Abstract
Transthoracic echocardiography (TTE) is a valuable non-invasive imaging research technique. In ovine models of cardiac disease, restraint for TTE often involves sedation even with currently available restraint equipment; our goal was to determine the feasibility of using a commercial restraint device, commonly known as the sheep chair, in minimizing animal stress and the need for sedation while achieving a complete TTE examination. A total of 10 healthy adult Dorset sheep were restrained in a sheep chair for TTE and observed for signs of stress. No animals displayed overt evidence of stress and none required sedation. While individual anatomic variation existed, image quality was sufficient to determine cardiac function. These observations suggest that a sheep chair is a useful aid in minimizing the need for sedation to acquire a full TTE study in ovine subjects.
Evaluation of the hemodynamics and structural anatomy of the heart by means of transthoracic echocardiography (TTE) is a vital non-invasive imaging technique for numerous species in both clinical and research settings. 1 Ovine models are increasingly popular research subjects due to ease of handling and their similarities to humans in microscopic and gross cardiac and hemodynamic parameters. 2 However, sheep will often exhibit signs of stress (struggling, tachypnea, tachycardia, vocalization, etc.) when held in a standing or laterally recumbent position, such as for TTE examination. 3 While there are published reference ranges for non-sedated sheep, most reports of TTE in ovine models involve sedation and produce good quality images mainly from the parasternal views.1,4,5 There are significant drawbacks of sedation for TTE examinations (i.e. medication cost and effects on cardiac function and blood pressure) which create a risk for the subjects and can alter echocardiographic measurements.
We aimed to find a less stressful TTE restraint technique where sedation would not be necessary and image quality would be maintained. Semi-recumbent restraint devices, called sheep chairs or sheep hammocks, are available to facilitate maintaining of sheep in a ‘sitting’ or ‘tipped’ position as for many husbandry procedures. 4 The use of a semi-recumbent restraint device for TTE has not been documented to the authors’ knowledge. Our goal was to determine if a sheep chair would provide safe and effective restraint and permit a complete TTE examination in non-sedated ovine subjects.
We observed Dorset sheep undergoing TTE in a separate study that, for its own statistical significance, used 10 animals. 6 Animals were obtained from a closed herd, displayed no clinical signs of brucellosis or foot rot, tested negative for Q fever, met the requirements of the United States Department of Agriculture Scrapie Program, were treated for Coccidia (using amprolium) and vaccinated for tetanus, as well as Clostridium perfringes types C and D. Only male animals (61 ± 4 kg, 8–10 months of age) were used due to their greater docility over females, and to avoid any sex-based variation in behavior; castration was performed as standardized from the supplier. Upon arrival at the research facility, the animals were acclimated in group housing for 7 days then individually housed post-operatively (for the primary study). 7 All procedures were performed in the morning without fasting and on a 12-hour light/dark cycle; animals were deemed healthy on daily veterinary examinations. Humane care was provided in compliance with the Principles of Laboratory Animal Care and the Guide for Care and Use of Laboratory Animals. The study protocol was approved by our local Institutional Animal Care and Use Committee.
The sheep chair consists of loosely woven synthetic fiber netting supported by a rectangular stainless steel frame with two legs and two hooked support beams (Premier 1 Supplies, Washington, Iowa, USA; Figure 1(a)). For the TTE, one handler and one sonographer lifted and tipped the sheep backwards into the chair to place the sheep in partial dorsal recumbency with their hindquarters supported by netting across the bottom of the chair. Approximately one minute was given for acclimatization to the chair. There was no need for more than one handler for the examination, in comparison with two or three handlers for standing examination or traditional sheep tipping. A single handler maintained a relaxed hand on the sheep’s neck or upper thorax and used the other hand to extend each of the sheep’s forelimbs to facilitate access to the imaging window (Figure 1(b)).
Commercial sheep chair (a) and a sheep during transthoracic echocardiographic examination (b).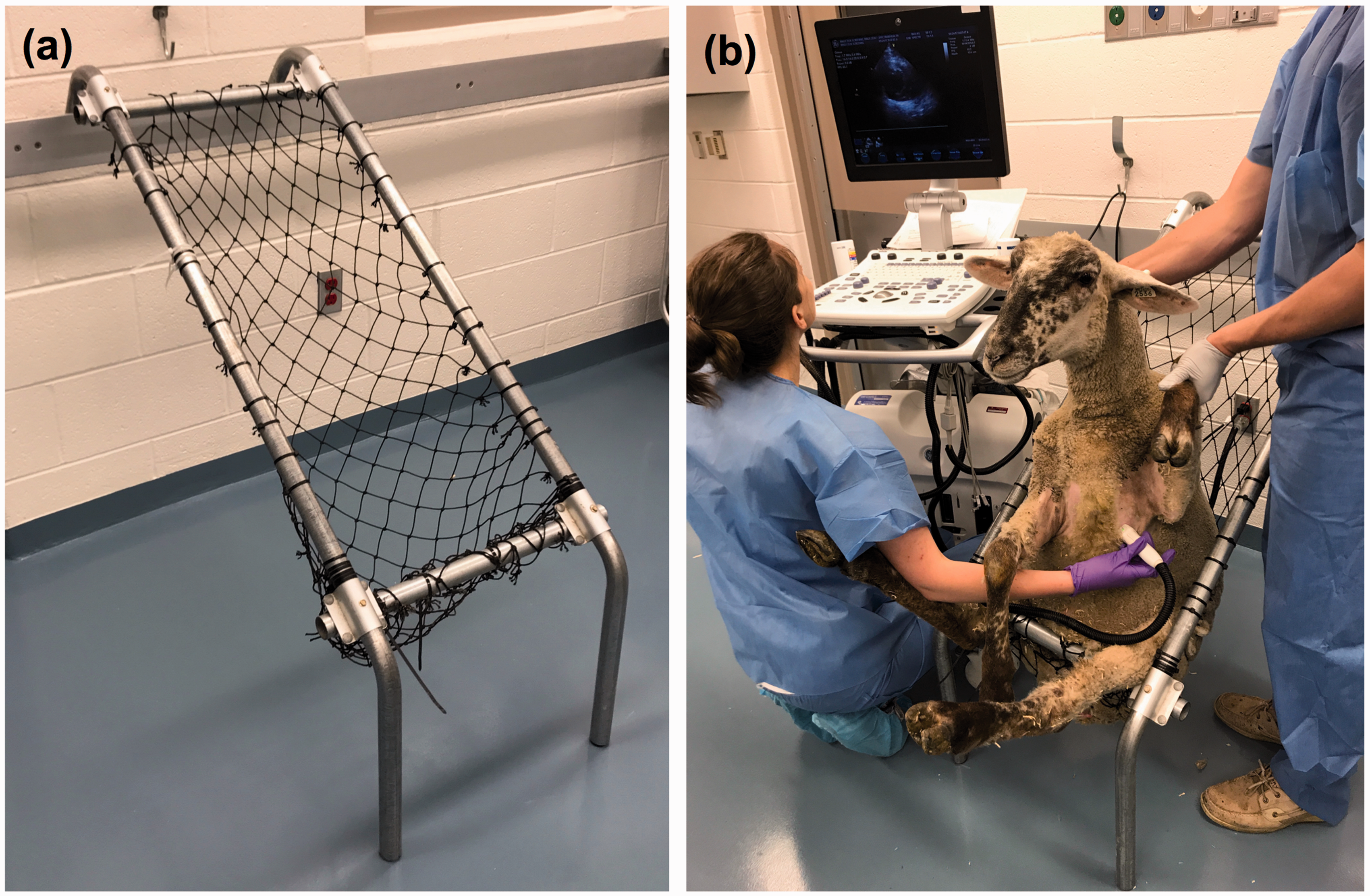
A General Electric Vivid S6 ultrasound machine (GE Healthcare, Wauwatosa, WI, USA), associated three-lead electrocardiogram, and M4S-RS sector phased array probe (1.5–3.6 MHz) were employed for all echocardiograms. Imaging was performed by cardiac sonographers with previous experience with TTE in animal research.
Two-dimensional images were acquired from the long and short axis of the left and right parasternal and apical windows (Figure 2). To acquire the left parasternal window, the probe was placed between the third and sixth intercostal spaces, medial to the axilla, with the indicator directed towards the shoulder (zero degree rotation). To acquire the left apical window, the probe was moved medially towards the sternum and caudally towards xiphoid between the fifth and seventh intercostal spaces. The far field of the beam was directed towards the base of the heart with the apex in the near field. The indicator was considered at zero degrees when a four-chamber view was created. These views were repeated on the right thorax.
Transthoracic echocardiographic image planes obtained from non-sedated sheep restrained in a sheep chair. Image planes: (a) parasternal long axis; (b) parasternal short axis; (c) apical four chamber (AP4CH); (d) apical two chamber (AP2CH); (e) apical three chamber (AP3CH); (f) schematic of short axis image planes providing orientation of probe indicator angulations. Ao: aorta; AoV: aortic valve; LA: left atrium; L: left ventricle; MV: mitral valve; RA: right atrium; RV: right ventricle; TV: tricuspid valve.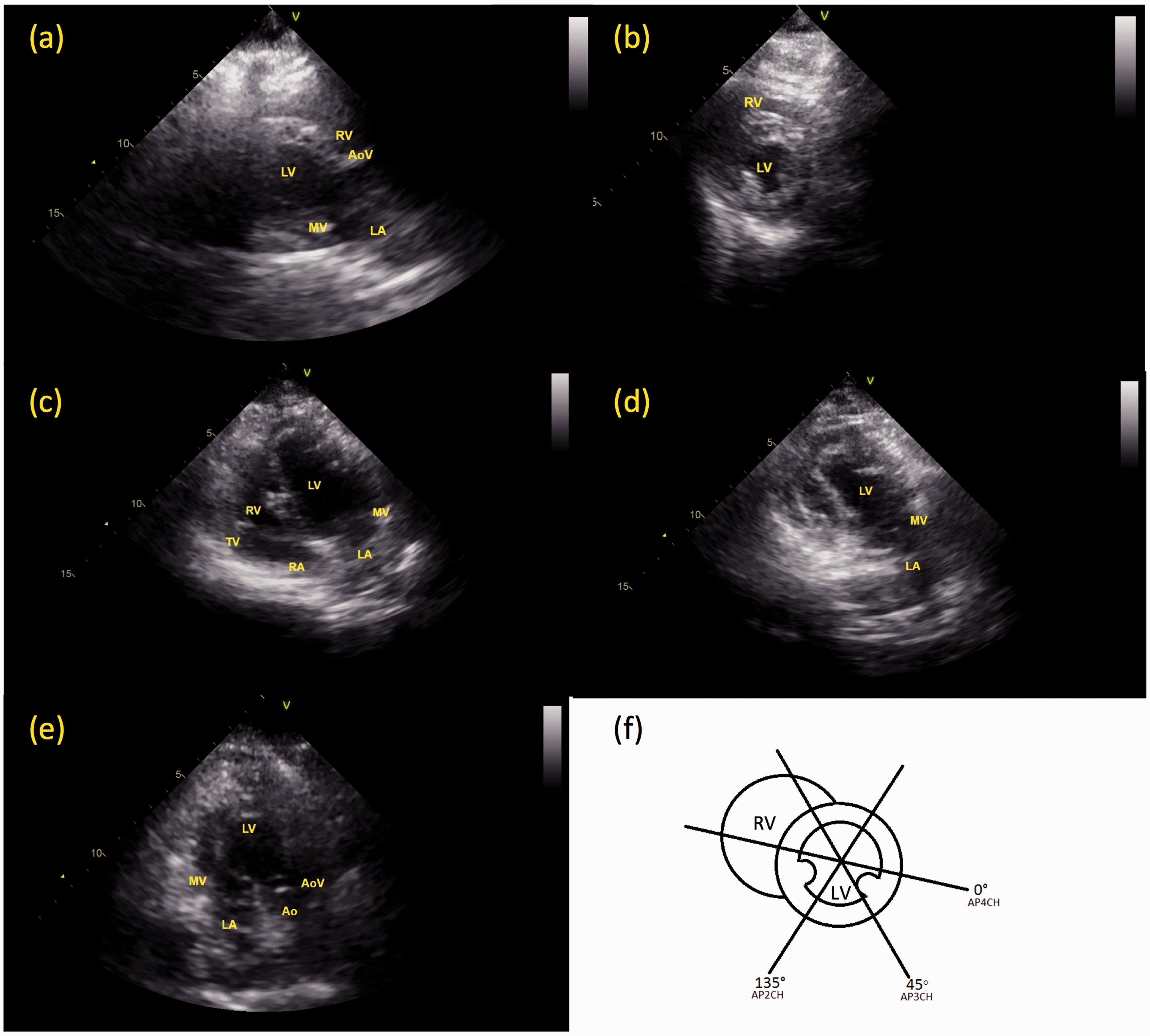
Using the sheep chair, the TTE image quality was sufficient to provide an assessment of cardiac function in each animal. The parasternal image planes and the apical four-chamber image plane were reliable in image quality, however the apical two and three chamber image planes had increased tissue interference (reducing ultrasound beam penetration) and anatomic variability that occasionally resulted in incomplete structure visualization. It was concluded that biventricular assessment was best evaluated from the parasternal windows; wall motion abnormalities, wall thickness, and ejection fraction measurements were appreciated. From the apical four-chamber view, right ventricular chamber assessment and Doppler interrogation of blood flow and annular tissue motion over mitral and aortic valves was achievable.
Herbivores are difficult to assess for behavioral signs of stress because both outward activities and quantitative measures (i.e. heart rate or plasma cortisol levels, are not consistently altered with stress). 6 However, we felt that in comparison with our previous TTE experiences in non-sedated, standing sheep, there was an improved level of animal comfort with the use of the sheep chair. On serial TTE examinations in the sheep chair there were no long-term adverse reactions observed based on their daily habits and close veterinary monitoring.
In conclusion, this was a promising preliminary observational study of the sheep chair as a means of restraint for acquisition of adequate TTE image quality while eliminating the need for sedation, reducing the number of handlers required, and lessening observable animal stress compared with traditional restraint methods. Future studies should quantitate the effectiveness of the sheep chair in stress reduction in comparison with traditional standing TTE, and derive normal echocardiographic reference ranges for non-sedated sheep in the described semi-recumbent position produced by the sheep chair.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Meijer Heart and Vascular Institute internal grant. Michelle Colpitts is the Cardiovascular Imaging Research Fellow and Marcin Malinowski is the Peter C and Pat Cook Endowed Research Fellow in Cardiothoracic Surgery.
