Abstract
We compared the efficacies of two different individually ventilated cage systems (Allentown and Tecniplast) for health monitoring (HM) of murine infectious agents using exhaust air particle (EAP) capture and real-time PCR. After three months of monitoring, both EAP capture media allowed detection of Helicobacter, Pasteurella and Entamoeba. Use of the EAP real-time PCR for HM reduces the number of mice used.
Health surveillance is important in contemporary mouse facilities. Previously, health monitoring (HM) of mice was performed primarily via soiled-bedding sentinels (SBS). Use of individually ventilated cage (IVC) systems, which provide biocontainment, 1 challenged reliable HM with SBS since transmission of infectious agents to SBS was sometimes insufficient. Recent reports showed higher detection of murine infectious agents with EAP real-time PCR compared with SBS.2–7 Since the air flow and the mode of capture of EAP in the Allentown and Tecniplast IVC systems are different, which might influence HM results, we compared their efficacies.
The NexGen IVC rack (Allentown, NJ, USA) has a top-mounted air handling unit (AHU) (Figure 1(b)), and holds 60 cages with 50 air changes per hour (ACH). Air is supplied at the cage floor level and exhausted at lid level (Figure 1(a)). The supply-exhaust ventilation system has horizontal manifolds at the cage level connected to a vertical plenum between manifold and the rack supply-exhaust ventilation system (Figure 1(c)). Room air is pre-filtered and H14-HEPA-filtered before reaching the cages; exhaust air is pre-filtered and H14-HEPA-filtered before returning to the room exhaust air system (Figure 1(b) and (c)). In this study, the rack was coupled with one rack-mounted AHU (EcoFlo). The Sentinel EAD (exhaust air dust) medium selectively captures particles by an electrostatic filter-like dual-fiber material. The air dynamics at the rack exhaust level (Figure 1(c)) allows transportation of micro-particles collected at cage level up the rack exhaust to the Sentinel EAD medium and its holder device (Figure 1(c) and (d)).
Allentown (above) and Tecniplast (below) IVC rack systems and capture media. In the racks, the blue arrows represent supply air, the red arrows depict exhaust air. (a) Cage ventilation, (b) NexGen IVC rack, (c) Horizontal manifolds and vertical exhaust plenum, (d) Sentinel exhaust air particle medium (top), swabbing (below), (e) Complete cages, (f) Cage ventilation, (g) Greenline GM 500 IVC rack and horizontal air-exhaust plenum (*), (h) Supply and exhaust air flow, (i) Insertion of the Interceptor and swabbing of the air exhaust plenum, (j) Complete cages.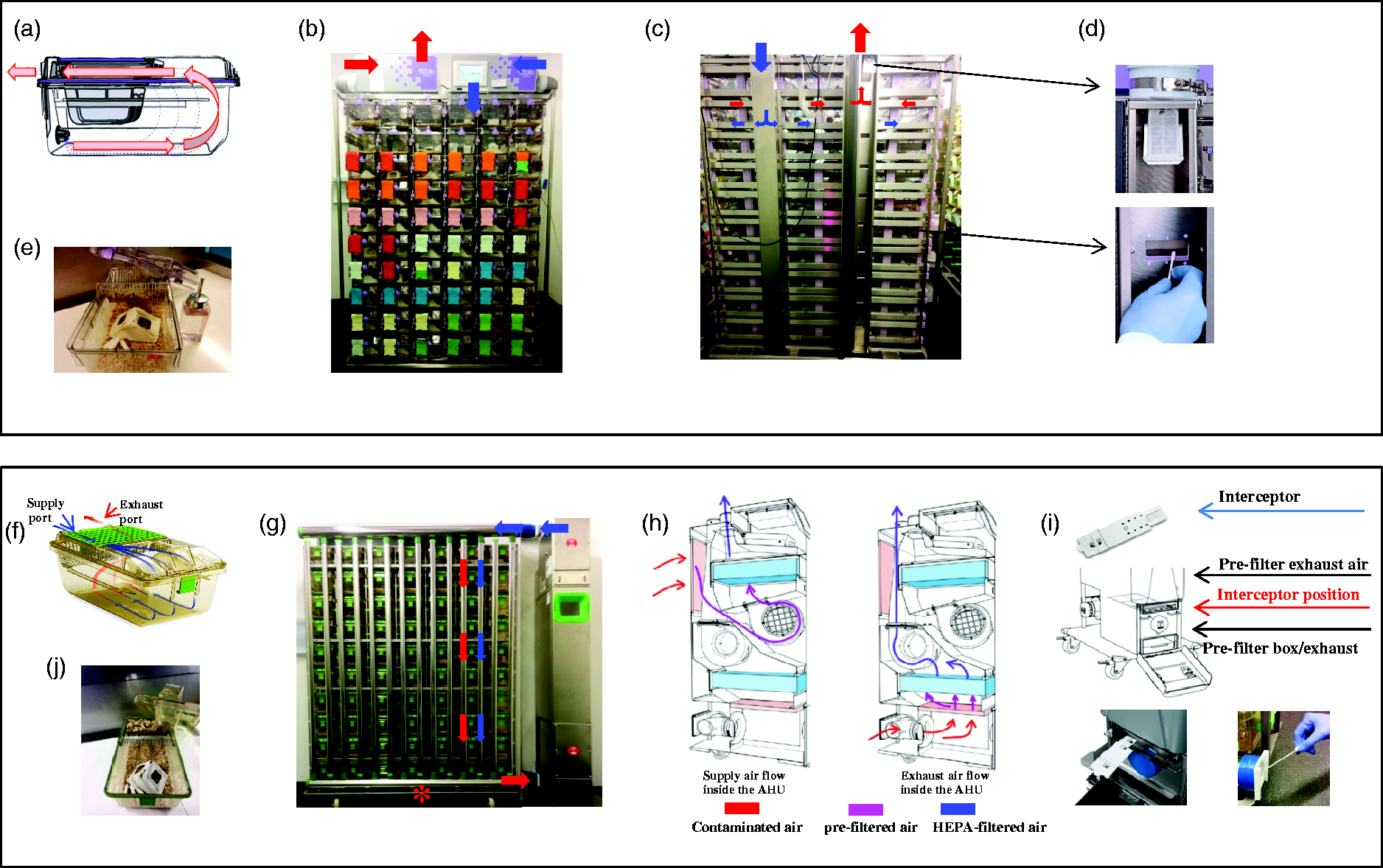
The Tecniplast system has a standing AHU (Figure 1(g), Greenline GM500, Buguggiate, Italy) and holds 70 cages with 75 ACH. Air is supplied and exhausted at lid level (Figure 1(f)). The vertical plenums allow micro-particles emitted from the cage to fall into the removable horizontal air-exhaust plenum (Figure 1(g)). Room air is pre-filtered and H14-HEPA-filtered before reaching the cages; exhaust air is pre-filtered and H14-HEPA-filtered before emission into the room exhaust air system (Figure 1(h)). In this study, one rack was coupled with one AHU (TouchSlim Plus). The Interceptor is an irradiated cardboard folder containing a sliding filter. The frame with slots for two interceptors is placed underneath the exhaust pre-filter of the AHU (Figure 1(i)). The exhaust plenum (Allentown) or the exhaust filter box (Tecniplast) is maintained in negative pressure, preventing emission of unfiltered contaminated air.
During the study, materials were decontaminated or sterilized. Since the autoclave did not accommodate complete racks, they were cleaned with Torvan (Johannes Kiehl KG, Odelzhausen, Germany) and decontaminated with Pursept-A Xpress (Schülke & Mayr GmbH, Norderstedt, Germany). The Sentinel capture medium (Figure 1(c) and (d)) and the Interceptor (Figure 1(i)) were introduced using new gloves. A sample was taken from the vertical plenum of the IVC rack from each row (Allentown, Figure 1(d)) and from the horizontal air-exhaust plenum (Tecniplast, Figure 1(i)) using sterile swabs (Copan Italia, Brescia, Italy, Cat-No.: 160C) and screened via real-time PCR to determine whether nucleic acid from murine infectious agents was present. The testing laboratories were unaware of the health status of the room prior to receiving samples. Complete cages were autoclaved with bedding (FS14, J. Rettenmaier and Söhne, Rosenberg, Germany), houses (Mouse Smart Homes, Datesand Group, Manchester, UK), and nestlets (Arbocell, Rettenmaier and Söhne) (Figure 1(e) and (j)).
Male mice (Mus musculus), primarily on a C57BL/6 background, were kept singly for rederivation and embryo cryopreservation and until lines were archived, and breeding of rederived lines was not compromised. Thus, our work did not warrant a special licence and is in compliance with the German Animal Welfare Act. Mice were kept in IVCs under positive pressure at 20–24℃, 50–70% relative humidity and a 12 h/12 h light/dark cycle. They received an autoclaved diet (1314P FORTI, Altromin, Lage, Germany) and autoclaved water ad libitum. Staff wore autoclaved suits, shoes, face masks, bonnets, and disposable gloves. Mice were handled under a class II biosafety cabinet (BS 48, Tecniplast) with forceps, which were disinfected after each cage change with 70% ethanol. The biosafety cabinet and the gloves were disinfected with Pursept-A Xpress after each cage change every two weeks.
As shown in Figure 2(a) and (b) and as tested by a different laboratory to Charles River Laboratories, the same mice positive for bacteria and their sentinel positive for Entamoeba spp. were kept in the Allentown system and then in the Tecniplast system for three months each. In both systems, 47 cages contained mice and 13/60 (Allentown) or 23/70 (Tecniplast) were free of mice. In the Tecniplast rack, three mice (3B, 5B, 5E) on the 1st day of this phase (Figure 2(b)) and four mice (3A 3F, 7C, 7F) two weeks after commencement of this phase were replaced by negative mice due to death or injury of mice used in Phase A. After the three-month period, the Sentinel capture medium or Interceptor was evaluated, as described for swabs, except that 1−1.5 mL of lysis buffer was used for the initial step.
8
FELASA-listed infectious agents and Bordetella bronchiseptica, Bordetella hinzii, Campylobacter genus, CAR bacillus, Corynebacterium bovis, Klebsiella oxytoca, Klebsiella pneumoniae, Proteus mirabilis, Pseudomonas aeruginosa, and Staphylococcus aureus were monitored. Afterwards, mice were euthanized by cervical dislocation.
Infectious agent status of mice used. (a) Phase A - Allentown rack, (b) Phase T - Tecniplast rack.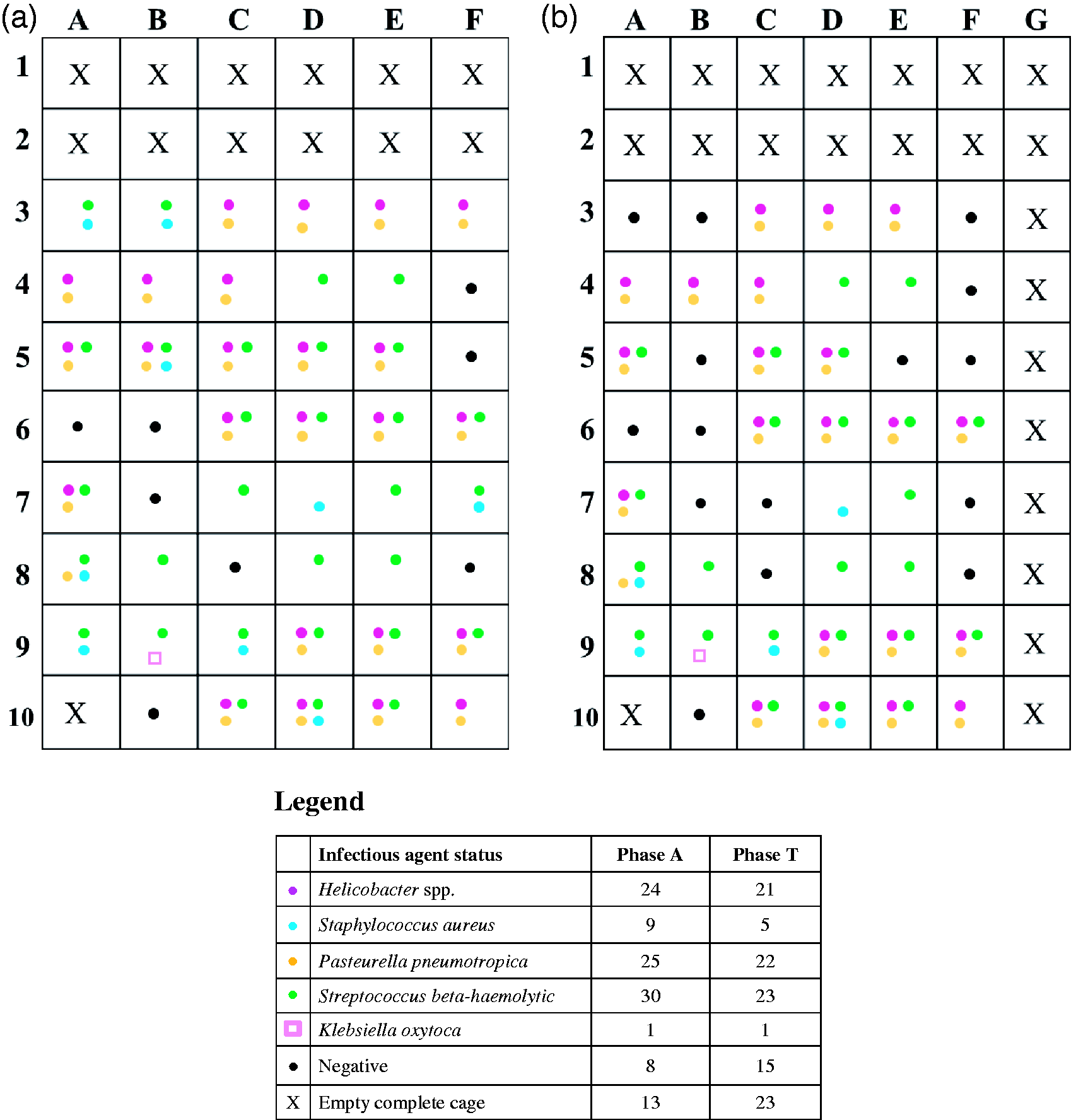
The swab samples taken immediately after rack decontamination tested negative for the relevant agents. The laboratory detected Entamoeba, Helicobacter hepaticus, Helicobacter typhlonius, Helicobacter ganmani, and Pasteurella pneumotropica, biotype Heyl and Jawetz in the Sentinel capture medium, and Entamoeba, Helicobacter hepaticus, Helicobacter typhlonius, Helicobacter ganmani, and Pasteurella pneumotropica, biotype Jawetz in the Interceptor. Neither IVC system detected Staphylococcus aureus and Klebsiella oxytoca, probably due to low prevalence and/or organism numbers, or beta-haemolytic Streptococcus because of absence of shedding or of group B. At the end of the study, beta-haemolytic Streptococcus, group B was not detected in any mouse, and seven mice were positive for Entamoeba spp. The few differences observed in the infectious agent status of mice used in both phases (Figure 2, legend) did not seem to influence the results obtained. A lack of detection of the biotype Jawetz in phase T may be due to cessation of shedding.
This is the first study to show that the use of both types of IVC systems with differing modes of EAP capture led to comparable results regarding the detection of the infectious agents in naturally infected mice monitored over a period of 3 months each. This means that routine husbandry with singly-kept mice, as practiced with quarantined mice, is conducive to detection of at least Helicobacter spp. and Pasteurella spp., which are more difficult to detect via SBS, and Entamoeba, which is easily transmitted to SBS. Our results confirm reports using the Tecniplast IVCs and extend these to include similar results with the Allentown system.3,4 We showed that appropriate cleaning and decontamination is sufficient to eliminate the presence of nucleic acids below the detection level even if an autoclave is not available. EAP real-time PCR technology can serve as an adjunct method in HM, leading to a reduction in the number of mice needed for routine HM.
Footnotes
Acknowledgements
The authors would like to thank Anika Lorenz and Heike Breuer from the CMMC Animal Facility, Malte Heykants, from the Research Group Comparative Medicine for excellent technical assistance, and Stefan Brux from Tecniplast and Jan Ludwig from Allentown for their technical support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Stephanie Durand and Kenneth Henderson are employed by Charles River Laboratories and provided testing for this investigation. Patrick Hardy is employed by Allentown Veterinary and Professional Services and contributed to the study design.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
