Abstract
The ventriculosinus shunt is a promising treatment for hydrocephalus. Currently, different shunt techniques exist, and it is not clear whether one is preferable. This pilot study reports on a non-hydrocephalic goat model (Saanen breed) that provides opportunities to evaluate and optimize several aspects of the ventriculosinus shunt technique. Analysis of the coagulation properties of 14 goats by a viscoelastic coagulation monitor showed that goats have a hypercoagulable state compared to humans. This property can be partially counteracted by antiplatelet drugs. During implantation of a ventriculosinus shunt, a pulsatile reflux of blood was observed. After implantation, the animals recovered to their preoperative state, and none of them developed a superior sagittal sinus thrombosis. Evaluation of the shunts after 16 days showed an obstructing luminal clot. Several model-related factors may have promoted this obstruction: the absence of hydrocephalus, the hypercoagulability of caprine blood and the smaller dimensions of the caprine superior sagittal sinus. However, the pulsatile reflux of blood, which is caused by the compliance of the shunt system distal to the valve, may have been an important factor as well. In conclusion, the non-hydrocephalic goat model limits animal suffering and simplifies the study protocol. This model allows researchers to evaluate their implantation technique and shunt hardware but not the efficacy of the treatment or shunt survival.
Introduction
Hydrocephalus, or the accumulation of cerebrospinal fluid (CSF) in the brain, is treated with ventriculo-peritoneal or ventriculo-atrial shunts. An important drawback of these techniques is siphoning, in which the CSF column within the catheter exerts suction through gravity when the patient is in a sitting or standing position. This effect leads to shunt-related intracranial hypotension and aspiration of the choroid plexus into the proximal catheter. 1 Intracranial hypotension causes headaches, nausea, vomiting and even subdural haematomas, while aspiration of the choroid plexus is a major cause of shunt obstruction. 2 Despite the usage of expensive resistance valves and anti-siphon devices, the rate of shunt failure – which requires surgical revision – remains as high as 50% in the first 2 years. 3
A more physiological, but still experimental, technique is the ventriculosinus shunt. This shunt drains CSF to a natural resorption site, such as the superior sagittal sinus, and theoretically reduces the risk of shunt failure in several ways.4,5 First, excessive drainage of CSF is prevented by the natural, self-regulating anti-siphon effect of the internal jugular vein. Second, the shunt system is shorter, less complex and confined to the skull, which minimizes the risk of mechanical failure and infection. 6
The efficacy of the ventriculosinus shunt was evaluated in a few clinical studies.7–10 Although the technique has been proven to be safe and effective,7–11 several issues and uncertainties persist. 11 First, authors do not agree on whether the tip of the shunt should be directed towards or against the direction of blood flow. 11 Second, problems with the implantation of the shunt system exceed 10% in most of the available studies.9,11 A technical advance using the Seldinger technique was published, but this method was evaluated in only one patient. 12 Third, when the shunt system is correctly implanted, obstruction of the intravascular catheter remains an issue.7,9,10 Based on their experience, different authors have proposed several prototypes of the ventriculosinus shunt with distinct features.13–15 It is not clear which of these shunt systems would be preferable. 11
To optimize the implantation technique and to evaluate the different prototypes of the ventriculosinus shunt, an animal model would be an indispensable tool. 16
The only model described to date to evaluate a ventriculosinus shunt is a hydrocephalic dog model that used only two animals, and has substantial shortcomings: the induction of hydrocephalus was not well-tolerated, and the implantation of the ventriculosinus shunt resulted in a thrombosis of the superior sagittal sinus. 16 The only way to prevent venous congestion and sinus thrombosis in a canine model is an extended miniaturization of the intravascular catheter to fit the small size of the superior sagittal sinus. 16 To facilitate the use of shunt systems that are similar in size and design to their human counterparts, a large animal model must be created. 17 A hydrocephalic sheep model to evaluate ventriculo-peritoneal shunts has been described in a previous study. 17
The purpose of this pilot study is to assess the suitability and feasibility of a goat model (specifically ‘Saanen breed’ goats, which are very similar to sheep) for evaluating the implantation technique and design of the ventriculosinus shunt. Since hydrocephalus is not required for addressing the research question, a non-hydrocephalic model was chosen.
The study consists of a cadaver anatomical study, an in vitro coagulation assay (Sonoclot Analyzer©) and an in vivo study. The cadaver anatomical study describes the relevant surgical anatomy of the caprine brain ventricles and superior sagittal sinus. The in vitro coagulation assay analyses the coagulability of the caprine blood. The in vivo study assesses the feasibility of the in vivo implantation of a ventriculosinus shunt.
Materials and methods
Animals
The species was selected based on anatomical characteristics. In pigs and cattle, frontal access to the ventricles and superior sagittal sinus may be impeded by the pronounced caudal extension of the frontal sinuses. The remaining possible models included sheep, goats, and horses or ponies. Ultimately, domestic dairy goats (Saanen breed) were chosen because of their manageability and short hair. All goats were purchased from dairy goat farms. The goats were 1–2 years old and weighed less than 65 kg. Only female goats were included because the more pronounced cornual processes of bucks could impede frontal ventricular access.
Since the investigation was a pilot study, small sample sizes were chosen. The study consisted of a cadaver study (9 goats), an in vitro coagulation assay (14 goats) and an in vivo study (3 goats). Three of the 14 goats included in the in vitro coagulation assay were also used for the in vivo study; thus, 23 goats were used in total. The studies were conducted serially, and the goats were allocated to the experiments in order of acquirement.
All experiments in this study were approved by the Ethics Committee of the Faculty of Veterinary Medicine of Ghent University (EC2012/187, EC2013/66, EC2013/129). The care and use of animals were in full compliance with the most recent national legislation (Belgian Royal Decree of 29 May 2013), 18 and the relevant European Directive (2010/63/EU). 19
For the cadaver anatomical study, the goats were euthanized directly after arrival at the research facility. The remaining goats were admitted to the animal facility at least 10 days prior to the start of the study for acclimatization and health checks (including clinical investigation, ultrasonographic examination of the abdomen and thorax, and blood and faeces analyses). The animals were housed in small groups (3–4 animals per cage). The cage measured 3 × 4 m2. The bedding consisted of straw, and cage enrichment (an elevated platform) was provided in all cages. Natural light was provided by translucent windows, and the cage temperature was maintained between 15 and 20℃. The animals were fed hay and water ad libitum and supplemented with 450 g of nutritional pellets daily.
Clinical evaluation of the animals was performed twice daily using a clinical scoring system. This scoring system was developed by the authors in cooperation with the Department of Internal Medicine of the Faculty of Veterinary Medicine of Ghent University. The score consists of objective (e.g. vital parameters, weight and food intake) and subjective (e.g. general impression and posturing) parameters, and it is based on existing pain scores.20,21 A perfectly healthy animal without signs of distress will score 0, while a very ill animal with signs of unbearable suffering will score 21. A total score of 6, or a maximum score for one of the parameters, was the threshold for therapeutic action to alleviate suffering.
All animals fasted for 24 h before surgery and received an intramuscular injection of 2.5 mg trimethoprim and 12.5 mg sulfadiazine/kg body weight (Borgal® 24%, Virbac, Barneveld, The Netherlands) starting one day prior to surgery and daily until 4 days after surgery. If needed, postoperative pain and fever were treated with 48 mg meloxicam subcutaneously (Metacam, Boehringer Ingelheim, Germany).
Euthanasia was performed by intravenous administration of 50 mg/kg sodium pentobarbital (20%) (Pentobarbital, Kela, Hoogstraten, Belgium) after intravenous premedication with 0.3 mg/kg midazolam (Dormicum, Roche Pharma, Brussels, Belgium) and 0.1 mg/kg morphine (Morphine HCL Sterop, Brussels, Belgium). The anaesthesia was intravenous propofol 2–4 mg/kg (Propovet, Parsippany-Troy Hills, NJ, USA).
Cadaver anatomical study
After euthanasia, the goats were frozen at −20℃ and decapitated. The heads were sawed in coronal slices at approximately a 1 cm thickness. The superior sagittal sinus was inspected for septa or trabeculae and measured at the confluens sinuum and 1 cm rostral to the confluence. The rostral horns of the lateral ventricles were measured at the level of the coronal suture. A point on the coronal suture was determined where a catheter, which is inserted perpendicularly to the skull in the coronal plane, would enter the ipsilateral ventricle. The length of the intraventricular trajectory of a shunt placed in that manner was measured.
In vitro coagulation assay
The goats were anaesthetized on the morning of the operation in the operation theatre. Eight animals received 75 mg clopidogrel (Plavix, Sanofi, Machelen, Belgium) and 80 mg acetylsalicylic acid (Asaflow, Takeda, Sint-Jans-Molenbeek, Belgium) perorally starting the day before the procedure. Six goats did not receive any antiplatelet drugs. Three millilitres of CSF and 15 ml of venous blood (citrated with 0.129 mole Na3-Citrate/l, Terumo, Heverlee, Belgium) were obtained by a sub-occipital puncture and a venipuncture, respectively. The samples were stored at room temperature and processed within 60 min. Because tissue factor is a potent activator of the extrinsic coagulation pathway, the first 1 ml of CSF and 3 ml of blood were not used for analysis.
To assess the impact of CSF on coagulation, different concentrations of CSF were added to the blood samples (0 µl CSF/ml blood and 100 µl CSF/ml blood). Subsequently, the mixture was recalcified with 40 µl of 0.25 mol/l CaCl2 and analysed by a Sonoclot Coagulation Analyzer© (Sienco, Arvada, CO, USA).
The Sonoclot Analyzer© is an in vitro method for the analysis of the coagulation process from the start of fibrin formation through polymerization of the fibrin monomer and platelet interaction and eventually to clot retraction and lysis. The system consists of an open-ended plastic probe, which vibrates vertically while immersed in a cuvette containing a 0.33 ml sample of whole blood, and the probe measures changes in the viscoelastic properties of whole blood during the clotting process. The curve or signature reflects the changes in viscoelasticity from a liquid to a solid state. 22 Three parameters are defined: the activated clotting time (ACT), clot rate (CR) and platelet function (PF). The ACT is an expression of how long the sample remains completely in the liquid phase and corresponds to the time necessary for fibrinogen to be converted to fibrin monomers. The CR is the slope of the second peak/plateau of the curve, which corresponds to the polymerization of fibrin monomers; a faster fibrin polymerization will be reflected by a steeper slope. The PF represents the attachment of platelets to fibrin and the retraction of the clot. 22
Statistical analysis was performed using SPSS Statistics® 22 (IBM Corp., Released 2013, IBM SPSS Statistics for Windows, Version 22.0, Armonk, NY, USA).
The distribution of the Sonoclot parameters was evaluated using QQ-plots and a Shapiro–Wilk test
To evaluate the effect of CSF on blood coagulation, the goats were considered one group independent of antiplatelet drug administration. Each of the Sonoclot parameters of pure blood was compared to the blood–CSF mixture using a paired-sample T test.
The effects of antiplatelet drug administration on the Sonoclot parameters were evaluated by comparing the Sonoclot parameters of the goats that received antiplatelet drugs with the parameters of the goats that did not receive antiplatelet drugs. An independent sample T test was used.
Statistical significance was set at 5%.
In vivo study
Shunt implantation
A ventriculosinus shunt was implanted in three goats under general anaesthesia. The procedure started in the morning and was performed in the operation theatre. The animals received 75 mg clopidogrel (Plavix, Sanofi, Machelen, Belgium), 80 mg acetylsalicylic acid (Asaflow, Takeda, Sint-Jans-Molenbeek, Belgium) and 40 mg pantoprazole (Pantomed, Takeda, Sint-Jans-Molenbeek, Belgium) perorally starting 24 h before surgery until the end of the study.
A surgical plan was made based on a contrast-enhanced brain CT scan the day before surgery. As shown in Figure 1, the head of the animal was secured in a custom-built frame. Under sterile surgical conditions, a 10 cm midline skin incision was made that reached from 2 cm rostral to the coronal suture to 2 cm caudal to the lambdoidal suture. A hole was drilled approximately 18 mm to the right of the midline on the coronal suture, and another hole was drilled on the midline just rostral to the lambdoidal suture. To introduce the ventricular catheter, an aiming device was used, which was similar to a device described in the literature.
23
Positioning and incision. (a) The goats were positioned prone with the head slightly elevated and fixated in a custom built head clamp. (b) A linear incision was made, starting 2 cm rostral to the coronal suture and ending 2 cm caudal to the lambdoidal suture. The foramen magnum and the arch of the atlas were also marked as these are the anatomical references for a suboccipital puncture. C: coronal suture; L: lamboid suture; FM: foramen magnum; C1: atlas.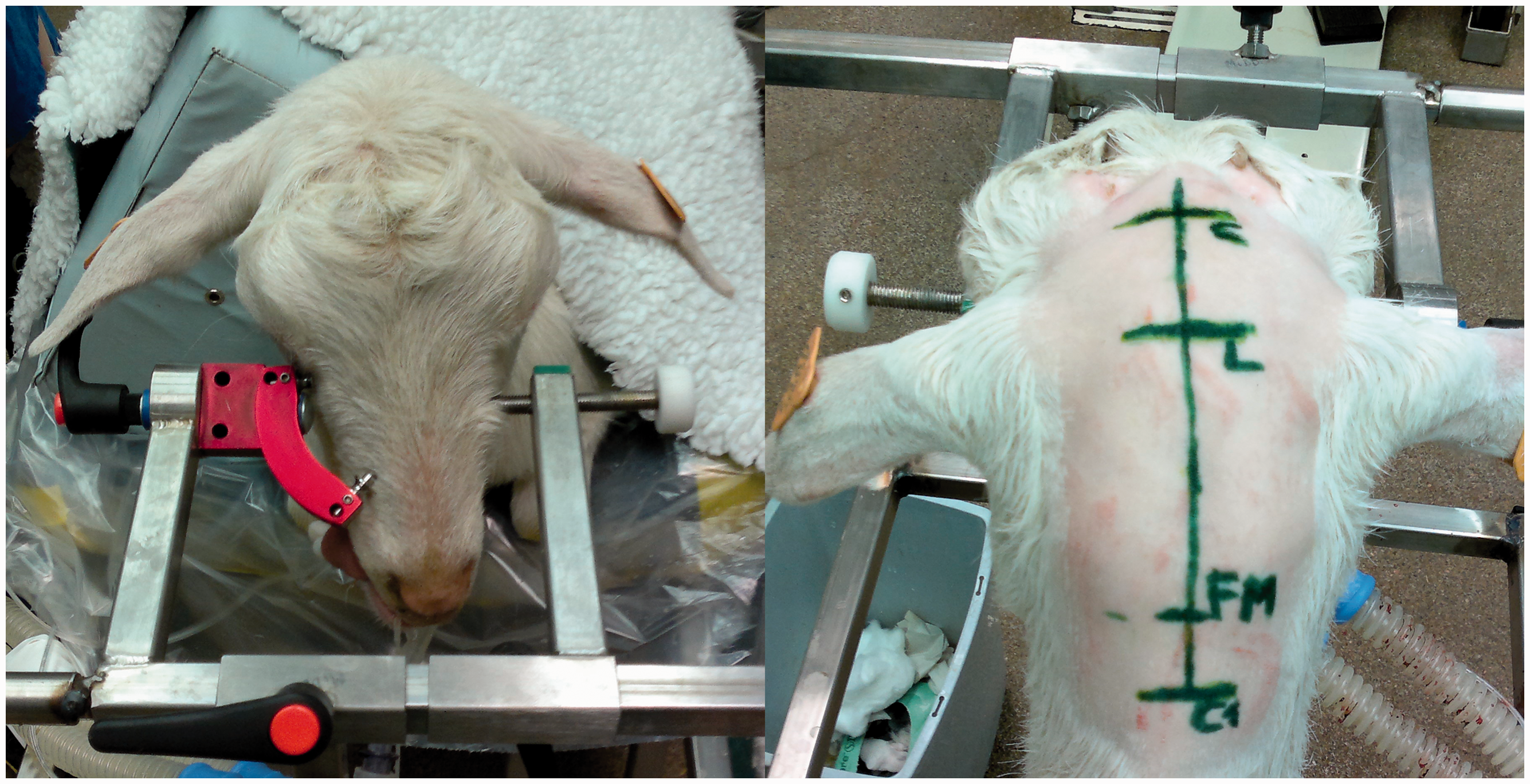
This device was placed over the burr hole parallel to the sagittal plane and perpendicularly to the surface of the skull in the coronal plane. In the sagittal plane, the appropriate angle was set, and the ventricular catheter (Codman Hakim Ventricular Catheter, inner diameter (ID) 1.4 mm and outer diameter (OD) 2.7 mm) was inserted into the rostral horn of the right lateral ventricle. The correct position was confirmed by the outflow of CSF. The catheter was connected to a Codman Hakim very low pressure valve® (Depuy Synthes, Codman Neuro, Raynham, MA, USA) and obstructed by a clamp.
The superior sagittal sinus was then identified. The roof of the sinus was punctured with an 18 G needle and/or incised with a surgical blade (no. 11). A peritoneal catheter (Codman Hakim Peritoneal Catheter, ID 1 mm and OD 2.2 mm) was introduced over a length of 3 cm in the superior sagittal sinus against the direction of blood flow.
The correct position of the sinus shunt was confirmed both by injection of a 0.9% sodium chloride solution and by aspiration of blood. The intravascular catheter was then connected to the valve after flushing the catheter with a 0.9% sodium chloride solution. The clamp was removed from the ventricular catheter, and the system was inspected for drainage of CSF to the sinus or reflux of blood to the catheter system.
In one goat, a spinal epidural catheter (Portex, Smiths-Medical, Hythe, UK, ID 0.55 mm and OD 1.03 mm), which was obliterated at its proximal end, was inserted in the superior sagittal sinus before implantation of the final catheter. After being inspected for blood reflux, the spinal epidural catheter was replaced by the final Codman Hakim peritoneal catheter.
Postoperative evaluation
Postoperatively, head computed tomography (CT) was acquired to verify the correct position of the shunt components and the patency of the superior sagittal sinus. The animals were evaluated daily until two weeks after the implantation. This follow-up period was chosen because full recovery or major complications, such as a superior sagittal sinus thrombosis or infection, are expected to manifest within this period. For clinical evaluation, the scoring system described above was used.
At the end of the follow-up period, the animals were anaesthetized, and the patency of the ventricular and intravascular catheters was assessed using water columns. At the end of this procedure, the animals were euthanized, and the superior sagittal sinus containing the intravascular catheter was explanted with the catheter left in place. The lateral wall of the superior sagittal sinus was opened to visualize the intravascular catheter. The correct position was verified, and the presence of clots was evaluated using a stereomicroscope. Subsequently, the catheters were removed along with the surrounding sinus wall, and the catheters were transversely cut in two equal parts and fixed in a HEPES buffer containing 2.5% glutaraldehyde solution (Sigma Aldrich, Steinheim, Germany). After fixation, the samples were rinsed with a 0.9% sodium chloride solution, air-dried, mounted on aluminium pin mounts and sputtered with platinum particles.
The samples were then observed with a JEOL JSM-5600 LV scanning electron microscope (SEM) with the lumen facing the camera and evaluated at different magnifications.
Results
Cadaver anatomical study
Dimensions (mean values and 95% confidence intervals) of the superior sagittal sinus (SSS) and lateral ventricles. The dimensions of the superior sagittal sinus are expressed as latero-lateral × dorso-ventral distances. The dimensions of the ventricles are measured at the level of the coronal suture.
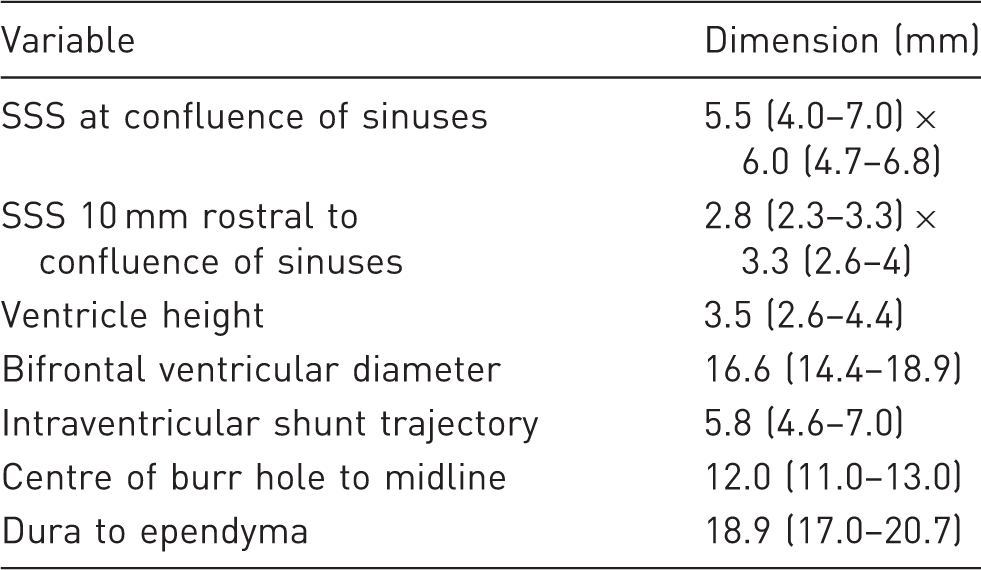
The confluence had variable dimensions. The mean latero-lateral diameter was 5.5 mm, and the dorso-ventral diameter was 6 mm. As can be seen in Figure 2(a), the confluence frequently contains septa and trabeculae.
Anatomy of the caprine superior sagittal sinus and lateral ventricles. (a) Septa in the confluence of sinuses. (b) Cross section of the superior sagittal sinus 1 cm rostral of the confluence of sinuses in the same animal. (c) A catheter, inserted 12 mm lateral to the midline perpendicular to the tabula externa of the skull, will enter the ipsilateral rostral horn of the lateral ventricle. (d) The tip of the ventricle catheter is inserted in the rostral horn of the right lateral ventricle (arrow).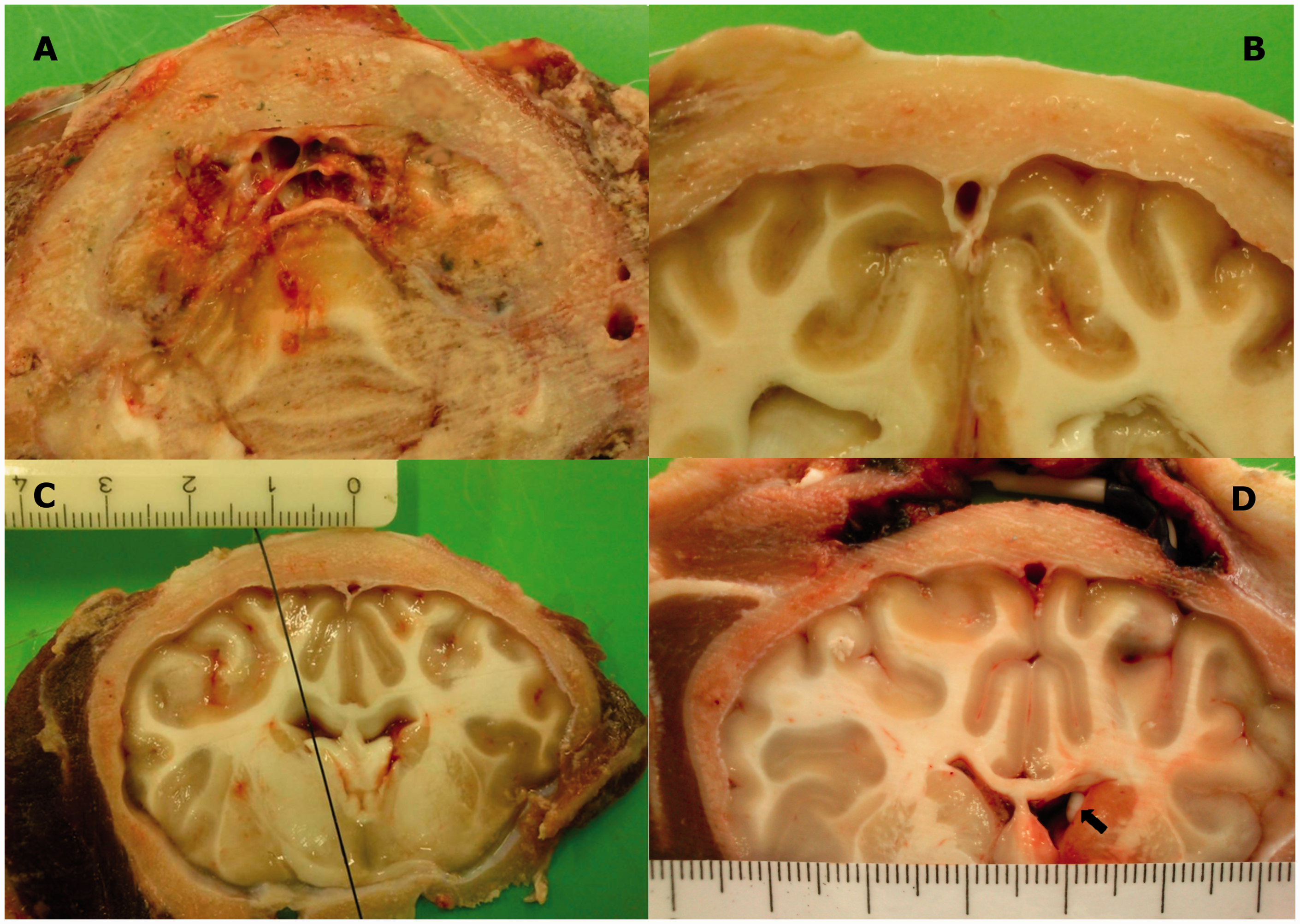
Ten millimetres rostral to the confluence, the cross section of the superior sagittal sinus has a triangular shape (Figure 2(b)). The mean latero-lateral diameter (base) was 2.8 mm, and the dorso-ventral diameter (height) was 3.3 mm on average. A septum was observed in only a few goats.
The mean height of the lateral ventricles at the level of the coronal suture was 3.5 mm. The mean bifrontal diameter at the same slice was 16.6 mm.
A burr hole at the coronal suture should be placed 12.0 mm lateral to the midline to ensure that a catheter inserted perpendicularly to the tabula externa of the skull will enter the rostral horn of the ipsilateral ventricle. Following this trajectory, the distance between the internal tabula of the skull and the dorsal ependyma of the rostral horn was 18.9 mm, and the intraventricular trajectory was 5.8 mm (Figure 2(c) and (d)).
In vitro coagulation assay
Coagulation properties of goat blood. The influence of in vitro addition of CSF to pure blood and oral administration of antiplatelet drugs on the ACT, CR and PF are shown (mean value and 95% confidence interval).
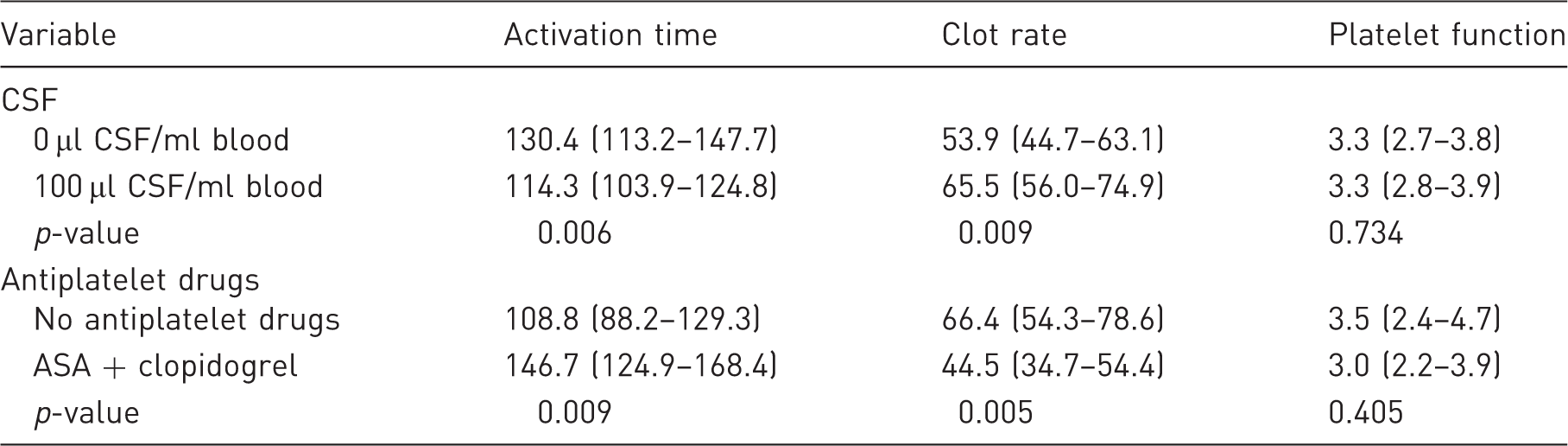
After the addition of 100 µl of CSF per millilitre of blood, the ACT shortened from 130 s to 114 s, and the CR increased from 54 units/min to 66 units/min. There was no significant impact on the PF.
After oral administration of antiplatelet drugs, the ACT increased from 109 s to 147 s, and the CR decreased from 66 units/min to 45 units/min. There was no significant impact on the PF.
In vivo study
Shunt implantation
No specific obstacles were encountered during implantation of the shunt. When the transparent part of the Codman Hakim low pressure valve was inspected, spontaneous drainage of CSF to the superior sagittal sinus was observed directly after implantation of the shunt. Through the wall of the distal segment of the silicone catheter, a pulsatile reflux of blood to the shunt system was observed. This reflux disappeared after obliterating the catheter 1 cm proximally to its entrance in the superior sagittal sinus. No reflux was observed after introduction of a more rigid catheter with a smaller inner diameter.
Clinical score
The average clinical score of the three animals during the last 5 days before surgery was 1.5. The clinical score increased to 3.2 immediately after surgery and to 3.5 on the first postoperative day. The average clinical score normalized starting on the second postoperative day (average score between day 2 and 5 after surgery: 1.33).
CT imaging
CT imaging after surgery showed a correct position of the ventricle catheter in two animals. In the third animal, the ventricle catheter was positioned too deep. This suboptimal position had no clinical consequences. The intravascular catheter was correctly positioned in all animals. The distal part was always located in the ventral angle of the triangular section of the sinus (Figure 3). Clinically and radiographically (contrast-enhanced CT), there were no indications of thrombosis of the superior sagittal sinus.
Position of the intravascular catheter in the superior sagittal sinus. Coronal (top left) and sagittal (top right) CT imaging of the caprine head showing the position of the intravascular catheter and the ventricular catheter. The intravascular catheter was always positioned in the ventral tip (just above the falx) of the superior sagittal sinus. In this positon the catheter touches the lateral walls of the sinus. The ventricular catheter was positioned too deep in this animal. A: intravascular catheter; B: ventricular catheter.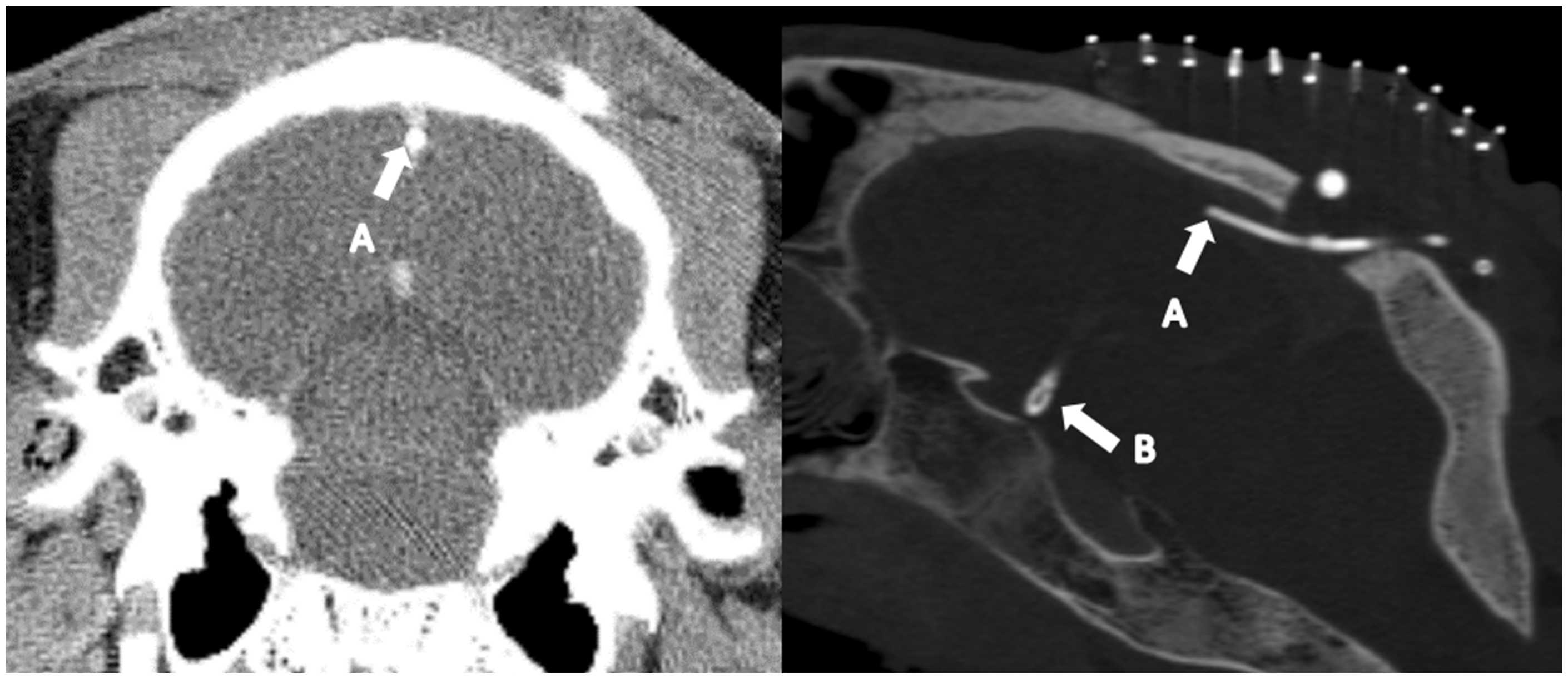
Assessment of shunt patency
The assessment of the shunt system after 16 days showed a patent ventricle catheter, but there was an obstructed vascular catheter in all animals.
Autopsy
Stereomicroscopic evaluation showed a correct position of the intravascular catheter in the three animals. However, the presence of a clot was visualized around all three catheters. This clot was situated at the site were the catheter enters the sinus and extended downstream (i.e. caudally) along the sinus roof (Figure 4(a)). The SEM evaluation of the lumen of the intravascular catheter showed the presence of an intraluminal clot in all catheters (Figure 4(b)).
Microscopic evaluation of the intravascular catheter. (a) Evaluation with a stereomicroscope showed the presence of a clot (arrow) originating at the entry site of the catheter and extending downstream (i.e. caudally) along the sinus roof. (b) Scanning electron microscopic (SEM) views of the obstructed intravascular catheter. Left: SEM evaluation of a transverse section through the intravascular catheter showing the obstructing clot. Right: Higher magnification of the intraluminal clot showing fibrin network with red blood cells and platelets. A: fibrin network; B: red blood cell; C: platelets.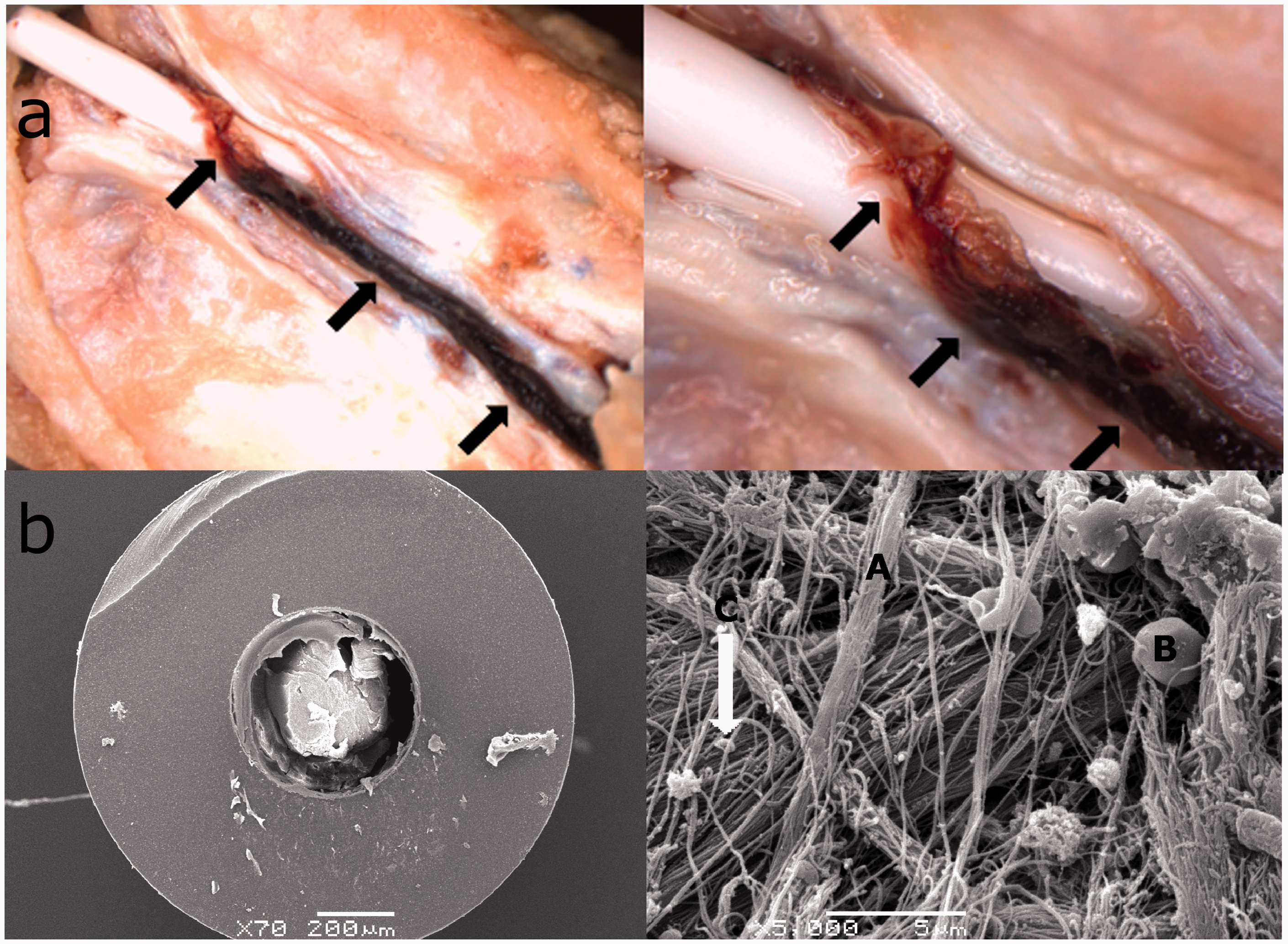
Discussion
Shunting CSF to its natural resorption site – the superior sagittal sinus – with a ventriculosinus shunt is a promising treatment for hydrocephalus.7,8,11 Different authors have evaluated this shunt in clinical series.7–9 Based on their experience, the authors have modified the technique and developed designated shunt hardware.12–15 Currently, it remains unclear which technique or shunt system is ideal. 11 To maximize the chance of success, the ventriculosinus shunt should be optimized by evaluating and combining the most valuable characteristics of the available techniques. This process requires a suitable animal model. 16
This pilot study reports on the characteristics and feasibility of a non-hydrocephalic goat model. The purpose of this model is to enable the evaluation and optimization of different implantation techniques and prototypes of the ventriculosinus shunt.
Strengths and limitations of the non-hydrocephalic caprine model
The implantation of a ventriculosinus shunt in goats has been found to be feasible and safe. The anatomy and dimensions of the caprine brain ventricles and superior sagittal sinus allow for the implantation of ventriculosinus shunts that are comparable in design and dimensions to shunts for human use. 8 These characteristics are in contrast to the previously described canine model, in which the implantation of a distal catheter with a comparable outer diameter resulted in venous congestion and sinus thrombosis. 16
Although the goat model resembles human anatomy, the dimensions of the caprine superior sagittal sinus are significantly smaller. 24 Depending on the prototype that will be tested, miniaturization of the shunt material might yet be necessary. The smaller superior sagittal sinus may also promote distal shunt obstruction, as the proximity of the sinus walls to the catheter tip may cause endothelial irritation and blood stasis, which are both known to enhance coagulation. 25
In contrast to previously described animal models,16,17 hydrocephalus was not induced in this study. Injection of kaolin by a sub-occipital puncture, which is the best documented method for the induction of hydrocephalus, causes chemical meningitis. 26 This technique is associated with a high morbidity and mortality.17,26–28 However, hydrocephaly is not required for the evaluation of the implantation technique and the design, dimensions and tolerance of current and potential new prototypes of the ventriculosinus shunt. The clinical impact of meningitis itself may even cloud the assessment of the tolerance for the shunt system. Thus, the use of a non-hydrocephalic model is an important refinement because it simplifies the study protocol and drastically reduces animal suffering.
A major limitation of the non-hydrocephalic model is the impossibility to evaluate the efficacy and survival of the shunt system. The preserved physiological drainage of CSF causes the flow through the shunt system to be at least limited and inconsistent. This inconsistency leads to impaired clearing of fibrin deposits, which are known to result in catheter obstruction. 29 This phenomena partially explains why all implanted shunts were found to be obstructed after two weeks.
In addition to the absence of hydrocephaly, the coagulation properties of the caprine model may promote shunt obstruction. Sonoclot analysis showed that goats have a hypercoagulable state that is comparable to humans. Compared to normal human values, 30 the ACT is 34% shorter, and the CR is 127% higher. This difference in coagulability was partly counteracted by administration of antiplatelet drugs, which reduced the relative difference in ACT to 11% and CR to 52%. The addition of CSF to blood enhances coagulation in humans. 30 This effect appears to be more pronounced in goats, which may further increase the risk of shunt obstruction.
Intra-operative evaluation of the ventriculosinus shunt
The current study shows that an intra-operative evaluation of the shunt system may be useful for clarifying the causes of shunt failure, even in the absence of hydrocephalus. One could observe a pulsatile reflux of blood to the shunt system despite the use of a competent one-way valve and a retrograde position of the shunt. This reflux is probably caused by a combination of the compliance of the silicone shunt system distal to the valve, the pressure pulsations in the superior sagittal sinus and the inertia of blood corpuscles that hit the CSF column at the distal shunt tip.
Pulsatile reflux of blood results in the development of a relatively static blood–CSF mixture in the distal shunt system. Both stasis of blood and the interaction with CSF promote clot formation, which finally results in shunt obstruction. 30
The reflux could be prevented by clamping the distal catheter close to its entrance in the superior sagittal sinus. In addition, reflux was not observed when a more rigid catheter with a smaller inner diameter was inserted in the sinus. Both manoeuvres reduce the compliance of the shunt system distal to the valve. These findings suggest that using a more rigid distal catheter with a smaller internal diameter may help prevent distal shunt obstruction.
In conclusion, the implantation of ventriculosinus shunts is feasible and safe in a non-hydrocephalic goat model. This model facilitates the intra-operative evaluation of the implantation technique, hardware and functions of the ventriculosinus shunt. The tolerance for the shunt system and possible postoperative complications can be monitored without interference of sequelae from the induction of hydrocephalus. However, in the absence of hydrocephalus, it is impossible to evaluate shunt efficacy and shunt survival. The development of a hydrocephalic goat model for this purpose may be used in future research.
Supplemental Material
Supplemental material for A non-hydrocephalic goat experimental model to evaluate a ventriculosinus shunt
Supplemental material for A non-hydrocephalic goat experimental model to evaluate a ventriculosinus shunt by Jelle Vandersteene, Edward Baert, Stijn Schauvliege, Kimberley Vandevelde, Frank Dewaele, Filip De Somer and Dirk Van Roost in Laboratory Animals
Footnotes
Acknowledgements
We express our gratitude to professor Paul Simoens for his enthusiastic support during this study and preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Industrial Research Fund for Ghent University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
