Abstract
The temporoparietal flap containing the temporal fascia has a wide spectrum of applications in head and neck surgery. The aim of this study is to demonstrate a temporoparietal fascial flap model that can be used in various head and neck defects in rats. Ten male Sprague-Dawley rats weighing 250–300 g were used in this study. Temporoparietal fascial flaps were elevated with the axial pedicle from the rats using microsurgical techniques. The arc rotations of the flaps on the pedicles were examined and the dimensions of the defects these flaps could cover were identified. The flaps were observed to be the right size for use with defects of the occipital, temporal and mastoid regions, ear, facial nerve and the mandible. The temporoparietal fascial flap is an experimental flap model, which can be useful in experimental studies related to complex head and neck defects.
The temporoparietal region is a structural unit of the utmost importance for clinical application in the fields of aesthetic and reconstructive surgery. This unit consists of seven layers: the skin, subcutaneous tissue, superficial temporal (temporoparietal) fascia, loose areolar tissue plane (innominate fascia), temporal fascia and temporal fad pads, temporalis muscle, and pericranium.1,2
A temporoparietal fascial (TPF) flap can both be used as a random local fascial flap as well as a superficial temporal artery-based axial flap. The single pedicle can be elevated together with the hairy skin, temporoparietal fascia, temporal fascia, temporalis muscle and parietal bone on it.2–5 Developments in microvascular surgery in parallel with progress in vascular surgery have extended the application possibilities of the TPF flap and enabled its potential use as free tissue.6–9
Although it is widely used in the clinical sphere, with reference in the literature, we have not found other experimental studies of the TPF flap. In this experimental study, a model of the TPF flap with axial pedicles in rats that can be used for studying various head and neck defects is discussed.
Materials and methods
All procedures were performed in accordance with the Helsinki Declaration, and a local ethics committee approval was obtained from Dicle University, Sabahattin Payzin Experimental Research Centre (DUSAM). Ten male Sprague-Dawley rats weighing 250–300 g were included in the study.
Surgical anatomy
When the anatomical structure of the temporal fascia in rats was examined, a layer measuring approximately 1–2 cm in diameter was observed, which comprised the area between the front of the ear at the back, the lacrimal bone and duct at the front, and the eye and frontoparietal bone at the top. This layer is rather thin and is rich in vascular networks.
3
A number of perforating blood vessels originate from this network.
10
The temporal fascia is located superficially right under the skin, below the hair follicles.3,10 This anatomical unit consists of seven layers: the skin, subcutaneous tissue, superficial temporal (temporoparietal) fascia, loose areolar tissue plane (innominate fascia), temporal fascia and temporal fad pads, temporalis muscle, and pericranium1,2 (Figure 1). In histological cross section, some differences are observed between the layers. In the outer surface, the temporoparietal fascia has thicker collagen fibers, whereas the inner surface has more commonly elastic fibers than the outer part. Additionally, abundant fat tissue can be found in the outer surface. However, the inner part of the temporoparietal fascia is also highly vascularized.
3
Besides, the loose areolar tissue plane is quite vascular and comprises very loose connective tissues.
2
A short description of the anatomic layers of the temporoparietal region.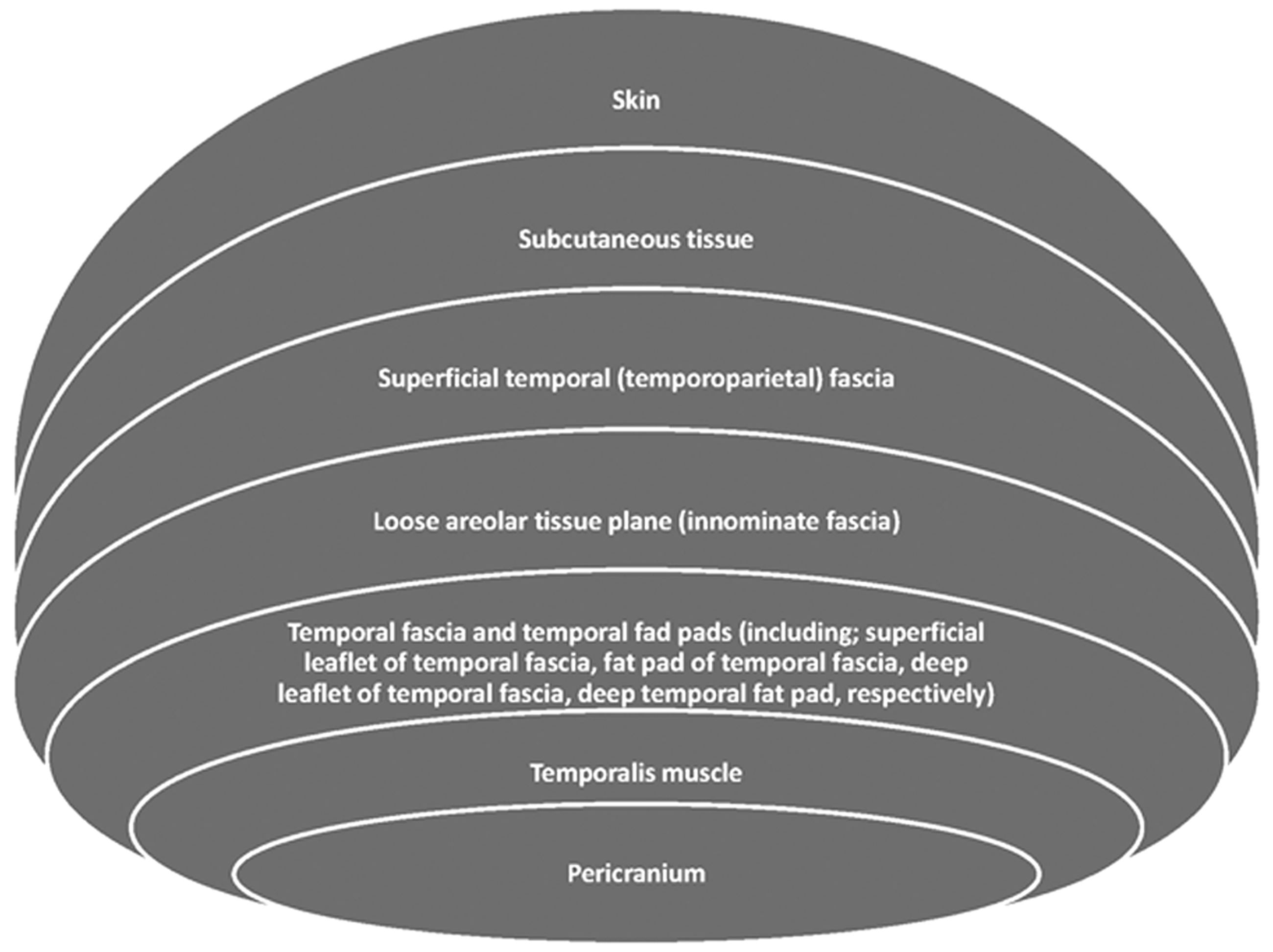
The temporalis muscle may be included to the TPF flap in order to prepare a myofascial flap. The TPF flap is nourished by the arteria temporalis superficialis originating from the external branch of the arteria carotis communis. Its venous drainage is enabled through the vena temporalis superficialis leading to the vena jugularis externa. 3 These blood vessels lie within the superficial and deep branches of the fascia and continue beyond the superficial fascia towards the fronto-parieto-occipital region.3,10 The artery and the vein are within the same perivascular sheath; still, the vein is located more superficially than the artery. These blood vessels are not limited to the temporal area but extend towards the frontal and parietal regions. 3
Surgical technique
A mixture of 90 mg/kg ketamine sodium (Ketalar® flacon; Pfizer Ltd, Istanbul, Turkey) and 10 mg/kg xylazine hydrochloride (Rompun® flacon, Bayer Inc, Leverkusen, Germany) was administered to the rats through the intraperitoneal cavity in order to induce anesthesia. The head region of the rat was shaved and disinfected using a 10% povidone iodine solution (Batticon®, Adeka Ilac Ltd Sti, Samsun, Turkey). A 1.5 cm incision in the shape of a ‘lazy S’ beginning past the eye and reaching until the back of the ear was made in the temporal and preauricular regions (Figure 2).
The skin incision planned in order to elevate a temporoparietal fascial flap.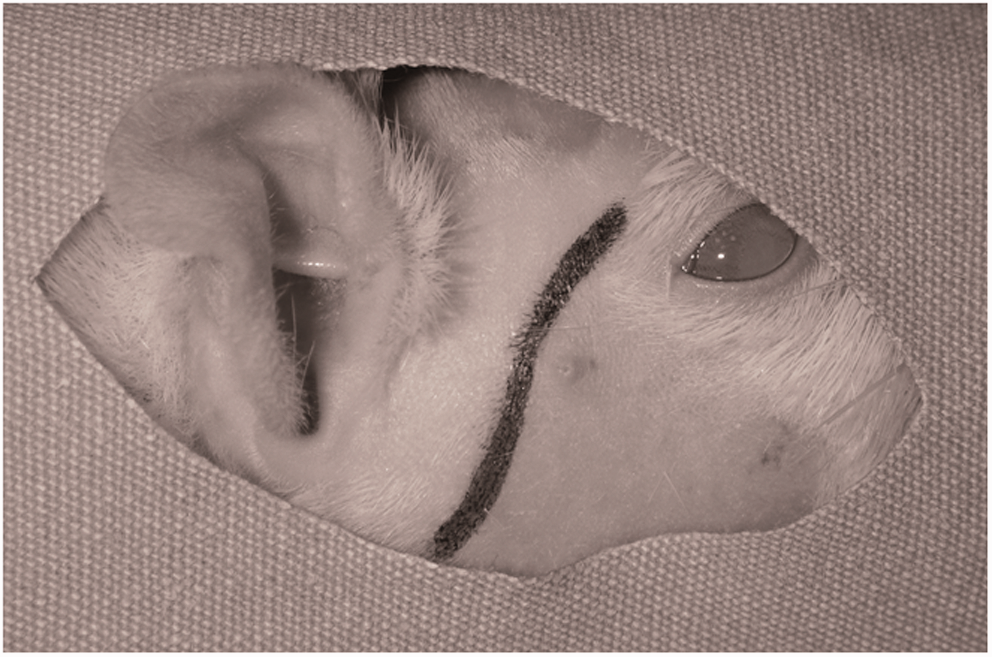
Following the skin incision, using microsurgical techniques, under 4× enlargements through a loupe, a superficial dissection was made in the temporal scalp region reaching right under the hair follicles in a manner which revealed these follicles. The anterior and posterior skin flaps were elevated from over the fascia. The fascial layer was revealed in the area beginning from behind the eye and the lacrimal bone at the front, the front of the ear at the back, the level of the frontoparietal bone at the top where the macroscopic fibrillar appearance of the temporal muscle ends, and the line of the zygomatic arc at the bottom. The top, bottom, back and front sides of the fascia were incised in line with these limits (Figure 3).
Anatomically schematized view of the incised section of the temporoparietal fascial flap (area within the dotted lines). a. = artery, v. = vein, n. = nerve.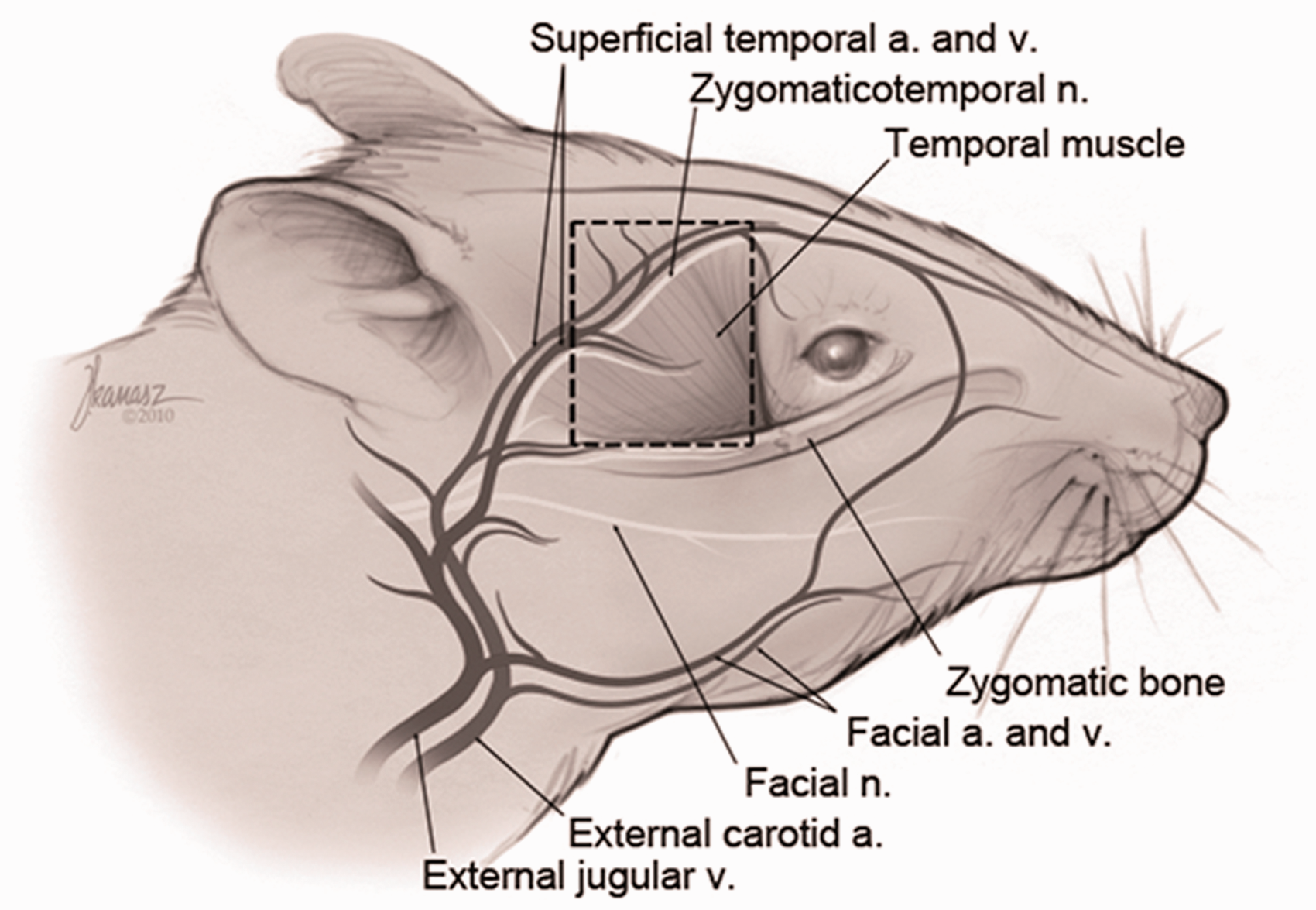
From the upper border of the incised fascia, a route was followed leading to the relatively loose area over the temporal muscle. The dissection continued along the edges of the fascia until the blood vessels on the lower side were observed. When the whole surface of the planned flap was opened, the superficial temporal artery and vein were observed towards the rear edge of the masseter and preserved. The pedicle was reached through the posterior corner of the incision. Since the walls of the blood vessels were rather thin, effort was made to perform a very careful dissection. The superficial temporal artery and vein were isolated in the inferior and posterior regions and the flap was prepared in an axial manner (Figure 4).
View of the flap from axially elevated on the pedicle.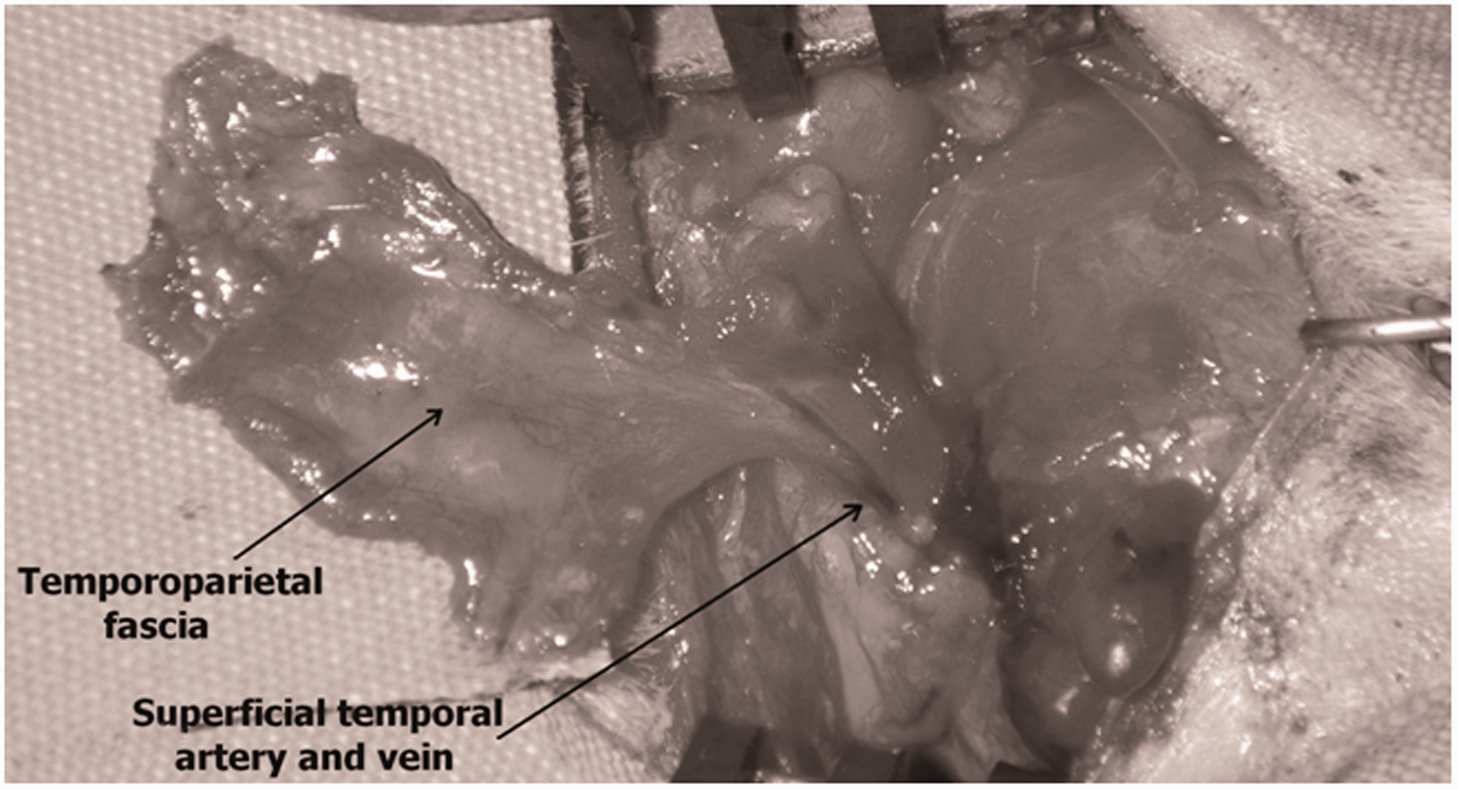
Subsequently, the blood vessels were followed until the point where the facial artery originates from the carotid artery (pivot point) and the dimension and distance of the defects that the flap can cover on this pivot point through arc rotation were measured. The flap was released on the pivot point by cutting off the blood vessels. In order to macroscopically observe the distribution of the pedicle trace within the pedicle, it was cannulated using a number 22 branulla and stained with methylene blue. Then, in order to clearly reveal the blood vessel network of the flap, 50 g of bullet oxide, 5 g of gelatine, 100 mL of 0.9% saline were mixed, a 50 cc mixture was prepared at 70℃, and drawn into a syringe. The blood vessels were stained by injecting the mixture through the cannula into the carotid artery. Following this, the flap was removed and kept at 4℃ for 4 h before microangiographic imaging with the help of a mammography device (Alpha ST®, Penta® Elektronik Medikal Sistemler AS, Istanbul, Turkey).
Results
It was observed that the flap could be rotated in every direction with minimal torsion on the pivot point (the origin of the facial artery from the arteria carotis communis) (Figure 5).
View of the rotation arc of the temporoparietal flap.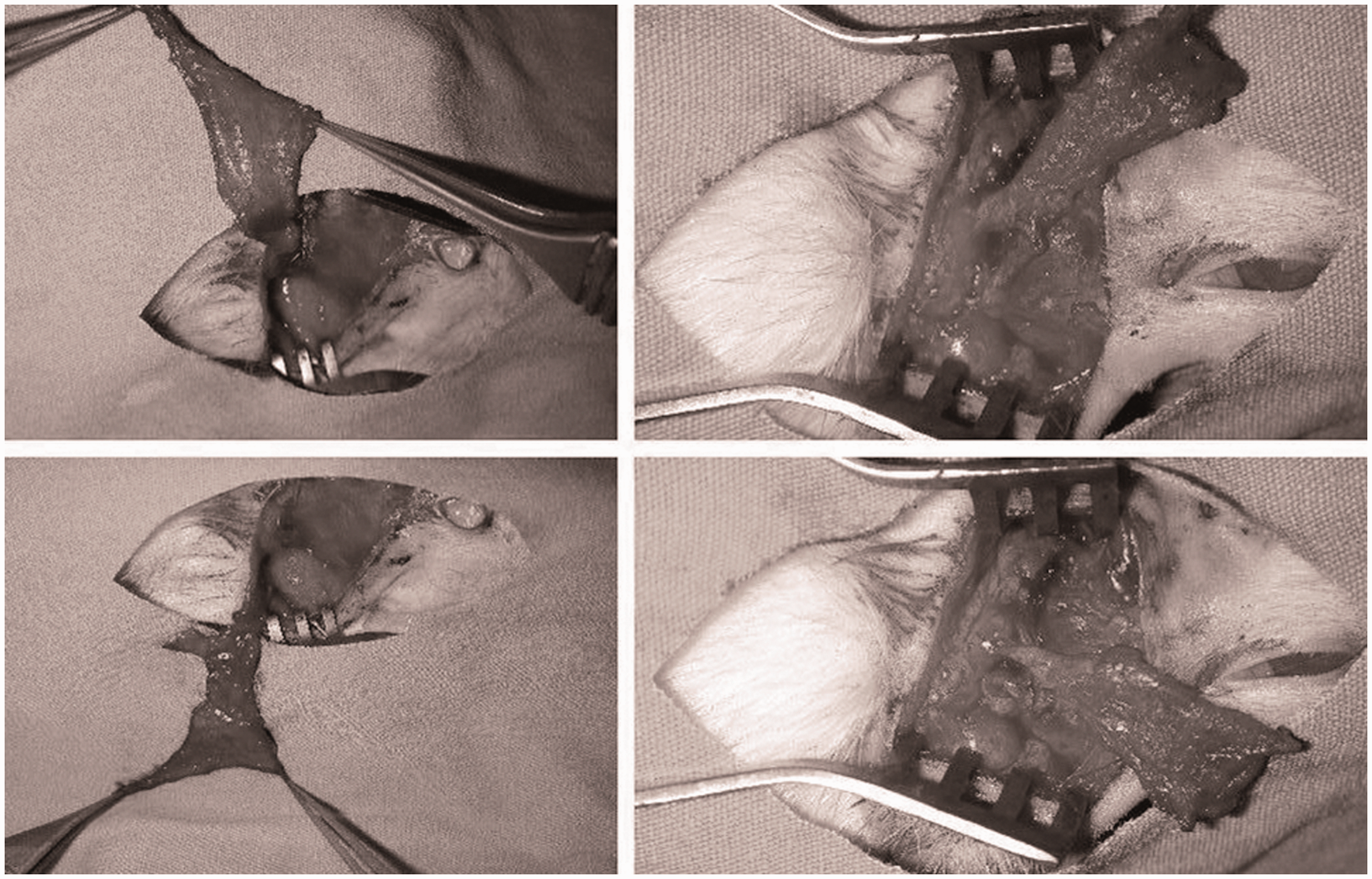
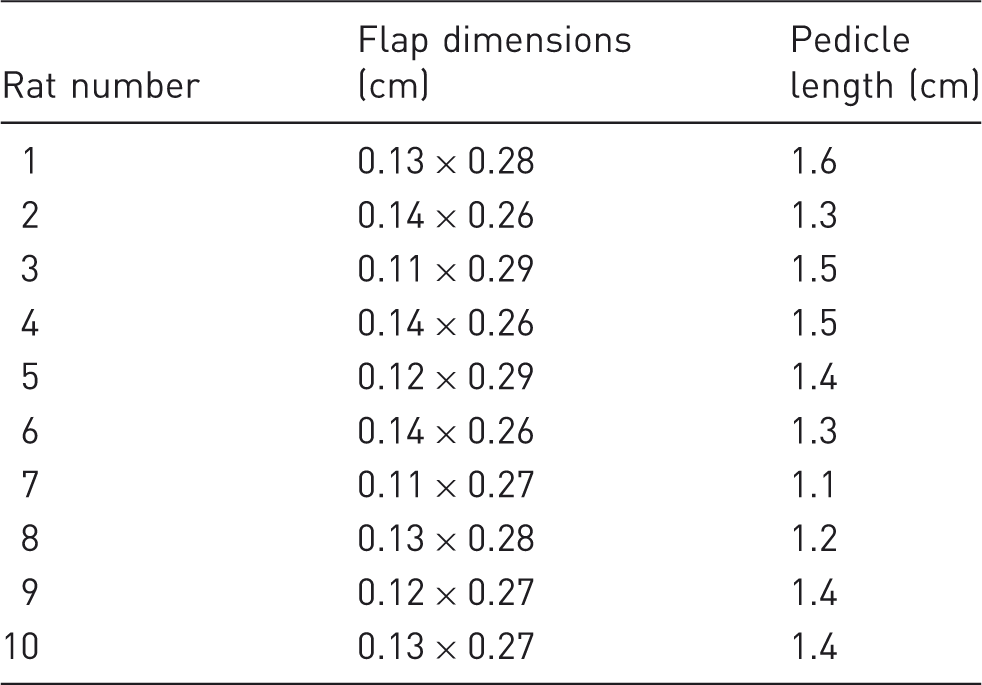
Distribution of the temporoparietal fascial flap dimensions and pedicle lengths.
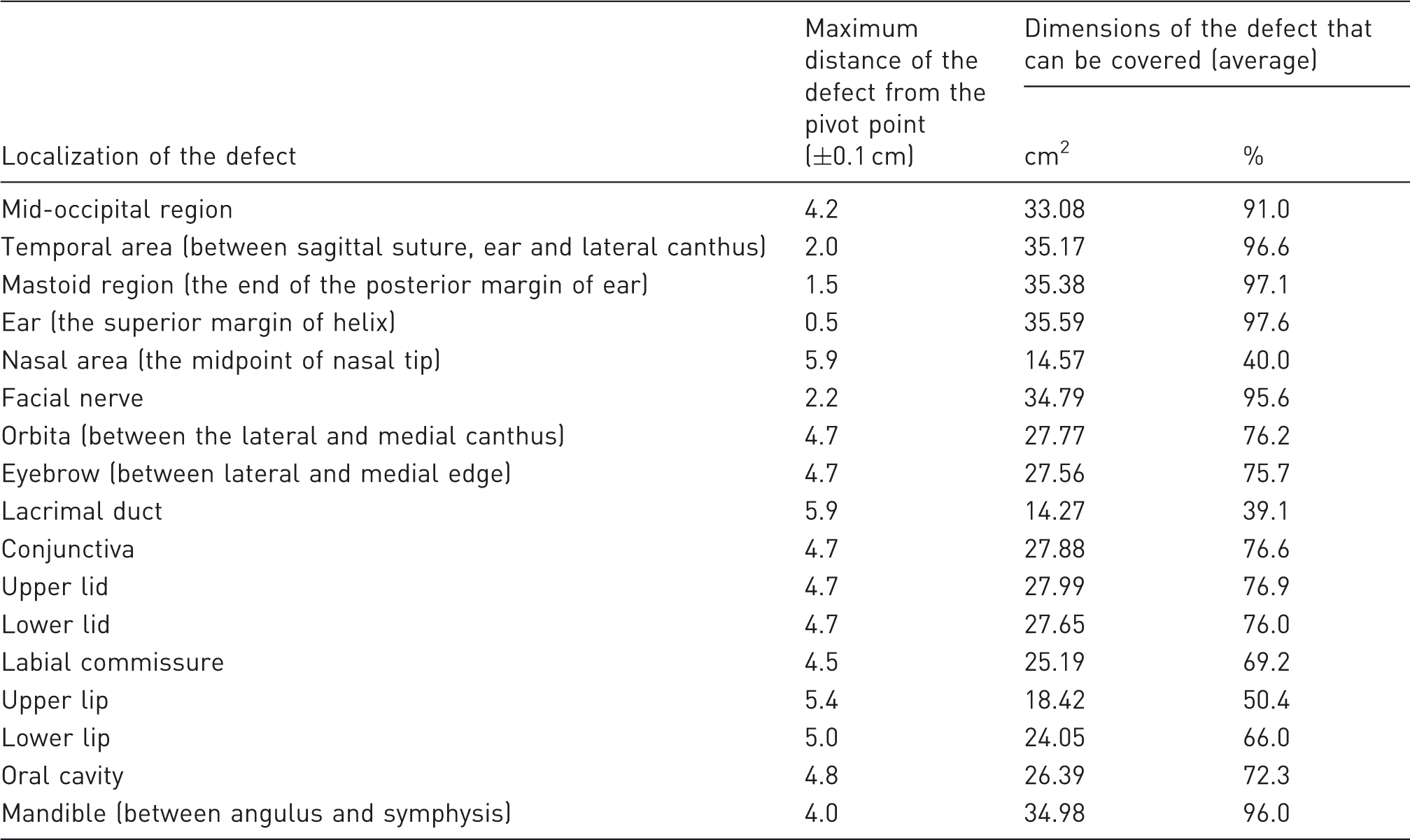
Results of the measurements of the temporoparietal fascial flap in different defect localizations (measurements were performed using calipers).
Following the methylene blue staining of the flap pedicle, it was macroscopically observed that the flap was stained in every area (Figure 6).
View of the flap pedicle stained with methylene blue.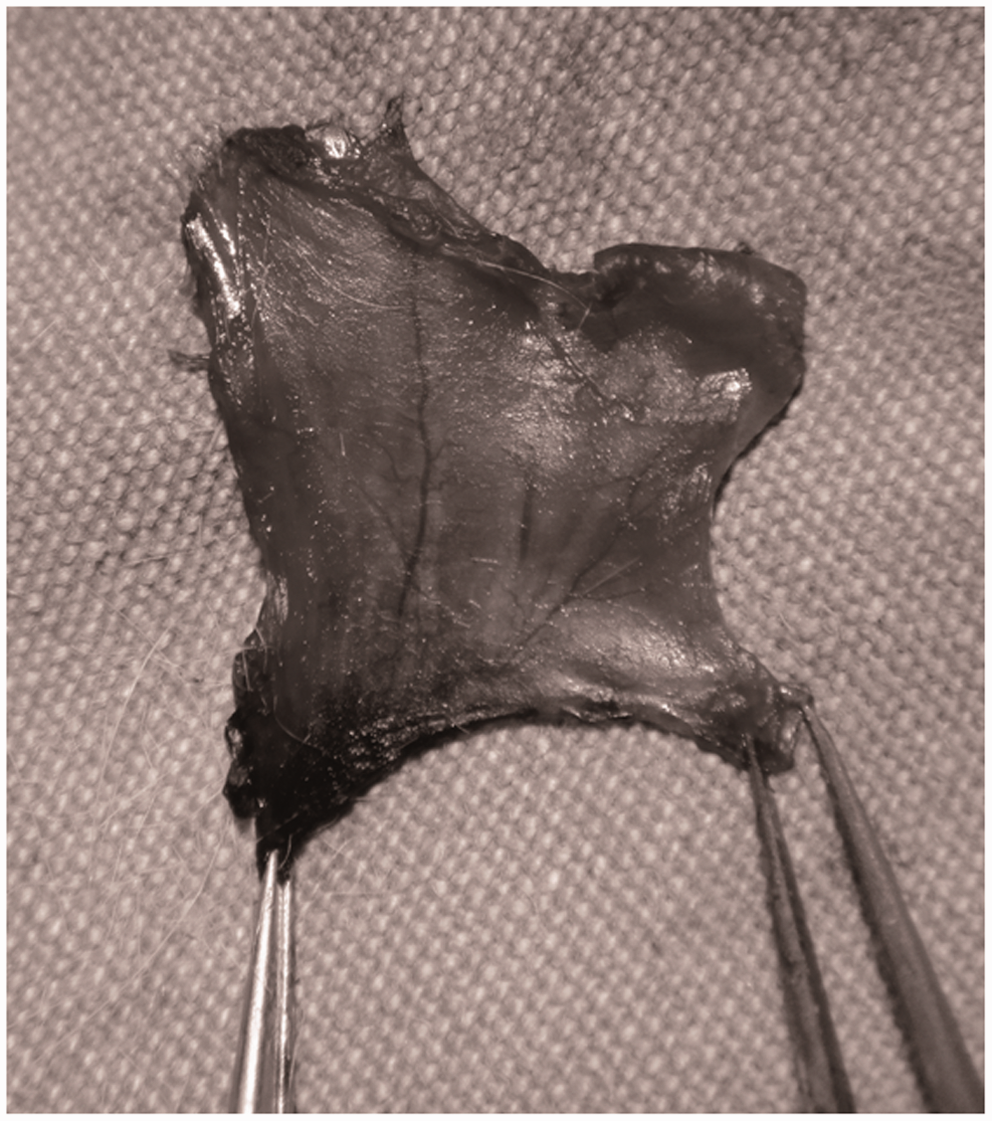
Microangiographic imaging indicated that the pedicle nourishes the whole flap and the circulation in the distal part of the flap was adequate (Figure 7).
Microangiographic view of the vascular structure of the flap.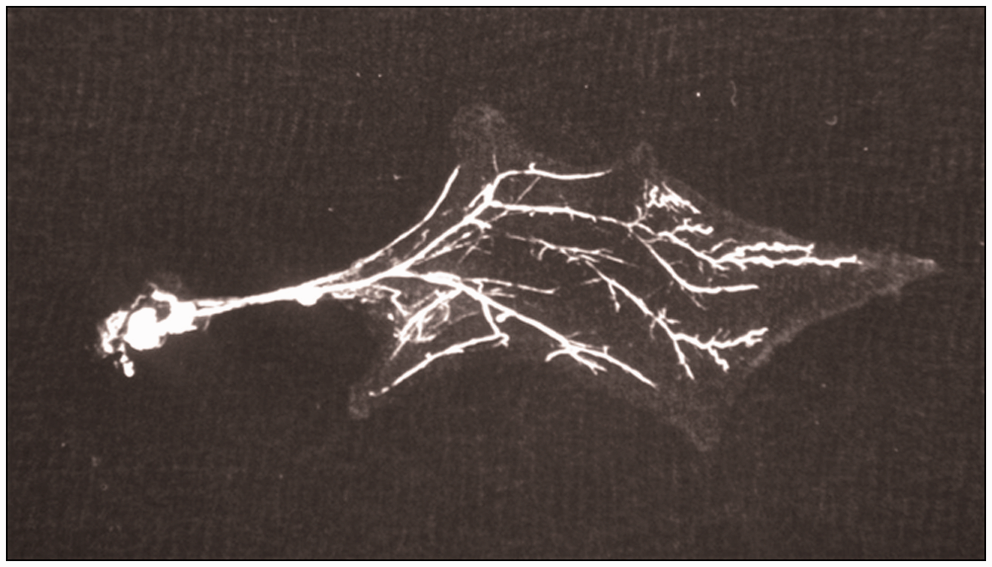
Discussion
The TPF flap has extensive uses in the clinical field.3,11–14 The application areas of this flap include the facial contour defects, 7 repair of the hairy or non-hairy skin defects of the temporal bone, 6 covers of ear cartilage, 15 tympanoplasty operations, 16 repairs of the mastoid cavity defects, eye socket reconstructions,17,18 repairs of the malar or oral cavity defects,19,20 oropharyngeal defect repairs, covers on bones such as the mandible or on the surfaces of other vital organs, 21 repairs of tissue defects on the areas subjected to radiotherapy, treatments of facial paralysis 22 and paralysed labial commissure, conjunctivopalpebral revisions, repairs of subcutaneous defects, inferior labial and orbitomalar reconstruction, malo-commissuro-labial reconstructions, repairs of the eyebrows and the lacrimal duct, reconstructions of the defects causing bone and tendon exposures, covers of calvarial defects, 23 Frey’s syndrome, 24 osteoradionecrosis, nasal septal perforations8,25 and pathologies of the temporal and mastoid bones. 26 Although it is widely used in the clinical sphere, with corresponding data in the literature, we have not found other experimental studies of the TPF flap.
Since the TPF flap is of a thin nature, it is also a very suitable choice for repairs of the ear.3,13,15,23 This way, it becomes possible to form a helical contour and an antihelical eminence. 15 In our experimental study, we observed that the temporoparietal flap is an alternative for covering ear defects adequately. Similarly, defects of the mastoid and temporal areas and the occipital region were also revealed to be greatly suitable for covering with this flap.
The TPF flap can also be used in a composite fashion to include muscle and bone tissue.3,21,23 This flap possesses all the advantages of vascularized osteofascial flaps. Therefore, a composite temporal osteomyofascial flap is a very versatile repair choice for the zygomatic arc, malar and mandibular reconstructions. 21 As we observed that the TPF flap provides adequate tissue coverage in the mandibular region, we believe that it may serve as a good vascular bed for prefabricated models which can be built using osteoinductive and osteoconductive agents.
If the auriculotemporal nerve is included within the flap, the TPF flap may turn into a sensorial flap. 19 Even if it is not prepared in the form of a sensorial flap, we believe that the TPF flap, which according to our observation extends to every point of the facial nerve, may be used as a tube or conduit in experimental facial nerve repairs. Considering that a fascial flap rotated towards the facial nerve region in our experimental study covers an average area of 34.79 cm2, it may be deduced that the flap enables the preparation of a conduit or a scaffold. Besides the conduit feature, the TPF flap may also make prefabrication with various neurotrophic and neurotropic substances possible as in the prefabricated nerve models.
In order to form an eyelid, the TPF flap may be elevated from the hairless region together with the skin island on top of it. Eyelash or eyebrow reconstructions may be performed using the hairy part of the skin island. This way, the negative results observed after skin grafting may also be prevented. 18 However, according to the results obtained from our study, we have observed that this flap applied in the form of a pedicled flap can cover approximately 75% of the rat orbita and eyelid defects. Thus, it can be claimed that through the use of this flap, the need for free tissue transfer is reduced. Also, since the distance of the conjunctival area from the pivot point of the flap is similar to the relevant distance in eyelid defect measurements, approximately 76% coverage is enabled on the conjunctiva. We are of the opinion that this ratio is an acceptable one for the coverage of conjunctival defects. The flap may be formed into a tube in order to build tear ducts. But since the nasal cavity lies rather distally, there may be difficulties according to the results of our study in extending the lacrimal duct into the nasal cavity, although the width of the flap is adequate for this purpose. Similarly, the TPF flap does not form an ideal model for nasal, labial commissure, upper and lower lip and oral cavity defects in rats for the reason that it could only provide limited coverage.
The TPF flap is a reliable flap that can be transferred in the shape of a pedicled, free, or composite tissue.6,13,18,22 Although it can be transferred as a free tissue in humans, since its pedicle is thin, the pedicled use is more common and versatile than the free tissue transplant in experimental studies.1,18 Besides the methylene blue staining, the microangiographic imaging in our study also revealed that the superficial temporal artery perfuses every point of the flap adequately. With meticulous microsurgery, it would be possible to freely transfer the TPF flap, which is relatively safe in terms of the vascular network.
The anatomy of the temporal fascia has been clearly demonstrated and understood in humans.3,23 From an anatomical point of view, this structure in rats closely resembles that in humans. In our study, no variations were noticed in the macroscopic and microscopic studies of the anatomy of the temporoparietal fascia in rats. Therefore, we can conclude that this flap has a consistent anatomy in rats. In humans, the superficial and deep TPF layers can be easily separated from each other. The dissection of the deep temporal fascia is easier than that of the superficial layer. Our experience from the study has also shown that these two layers are separated with a rather thin and loose layer in rats and that it is similarly more difficult to dissect the superficial fascia compared with the deep fascia. Therefore, it is possible to claim that a dissection including both the superficial and the deep layers of the temporoparietal flap during flap elevation can be performed more easily. After the flap is elevated, the dissection of the fascial layers can be executed separately. But since the deep temporal fascia is nourished directly by the specific arterial branches of the innominate fascia, the innominate and deep fascia must not be separated. 3
The possibility of dissecting the flap until the originating point of the facial artery from the carotid artery may extend the rotation arc considerably. Thus, a safe rotation and transposition becomes possible. Although in our study the degree of the curving occurring after the rotation of the pedicle has been subjectively assessed as minimal, we acknowledge the need for further research on this point.
The most important disadvantage of this study is that it evaluates the versatility of the TPF flap, which has a wide range of clinical applications, in an experimental setting. Still, the limited number of experimental studies performed on the subject has underscored the need for this study in spite of this disadvantage. Another disadvantage of the study is that the viability of the flap following the postoperative period could not be observed intravitally in the short and long term. In order to collect data in this respect, advanced experimental devices enabling the measurement of blood flow need to be made available.
The reason for the limited number of the experimental studies performed on this flap may be associated with the thinness of the flap pedicle and the difficulty in its dissection. This study attempted to evaluate the versatility and the feasibility of the TPF flap in various defect models in the head and neck areas in rats. In this respect, the TPF flap is an experimental flap model that can be conveniently used in defect models, especially of the occipital, temporal and mastoid regions, ear, facial nerve and the mandible.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
