nocturnal and consume two-thirds of their total food intake during the night. It is argued that overnight fasting of mice is not comparable with overnight fasting of humans because the mouse has a nocturnal circadian rhythm and a higher metabolic rate. It is suggested that because many physiological parameters are regulated by circadian rhythms, fasting initiated at different points in the circadian rhythm has different impacts and produces different results.
Fasting of mice is considered to be a standard procedure used in association with many different types of experiments.1–3 Fasting is primarily used as a way of standardizing tests or before surgery.
Motivations for fasting mice include: Overnight fasting before measuring blood glucose to reduce variability in basal blood glucose, to assure a more uniform drug absorption by preventing mixing test drug with food; overnight fasting for toxicology testing; fasting-induced feeding to investigate anorexigenic compounds, in which case mice are usually fasted for 4 h up to overnight; and pharmacokinetic studies in general.1,2,4–7
In this review the term ‘fasting’ refers to a state where the animal is completely deprived of food but has access to water, which is similar to the study designs of most of the references. In order to facilitate comparison of quantitative data, values are given in international units, and have been converted where required. 8
The aim of this review is to summarize present knowledge about the effects of fasting mice to facilitate an optimization of the fasting procedure and thereby secure better scientific data, minimize the discomfort of the animals, and hence increase animal welfare.
Physiology of non-fasted mice
Food and water intake
Circadian rhythms regulate many aspects of the body physiology, including cellular and physiological systems such as hormone secretion rhythms, body temperature, hepatic energy metabolism, and sleep–wake cycles. The circadian rhythms are primarily paced by the suprachiasmatic nucleus (SCN) of the anterior hypothalamus in mammals. External stimuli such as light and dark influence circadian rhythms.9,10 Light is the most potent stimulus, entraining the SCN via the retinohypothalamic tract. 11 It has been shown in rats that food intake and feeding pattern are disrupted if the SCN is lesioned. 12
The sleep–wake pattern of mice has a characteristic nocturnal pattern; mice sleep more during photophase (light period) than during scotophase (dark period). 13
Food intake and metabolism are closely associated with the function of the hypothalamo–pituitary–adrenal system (HPA).14,15 HPA activity is normally of low amplitude at the beginning of photophase and of high amplitude at the beginning of scotophase, corresponding with the high activity level of the mouse in scotophase and the low activity level in photophase.
Food and water intake in mice.

Total food and water intake (mean ± SD) in eight male, 12-week-old C57BL/6 J during photophase (light phase) and scotophase (dark phase). Food: powder CE-2 diet. Data from Kurokawa et al. (2000). 20
Glucose concentrations of non-fasted mice.
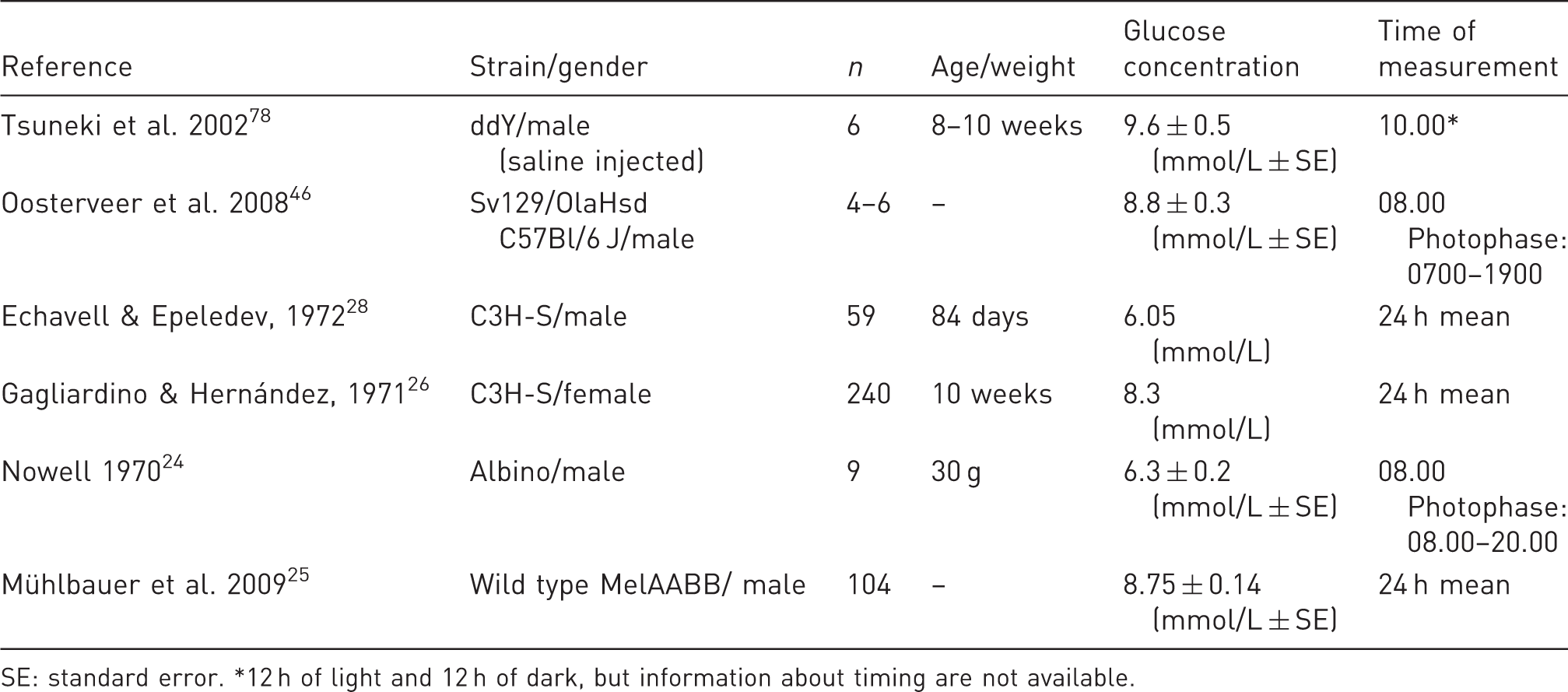
SE: standard error. *12 h of light and 12 h of dark, but information about timing are not available.
A significant (
Deviations from these diurnal rhythms in eating behaviour and glucose fluctuations have been found, though Petersen demonstrated a significant (
In a study with eight mice, it was shown that the feeding and drinking events were synchronized, but that the mice drank infrequently without eating. 20 Kurokawa et al. did not provide data of mean meal time, mean drinking time, mean inter-meal interval time or mean interval between drinking time. 20 Another study found that mice had an average of 36 food bouts and 32 water bouts during one day and night, lasting approximately 3.00 min and 0.88 min, respectively. The average interval between food bouts was 34 minutes and between water bouts it was 42 minutes. Drinking typically followed eating within 5 min. 18 Meal patterns were analyzed for seven days in a study, where the mice were fasted for 5 h in the photophase and fed ad libitum for the remaining 19 h. The average number of meals per day and night was approximately 20, lasting approximately 5 min and with an approximately 40 min inter-meal interval. 31
The different results in the above-mentioned studies might be due to the differences in experimental design, regarding strain and age of mice, diet, housing and method of measuring. Information about bedding and ambient temperature were not given by Gannon et al. 18 Type of food was not reported by Kurokawa et al., although it has been shown to affect eating patterns.20,31
Ad libitum fed mice kept at thermoneutrality at 33℃ ate about 50% less than mice kept at 23℃. 32 In a study by Bronson, mice housed at 11℃ had a 37% higher food intake compared with mice housed at 22℃. 33 The mice housed at 11℃ had cotton nests with a core temperature of 18℃ and thus were only exposed to the cold when feeding. Similarly, Overton and Williams reported a reduction in food intake when ambient temperature increased from 23℃ to 30℃. 34 Mice housed at 20℃ had a significantly higher food intake than mice housed at 25℃, demonstrating that a 5℃ difference in ambient temperature can cause a significant difference in food intake. 35
Mean food and water intake of mice.
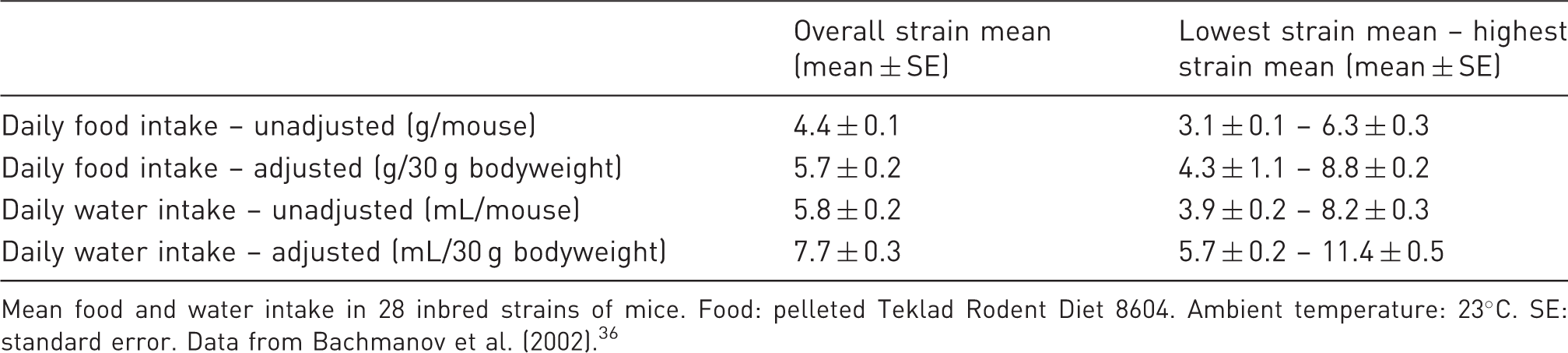
Mean food and water intake in 28 inbred strains of mice. Food: pelleted Teklad Rodent Diet 8604. Ambient temperature: 23℃. SE: standard error. Data from Bachmanov et al. (2002). 36
The gastric content in ad libitum fed mice shows a circadian rhythm, being highest in the middle of scotophase and lowest towards the end of photophase, which is analyzed using measurements every fourth hour.37,38 The gastric content was similar for animals with and without bedding. 38
In conclusion, the food and water intake in mice thus seem to be following a pattern with small, frequent meals associated with drinking. Food intake is dependent on housing temperature and strain and two-thirds of the food are eaten during the dark phase.
Hormones
Adrenal corticosterone is produced when the adrenal gland is stimulated by the adrenocorticotropic hormone. 39 Several studies show that the serum corticosterone level peaks near the transition from photophase to scotophase, followed by a gradual decrease during scotophase and stays at base level during photophase until the transition to scotophase. The lower peak is near the transition from scotophase to photophase.39–45 There are significant differences in corticosterone levels over 24 h.40,44 Corticosterone leads the activity level of the mouse, increasing as the activity level increases in scotophase and decreasing as the activity level decreases in photophase. 40
Food intake stimulates the pancreatic release of insulin. In a study by Ahrén, plasma insulin displayed diurnal variation with a nocturnal peak in NMRI mice, and the plasma insulin level reached a significantly higher peak in males than in females, but there was no difference between gender regarding the relative increase. 29 Other investigators have found a circadian rhythm characterized by an insulin peak (6 ng/mL) at or just before the transition from photophase to scotophase.25,26,43 The insulin level at approximately one hour into the photophase is 1.59 ng/mL. 46
The ghrelin hormone is mainly produced in the stomach and regulates feeding activity, being elevated during fasting and suppressed after food intake. Food intake is stimulated by activating neuropeptide Y-ergic neurons in the hypothalamus. 13 Other hormones regulating metabolism include adrenalin, noradrenalin, glucagon, growth hormone, thyroid hormones, leptin and resistin. 47
Body temperature
The body temperature of mice follows a circadian rhythm, with a peak body temperature at the beginning of scotophase and the lowest body temperature in the middle of photophase.13,48 It has been shown that the 24 h mean temperature is about 37.7℃, the lowest hourly mean 36.7℃ and the range of hourly means 1.9℃.49,50 Corresponding with these results, Swoap reported a photophase mean body temperature of 36.7℃ and a scotophase mean body temperature of 37.8℃. 51 As in other mammal species, the temperature is highest in the activity period and lowest in the resting period.
Fasting of mice: physiological changes
Non-food and water intake
Mice housed on solid flooring have the opportunity to consume faeces (coprophagia) during fasting periods, as well as bedding, environmental enrichment, biting sticks and nesting material if provided. Mice usually have access to water ad libitum during fasting, but since the water intake, as earlier described, is related to food intake, fasting also influences water intake.
In a study by Kutscher, mice fasted for up to five days (they could not survive any longer) displayed different drinking patterns: consistent polydipsia, sporadic polydipsia, or polydipsia restricted to the beginning or the end of the fast. 16 In total 35% had polydipsia and 42% had decreased drinking activity (compared with their intake measured while not fasted). Five of the polydipsic mice showed this pattern on day 1. Both polydipsic and non-polydipsic mice kept their nocturnal drinking pattern during three days of fasting, except that non-polydipsic mice seemed to have a disturbed drinking pattern after three days of fasting.
Some strains of inbred mice show polydipsia during fasting, whereas other strains show hypodipsia. In a study where the circadian rhythm of water intake during fasting was measured, the fasted mice showed a pronounced circadian rhythm in their water intake.
52
Significant (
Hormones
Among others, the corticosterone hormone is known to be stress responsive, and is therefore relevant when exploring the effects of fasting. Stress has been defined as a response of an organism to external stimuli or change. 45 It is characterized by the activation of the autonomic nervous system and HPA axis and thereby an increase of glucocorticoids, such as corticosterone.45,54
Fasting also stimulates the sympathetic nervous system. In rats it was shown that increased noradrenalin turnover caused an elevated sympathetic activity, resulting in fatty acid release. 55
Mice of the C57BL/6 J strain, fasted for 2, 4, 6 and 12 h had significantly (
By comparison, mice of the BALB/c strain, (gender not given), stressed by swimming until exhausted, had a significant (
Corticosterone levels are complex to compare, since there are gender differences, as shown by Champy et al., and there might be strain differences. 56
Leptin is a hormone secreted from adipose tissue in proportion to fat storage reserves, regulating the whole-body energy balance. Mice fasted for 2, 4 and 6 h had a significantly (
Neuropeptide Y (NPY) is down-regulated as a result of increase in adipose tissue and leptin secretion. NPY is appetite stimulating. Mice fasted for 48 h had a significant (
Ghrelin is elevated in response to fasting and hunger. The high ghrelin production during fasting might be associated with an increase in glucagon. Ghrelin stimulates feeding behaviour via the nervous system and is antagonistic to leptin. 62 Ghrelin gene expression in the stomach of mice was increased by 48 h of fasting. 63
Resistin is, like leptin, a hormone secreted by adipocytes. Resistin increases with high-fat feeding and causes decreased sensitivity to the effects of insulin. It is decreased by 48 h of fasting. 64
An insulin drop is considered to drive the metabolic adaptation to fasting.
65
There was no significant difference in plasma insulin levels between mice fasted for 16 h and mice fasted for 4 h. When measured under hyperglycaemic conditions, 16 h fasted mice had a significantly (
Thyroxine (T4) and triiodothyronine (T3) are regulators of basal metabolic rate. Both were significantly (
Testosterone was significantly (
In summary, corticosterone, NPY, ghrelin and resistin increase and T4, T3, testosterone, insulin and leptin decrease in response to fasting.
Weight loss
Relative (%) body weight loss of fasted mice.
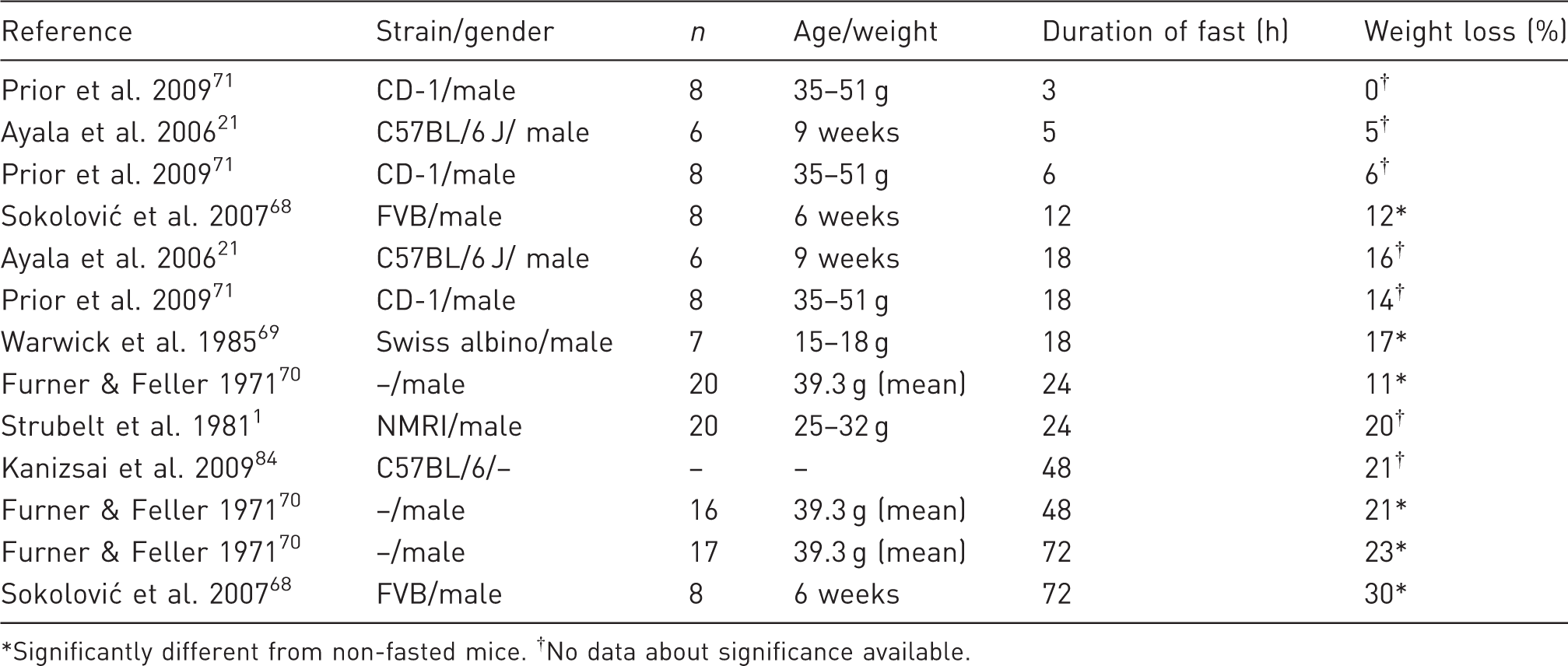
Significantly different from non-fasted mice. †No data about significance available.
Mice lost significantly more weight after 16 h of fasting compared with 4 h, and a significantly (
Mice are typically used for experiments at a relatively young age, and normally they would gain weight through natural growth, thus the relative weight loss is more severe if corrected for expected weight gain, depending on the duration of the fast.71,72
Mice fasted for 18 h had significantly (
Fat storages were dramatically decreased after 28 h of fasting, and not much different from the decreases seen in mice dead from starvation. 33
Gastrointestinal emptying
An empty stomach, small intestine and/or large intestine might be required in some studies including surgery or investigation of oral/gastrointestinal absorption. In a study by Prior et al., mice were fasted for 0, 3, 6 and 18 h. 71 There was no significant difference in the gastric content weight between 6 and 18 h fasted mice, and no difference between 0 and 3 h fasted mice, but there was a significant increase between 0 or 3 h, compared with 6 or 18 h. The same pattern was observed for intestinal transit time of charcoal. It was concluded that fasting for 6 h gave similar results to fasting for 18 h regarding gastric emptying and intestinal transit time of charcoal.
Another author showed that mice fasted for 4 and 8 h, with the start of fasting from 4 h before photophase, had significantly lower gastric contents than non-fasted mice. Interestingly, mice fasted for 12 h, reported as a complete emptying of the stomach, did not have a significantly lower content than non-fasted mice. Non-fasted mice had a spontaneous decline in their feeding activity, resulting in a low gastric content at a point coinciding with the mice fasted for 12 h, and therefore no significant difference was seen after 12 h of fasting. 37 Bronson reported that most of the food eaten in the scotophase cleared the stomach within 6 h of fasting in the photophase, and after 14 h of fasting in the photophase, the small intestines were essentially empty. 33
During three days of fasting, mice excreted boluses of their bedding material, sterolit (a granular clay material), mean weight being 1.8, 1.1 and 0.5 g, respectively, for the first three days of fasting, indicating intake of bedding.
16
Yeung et al. found that mice fasted for 24 h on grid floor had a significantly (
The mean residual gastric content after 18 h of fasting was 0.076 g. 74
Fasting for 29 h resulted in a significantly lower gastric content compared with non-fasted mice. The gastric content of fasted mice showed a circadian rhythm similar to that of non-fasted animals. Mice that had bedding had a 72% mean drop in gastric content, while mice without bedding only had a 50% drop. 38 It was emphasized that the relative variation was identical under both fasting circumstances, but no explanation of the difference in the total decrease in the mean gastric content was given. The gastric content of the 29 h fasted mice might be secretions from the stomach.
Gastric emptying after administration of a simple, oral solution was faster in 24 h fasted mice than in non-fasted mice, although no information about significance was given. 75 Gastric emptying time is associated with stomach volume and body size. 76
In conclusion, gastric content of mice follows a circadian rhythm associated with food intake. Shorter periods of fasting do not necessarily lead to less gastric content than non-fasted mice would experience at certain points in the circadian rhythm. There is still some form of gastric content after 29 h of fasting.
Metabolism
Blood glucose decreases during fasting due to lack of food intake and thereby to lack of glucose absorbed from the intestines.
Mice fasted for 9 h had significantly (
Glucose concentrations of fasted mice.
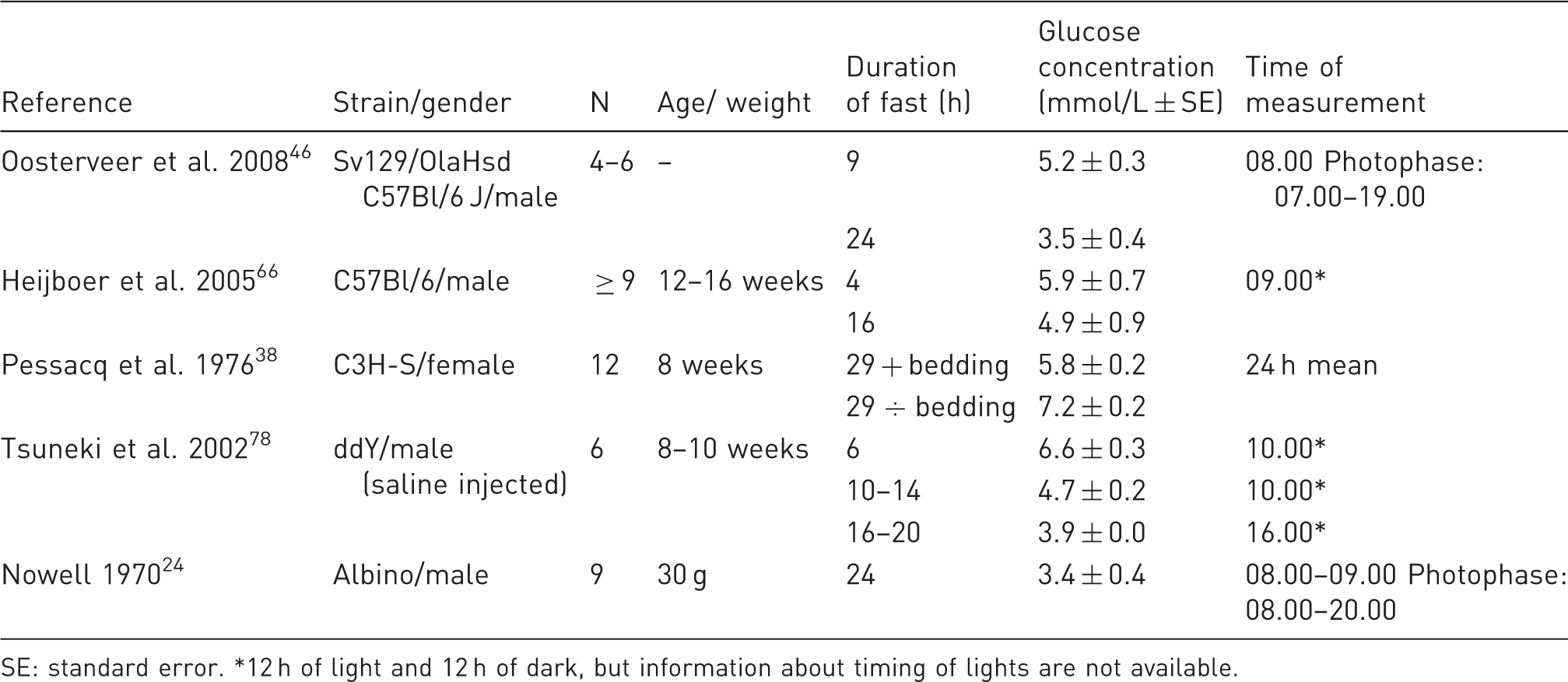
SE: standard error. *12 h of light and 12 h of dark, but information about timing of lights are not available.
Hepatic glucose production was not significantly different from 4 to 16 h of fasting. 66
Muscle specific glucose uptake under hyperinsulinaemic conditions was significantly (
Glucose can be stored as glycogen in the liver, kidneys, heart and muscles. This reservoir is mobilized by glycogenolysis during the early stage of fasting, in order to maintain blood glucose concentration. In mice, hepatic glycogen is significantly (
There was no significant (
Prolonged fasting induces an increase in plasma glucagon, plasma glucocorticoids, plasma (nor) epinephrine and a decrease in plasma insulin concentrations. This leads to hydrolysis of triglyceride (TRIG) in adipose tissue, which causes a rise in plasma free fatty acid (FFA) concentration.65,79 There was a significantly (
The FFAs are taken up by the liver, and either re-esterified or oxidized (β-oxidation). Re-esterification leads to synthesis of TRIG. These are secreted as very low density lipoproteins, which can provide FFAs for other tissues. Hepatic steatosis is a pathological increase in hepatic TRIG. Hepatic TRIG was six-fold higher in 16 h fasted mice than in 4 h fasted mice (
The majority of the FFAs that are oxidized by the liver are only partially oxidized, to acetyl-CoA, which condenses itself into ketone bodies, providing an energy source for the brain.65,79
Plasma ketone bodies were significantly (
The FFAs fully oxidized by mitochondria, provide energy for hepatic gluconeogenesis, using substrates as glycerol from hydrolysis of TRIG in adipose tissue, lactate from glycogenolysis in skeletal muscle and amino acids. 79 Plasma lactate levels decreased gradually during 72 h of fasting. 77 Hepatic mRNA expression levels of transcription factors and related proteins involved in gluconeogenesis were elevated, while those for glycogenolysis were lowered, in 16 h fasted mice compared with 4 h fasted mice. 66 The latter is because the liver glycogen depots are decreasing, thereby depleting the substrate for glycogenolysis. Total hepatic glucose production was not altered. 66 No change in urea (a product of protein catabolism) was seen after 24 h of fasting or during 2 to 16 h of fasting.56,57 These results indicate that glycogen is rapidly utilized, lactate and glycerol are utilized for gluconeogenesis, but protein breakdown is retained after 24 h of fasting.
In conclusion, the metabolic state of fasting can be described as catabolic or post-absorptive characterized by mobilization of glycogen providing glucose and increased mobilization of adipose tissue, again providing FFAs, utilized for production of ketone bodies and TRIG and providing energy for gluconeogenesis using lactate, amino acids and glycerol as substrates.
Liver parameters
Hepatic steatosis is seen after 16 h of fasting in mice. 66 After 48–72 h of fasting subtle steatosis is seen in the liver in centrizonal hepatocytes. After 66–72 h, the fatty change was minimal with few microvesicular lipid droplets. After 96 h, no fatty change was seen, and the hepatocytes revealed features of cytoplasmic atrophy. 77
No significant difference in cytochrome P-450 level was seen after 12 h of fasting, but was significantly (
Reduced glutathione is an antioxidant which is significantly decreased after 17 h and 24 h of fasting.1,83
Body temperature
Mice fasted for 24 h had a significantly (
Torpor has been defined as a suppression of metabolic rate to a level as low as 30% of the basal metabolic rate for a period of several hours by the lowering of the body temperature. Torpor has also been defined as a body temperature of less than 31℃.85,86
Non-fasted mice do not normally exhibit torpor.87–89 Mice can become torpid after about 7 h of fasting, with a body temperature drop of up to 15℃.60,89,90 The mean time from initiation of fasting to first torpor bout was reported to be 13.3 h in a study by Brown and Staples. 89 It has been stated that torpor typically occurs after 8 h of fasting, and/or at the end of the dark cycle which is coincident with the peak of circulating ghrelin. Ghrelin is able to deepen torpor. 91 A food restriction of 50% for three days also induced torpor. 51
Fasting-induced torpor has been recognized in both sexes, young and adult, individually- and group-housed animals and in several strains of mice, both inbred and outbred.86,88 Free-living mice in nests have also been found torpid during the cold months. 92 After 24 h of fasting 35% of mice were torpid, and after 48 h of fasting 100% were torpid. 88 Group-housed mice had a significantly greater torpor incidence than singly-housed mice. Mice that were fasted several times all experienced torpor at some point. 86
The minimum possible body temperature in mice is 16–19℃. 87 During three days of fasting, mice typically undergo at least one bout of torpor each day, usually beginning at the end of scotophase. Swoap et al. found the rate of temperature decrease to be 0.16℃ per minute. 60 Some mice become hypothermic, characterized by an apparent inability to spontaneously arouse without assistance, and are likely not able to survive as long as those undergoing a daily torpor. 89 Emergence from torpor is typically finished within 30–45 min of the initiation from recovery, maximally gaining 0.26℃ per minute. 90 Handling of mice for subcutaneous injection or insertion of a rectal probe causes immediate arousal from the torpid state.88,90 Mice are also reported to arouse spontaneously and as a result of noise or moving of the cage.86,87
It is suggested that entry into torpor is a consequence of a cold environment or fasting. Norepinephrine release induces β3-mediated sympathetic activation of white adipocytes, causing suppression of leptin secretion and an elevation of lipolysis, resulting in a liberation of FFAs to the blood. 90 Torpor is likely regulated by a complex interaction among ghrelin, leptin, insulin and hypothalamic neurons. 91 Mitochondrial respiration is actively suppressed during daily torpor in fasting mice. Mitochondrial respiration rate is a measure for the activity of the components of oxidative phosphorylation that control it. 89
In conclusion, torpor seems to be a way of reducing the energy expenditure during times of actual or predicted food shortage, and mice can enter torpor when temperatures are low or during times of fasting.
Cardiovascular parameters
A negative energy balance leads to decreased heart rate and blood pressure.
51
Mean arterial pressure is significantly (
Bradycardia and hypotension are suggested to be part of the torpor response. The bradycardia patterns were noticed within 6 h of fasting. 53 The gene that was mostly up-regulated after 24 and 48 h of fasting was cytochrome P-450 4A14. Disruption of this gene has been associated with hypertension. 81
The obtainable blood volume decreased significantly after 24 h of fasting and a further significant decrease was reported after five days of fasting. 93
White and red blood cells, haemoglobin, mean cell volume and platelets were significantly decreased after 2 h of fasting compared with 16 h of fasting, whereas haematocrit was significantly (
Toxicology
In tests with bunitrolol or pindolol (β-adrenoceptor blockers), mice fasted for 17–20 h developed clinical signs comparable with non-fasted mice about 10 min earlier, acute oral toxicity was significantly higher and death was hastened compared with non-fasted mice. 94 The blue metabolite of pindolol (β-adrenoceptor blocker) appeared in the urine of non-fasted mice after one hour, but in the urine of 17–20 h fasted mice, after half an hour. 94
In a toxicity study testing the hepatotoxic response to carbon tetrachloride, no significant difference in aspartate aminotransferase (AST), alanine aminotransferase (ALT) and sorbitol dehydrogenase (SDH) was seen after 6 h of fasting, but after 12, 24 and 36 h of fasting AST, ALT and SDH were significantly increased in fasted mice compared with non-fasted mice. The increase was enhanced after 24 h but not after 36 h. Similarly, in 24 h tests with thioacetamide, paracetamol, bromobenzene, allyl alcohol and phalloidin, a significant increase was seen in at least one of the parameters AST, ALT and SDH compared with non-fasted mice. 1
Carbon tetrachloride caused massive hepatic centrilobular necroses in 24 h fasted mice, where non-fasted mice only showed moderate toxic alterations. Fasting did not influence enzyme concentrations after saline, alpha-amanitin and praseodymium treatment. In vitro protein binding of both carbon tetrachloride and paracetamol to hepatic microsomal protein remained unaffected upon 24 h of fasting. Total hepatic amount of carbon tetrachloride was significantly (
Ethylmorphine, p-nitroanisole and aniline are drugs which undergo oxidative enzymatic degradation by hepatic microsomes. The metabolism of ethylmorphine and p-nitroanisole was significantly elevated after two and three days of fasting, but not after one day. Aniline metabolism was significantly elevated after one, two and three days of fasting. 70
Mice fasted for 12 h overnight prior to oral administration of paracetamol (e.g. acetaminophen), a drug that can cause hypothermia, liver necrosis and hepatic congestion in mice, produced a significantly (
In a study examining intravenously administered digitoxigenin toxicity mortality was significantly (
In conclusion, fasting is capable of influencing the toxicity of drugs, either by increasing or decreasing toxicity.
Discussion
Fasting of mice is often used for evaluation of blood glucose and insulin in association with metabolic diseases, such as diabetes. Glucose tolerance tests, insulin tolerance tests as well as hyperglycaemic, hyperinsulinaemic–euglycaemic and hyperinsulinaemic–hypoglycaemic clamps include commonly used tests in which a fasting duration of 5–6 h has been recommended. Fasting for 5–6 h instead of overnight (16–18 h) might offer a better comparison to humans, who are usually fasted overnight before similar tests. Blood glucose and insulin levels in humans after an overnight fast represent ‘a basal steady-state’, whereas overnight fasting of mice cannot be considered a routine event, due to their nocturnal eating pattern. 95
Fasting of mice changes several physiological parameters and moreover, these changes are dependent on a variety of environmental factors.
Glucose, insulin and corticosterone among others, all fluctuate with circadian rhythms in mice. To compare these parameters between mice in the same study or between studies, it is necessary to have information about light/dark cycles (durations and timing of scotophase and photophase), and exactly when the samples are collected. If there is lack of information, reported differences might be completely or partially caused by normal fluctuations in circadian rhythm, or actual differences could be masked.
When animals are fasted in a study, it is necessary to report when the animals were fasted and for how long, otherwise comparison is difficult or almost impossible. Since the normal temperature in an animal room is 20–24℃, mice are housed under less than their thermoneutral temperature of approximately 30℃.96–98 Ambient temperature affects the food intake of mice, and the food intake affects other aspects of the metabolism, hence knowledge about ambient temperature is also important.
Some parameters oscillate during fasting, therefore, it cannot be assumed that, for example, an increase from one point to another is equal to a gradual increase. 56
It is complex to assess whether animal welfare is influenced by fasting. Duration and timing of the fast are important factors. It seems difficult to prove when the mouse recognizes that it is fasting, since this is probably individualistic and associated with habits and external factors. The longest voluntary inter-meal interval reported in this review was of 5 h. 20 This offers an idea of when the mouse starts to experience that it is being fasted, but this interval was registered in the photophase, and cannot be used as an argument for fasting mice for 5 h during scotophase. Secondly, strain differences must be expected. It has been stated that many wild mice only feed and forage in the night, or that they often encounter food restriction or deprivation.33,99 If fasting is considered a part of the mouse’s daily cycle, one can argue that the changes resulting from fasting can be considered to be physiologically normal. This can only be claimed for fasting in the photophase, there is no evidence that mice would omit to eat in the scotophase. Experiments are mostly done in the photophase, which means that the mice will usually be fasting during scotophase because it is easy in practice to remove the food the afternoon before, except when the fasting period is only for a couple of hours.
Mice held under an opposite light/dark cycle can easily be fasted during photophase. Keeping the mice on an opposite light/dark cycle could also provide advantages regarding the fact that cleaning, feeding and activities in general would be done during the activity period of the mice. Disadvantages include trouble and discomfort working in the dark: in cases of large animal rooms everybody needs to agree upon having opposite light/dark cycles and extra time spent when mice have to adapt to the opposite light/dark cycle; for example corticosterone levels take about six days to normalize. 42 If the mice have to be transferred to a lightened room for experimental work, and then put back into the scotophase, it must be considered whether these changes between light and dark may be more disturbing than being disturbed in the photophase. A previous investigation assessed that up to 24 h of fasting is minimally stressful, but reported that data on mice are lacking. 100
Feeding sucrose cubes to rats and mice has been presented as an alternative to fasting them. This feeding can produce marked changes in gastrointestinal tract weight and pancreatic and hepatic structure and function. Using sucrose feeding for toxicology studies or any studies examining hepatic function is therefore not recommended. 101 It is obviously not a valid alternative if the purpose of fasting is to evaluate blood glucose level.
Placing the mice on a grid floor during fasting to avoid intake of bedding material has been recommended. 102 It also precludes the mice from coprophagia. The animal welfare implications of changing the environment by placing the mice on grid floor must be assessed as well as the consequences of the possibility of mice eating bedding or faeces. In a study by Llanos and Nash, gastric content after fasting for 12 h was no different from gastric content of non-fasted mice at a particular time of day (at the end of photophase), and the authors discussed that fasting is not always necessary. 37 The feeding activity of the mice was restricted to the scotophase, thus these results may not be applicable for mice eating in the photophase as well. There was no description of how the feeding activity was measured. To optimize animal welfare, an alternative to feed the mice a hydrated diet, which increases the gastric emptying rate has been provided. 73
There is little evidence of how the mouse behaves during a fast, but stress and aggression requiring single-housing have been mentioned. 71 One study showed that fasted mice increased their general activity significantly compared with non-fasted mice and discussed that the increase might be due to motivation to find food. 99 Another study found increased activity of fasted mice during scotophase, and activity levels similar to non-fasted mice during photophase, while other investigators found photophase hyperactivity during a 24 h fast.34,84 Williams et al. reported activity during fasting to be increased, but not significantly increased, during photophase. 53 It has been suggested that an increase in locomotor activity associated with fasting can be interpreted as food foraging behaviour or exercise-induced thermogenesis. 103
Conclusion
Effects of fasting.

The arrows indicate whether the parameters increase, decrease, do both or stay level during fasting.
= decrease, ↑ = increase, ↕ = decrease and increase, ↔ = no change. See references for information about duration of fasting. Genetics and toxicology are excluded.
Footnotes
Declaration of Conflicting Interests
The authors declare no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
