Abstract
The cannulation of the cisterna magna in rats for in vivo sampling of cerebrospinal fluid serves as a valuable model for studying the delivery of new drugs into the central nervous system or disease models. It offers the advantages of repeated sampling without anesthesia-induced bias and using animals as their own controls. An established model was retrospectively reviewed for the outcomes and it was hypothesized that by refining the method, i.e. by (1) implementing pathophysiological-based anesthesia and analgesia, (2) using state-of-the-art peri-operative monitoring and supportive care, (3) increasing stability of the cement–cannula assembly, and (4) selecting a more adaptable animal strain, the outcome in using the model – quantified by peri-operative mortality, survival time and stability of the implant – could be improved and could enhance animal welfare. After refinement of the technique, peri-operative mortality decreased significantly (7 animals out of 73 compared with 4 out of 322; P = 0.001), survival time increased significantly (36 ± 14 days compared with 28 ± 18 days; P < 0.001), as well as the stability of the cement–cannula assembly (47 ± 8 days of adhesion compared with 33 ± 15 days and 34 ± 13 days using two other cement types; P < 0.001). Overall, the 3R concept of Russell and Burch was successfully addressed and animal welfare was improved by (1) the reduction in the total number of animals needed as a result of lower mortality or fewer euthanizations due to technical failure, and frequent use of individual rats over a time frame; and (2) improving the scientific quality of the model.
Cerebrospinal fluid (CSF), produced mainly by the choroid plexus and ependymal layer of the brain ventricular system and drained through the intracranial and spinal arachnoid villi into venous blood, plays an important role in the homeostasis of the central nervous system (CNS). In preclinical research, drug concentrations in CSF are used as a surrogate measure of their availability in the CNS,1,2 and CSF penetration studies in animals remain a practical option for assessment of CNS drug delivery in early preclinical drug development, 2 be it by direct injection into the CNS or distribution to the CNS after parenteral administration. Further, analysis of CSF allows the study of different disease models, such as the accumulation of extracellular β-amyloid plaques which is characteristic of Alzheimer’s disease, 3 or damages to the blood–brain barrier (BBB) and the blood–CSF barrier in multiple sclerosis patients. 4
In an experimental setting, CSF sampling may be performed at either the ventricles, cisterna magna (CM) or lumbar thecal sites. Different in vivo CSF models, whether acute or chronic, exist either in anesthetized or in conscious animals. Acute CSF sampling in rats via a lumbar puncture through a skin incision,5,6 and through a puncture of the CM with or without skin and muscle preparation at the dorsal surface of the neck have been described. For puncture of the CM, various models have been used in both anesthetized7–11 and in awake 12 rats, with or without stereotaxic fixation of the animals. Both techniques present pros and cons as repeated anesthesia as well as repeated puncturing of the CM in awake animals could be distressful, and anesthesia could bias experimental results, depending on the readout. Also, manual puncture of the CM is highly dependent on a researcher’s skills in order to unambiguously identify the puncture site and controlling the depth of needle insertion, which could result in blood vessel/tissue damages and consequently CSF contamination. 10 Chronic animal models most often involve different techniques of surgical implantation of a cannula into the lateral ventricle, 13 or into the CM either through the occipital bone 14 or through the interparietal bone.3,15–18 The cannula allows repeated sampling of CSF in conscious, freely moving animals, thereby increasing animal welfare and allowing analysis of samples without potential bias due to anesthesia. The use of one fixed cannula also reduces the risk of contaminating CSF samples with blood, as would likely occur if a new perforation has to be performed at each sampling.
An established model was retrospectively reviewed for the outcomes. The technique described here is of a chronic CSF rat model based on studies by Bouman and Van Wimersma Greidanus 15 and El Mouedden et al., 3 in which rats underwent stereotactic surgery with implantation of a cannula in the CM. The cannula was fixed and stabilized onto the skull using dental cement. After a week of recovery, CSF could be collected in the conscious rat. It has been used both for validation and optimization of novel BBB transporters and for the development of new drugs for Alzheimer's disease.
We hypothesized that by refining the method used in their study, i.e. by (1) implementing pathophysiological-based anesthesia and analgesia, (2) using state-of-the-art peri-operative monitoring and supportive care, (3) increasing stability/adhesivity of the cement–cannula assembly, and (4) selecting a more adaptable animal strain, the outcome in using the model – quantified by peri-operative mortality, survival time and stability of the implant – could be improved and animal welfare enhanced.
Materials and methods
Animals
The rats were housed in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International accredited facility in Eurostandard type IV polycarbonate open-top cages (Tecniplast, Buguggiate, Italy) equipped with dust-free bedding, nesting material (paper tissue) and wooden gnawing devices. Room temperature was maintained at 22 ± 2℃. Rats were kept in a 12 h light/dark cycle. Food and water were provided ad libitum. Up to the date of surgery, the rats were group-housed; after surgery they were individually-housed to avoid removal of the cannula by cage mates.
The peri-operative records of 379 male adult rats (40 Wistar (Crl:WI, body weight [BW] 17 ± 67 g) and 339 Wistar Han IGS (Crl:WI(Han), BW 259 ± 33 g)) were reviewed retrospectively, and the following outcome indicators were compared between the original (‘pre-refinement’; 40 WI and 22 WI(Han)) and refined surgery (‘post-refinement’; 317 WI(Han)) procedures:
Peri-operative mortality: defined as death under anesthesia and within 24 h after surgery. Animal survival time: defined as days of use of animals. Rats with survival time of between day 1 up to day 60 post-surgery were considered for statistical analysis. Stability of the cannula–cement assembly: defined as days of firm adhesion of the assembly to the animal’s skull. Visible instability (noticed while manipulating the animals during CSF flow checks) or complete detachment of the cannula–cement structure was defined as loss of assembly. Duration of surgery: defined as time in minutes between incision of the skin and the finished suture. Intraoperative positivity: defined as free CSF flow out of the cannula after positioning and before applying dental cement.
The desired prospective methodical changes targeting the refinement of the model (‘protocol B’) were amended in the formerly accepted research protocol (‘protocol A’) and approved by the cantonal veterinary authorities.
The original technique (‘protocol A’) has been described extensively. For the refined technique (‘protocol B’) only aspects that have been optimized are detailed.
Protocol A
Intraoperative anesthesia, analgesia and monitoring
Before surgery, the actual BW of each rat was recorded in the peri-operative record.
The rats were anesthetized using a mixture of 100 mg/kg ketamine and 10 mg/kg xylazine (KX) (Ketasol® and Xylasol®; Graeub AG, Bern, Switzerland) given by intraperitoneal (IP) injection. Animals were given 0.025 mg/kg buprenorphine (Temgesic®, Reckitt Benckiser Switzerland AG, Wallisellen, Switzerland) subcutaneously (SQ) before surgery as pre-emptive analgesia.
The rats were breathing room air spontaneously, and peri-operative monitoring consisted of rectal temperature and anesthetic depth (by pinch reflex).
Preparation
Eye ointment (Bepanthen®; Bayer AG, Zürich, Switzerland) was applied over the cornea to avoid desiccation. The surgical area over the skull was clipped and thoroughly washed and disinfected with iodine soap (Betadine® soap; Mundipharma Medical Company, Basel, Switzerland). For local anesthesia, 0.1 mL lidocaine (Lidocain 2%; Streuli Pharma AG, Uznach, Switzerland) was infiltrated SQ into the area of the surgical cut. The rat was then fixed in a stereotaxic apparatus (David Kopf® Model 900 Small Animal Stereotaxic Instrument; David Kopf Instruments, Tujunga, CA, USA) on top of a temperature-controlled heating mat, with the head positioned in order to achieve a flat skull. The skin was then sprayed with alcoholic iodine solution (Betaseptic®; Mundipharma Medical Company, Basel, Switzerland).
Draping
No sterile draping was used.
Surgery
Surgical implantation of the cannula (manufactured from a 20 gauge hypodermic needle with a fitting stainless-steel wire as stylet) in the CM was performed under aseptic conditions. A 2–3 cm incision was made in the skin of the skull. The skull was carefully prepped free of the muscles and periosteum using a scraping tool. The skin and subcutaneous tissue were held back using four bulldog forceps.
Using the sagittal suture as well as the external occipital crest as landmarks, one 1.6 mm hole for the placement of the cannula was drilled into the interparietal bone of the skull, located on the sagittal midline immediately rostral to the interparietal–occipital bone suture. Two additional holes with a diameter of 0.6 mm each were drilled slightly rostral and lateral of the cannula hole in the darker area of the interparietal bone, ensuring that these were not too close to the interosseous sutures to avoid massive bleeding. Two stainless steel anchoring screws (1 mm diameter × 3 mm length) were placed in these holes (Figure 1).
Rat skull prepared for insertion of the cannula into the cisterna magna (CM). In this picture sterile draping is in place (described in protocol B). Arrows point to anchoring screws (a), hole for placement of cannula (b) and Tygon tubing sealing the cannula (c).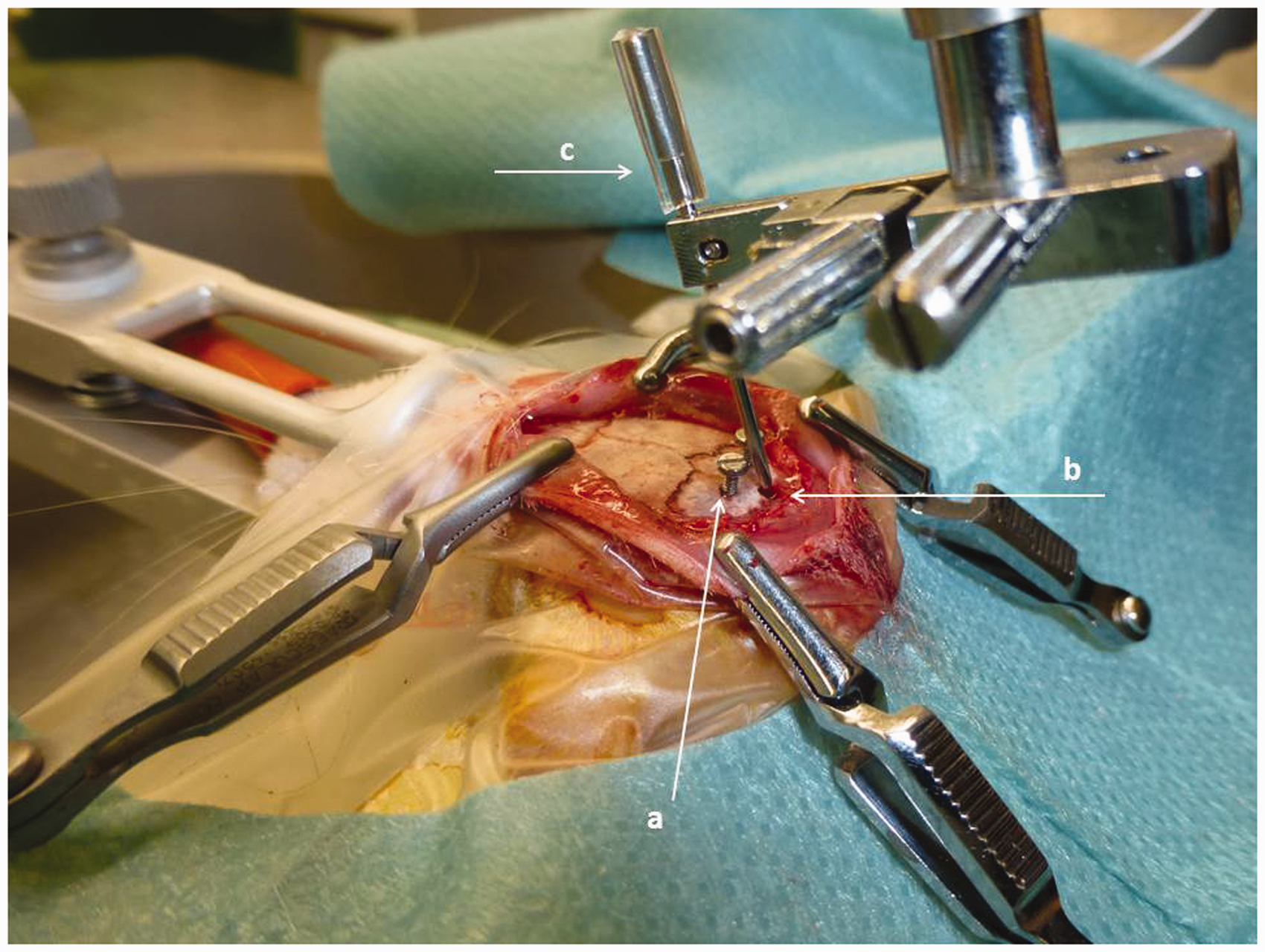
The cannula with the stylet was fixed in the cannula holder of the stereotaxic apparatus and set at an angle of 70° to the surface of the skull. Under visual guidance the tip of the cannula was placed into the middle of the drilled hole and the cannula was lowered 8.5 mm from the starting point using the worm-gear-driven VD drive of the stereotaxic apparatus. The correct placement of the cannula was checked by carefully removing the stylet from the cannula. When the cannula was correctly positioned a spontaneous flow of CSF occurred.
Thereafter the cannula was fixed to the skull by applying dental cement around the cannula itself and over the anchoring screws. A head cap was molded with two-component cement as used in human dentistry (Paladur®; Heraeus Kulzer Ltd, Hanau, Germany). A series of surgeries were also performed using glass ionomer cement (GC FujiCEM®; GC Austria Ltd, Flums, Switzerland).
The skin wound was sutured with a 5-0 synthetic absorbable suture (Monocryl®; Ethicon, Neuchatel, Switzerland).
Recovery and post-operative analgesia and care
The rats were placed in recovery cages under a heating lamp until return to full consciousness. Anesthesia was not reversed. Post-operative analgesia was covered by 1 mg/kg meloxicam (Metacam®; Boehringer Ingelheim Switzerland Ltd, Basel, Switzerland) SQ after surgery and continued per os once a day for three days. Rats were allowed to recover for at least one week before their use in experiments.
A daily general assessment of well-being (overall clinical appearance, wound healing and behavior) was carried out for up to seven days. BW and positivity of the implanted cannula were checked one week post-operation, and thereafter weekly if the animals were not used in an experiment.
Protocol B
Intraoperative anesthesia, analgesia and monitoring
Rats were anesthetized with a mixture of 0.15 mg/kg medetomidine (Dorbene®; Graeub AG), 2 mg/kg midazolam (Dormicum®; Roche Pharma AG, Reinach, Switzerland) and 5 µg/kg fentanyl (Fentanyl–Curamed®; Actavis Switzerland AG, Regensdorf, Switzerland) (MMF), diluted to achieve a volume of 0.065 mL/100 g BW administered SQ. Generally, the surgical anesthetic depth is reached 20 min after injection.
Once the rats were positioned in the stereotaxic apparatus, oxygen was supplied via a nose mask. Isoflurane (Forene®; AbbVie AG, Baar, Switzerland) 0.5 to 1% in oxygen was provided as needed if surgical depth was judged to be inadequate or if it started weaning off in the case of a lengthy surgery. Intraoperatively, heart rate and oxygen saturation of blood (SpO2) were continuously monitored via a pulse oximeter (Nonin Palm® Model SAT 2500A Vet; Nonin Medical BV Europe, Amsterdam, The Netherlands) in addition to body temperature and anesthetic depth (assessed compositely by response to toe pinch, heart rate and whisker movements).
Draping
The surgical field was draped with a sterile adhesive film (Tegaderm® film; 3M Switzerland Ltd, Rüschlikon, Switzerland) and a sterile self-adhesive drape (Foliodrape®; IVF Hartmann AG, Neuhausen, Switzerland).
Surgery
Once the skull was prepared free of the muscles and periosteum, 40% of phosphoric acid etching gel (GC Austria Ltd) was applied to the skull with a felt applicator and left to act for 10 s before being removed with a swab. The skull was then thoroughly rinsed with sterile water and dabbed dry. Bonding agent (G-ænial Bond®; GC Austria Ltd) was applied with a felt applicator and left to act for 10 s before drying the surface with compressed air and light-curing the bonding agent for 10 s with a light-emitting diode (LED) curing light (Silverlight®; Mectron Medical Technology, Carasco, Italy). After drilling the holes and placing the cannula, a dual-cure self-adhesive universal resin cement (G-CEM®, GC Austria Ltd) was used making sure to light-cure for 10 s from different angles between layers of cement. Once the cement hardened, the cannula holder was carefully removed and a final cement layer applied and light-cured.
Recovery and post-operative analgesia and care
The rats were placed in clean recovery cages under a warming blanket (adult size) with a forced air warming device (Bair Hugger® Model 750; 3 M Switzerland Ltd) to recover until fully awake. Anesthesia was reversed with a mix of buprenorphine 0.05 mg/kg ((Temgesic®; Reckitt Benckiser Switzerland AG), 0.75 mg/kg atipamezole (Alzane®; Graeub AG) and 0.2 mg/kg flumazenil (Anexate®; Roche Pharma AG) administered SQ. Analgesia was guaranteed by buprenorphine in the antagonization mix and 1 mg/kg meloxicam (Metacam®; Boehringer Ingelheim Switzerland Ltd) administered SQ immediately before antagonization and continued per os once a day for three days.
Moist food was provided in the cages for three days.
Statistical analysis
Parameters were analyzed for normality (Kolmogorov–Smirnov test), and parametric tests were chosen because of the distribution and small sample size. Results are expressed as mean and standard deviation (SD).
Fisher’s exact test was used to compare peri-operative mortality before and after refinement of the technique. Animal survival before and after refinement was analyzed using the unpaired t-test with Welch’s correction to account for the unequal sample size and variance. Differences in stability and surgery duration between cements were analyzed with the one-way analysis of variance (ANOVA) test. Post hoc, Bonferroni test for multiple comparisons was applied where appropriate. Fisher’s exact test was used to compare peri-operative intraoperative positivity before and after refinement. Overall significance was set at a value of P < 0.05. Data were analyzed using commercially available computer programs (GraphPad Prism software; GraphPad Software Inc, La Jolla, CA, USA).
Results
In this retrospective study, data from both Wistar (Crl:WI) and Wistar Han IGS (Crl:WI(Han)) rats were included. The reason for the change of strain was because of the rather steep increase in BW of the WI rats, rendering them quite large to be handled safely. To overcome this problem, WI(Han) rats were selected, which gained weight at a much slower rate than WI rats. As there were no significant differences observed between the strains for peri-operative mortality, animal survival time, stability of the cannula–cement assembly, duration of surgery, or intraoperative positivity, data from both strains were combined for these parameters in order to increase statistical value.
Peri-operative mortality (within 24 h post-operation) decreased significantly from seven animals (six WI and one WI(Han)) out of 62 animals (40 WI and 22 WI(Han)) before refinement to three rats out of 317 after refinement (all WI(Han)) (P = 0.0002) (Figure 2).
Peri-operative mortality (defined as death under anesthesia or in the first 24 h post-surgery) before and after refinement was compared with Fisher’s exact test. ***Difference is statistically significant according to Fisher’s exact test (P = 0.0002).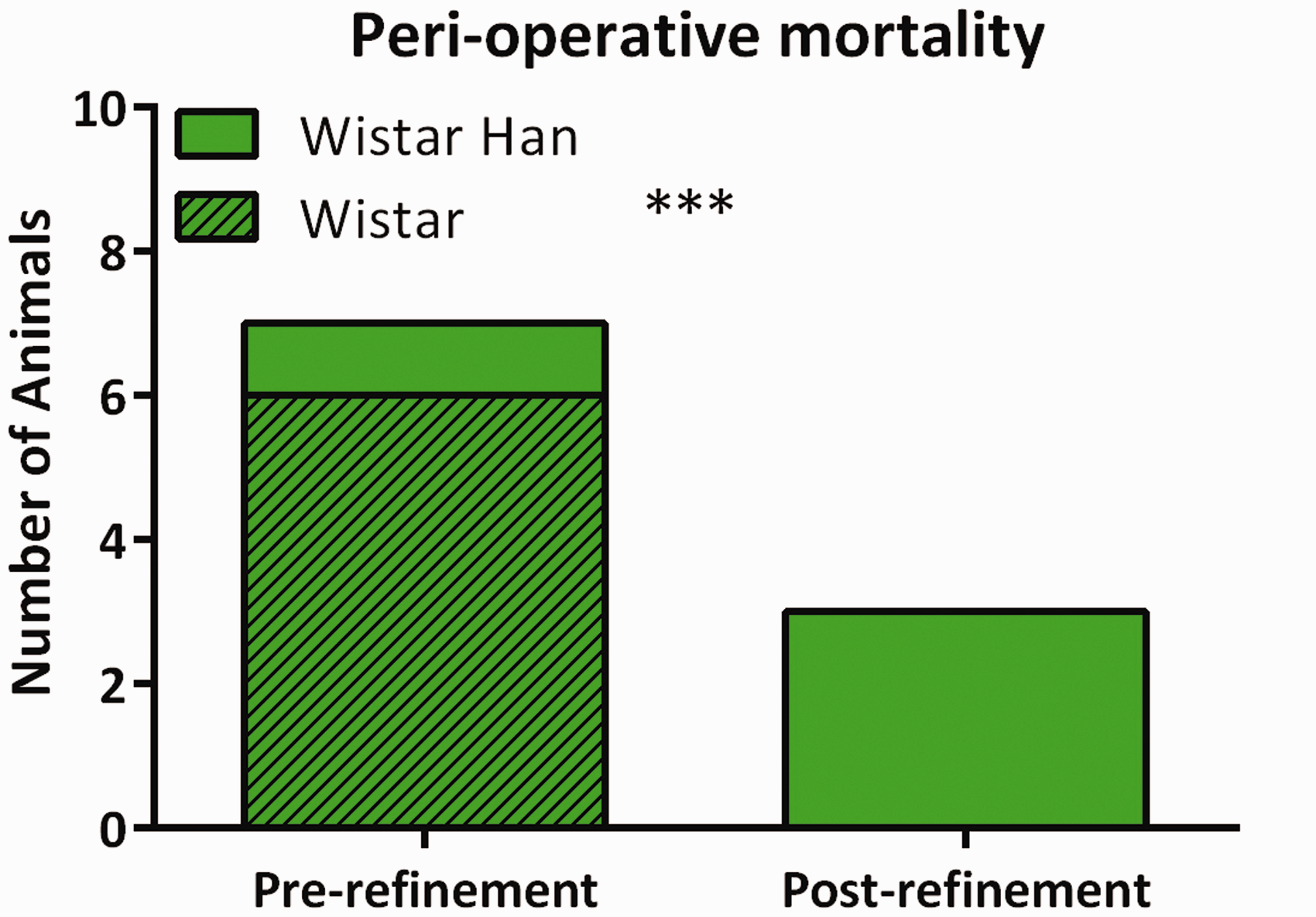
A significant (P = 0.03) increase in the survival time (defined as days of use of animals; rats with survival time of between day 1 and up to day 60 post-surgery were considered) from 31 ± 16 to 36 ± 14 days could be achieved with the refined technique (Figure 3). In the pre-refined setting, there were no significant differences in survival time between the different strains according to the unpaired t-test (30 ± 15 days for WI versus 34 ± 18 days for WI(Han), P = 0.30).
Survival time before and after refinement was analyzed using the unpaired t-test with Welch’s correction (*P < 0.05).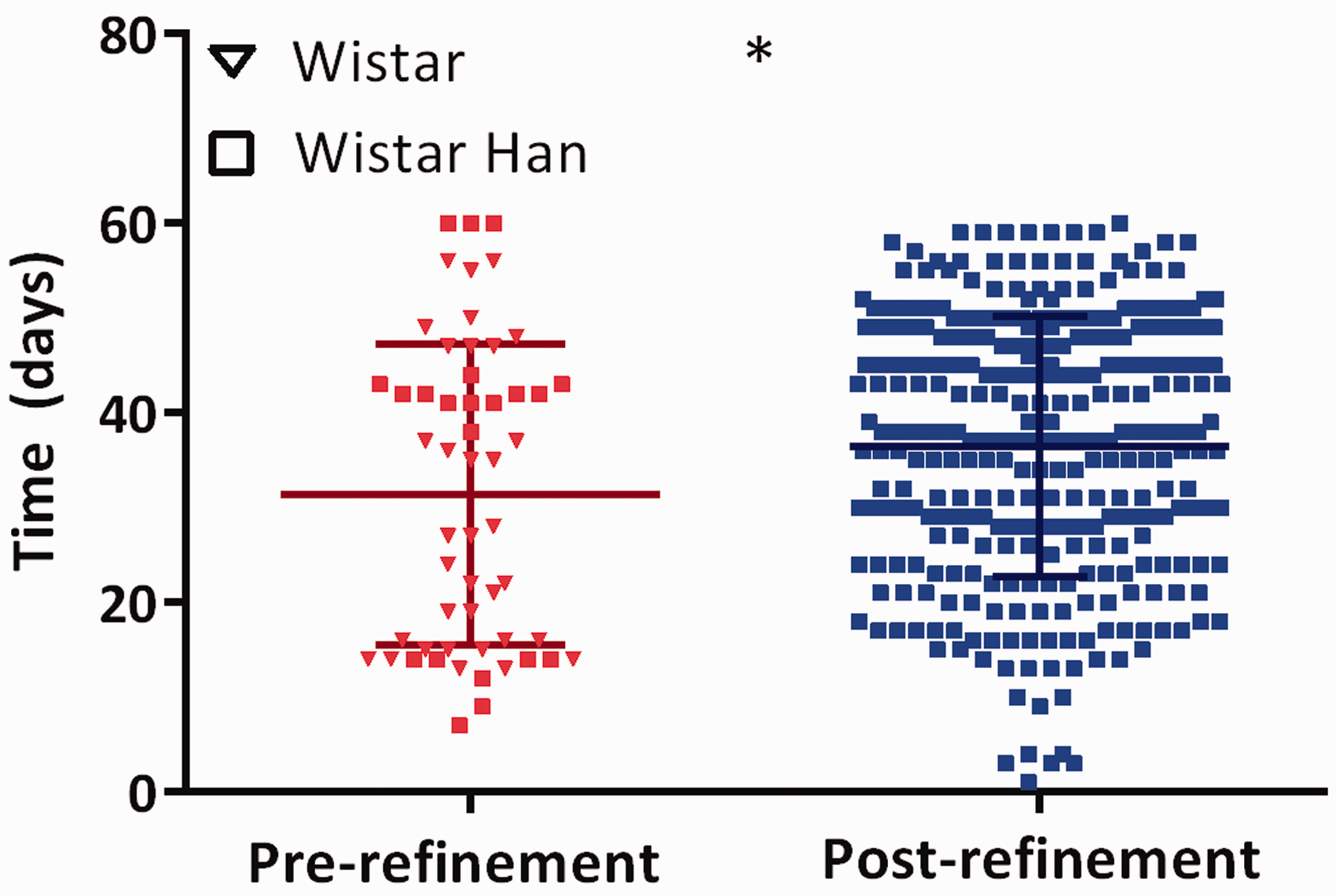
A significantly higher stability (P < 0.0001) of the cement–cannula assembly was achieved (47 ± 8 days of adhesion) using the new cementing technique described in protocol B which combined etching of the skull, bonding agent and light-curing cement (G-CEM®) compared with Paladur® used in protocol A (33 ± 15 days of adhesion; 30 ± 15 days for WI, 34 ± 15 days for WI(Han)) and GC FujiCEM® used in a series of surgeries (34 ± 13 days of adhesion) (Figure 4). No significant differences were found for cement stability between the different strains.
Stability of the cement/cannula assembly. Difference in stability (days) of the cement/cannula assembly between cements was analyzed with the one-way analysis of variance (ANOVA) test with post hoc Bonferroni correction (****P < 0.0001).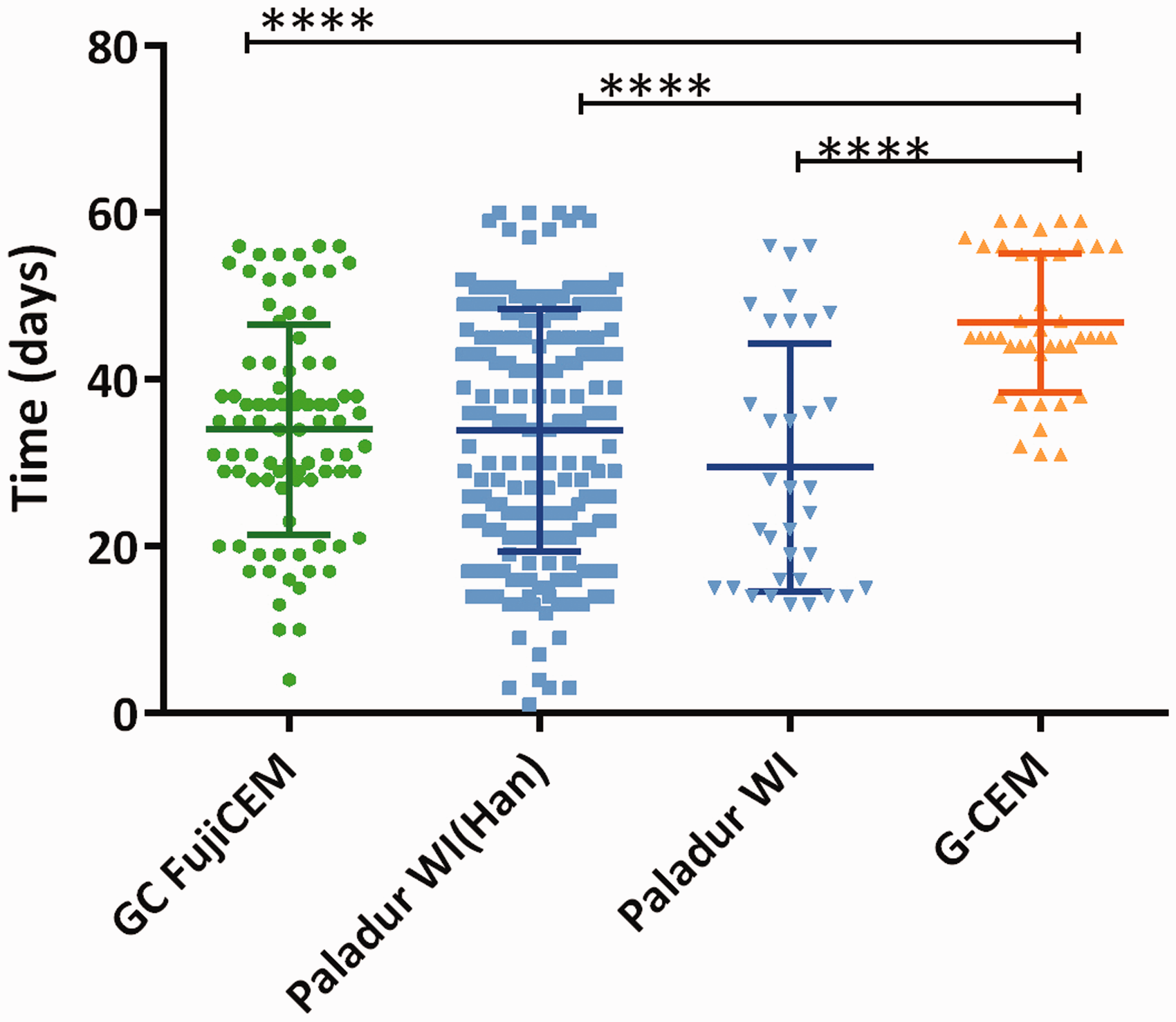
Surgery duration was significantly longer due to some additional intermediate steps (e.g. several times light curing) using the new technique with G-CEM® (45 ± 11 min; P < 0.0001) compared with the GC FujiCEM® (35 ± 8 min) and Paladur® (42 ± 9 min) techniques (Figure 5). This was also true when corrected for the surgeon variable (not shown). No significant differences were found across the strains in the Paladur® group.
Surgery duration. Difference in duration of surgery between cements was analyzed with the one-way analysis of variance (ANOVA) test (*P < 0.05, ****P < 0.0001).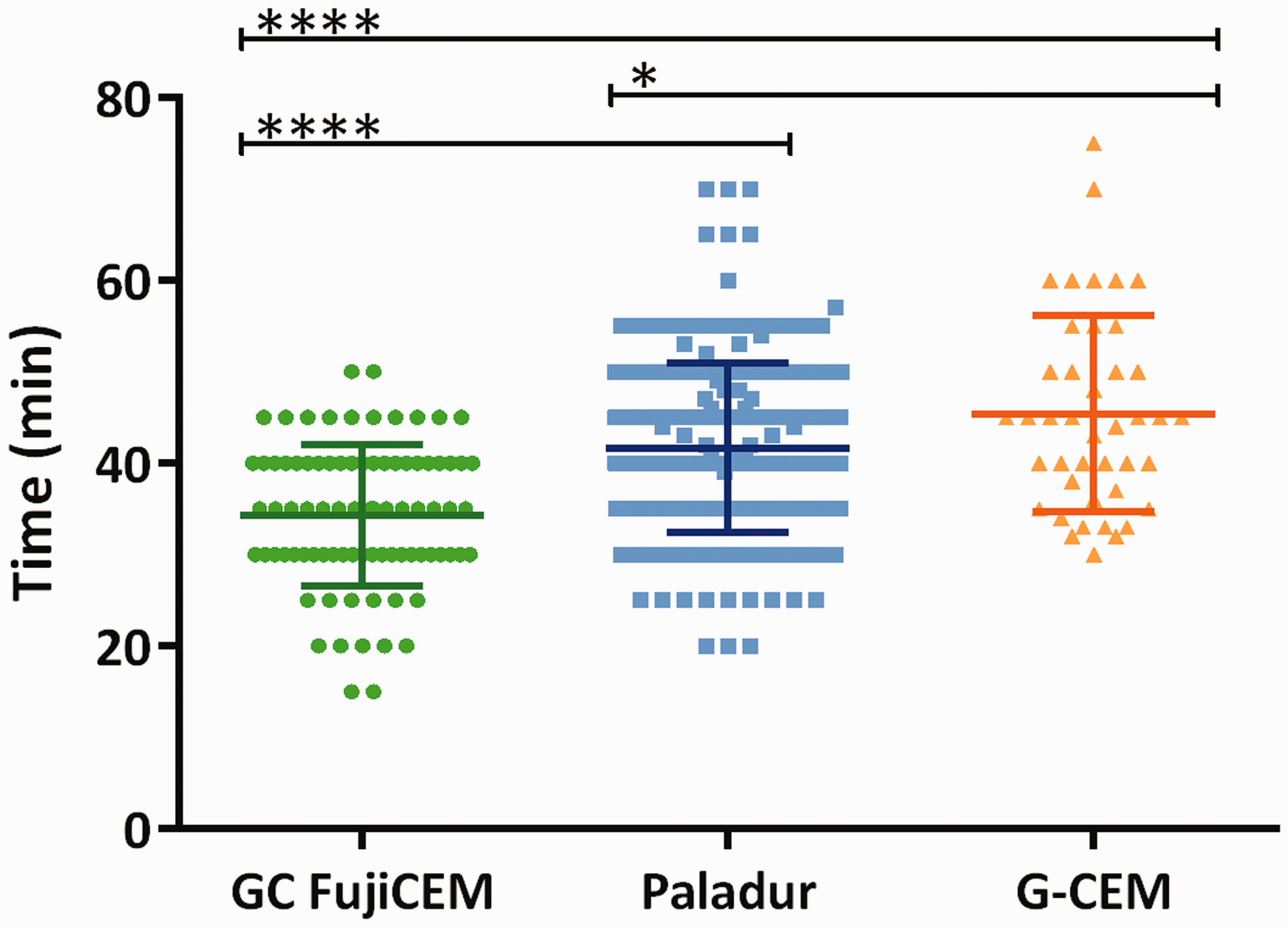
There were no significant differences in intraoperative positivity rates (defined as free CSF flowing out of the cannula after positioning and before applying the dental cement) between the two protocols. Overall, intraoperative positivity rate reached 78.63%.
Discussion
The holistic, pathophysiological-driven and model-tailored refinement of the method used improved the outcomes in the rat model, as well as enhanced animal welfare by significantly reducing mortality, increasing survival time, and improving stability of the cement/cannula assembly.
To refine anesthesia, the KX protocol was replaced by a balanced anesthesia, consisting of a mixture of MMF; oxygen was provided throughout and isoflurane if needed. The latter protocol has many advantages:
SQ administration reduces the risk of damages caused by intra-organ injections. It is also better tolerated by animals and results in a more constant onset of action. All anesthetic drugs are reversible allowing for a targeted anesthesia time. At the end of surgery animals are fully antagonized (except for the analgesia) and recover within minutes. Animals typically experience a smooth and short recovery phase compared with KX where the recovery time is very long as it relies on redistribution and metabolism. This is most important in small mammals where a fast return to normophysiology reduces morbidity and mortality.
19
The cannulation of the CM potentially increases intracranial pressure (ICP) with consequent risks of brain herniation and death. The choice of the anesthetics for neurosurgery, especially if there is a risk of increased ICP, should therefore target the maintenance of cerebral perfusion and oxygenation.
First, normotension should be achieved in order to maintain an adequate cerebral perfusion pressure. Unfortunately blood pressure cannot be maintained routinely in rodent anesthesia due to lack of monitoring and venous access. The choice of an anesthetic which impairs the cardiovascular system minimally is therefore mandatory, and MMF is superior in maintaining blood pressure compared with KX.
20
Second, drugs which have the potential to increase cerebral metabolic rate of oxygen (CMRO2) and ICP should be avoided. A typical example is ketamine – a dissociative N-methyl-D-aspartate (NMDA) agonist anesthetic, although this is becoming controversial – at least when used at low doses. Still the use of high-dose ketamine increases cerebral blood flow,21,22 resulting in increased ICP.22–25 For the model in this study, this would be an undesired side-effect, therefore MMF is advantageous compared with KX because of the minimal interference with the cerebral autoregulation mechanisms. Benzodiazepines and opioids reduce CMRO2 and cerebral blood flow (CMRO2–CBF coupling) topped by the vasoconstricting action of medetomidine, resulting in a beneficial reduction in cerebral vascular volume and ICP. 26
The state-of-the-art administration of oxygen supplied via a nose mask counteracts peri-anesthetic hypoxia which occurs in anesthetized rats breathing room air only, contributing to the maintenance of an adequate brain oxygenation and thereby reducing peri-operative mortality. 19 Additionally this allows the easy administration of isoflurane at 0.3 minimum alveolar concentration (MAC) 27 if the anesthetic depth is not adequate or if surgical depth is weaning off in the case of a prolonged surgery, without interfering with cerebral autoregulation mechanisms at this very low dose. 28 This makes the anesthetic very adaptable to the needs of the individual animals, length of the procedure, and the operator’s skills.
According to the recommendations for the standards of intraoperative monitoring, 29 in protocol B heart rate and SpO2 were constantly monitored via a pulse oximeter in addition to the body temperature. This allowed us (1) to indirectly assess the anesthetic depth/nociception according to the sympathetic stimulation (heart rate change), and to titrate anesthesia to effect; (2) to measure SpO2 which when > 90% is a guarantee of adequate oxygenation of the arterial blood (PaO2) and tissues (SpO2 ≅ PaO2), with special emphasis on the brain. Constant monitoring also allowed us to undertake corrective measures in case of desaturation.
A multimodal, mechanism-based balanced analgesia protocol targeting all nociception processes (transduction, transmission, modulation and perception) was implemented for peri-operative analgesia. Intraoperative transmission of nociceptive stimuli at the incision site was blocked by infiltrating the area with lidocaine SQ. Medetomidine and midazolam exerted their analgesic actions by modulating the ascending nociceptive pathways, and fentanyl and medetomidine also reduced pain perception at the central level. Post-operatively, animals received buprenorphine which antagonized the sedative effects of the fentanyl while providing additional analgesia. The COX-2-selective non-steroidal anti-inflammatory drug (NSAID) meloxicam administered immediately before antagonization and per os for three days after surgery contributed to pain relief as a result of the reduction of transduction at the peripheral level.
Taken all together the aforementioned pathophysiological-driven refinements proved to be valid as a way of reducing peri-operative mortality in this model. The more obvious limitation in this retrospective study is the absence of objective measures of cardiovascular and cerebral functions and anesthetic depth, allowing comparisons of these readouts in protocols A and B. These would also be valuable for assessing whether buprenorphine given before KX added to the additional respiratory or cardiovascular depression and secondary decrease in oxygen delivery, contributing to the higher mortality rate.
In protocol A, surgical gloves were worn, but no sterile draping was used. After refining the procedure, and following recommendations from the Guide for the care and use of laboratory animals, 30 aseptic techniques were applied to reduce microbial contamination to the lowest possible practical level. Before starting surgery, the surgical field was aseptically prepared and draped. By observing these recommendations a higher success rate with good outcomes could be achieved, thereby reducing morbidity and mortality while at the same time increasing reproducibility of the model and refining the method.
The Paladur® cement used in protocol A has the drawback that it is based on methacrylate copolymers and emanates acrid fumes which are toxic to the personnel. Also, Paladur® is quite liquid and is therefore not easy to handle when freshly mixed, making the application on the skull technically demanding.
As an alternative, GC FujiCEM®, a glass ionomer cement, was used in a series of surgeries. This cement is odorless and is very clean and easy to handle, but is quite aggressive to the skin, leading to skin necrosis around the head cap. Its major drawback, however, is its poor adhesivity to the skull of the rats.
Finally, a new technique was adopted by first preparing the skull surface with a 40% phosphoric acid etching gel before applying a light-curing bonding agent. The cannula was secured using an odorless dual-cure self-adhesive universal resin cement which was then light-cured. Care was taken to ensure that these components (etching gel, bonding agent and cement) were compatible with each other. Even though the additional intermediate steps resulted in a longer surgery duration, the new technique is superior in terms of occupational health and safety by avoiding exposure of personnel to acrid fumes, ease of handling, adhesivity, and a significantly higher stability of the cement–cannula assembly.
An additional benefit of increased stability of the cannula might be as a result of a reduction in inflammatory reaction in the brain which could bias research data. This parameter would have to be evaluated separately, as this is not feasible in a retrospective study.
In protocol A, no special attention was given to post-operative feeding. After careful observation of the animals, it was noticed that immediately after surgery, the rats seemed to have problems gnawing and biting standard rat chow and it was presumed that they were experiencing discomfort at the surgical site when applying pressure with their teeth. After reviewing the technique, rats were given a soft chow after surgery, in addition to the standard pellets, for up to three days. Rats were observed to readily accept the soft chow, although BW checks one week after surgery did not show any significant differences. It is possible that BW change after one week is not a sensitive enough indicator and that BW at an earlier time point (i.e. 2 or 3 days post-operation) should be taken instead, or the weight changes during the first week should be analyzed to provide a clearer picture (data not shown). Nevertheless, this change has also been considered as a refinement by the animal caretakers observing the animals daily.
In conclusion, after retrospective review, the in-house performance of the model was considered to be low, and the processes were analyzed and consequently refined to obtain better outcomes. The peri-operative mortality rate could be successfully reduced and survival time increased by refining and implementing state-of-the-art sterility, anesthesia, analgesia, peri-operative monitoring, and care. The stability of the cement–cannula assembly was successfully optimized by refining the technical procedures.
Overall it can be concluded that the 3R concept of Russell and Burch31,32 was successfully addressed and animal welfare was improved by (1) the reduction of the total number of animals needed as a result of lower mortality or fewer euthanizations due to technical failure, and frequent use of individual rats over a time frame; and (2) improving the scientific quality of the model.
Footnotes
Acknowledgments
We would like to thank Ms Véronique Dall`Asen and Dr Eduard Urich for their valuable help in performing part of the surgeries, and Ms Larissa Kohler for her assistance in processing data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
