Abstract
Catheterization of laboratory mice is commonly performed in biomedical research to infuse substances and for blood sampling. One approach is to catheterize the right common carotid artery and advance the catheter until the tip is positioned in the aorta or the proximal brachiocephalic trunk. Owing to the small body size of the mouse, a catheter tends to occupy a great part of even the larger vessel lumens, and this may increase vascular resistance with potential pathophysiological impacts on the heart. The present study compared cardiac function of catheterized mice, with catheter tip placement in the brachiocephalic trunk, with sham-operated mice and non-operated control mice. During four weeks post-catheterization, M-mode echocardiography measurements of the thickness of the left ventricular anterior wall, left ventricular inner diameter and the thickness of the left ventricular posterior wall were performed. The left ventricular volume, ejection fraction and fractional shortening were calculated. Moreover, aortic recordings of the thickness of the medial and lateral walls as well as the inner diameter were measured. Terminally, histological analysis of the hearts was conducted, and body weights and heart weights were compared between groups. No effects on echocardiography parameters, histology, body weights or cardiac weights could be found between groups. In the present study, implantation of a carotid catheter with catheter tip placement in the proximal brachiocephalic trunk had minimal influence on cardiac and aortic physiology and did not induce significant cardiac changes.
Vascular catheterization of laboratory mice is frequently used for the infusion of substances and for blood sampling. Catheterization of the right common carotid artery, where the catheter tip is advanced towards the aortic arch1–3 is a common method. Positioning the catheter in a major vessel is good surgical practice, as it improves blood flow around the catheter and minimizes endothelial damage. The benefits, compared with catheterizing smaller vessels, are improved catheter patency and a reduced risk of complications such as thrombophlebitis and thromboembolism. 4
We routinely catheterize mice in our laboratory and have previously demonstrated minimal surgical stress and good recovery with this technique.5,6 However, histological data from an unrelated study have revealed signs of myocardial degeneration of the carotid artery only four days post-catheterization. 7 Increased aortic turbulence may increase cardiac work load and result in a negative impact on cardiac function. 8 This risk increases over time with a sustained increase in cardiac work load. 9 As vascular catheters are frequently intended for long-term usage, the potential impact on cardiac function from altered hemodynamics may thus increase with the length of the study period. We considered whether placement of the catheter tip close to the aortic arch would affect cardiac function adversely due to a disruption of aortic hemodynamics, as seen in other aortic pathologies such as aortic stenosis 10 and aortic regurgitation, 11 and whether long-term carotid catheterization would result in hypertrophic cardiomyopathy with cardiac dysfunction in these mice.
To our knowledge, no studies have investigated cardiac function in mice with a carotid catheter. A thorough assessment of the impact of carotid catheterization on cardiac function in mice is particularly important, as even subclinical pathological heart conditions may affect animal welfare and the experimental outcome.
The present study investigated cardiac function in mice with a carotid catheter through echocardiography for four weeks post-catheterization. Morphometric and functional parameters were compared with sham-operated mice and non-operated control mice. Body weights (BWs) and cardiac weights were compared between groups, and histopathological analyses of the hearts were conducted to assess tissue level effects from the catheterization. The hypothesis was that catheterized mice would demonstrate impaired cardiac function on echocardiography, and present with histological evidence of cardiomyopathy in contrast to sham-operated and control mice.
Animals and housing
The experiments were approved by the Animal Experiments Inspectorate under the Danish Ministry of Food, Agriculture and Fisheries (license number: 2014-15-2934-01055) and by the local animal welfare body. All animals were handled by trained personnel in accordance with the Guide for the care and use of laboratory animals 12 in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility.
Twenty-one male BomTac:NMRI mice were purchased from Taconic, Ry, Denmark. The animals were 7–8 weeks old and were acclimatized for one week before starting the experiments. The specified gender, stock and number of mice were used to test the hypothesis, based on histological analyses and practical experience from previous catheterization studies.6,7 We generally use outbred NMRI stock in our catheterization studies, as these are commonly used in general biological research. 13 Furthermore, they are genetically uniform in comparison to the wide variability of a species but without lacking genetic diversity, as do the genetically defined inbred mouse strains. 14
The mice were randomly distributed into three groups: one control group (CONTROL, n = 7), one group in which a catheter was surgically implanted in the common carotid artery (CATH, n = 7) and one sham-operated group, in which the carotid artery was ligated but not catheterized (SHAM, n = 7). One catheterized mouse did not recover well and was euthanized as it had reached a humane endpoint, leaving six mice in the CATH group.
The mice were housed in Eurostandard type II cages (Tecniplast, Buguggiate, Italy) in an individually ventilated cage (IVC) system, with aspen chips (Tapvei Oy, Kortteinen, Finland) as bedding. To avoid conspecific interference with the catheters, all mice were singly housed from the day of arrival. Bite bricks (Tapvei Oy), cardboard tubes (Lillico, Horley, UK) and shelters (JAKO Shelter, Molytex, Glostrup, Denmark) were used as environmental enrichment. Wood wool (Tapvei Oy) and Nestlets (LBS Biotechnology, Horley, UK) were used as nesting material. Feed (Altromin 1314; Altromin GmbH & Co. KG, Im Seelenkamp, Germany) and acidified tap water were provided ad libitum. A diurnal rhythm was maintained through a 12:12 h light–dark cycle starting at 06:00 h, and with a 30 min twilight before lights off or on. Cage temperature was kept at 22℃ (±2℃), with a relative humidity of between 45 and 65%, and approximately 75 air changes per hour. The facility followed the Federation for European Laboratory Animal Science Associations (FELASA) guidelines for health monitoring in rodent facilities, 15 where sentinel mice had tested positive for Helicobacter spp. but none of the other pathogens on the FELASA list.
Materials and methods
Surgery
Outline of the project.
All mice were subjected to echocardiography in weeks 1, 4 and 6 of the experiment. In the second week, catheterized (CATH) and sham-operated (SHAM) mice were subjected to surgery, while control mice were left undisturbed. Body weights (BWs) were recorded weekly for all mice. At the end of the study, all mice were euthanized and the hearts were sampled for histopathology. CATH = mice were subjected to surgery with implantation of a carotid catheter. SHAM = mice were subjected to surgery with ligation but not catheterization of the carotid artery. CONTROL = control mice, not subjected to surgery.
The sham-operated mice were prepared for surgery in a similar manner to the catheterized mice. Using the same approach, the right common carotid artery was carefully dissected and ligated with a single suture without implantation of a catheter. The skin was sutured in the same manner as described above.
All surgeries were completed before noon and performed under aseptic conditions. The mice were given pre-emptive analgesia by means of 1 mg/kg BW buprenorphine (Temgesic; Schering-Plough, Brussels, Belgium) mixed in a nut paste (Nutella®; Ferrero, Pino Torinese, Italy) one hour prior to surgery and then once daily for two days post-surgery, as described previously.17,18 To ensure adequate analgesia on the day of surgery the mice were injected with 0.1 mg buprenorphine/kg BW subcutaneously in the flank before terminating anesthesia.
After surgery, the mice were allowed to recover in a quiet room on a heating pad set at 28℃. The well-being of all mice was monitored through daily inspections and recordings of food intake and BWs (data not shown) from three days prior to surgery to three days post-surgery, and then weekly throughout the experiment.
Echocardiography
In weeks 1, 4 and 6 of the study, all mice were subjected to echocardiography (Vevo 770, VisualSonics®; VisualSonics BV, Amsterdam, The Netherlands) under isoflurane anesthesia. To avoid biased measurements from unequal depths of anesthesia, the isoflurane concentration was set such that resting heart rates varied between 300 and 400 beats per minute, which corresponded to 2.5–3.5% of isoflurane delivered in 100% oxygen. During echocardiography, heart rate and respiration were monitored automatically by the software and body temperature with a rectal thermometer (ThermoWorks, American Fork, UT, USA).
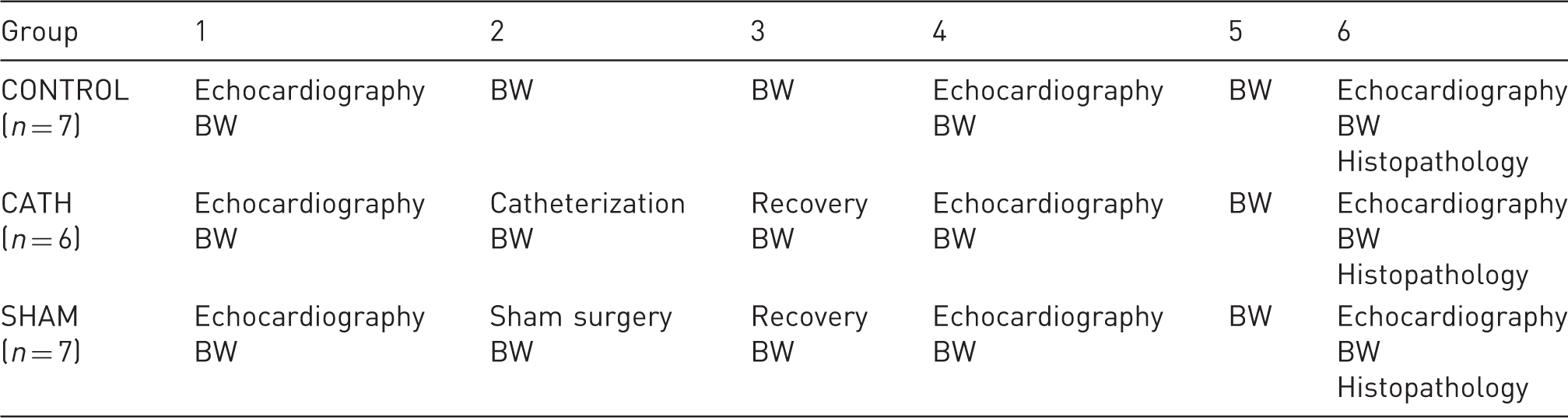
Three M-mode measurements were obtained from the heart, two in a longitudinal view and one in a cross sectional view (Figure 1), to calculate an average approximate thickness of the left ventricular anterior wall (LVAW), left ventricular inner diameter (LVID) and the thickness of the left ventricular posterior wall (LVPW) in diastole and systole. From the aorta, recordings of the thickness of the medial wall (AoMW), lateral wall (AoLW) and the inner diameter (AoID) were obtained in diastole and systole. From these measurements, left ventricular volume in diastole (LV Vol-d) and systole (LV Vol-s), ejection fraction (%EF) and fractional shortening (%FS) were calculated using the software. Correct catheter tip placement was confirmed for all catheterized mice using the first post-surgical echocardiography (Figure 2). All echocardiography recordings were completed within 20–30 min between 09:00 and 12:00 h.
Echocardiography. Examples are shown of M-mode ultrasound measurements of the heart in a cross-sectional view (A) and aorta (C). Estimations of the thickness of the left ventricular anterior wall (LVAW), left ventricular posterior wall (LWPW) and left ventricular inner diameter (LVID) are demonstrated in (B). From the aorta, estimations of the thickness of the medial wall (AoMW), lateral wall (AoLW) and the inner diameter (AoID) were equally obtained, as demonstrated in (D). Catheter tip placement in the proximal brachiocephalic trunk. The top picture shows a B-mode echocardiography displaying the heart (H), ascending aorta (Ao) and brachiocephalic trunk (Br) of one control mouse. The bottom picture shows the same anatomic structures and correct catheter placement (arrow) in one catheterized mouse.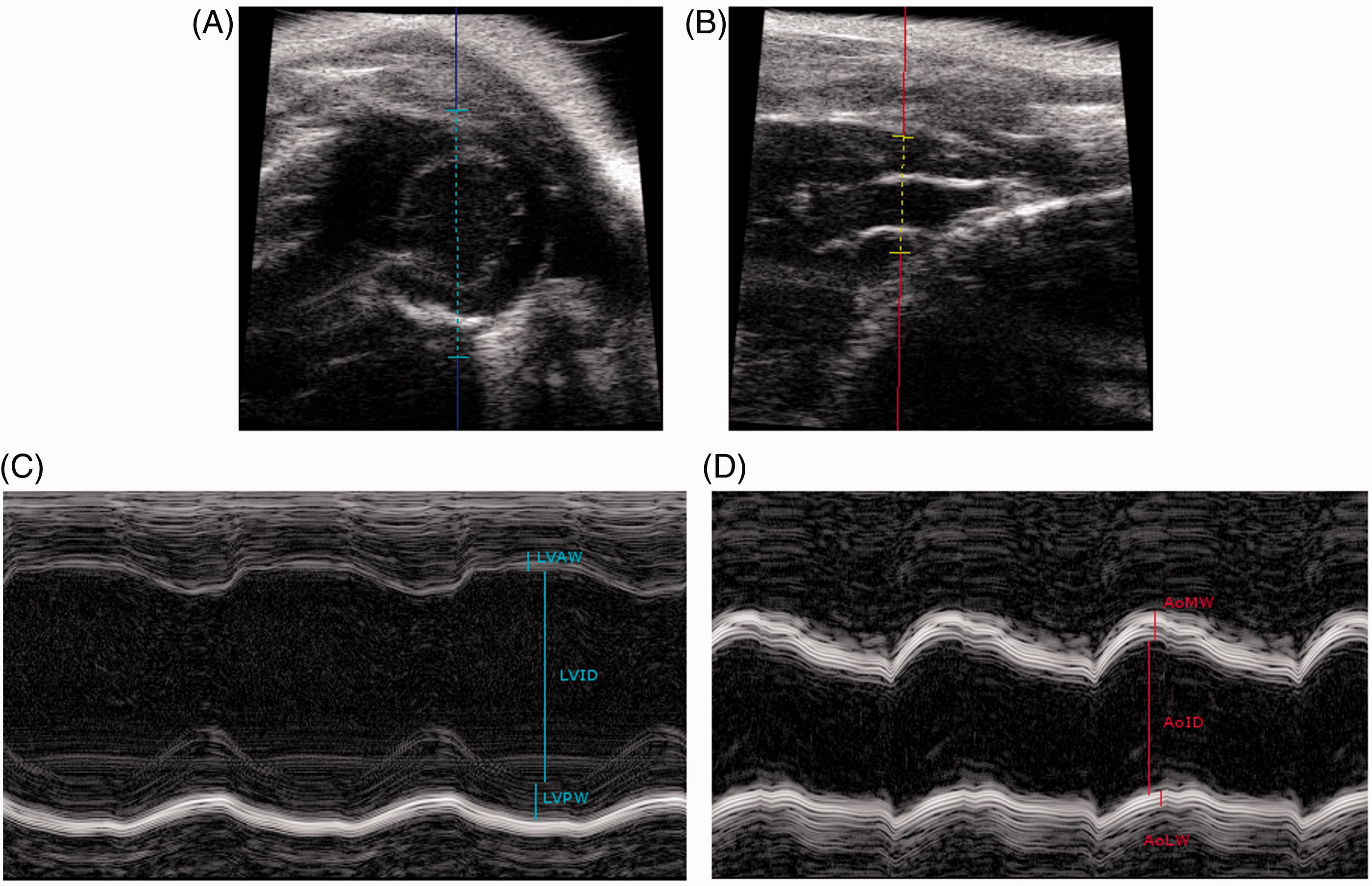
Pathology
Four-point score system for histological grading of cardiac sections.

Hearts were sampled for histopathological evaluation and serial transverse sections were evaluated from each heart.
Each section was graded with respect to six parameters: inflammation, perivascular inflammation, cardiomyocyte vacuolization, cardiomyocyte hyalinization, necrosis and fibrosis. Cardiomyocyte vacuolization was defined as a degenerative change with varying degrees of sarcoplasmic vacuolization. Cardiomyocyte hyalinization was similarly regarded as a degenerative change, where cardiomyocytes lost their patterns of cross striations and attained a homogenous eosinophilic appearance. Necrosis of cardiomyocytes was defined as the presence of cells with homogenous sarcoplasm and pyknotic or karyorrhectic nuclei. Inflammation was characterized by the infiltration of inflammatory cells within and/or between cardiomyocytes. The accumulation of perivascular inflammatory cells was scored as a separate parameter (perivascular inflammation). Fibrosis was defined as the presence of cells with ovoid to elongated nuclei (fibroblasts) lying within bands of eosinophilic fibrillar collagen, replacing lost cardiomyocytes.
Statistics
Statistics of histology data.
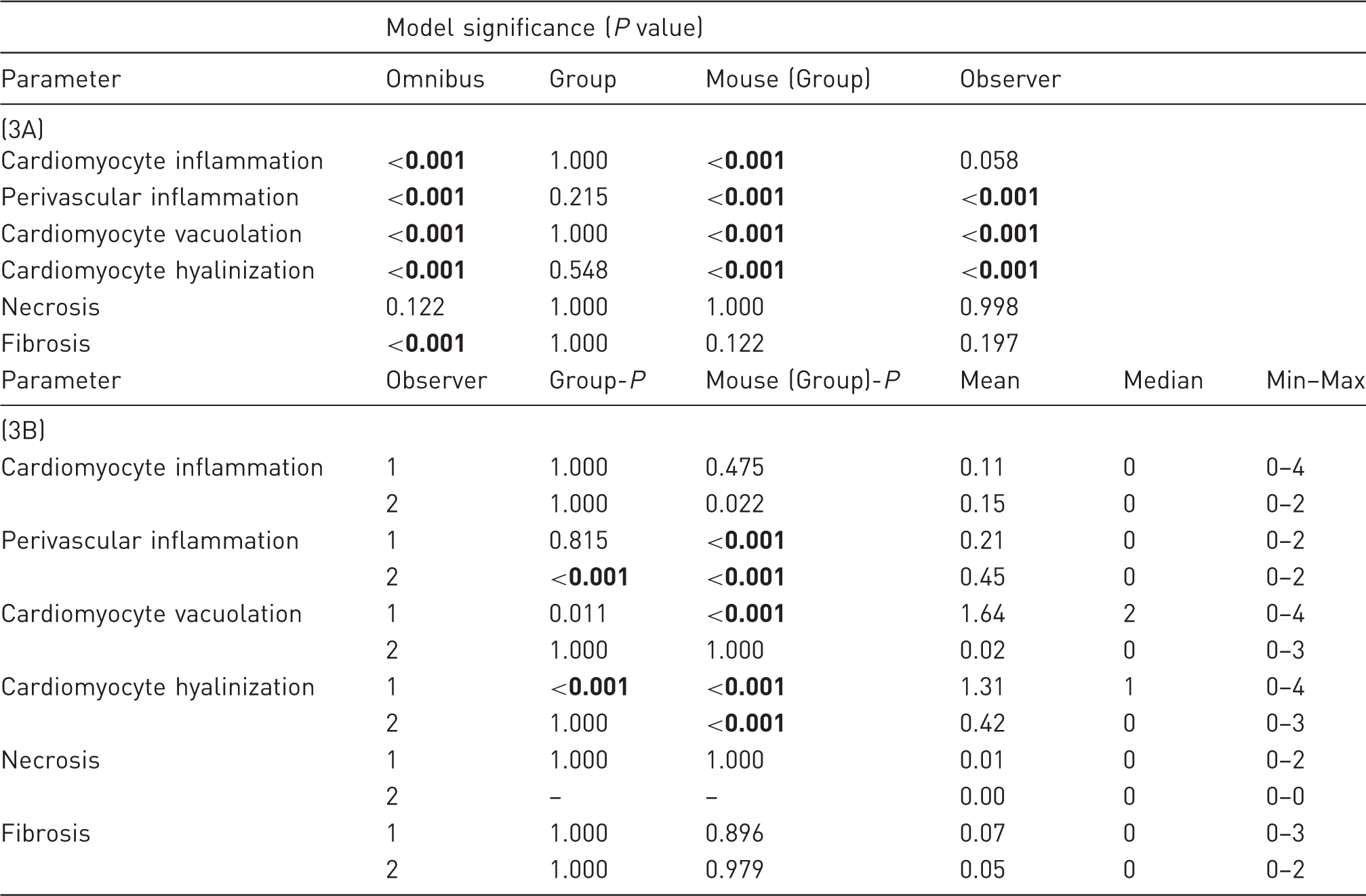
The top section (3A) presents the P values for each analyzed parameter, overall (omnibus) and for each model factor in a generalized linear model, where Group, Mouse and Observer were defined as model factors and where Mouse was defined as nested within groups. The bottom section (3B) presents P values, mean, median and minimum (Min) and maximum (Max) values for each of the parameters, separated by observer. To account for multiple comparisons, Bonferroni’s corrections were applied to avoid type 1 errors, resulting in an adjusted P value of 0.008. Significant P values are set in bold.
To account for multiple comparisons, Bonferroni’s correction was applied to the echocardiography and histology data to avoid type 1 errors, resulting in an adjusted critical P value of 0.008, below which these data were considered to be significant. As for the weight data, P values below 0.05 were considered to be significant.
Retrospective power calculations were performed using echocardiography data from the control group against the catheterized group, where the highest data variation and the smallest difference between groups were expected. With β = 0.20, α = 0.05 and standard deviations (SDs) at 0.33 (cardiac measurements) and 0.08 (aortic measurements), respectively, detectable effect sizes were estimated to be 50% for cardiac measurements and 13% for aortic measurements, which were considered to be biologically relevant.
Results
Echocardiography
Please refer to Supplementary file 1 (supplementary material can be found online at http://journals.sagepub.com/home/lan) for the descriptive statistics. The measured variables (LVAW, LVID, LVPW in diastole and systole, respectively) did not differ between groups (F(12, 92) = 1.243, P = 0.267) or over time (F(12, 92) = 1.416, P = 0.173). Neither did the calculated variables, LV Vol-d and LV Vol-s, differ between groups (F(12, 92) = 0.384, P = 0.966) or over time (F(12, 92) = 1.357, P = 0.201).
The aortic measurements also did not differ between groups (F(12, 94) = 0.954, P = 0.498) or over time (F(12, 92) = 1.374, P = 0.192).
Weights
Body weights (BWs), cardiac weights and heart–BW ratio.

The table shows the mean (mean ± SD) BW and cardiac weight (g) of control mice (CONTROL, n = 7), catheterized mice which had an arterial catheter implanted in their right common carotid artery (CATH, n = 6), and sham-operated mice which had their right common carotid artery ligated but not catheterized (SHAM, n = 7) as well as the heart–BW ratio. No significant group differences were found.
Histology
Comprehensive histological grading data are available through Figshare.com (https://figshare.com/s/0e15c0c4716801045643; DOI: 10.6084/m9.figshare.4263569). Histological examples are illustrated with different parameters in Figures 3 and 4.
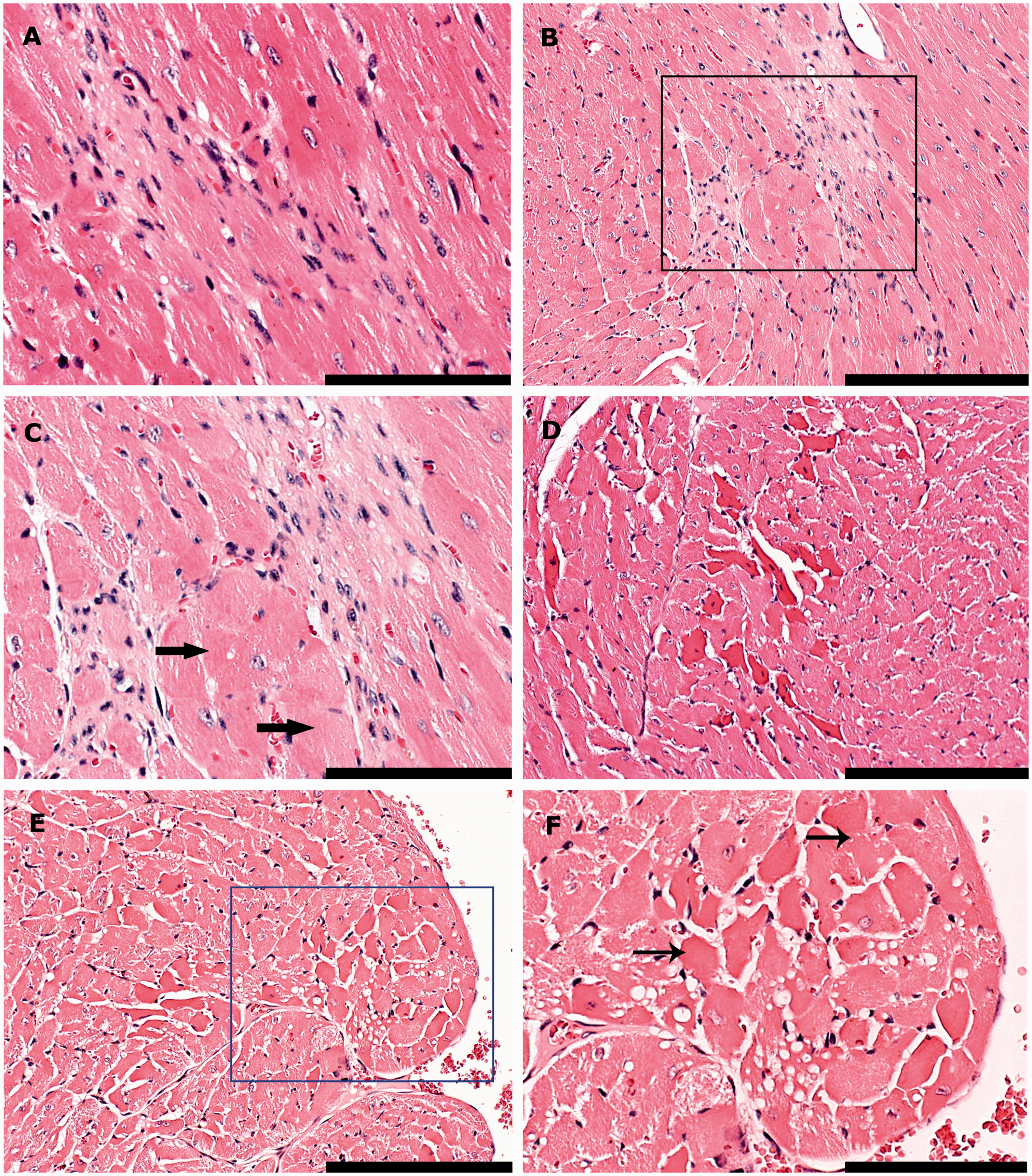
Histological examples of inflammation. (A) Left ventricle. The perivascular spaces of two vessels are minimally infiltrated by few, countable mononuclear cells (perivascular inflammation, Grade 1). (B) Left ventricle. The lymphocytic infiltrate surrounding this vessel is mild, consisting of more than a few countable cells but occupying less than 250 × 250 µm (Grade 2). (C–D) Base of heart, interventricular septum and aortic valves. The myocardium is severely infiltrated by lymphocytes and histiocytes, admixed with lesser numbers of neutrophilic granulocytes. The focus is larger than 500 × 500 µm (Grade 4). The area in the black box is shown in a higher magnification (40×) in (D). Bars = 100 µm in (A–B), bar = 200 µm in (C) and bar = 50 µm in (D). Histological examples of degenerative lesions. (A) Interventricular septum. A mild focus, occupying more than the space of 10 cardiomyocytes, but less than 250 × 250 µm (Grade 2), consists of cells with ovoid to elongated nuclei (fibroblasts) lying within eosinophilic, fibrillar collagen (fibrosis). (B–C) Left ventricle. This area of fibrosis is moderate (larger than 250 × 250 µm but less than 500 × 500 µm, Grade 3). Fibroblasts lie within branching bands of fibrillar collagen and degenerative to necrotic cardiomyocytes (thick arrows). The area in the black box is shown in a higher magnification (40×) in (C). (D). Right ventricle. Focally cardiomyocytes appear to be hypereosinophilic with loss of the normal pattern of cross striation (cardiomyocyte hyalinization, Grade 2). (E–F) Right ventricle. Focally, cardiomyocytes express varying degrees of sarcoplasmic vacuolation and necrotic changes, distinguished by the attainment of a homogenous appearance, sarcoplasmic fragmentation, pyknosis or karyorrhexis (thin arrows) (Grade 3). The area in the blue box (E) is shown in a higher magnification (40×) in (F). Bars = 100 µm in (B, D and E) and bars = 50 µm in (A, C and F).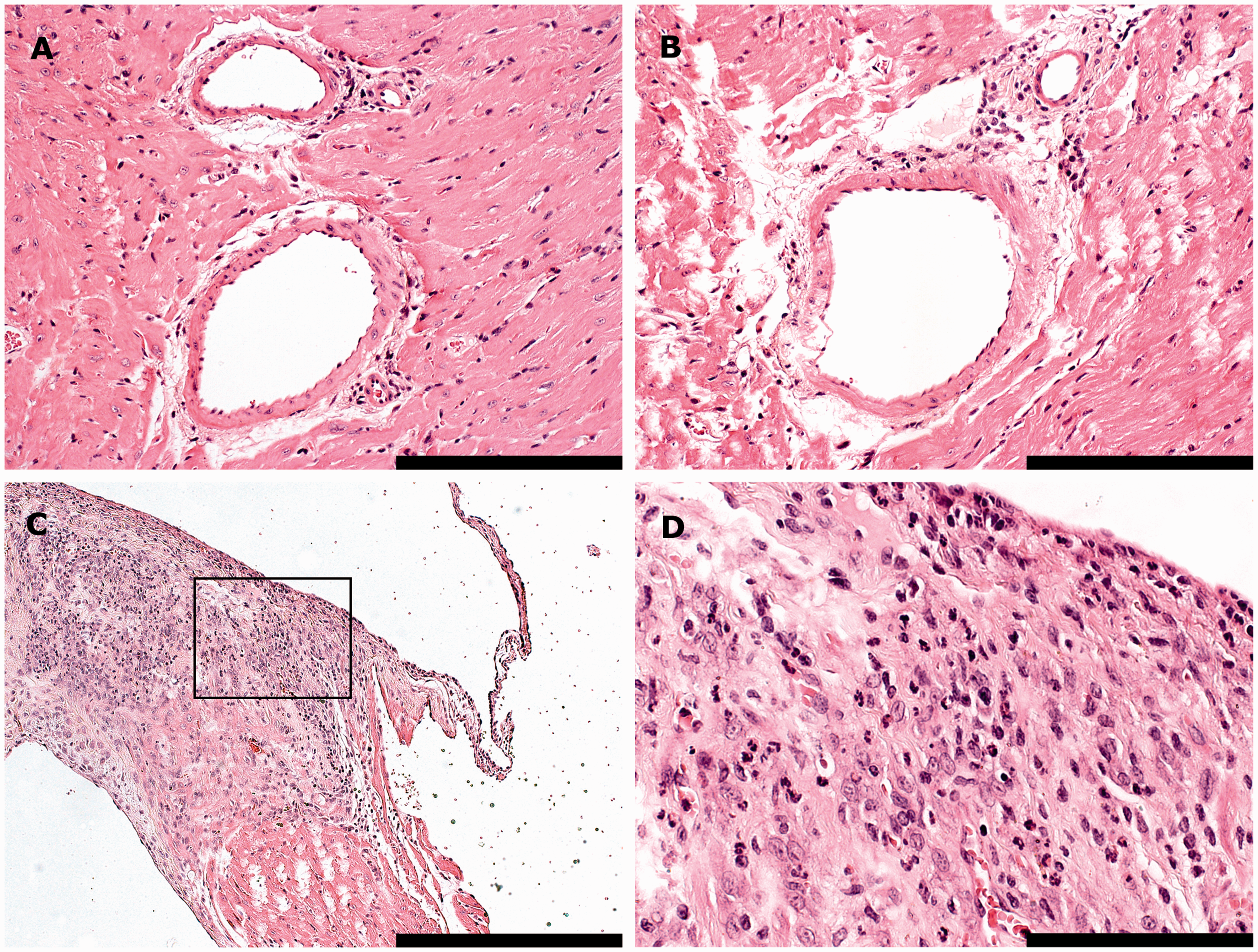
The omnibus test found overall significant effects in the dataset for all the histological parameters, except for cardiomyocyte necrosis (Table 3). These effects were attributable to variation within groups of mice rather than between experimental groups. One parameter (fibrosis) was found to be significant in the omnibus test but not within or between groups of mice. This effect was attributed to an interaction between Groups and Observers. No differences could be identified between groups for any of the parameters.
For three of the parameters (perivascular inflammation, cardiomyocyte vacuolation and cardiomyocyte hyalinization) significant differences in the scorings were found between the observers, with Observer 2 generally scoring lower. When analyzing these parameters separately for the two observers, an overall group difference in perivascular inflammation was found by Observer 2, which was not significant for any of the model factors. For cardiomyocyte hyalinization, a significant group difference could be identified by Observer 1, where the control mice had significantly higher scores compared with the catheterized (P = 0.001) and sham-operated (P = 0.002) mice.
Discussion
Quick and easy vascular access is important in many mouse models, especially in drug discovery and safety testing. Vascular catheters in mice, however, may be associated with complications that threaten survival, complicate post-surgical catheter maintenance and impair animal welfare.7,16,23 Thus, further efforts must be made to minimize deleterious effects on the models, and the redundant use of animals in research.
Carotid catheterization is a well-established procedure in mice.1,5,24 During surgery, the catheter is advanced such that the tip is placed into a major vessel in order to improve blood flow around the catheter and to minimize catheter patency failure. 4 The catheter is advanced via the right common carotid artery down the brachiocephalic trunk, with the tip positioned in the proximal brachiocephalic trunk (as in the present study) or in the ascending or descending aorta.3,25,26 Most published literature concerning this technique, however, does not describe precise placement of the catheter tip, and thus some methodological differences may exist between laboratories.
In the present investigation, a commercially available standardized catheter was used, which has been described previously. 16 This catheter had a retention bead one cm from the tip for securing the catheter inside the vessel during surgery. Based on our experience, this also positioned the catheter tip in the proximal brachiocephalic artery, adjacent to the aortic arch in NMRI mice of the applied age group (7–8 weeks old). Correct catheter placement was confirmed by echocardiography (Figure 2). The aorta was avoided due to the natural turbulence caused by blood ejected from the left ventricle during systole, and in order to minimize direct effects on aortic arch baroreceptors. In small animals such as the mouse, however, a catheter tends to occupy a great part of even the larger vessel lumens, and this may increase vascular resistance systemically, leading to increased cardiac pressure and a decrease in cardiac output. 27
Pathological cardiac hypertrophy is a well-known consequence of altered hemodynamics, pressure or volume overload, 28 such as that caused by aortic constriction.29,30 LV hypertrophy is one adaptation to chronic hemodynamic overloads on the heart, 31 and may lead to congestive heart failure and contractile impairment.9,30 The present study was conducted to examine potential pathological effects of carotid catheterization on cardiac physiology and function, due to an assumed effect on aortic hemodynamics from the presence of the catheter. The sham-operated group, which had the carotid artery ligated but not catheterized, was included to account for the isolated effects of carotid ligation on hemodynamics and baroreceptor activity.
Grossly, no differences in cardiac mass were found between groups, either when comparing heart weights or the heart–BW ratios, revealing no macroscopic evidence of cardiac hypertrophy for any of the mice.
Echocardiography recordings of the mouse hearts were used to assess potential physiological effects from the long-term catheterization. In contrast to our hypothesis, no group differences on the cardiac and aortic parameters could be identified using echocardiography. Anesthesia is known to relax vascular tone and baroreceptor reflexes, resulting in hypotension32,33 which may potentially affect the measurements. However, isoflurane anesthesia is generally known to have minimal cardiodepressant effects in comparison to other anesthetic agents,34,35 and as anesthetizing the mice for the echocardiography procedures was necessary, a minor relaxation of vascular tone was accepted and was assumed to be equal across groups. Vascular tone was maintained steady by controlling the anesthetic depth during echocardiography.
On a cellular level, cardiomyopathy in mice normally follows a pattern of progression, beginning with cardiomyocyte degeneration, which then advances to necrosis, loss and replacement by fibrosis. 21 In the present study, lesions of degeneration and necrosis were often seen without concurrent fibrosis (Figures 4D–F), indicating that spontaneous alterations were more likely to be a cause of normal background changes. However, fibrosis, indicating replacement of lost cardiomyocytes, 36 was also noted on histology and was always evaluated as potential cardiomyopathy with or without concurrent degeneration or necrosis. The presence of inflammatory infiltrates along with clear changes of cardiomyopathy is less common in mice, 21 but was included as a parameter in the present study due to the implantation of a vascular catheter in these mice, which was regarded as a potential source of infection.
Contrary to our hypothesis, no effects of the experimental procedures could be identified with histology. Although the generalized linear analysis found overall significant effects in the model, these effects were attributable to variation within the groups of mice and discrepancies between the two observers’ scorings for three of the parameters. For only one of these parameters (cardiomyocyte hyalinization) a significant difference was found between groups, where the control group was scored with a higher degree of cardiomyocyte hyalinization compared with the two experimental groups. Cardiomyocyte hyalinization was, in conjunction with cardiomyocyte vacuolation, included as a measure of myocardial degeneration and initial cardiomyopathy. Multifocal myofiber degeneration may, however, occur spontaneously. 36 Thus, the higher scorings of cardiomyocyte hyalinization in control mice were attributed to normal background variation. Although an observer discrepancy was identified for three histological parameters (perivascular inflammation, cardiomyocyte vacuolation and cardiomyocyte hyalinization), no significant group differences related to the experimental technique could be identified either collectively or in the observers’ respective score sets, and it is unlikely that this discrepancy should have produced false negative results.
Based on the results in the present study, implantation of a carotid catheter with the tip positioned in the proximal brachiocephalic trunk has minimal influence on cardiac and aortic physiology, and does not seem to induce significant histopathological changes in the heart. These results are in contrast to histological analyses obtained in a previous study, 7 where early signs of myocardial degeneration with fibrosis were seen only four days post-catheterization. In this study, catheterized mice were connected to an automated blood sampling device following surgery, and were subjected to computer-driven blood sampling and saline infusions. The present study also aimed to investigate the isolated effects of long-term carotid catheterization, and why the catheter was capped following surgery and not accessed until euthanasia. It is possible that the frequent access and utilization of the catheter in the previous study increased cardiac strain, and further research may be warranted to fully examine the cardiac effects of catheterization with regular access through the catheter.
In conclusion, long-term carotid catheterization with catheter tip placement in the proximal brachiocephalic artery had no detectable effects on cardiac or aortic function in the present study.
Footnotes
Acknowledgements
The authors would like to thank Dr Cory Brayton and Dr Otto Kalliokoski for thoroughly reading and commenting on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
