Abstract
Accurate knowledge of the health status of experimental animals is pivotal to high scientific and ethical standards in biomedical research. Individually ventilated cages (IVCs) are becoming the predominant system for housing laboratory mice, as they prevent cage-to-cage infections. However, this feature constitutes a major drawback for hygienic monitoring of mouse colonies, as traditional screening programs build on reliable transmission of infectious agents from experimental animals to sentinel mice commonly tested as representatives for the mouse colonies. In recent years, the laboratory animal community has realized that sentinels are ineffectual for screening mouse colonies in IVC systems because infections are often not transmitted to sentinels and therefore remain undetected. Furthermore, sentinel monitoring results in high numbers of used animals. In contrast, environmental monitoring provides a more reliable approach to identify and exclude pathogens in rodent colonies. In recent studies we provided evidence that polymerase chain reaction analysis of exhaust air particles is superior to soiled bedding sentinels for different agents. In this study, we show that testing pooled environmental samples generates more meaningful information compared to soiled bedding sentinels during routine hygienic monitoring in different barriers.
Introduction
Analysis of environmental samples from individually ventilated cage (IVC) systems is a relatively new and promising approach to improve hygienic monitoring in laboratory animal facilities. Still, most hygienic monitoring programs are based on soiled bedding sentinels (SBS) tested as representatives for colony animals. Sick animals might be used in addition for direct testing but many infections are subclinical and infected animals are inconspicuous.
1
After an adequate exposure time sentinels are commonly euthanized and tested for pathogens using different testing methods. This leads to high numbers of mice sacrificed for health monitoring. Detection of unwanted organisms relies on infection of the sentinels via soiled bedding, yet it is well investigated that not all agents are transmitted via soiled bedding. Viruses, such as lymphocytic choriomeningitis virus, Sendai virus, or mouse norovirus, bacteria, such as
Material and methods
Animals
All SBS used in the study were 8–9 weeks old female SOPF RjOrl:SWISS obtained from Janvier Labs (Laval, France) and housed in groups of three animals per cage. Breeding and research colonies in different barriers consisted of mice of both sex and of various strains, immune status, age, and genetic background. Mice were housed in sex-matched groups of 2–5 animals per cage or as breeding pairs with or without offspring. Exceptionally, single animals were housed separately for a short time. Animal housing was performed in strict accordance with the Directive 2010/63/EU. All animals were housed in IVCs (Sealsafe plus, GM500, Tecniplast, Buggugiate, Italy) under specific pathogen-free conditions with a maximum cage density of five adult mice per cage (>100 cm2 floor area per mouse) and environmental conditions according to Directive 2010/63/EU (12 h light/12 h dark, 20–24℃, and 45–65% relative humidity). Autoclaved wood chips (Lignocel select fine, J. Rettenmaier & Söhne GmbH, Rosenberg, Germany) and paper strips (Arbocel crinklets natural, J. Rettenmaier & Söhne GmbH) were used as bedding and nest material. Sterile-filtered tap water and an irradiated standard diet for rodents (Altromin 1314, Altromin Spezialfutter GmbH, Lage, Germany) were available ad libitum. Access to the barriers involved air shower or wet shower and a complete change of clothes and shoes as well as wearing gloves, surgical masks, and bonnets the entire time. All cage changes and sample collections were performed in a HEPA-filtered cage-changing station.
Soiled bedding sentinel monitoring
Three SOPF SWISS mice were housed per sentinel cage. Each sentinel cage was assigned to a single room containing one to five IVC racks, each containing 63 cages. Once per week, 10 mL soiled bedding was collected from each cage of one of the IVC racks during routine cage changing process in a rotating system. The sentinel cage was filled with the soiled bedding mixed with an equal amount of fresh bedding. Sentinels were exposed to soiled bedding of each rack within the animal room at least two times per monitoring period of 12 weeks. After 12 weeks exposure time two sentinels per cage were killed and used for hygienic monitoring examination performed by an external diagnostic laboratory. The third mouse was kept as back-up in case of unexpected findings or test failure. Sentinel mice were tested using ELISA, PCR, bacterial culture, and microscopy for all FELASA listed agents as well as Hantavirus, CAR bacillus,
IVC air handling units and sample collection
Two different IVC air handling units (SKY FLOW and SMART FLOW, Tecniplast, Buggugiate, Italy) were used at 60 air changes per hour in positive pressure mode (15–22 Pa). For each EAP analysis, two autoclaved gauze pieces (ES Kompressen, Paul Hartmann AG, Heidenheim, Germany), each four layers thick and 2 × 2 cm in size, were pinned onto the “dirty side” of the exhaust air prefilter directly above the exhaust air hose opening and stayed there for three weeks. Dust loaded gauze samples were collected in 2 mL microcentrifuge tubes using disposable forceps. Up to 10 individual gauze pieces, each representing all cages connected to one air handling unit, were pooled in a 50 mL centrifuge tube and sent to a commercial diagnostic laboratory. Samples were tested by PCR for the same panel of agents as described for the SBS. 1 The second gauze piece from each air handling unit was stored as a back-up sample for further testing.
Study design
Study A
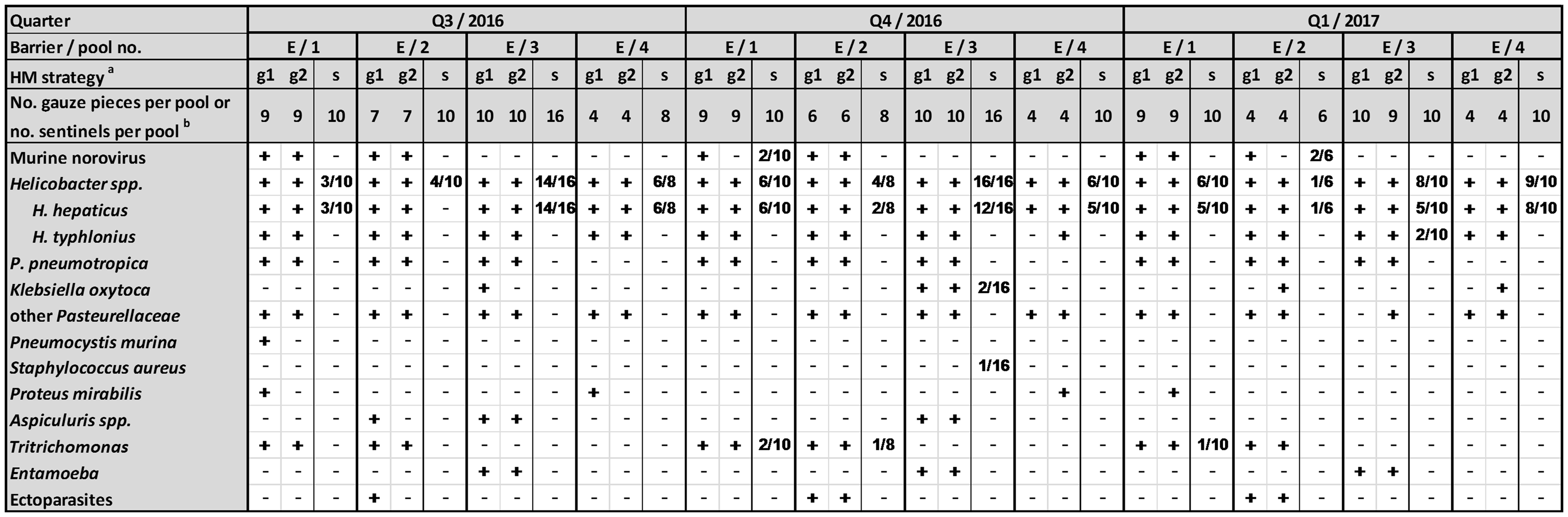
Health monitoring via SBS was compared to EAP PCR in an experimental barrier over three consecutive monitoring periods each with a sentinel exposure period of 12 weeks. The barrier consisted of 16 rooms with a total of up to 30 air handling units connected to 5004 cages in 78 racks, represented by maximal 44 sentinels or 30 EAP samples assigned to four pools. Rooms were assigned to the four pools based on previous SBS monitoring results, individual investigator ownership, and whether colonies were sanitized or not. Gauze pieces were pinned onto the “dirty” side of the prefilter for three weeks. About half time and at the end of each monitoring period, gauze pieces were collected and pooled for EAP PCR. Exposure times of SBS and gauze pieces are shown in Figure 1. In order to be able to compare which of the monitoring methods provided the most meaningful outcome for screening the colonies, results of pooled EAP samples and the corresponding SBS results were integrated into Figure 2.
Exposure time of soiled bedding sentinels and gauze pieces during studies A and B. Fields highlighted in gray depict dirty-bedding sampling and installation period of gauze pieces. Results of conventional soiled bedding sentinel monitoring compared to exhaust air dust analyses over a period of three consecutive quarters in one experimental barrier. Only agents identified by either method are shown in column one. For the sake of clarity agents with negative test results were skipped from the list. Pool number: rooms of the experimental barrier were divided into four pools.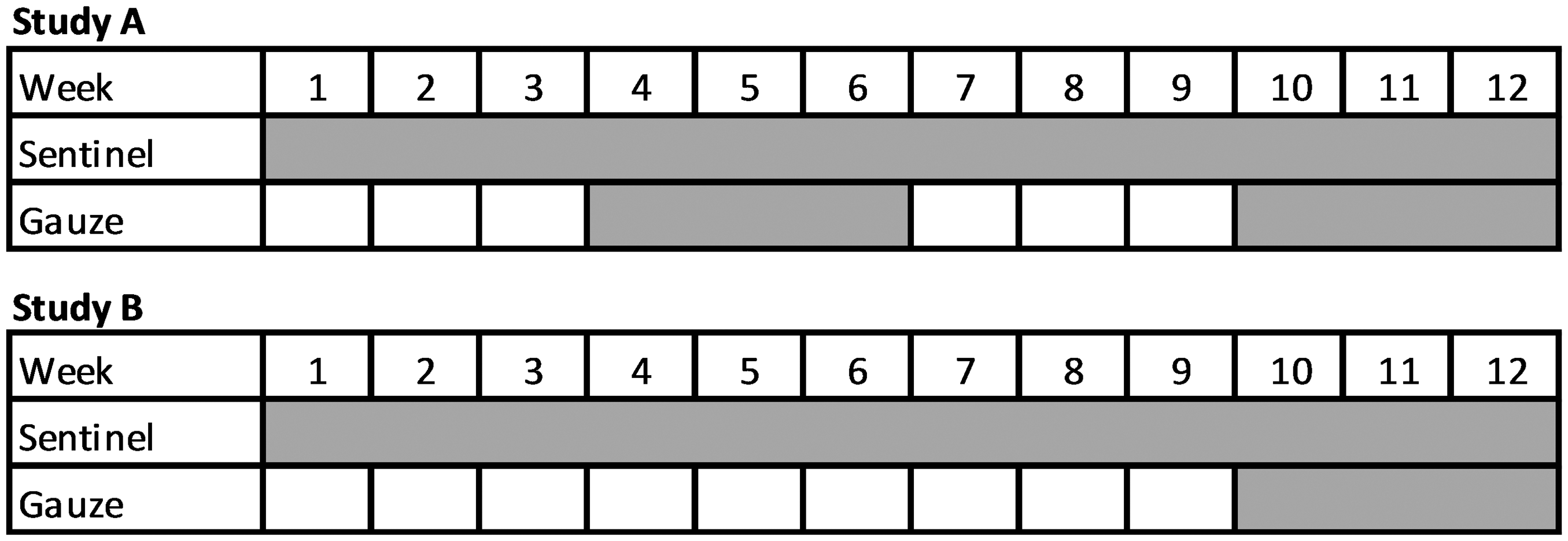
Study B
The study was then expanded to eight different breeding and experimental IVC barriers using the same experimental outline. The eight barriers consisted of 54 rooms with a total of 85 air handling units connected to 19,483 cages, represented by 152 sentinels or 85 EAP samples assigned to 13 pools. Large barriers with more than 10 air handling units were divided into two separate pools to analyze a maximum of 10 gauze pieces per pool. This time, dust samples were only taken during weeks 9–12 of the 12 week sentinel exposure period (see Figure 1). Results are shown in Figure 3.
Results of conventional soiled bedding sentinel monitoring compared to exhaust air dust analyses in eight experimental and breeding barriers.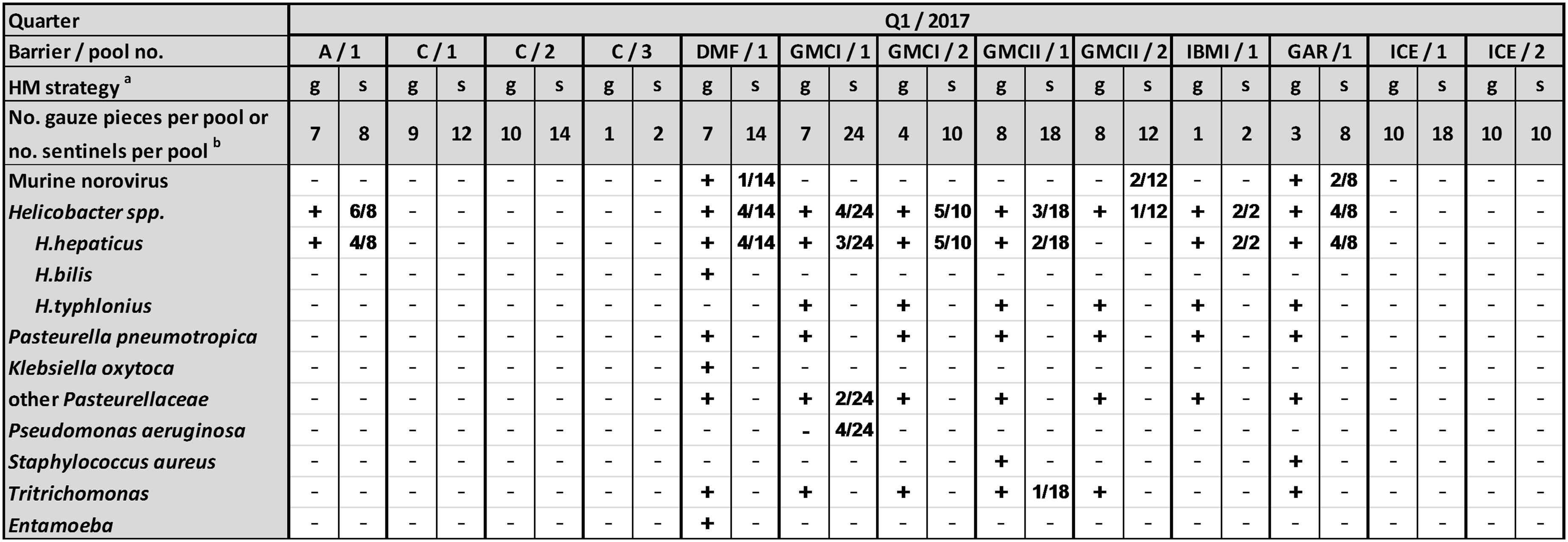
Results
In study A, the results of conventional soiled bedding sentinel monitoring and of EAP PCR were compared over a period of three quarters in an experimental barrier. For the following pathogens one or both methods revealed positive results at least once: MNV,
Eight experimental and breeding barriers, containing 85 air handling units and represented by 152 SBS were included in study B. Results of 13 pools of gauze pieces, consisting of 1–10 gauze samples were compared to results generated by the corresponding 13 groups of SBS (2–24 SBS per group) (Figure 3). Agents detected by either method, sentinel mice or EAP PCR, were MNV,
Discussion
Knowing the accurate microbiological status of laboratory mice is crucial for their use in biomedical research. Undetected pathogens represent an uncontrolled variable that might interfere with animal experimentation.
11
Soiled bedding sentinel based hygienic monitoring, still state-of-the art in the field, is flawed by serious drawbacks and many different pathogens might be overlooked. To bypass these drawbacks, EAP PCR was investigated for agents that are known to be badly or not detectable by SBS. The studies showed a clear superiority of EAP PCR compared to SBS even at very low prevalence.2,4,7,9 To investigate if environmental samples might completely make sentinels disposable in our routine hygienic monitoring program, both monitoring strategies were performed in parallel in different breeding and experimental barriers in our facility. In case SBS monitoring would not provide any additional meaningful information in comparison to environmental monitoring by EAP PCR, SBS might be replaced in future. Our routine microbiological monitoring is based on several layers of testing and decision making. The first layer aims at screening for potential infections within the mouse colonies with sufficient sensitivity at the lowest possible amount of testing. This is a matter of economics. Ideally, a single cage of infected animals within an otherwise negative colony is detectable by that first layer. In parallel, sick animals from the colonies are tested individually by classic methods like serology, bacteriology, parasitology, and postmortem analysis. Once an infection is detected, a decision is taken if this might be accepted, for example, because the agent is not on an exclusion list, or if the result warrants further testing. Further testing is based on a series of pooled tests where positive pools are broken down to smaller pools as long as all infected animals can be identified. During study A, EAP samples unexpectedly detected
Residual nucleic acids pose a risk of false positive results in future monitoring periods even if the infected animals have been removed from the colony. Discriminating between previous and current infections might not be possible then. Therefore, after an elimination attempt, careful sanitization of the equipment is necessary to remove all living or dead infectious agents, including all contaminating residual nucleic acids. We recommend washing and autoclaving the contaminated rack and all tubing, washing and spraying the prefilter case with disinfectant and exchange of the exhaust prefilter and to perform baseline testing for the agent in question before adding any new cages with animals to the rack system. We installed and sampled always two EAP samples on each prefilter. Individual retesting of the back-ups allowed not only to trace down infections quickly, it also allowed us to save money, because testing for single agents is much less expensive than testing for a whole panel of agents.
Pooling of samples is cost effective and suitable to monitor different barriers by using reasonable sample numbers. Nevertheless, false-negative results due to the dilution of nucleic acids cannot be ruled out, especially for infections with low prevalence. In this study the focus was on the comparison of EAP-PCR and SBS monitoring. The clear result was, even at a high level of pooling, that EAP PCR detected nearly all infections found by the SBS monitoring plus more agents not detected by SBS. Further studies are necessary to define the maximum level of pooling that combines economic aspects and an acceptable low probability of false negative results.
During our studies positive SBS results could not be confirmed by exhaust air dust PCR in three instances. In study A, one sentinel was positive for
In our studies, 8–9 weeks old female SOPF SWISS mice were used as dirty-bedding sentinels. Optimal age of sentinels is discussed controversial. Besselsen et al. showed with two serologic assays that ICR mice inoculated at four and eight weeks of age seroconverted, but were generally unable to seroconvert when infected at 12 weeks of age.
12
In contrast, a recent study demonstrated that transmission of agents such as MPV, MHV, MNV, Theiler's murine encephalomyelitis virus (TMEV), and pinworms did not differ in regard to the age of Swiss Webster sentinels.
13
Each sentinel cage consisting of three mice was assigned to a single room containing one to five IVC racks within different barriers. The number of colony cages (63 to a maximum of 315 cages per room) was higher in some rooms than recommended by the FELASA.
1
Sentinels were exposed to dirty bedding from individual racks at least twice during the quarterly screening. Insufficient transfer of infectious agents is a general drawback of dirty-bedding sentinel monitoring. Regardless of the amount and frequency of dirty-bedding transfer, there are various reasons for a reduced transmission of agents via soiled bedding, like low viability of bacteria such as
As shown here sentinel-free monitoring based on EAP PCR analysis in IVC reared mouse colonies is feasible. We provided evidence that our sentinel mice are completely replaceable by environmental samples. In addition to routine environmental monitoring, examination of sick mice showing clinical signs of illness should be a fixed component of health monitoring programs. This change of paradigm in rodent microbiological monitoring highly contributes to the 3R principle. First of all it allows replacing laboratory mice, in our facility about 1000 sentinel mice per year. The new technology provides much better information regarding the microbiological status of rodent colonies. This information allows us to better react on new infections and reduce the number of sick animals, which is refinement. Better knowledge of the microbiological status allows better standardization and therefore contributes to the reduction of animals by providing more statistical power.
Footnotes
Acknowledgements
The authors would like to thank the animal caretaker teams and Michael Opitz for excellent support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
